Abstract
Background:
Urinary tract infections (UTIs) are the most common bacterial infections in children. The high variability in pathogen susceptibility rates leads to the lack of clear guidelines for empiric and targeted therapies. In this view, local microbiological surveillance and locally adapted stewardship interventions need to be implemented.
Objective:
The study aims to describe the results of a pediatric antimicrobial stewardship program on antibiