Abstract
Objective:
The aim of this study was to determine the prevalence and antimicrobial susceptibility pattern of Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi among febrile patients at Karamara Hospital, Jigjiga, eastern Ethiopia.
Methods:
A cross-sectional study was conducted among 203 febrile patients presumptive of enteric fever (Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi) at Karamara Hospital from 15 February to 20 March 2016. Venous blood was collected, cultured, and biochemical tests were performed. Antimicrobial susceptibility testing was performed for each isolate using modified Kirby–Bauer disk diffusion technique.
Results:
The overall prevalence of enteric fever (Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi) was 11%. The prevalence of Salmonella enterica serovar Typhi (7%) was higher than Salmonella enterica serovar Paratyphi (4%). The odds of having enteric fever were higher among the study participants aged 31–45 years and with previous history of enteric fever. Most of the Salmonella enterica serovar Typhi isolates were sensitive to tetracycline (78.6%), gentamycin (64.3%), and ceftriaxone (64%), while most of the isolates of Salmonella enterica serovar Paratyphi were sensitive to tetracycline (100%), gentamycin (100%), and ciprofloxacin (62.5%). All the isolates were resistant to ampicillin and chloramphenicol. Multidrug resistances were found among most of the isolates.
Conclusion:
A high prevalence of enteric fever and drug resistance to most commonly prescribed antimicrobials were observed in this study. Those of old age with previous history of enteric infection were more affected by enteric fever. Health information should be given about the transmission, prevention of enteric fever, and antimicrobial use. The treatment of enteric fever should be supported by antimicrobial susceptibility tests in the study areas.
Background
Enteric fever (typhoid fever and paratyphoid fever) is caused by Salmonella enterica serovar Typhi and various strains of Salmonella enterica serovar Paratyphi. It is a major public health problem in developing and developed countries. About 21.6 million people become ill every year, and it results in 216,500 deaths.1,2Almost 80% of the cases and deaths were reported from Asia, while the rest (20%) of them were from Africa and Latin America. 2
Salmonella enterica serovar Typhi was believed to be the major cause of enteric fever worldwide. Salmonella enterica serovar Paratyphi is also becoming a substantial cause of emerging cases of enteric fever from many Asian countries.2,3 Low socio-economic status and poor hygiene conditions1,4 are responsible for the spread of the infection.5,6
The prevalence of enteric fever was reported at varying rates in different studies. Some of them reported an enteric fever rate of 23.1% in Nepal, 14.3% in India, 30.7% in Bangladesh, 14.3% in Nigeria, and 4.1% in Addis Ababa, Ethiopia. Majority of the isolated causative agents were Salmonella enterica serovar Typhi.5–11
The advent of antimicrobial therapy reduced the morbidity and mortality associated with enteric fever, but the emergence of antimicrobial resistant strains jeopardizes these advances. Salmonella enterica serovar Typhi is now known to be markedly resistant to commonly used antimicrobial by patients.12,13 There is also increasing concern about the prevalence of multidrug resistance (MDR) Salmonella enterica serovar Typhi 14 and Salmonella enterica serovar Paratyphi in developing countries.15,16 MDR of Salmonella enterica serovar Typhi to chloramphenicol, amoxicillin, co-trimoxazole,17,18 and ciprofloxacin 14 had been reported as an emerging new challenge in different studies.
The magnitude of enteric fever causative agents and its resistance to commonly used antimicrobial has been reported in few different studies conducted in Ethiopia.11,19–23 Unpublished reports from different health institutions in Jigjiga revealed higher number of clinically presumptive and Widal positives cases. However, the difference in the interpretation of the Widal test with different cut-off values in a variety of geographical areas remains problematic in the diagnosis of enteric fever to this day. The test kit also has low sensitivity and specificity and shows cross reactivity with other diseases. 24 Therefore, this study was tried to determine the prevalence, antimicrobial susceptibility pattern, and associated factors of Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi among febrile patients at Karamara Hospital, Jigjiga, eastern Ethiopia.
Materials and methods
Study setting
Jigjiga is the capital city of Ethiopian Somali Regional State and is located 585 km east of Addis Ababa, the capital city of Ethiopia. The city had a total population of 277,560. It has two public hospitals, two health centers, 25 private clinics, and 11 health posts. Karamara Hospital is one of the public hospitals which gives services to an average of 300 patients per day. This hospital-based cross-sectional study was conducted among febrile patients presumptive of enteric fever at Karamara Hospital from 15 February to 20 March 2016.
Population
All febrile patients, whose age was greater than 5 years attending at Karamara Hospital, were source population. The study population consisting of all febrile patients who came to the outpatients department with complaints of enteric fever, including temperature >39°C, were screened as presumptive enteric fever based on clinical diagnosis and were sent to the hospital laboratory for Widal test. Those febrile patients who had received any antimicrobial treatment for the last 2 weeks and during the study were excluded from the study.
Sample size and sampling technique
Sample size was determined using Epi Info™ version 7.1.6.0 by assuming a power of 80%, ratio of 1:1, 16% typhoid fever infection among patients who have good hand washing habits, and 35.3% among patients who have poor hand washing habits from previous study conducted in northwest Ethiopia. 21 Then, the final sample size after adding 15% non-response rate was 203. Purposive sampling technique was applied to select the study participants.
Data collection methods
Data were collected by the following methods.
Face-to-face interviews
All the study participants were interviewed using pre-tested structured questionnaire by trained health officers. The questionnaire contains information like socio-demographic characteristics, previous antibiotics use, source of drinking water, hand washing habits, and source of food.
Blood specimen collection, culturing, and identification
A venous blood sample (5 mL from adult and 2 mL from children) from each study participant was collected using sterile disposable syringes by trained laboratory technicians as described. 25 It was transferred into the blood culture bottle containing equal volume of tryptic soy broth (OXOID, England) and transported to the Somali Regional Health Public Health Research Laboratory for analysis.
In the laboratory, the inoculated tryptic soy broth was incubated for less than 7 days at 37°C and observed every day for bacterial growth. Those showing growth were removed and subcultured onto MacConkey agar (OXOID, England), xylose lysine deoxycholate (XLD) agar, and salmonella shigella (SS) agar. Phenotypic characterization of the isolates were performed using colony morphology; Gram’s staining; and biochemical tests such as motility test, catalase test, oxidase test, sugar fermentation, indole test, methyl red test, Voges–Proskauer test, citrate utilization test, triple sugar iron test, and urease test.3,5,26
Antimicrobial susceptibility testing
The antimicrobial susceptibility testing was performed using modified Kirby–Bauer disk diffusion technique, according to the Clinical and Laboratory Standard Institute (CLSI) guidelines. 27
In brief, a standard inoculum equivalent to 0.5 MacFarland standard was prepared by mixing three to five morphologically identical colonies of bacteria from pure cultures with 5 mL physiological saline (0.85% NaCl). The suspension was uniformly inoculated onto Mueller–Hinton agar (OXOID, England) plates. Using sterile forceps, antimicrobial disks such as ampicillin (10 µg), norfloxacin (10 µg), ceftriaxone (30 µg), gentamycin (10 µg), tetracycline (30 µg), ciprofloxacin (5 µg), nalidixic acid (30 µg), chloramphenicol (30 µg), and amoxicillin-clavulanate (20/10 µg) were applied to the plates at a distance of 15 mm away from the edge and 24 mm apart from each other. The plates were left at room temperature by inverting upside down for 5 min to allow for the diffusion of the antimicrobials into the agar medium and incubated at 37 C for 24 h. The diameters of the zone of bacterial growth inhibition around the disks were measured to the nearest millimeter using a digital caliper and interpreted as sensitive, intermediate, or resistance based on the CLSI criteria. 27
Quality control
The questionnaire was initially prepared in English and translated into local languages (Amharic and Somali) by a language expert and back to English by another language expert to check it’s consistency. A questionnaire was pre-tested on 5% of the study population at Hiwot Fana Specialized University Hospital, Harar, Ethiopia. A 3-day training was given to data collectors on the study protocol, media preparation,inoculation and antimicrobial susceptibility testing. The questionnaire was checked every day for completeness by the supervisors. Standard operational procedures (SOPs) were followed during sample collection and processing. The American Type Culture Collection (ATCC) reference strains such as Escherichia coli (ATCC-25922), Staphylococcus aureus (ATCC-25923), and Pseudomonas aeruginosa (ATCC-27853) were used to check the quality culture media and antimicrobial disks.
Data processing and analysis
Data were coded, entered, and cleaned using Epi-data (version 3.1) and exported to Statistical Package for the Social Sciences (SPSS; version 16) for analysis. The prevalence was calculated by dividing the frequency of positive samples by the total number of samples examined. Bivariate and multivariate logistic regression analysis was performed to assess factors associated with the outcome variable. Variables with p-value less than 0.3 in bivariate analysis were considered for multivariate logistic regression. Those variables with p ⩽ 0.05 at 95% confidence interval (CI) in multivariate logistic regression were considered as statistical significance factors associated with the outcome variable.
Results
Characteristics of the study participants
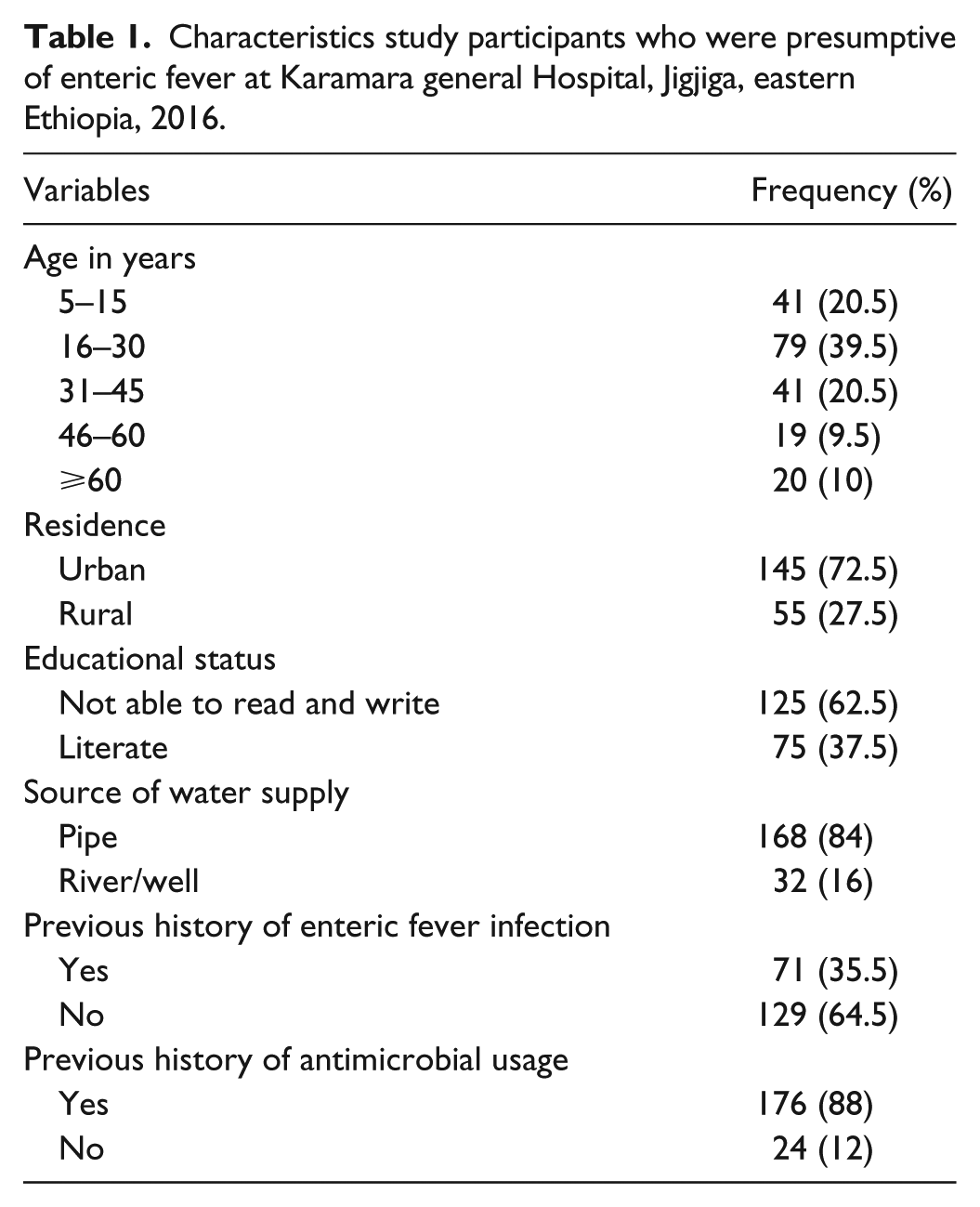
A total of 200 study participants were enrolled in this study with an overall response rate of 98.5%. The age of the study participants ranged from 5 to 90 years. Majority of the participants were 16–30 years (39.5%), males (62.5%), not able to read and write (62.5%), and urban dwellers (72.5%). A large proportion (84%) of the participants acquires drinking water from pipe. The majority (64.5%) of the study participants have not experienced enteric fever before. More than 88% of the participants received antibiotics (Table 1). About 3.5%, 53.5%, and 43% of the study participants used food from street vendors, homemade, and both homemade and street vendors, respectively. About 23.5% of the study participants wash their hands with water and soap before eating, while 76.5% of them wash with water only.
Characteristics study participants who were presumptive of enteric fever at Karamara general Hospital, Jigjiga, eastern Ethiopia, 2016.
Prevalence and factors associated with causative agent of enteric fever
The overall prevalence of enteric fever (Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi) was 11% (95% CI: 7.3–16.1). The majority of enteric fever was caused by Salmonella enterica serovar Typhi (7%; 95% CI: 4.2–11.4).
Those study participants aged 31–45 years (19.5%), male (13.3%), rural (12.7%), literate (11.2%), eat food from street vendor (14.3%), using pipe water supply (11.3%), washing their hands before eating by water (11.2%), previous history of antimicrobial usage (11.9%), and previous history of enteric fever (19.7%) were more infected with enteric fever. Study participants’ age, sex, previous history of antimicrobial usage, and previous history of enteric fever were selected from bivariate for multivariate analysis (p < 0.3).
In multivariate analysis, those study participants in the age group of 31–45 years (AOR: 10, 95% CI: 1.07–93.12) and with previous history of enteric fever (AOR: 4.83, 95% CI: 1.73–13.49) were found to have high odds of enteric fever (Table 2).
Factors associated with prevalence of enteric fever (Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi) among presumptive enteric fever patients at Karamara general Hospital, Jigjiga, eastern Ethiopia, 2016.

p ⩽ 0.05; **p ⩽ 0.01.
Antimicrobial susceptibility patterns of Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi
All Salmonella enterica serovar Typhi (100%) were resistant to ampicillin, chloramphenicol, and amoxicillin, while sensitive to tetracycline (78.6%), gentamicin (64.3%), and ceftriaxone (64.3%). However, 50% of them were sensitive to norfloxacin and intermediate to ciprofloxacin.
Salmonella enterica serovar Paratyphi were 100% resistant to ampicillin and chloramphenicol, 75% to amoxicillin, and 50% to nalidixic acid. Most of them were sensitive to tetracycline (100%), gentamycin (100%), and ciprofloxacin (62.5%). However, 50% of them were sensitive to ceftriaxone (Table 3).
Antimicrobial susceptibility pattern of Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi isolated from presumptive enteric fever patients at Karamara general Hospital, Jigjiga, eastern Ethiopia, 2016.

MDR (resistance to ⩾2 antimicrobials) was observed in 100% of Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi. Majority of the Salmonella enterica serovar Typhi (42.86) isolates were resistant to ampicillin, amoxicillin, and chloramphenicol. About 25% Salmonella enterica serovar Paratyphi were found resistant to different combinations of three and four antimicrobials (Table 4).
Multiple drug resistances pattern of Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi isolated from presumptive enteric fever patients at Karamara general Hospital, Jigjiga, eastern Ethiopia, 2016.

Amp: ampicillin; Amc: amoxicillin; Nor: norfloxacin; Nal: nalidixic acid; Cip: ciprofloxacin; C: chloramphenicol; Gen: gentamycin; Cef: ceftriaxone.
Discussion
In this study, the overall prevalence of enteric fever (Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi) was 11%. This was similar to the study conducted in Bahir Dar, Ethiopia (10.3%) 19 and Zaria, Nigeria(14.3%). 10 While it was higher than other studies conducted in Addis Ababa, Ethiopia (4.1%), 11 Gondar, Ethiopia (4.2%), 20 Ghana (2.4%), 28 and Nepal (8.9%) 7 , it was lower than the report from India (14.3%), 8 and Bangladesh (30.7%). 9
Salmonella enterica serovar Typhi was found higher than Salmonella enterica serovar Paratyphi in this study. The prevalence of Salmonella enterica serovar Typhi was 7%. This was similar to report with the finding from northwest Ethiopia (10.3%), 19 northern Ethiopia (4.2%), 20 and Kenya (6.4%). 29 However, it was higher than reports from Addis Ababa, Ethiopia (4.1%) 11 and Ghana (1.7%). 28 The difference might be attributed to socio-demographic characteristics, environmental sanitation, water supply, level of drug resistance, sample size, study period, method of laboratory examination, and inclusion of study population. For instance, Dagnew et al. 20 included all febrile patients without clinical screening for possibility of enteric fever. This might make the overall prevalence of typhoid fever lower in his study.
The age group of 31–45 years was at higher odds of enteric infection in this study. This finding was comparable with the study conducted in Ethiopia and Nigeria.11,30 However, it was different from other studies conducted in India, Nepal, and Pakistan.6,31,32 This could be due to the difference in proportion of study participants’ in each age group, study period,29,30 geographical location, and others.
In this study, those participants who had previous history of enteric fever have higher odds of enteric fever infection. This might be due to the reactivation of it from the previous infection.5,6 They might not also be cured at the time of treatment because of inadequate/improper treatment or drug resistance.
Optimum antimicrobial treatment of patients with enteric fever depends on the understanding of local patterns of antimicrobial susceptibility of isolates. In this study, most of the Salmonella isolates showed higher resistance to ampicillin, chloramphenicol, and amoxicillin. This was similar to the study conducted in Kenya 33 Ethiopia 20 and one systematic review on antimicrobial susceptibility. 34 This could be due to the ease of availability and usage of these drugs from drug shops/pharmacy and lack of awareness in the administration of antimicrobials. In addition, lower sensitivity of Salmonella enterica serovar Typhi to ciprofloxacin and norfloxacin was observed. This was comparable to the study conducted in Kenya. 33
In this study, almost all the isolates were resistant to two or more antimicrobials. This was similar to the report from Kenya, in which 97.9% of the isolates were resistant to more than two antimicrobials. 35 There is also an increase in MDR of salmonella reports in other studies from developing countries.14–16,33 This is an indication of the indiscriminate use of drugs and the transfer of resistant genes among different species of Salmonella. MDR among Salmonella enterica serovar Typhi isolates possess hazardous effects to human beings due to in-vivo transfer of antibiotic resistant genes to normal microflora of the intestine. This could lead to difficulty in the treatment of infection. 14 Thus, routine screening of antimicrobial susceptibility before prescription to patients is important in order to reduce the spread/development of resistant strains and to improve the prognosis of the patient.
This study has limitations in that it has a low sample size and did not perform serotyping and genotyping of bacterial isolates. In addition, the minimum inhibitory concentration of antimicrobial was not performed for each of the isolates.
Conclusion
The overall prevalence of enteric fever was 11%. The higher prevalence of Salmonella enterica serovar Typhi is of major concern. The age group of 31–45 years and previous history of enteric fever were identified factors related to enteric fever. Most of the Salmonella isolates were sensitive to tetracycline, gentamycin, and ceftriaxone and resistant to ampicillin, chloramphenicol, and amoxicillin. All Salmonella enterica serovar Typhi and Salmonella enterica serovar Paratyphi were multidrug resistant. Health information dissemination should be given to a community about the transmission, prevention of enteric fever, and antimicrobial use. Culture and antimicrobial susceptibility testing facilities should be employed as part of the routine laboratory practice in the study area. Health professionals should also give attention to prescribing antimicrobials for treating enteric fever based on antimicrobial susceptibility test. Tetracycline, gentamycin, and ceftriaxone can be used as part of the empirical treatment for enteric fever in the study area. Further study, which includes serotyping, genotyping, and antimicrobial susceptiblity tests using minimum inhibitory concentration and different risk factors of salmonellosis with a large sample size from different parts of the country, is recommended.
Footnotes
Acknowledgements
The authors are grateful to the study participants. They would also like to acknowledge the Somali Regional Health Bureau for facilitating this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical clearance was obtained from Haramaya University Institutional Health Research and Ethical Review Committee (IHRERC29/2016). Permission was obtained from the Ethiopian Somali Regional Health Bureau.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Haramaya University.
Informed consent
Information about the study was given to each study participant whose age was greater than or equal to 18 years of age and to their parents or guardian for those children less than 18 years of age. Written informed consent/assent was obtained from each voluntary study participant, parent, or guardian before data collection. Those participants who were found positive were treated free of charge according to their drug susceptibility result.
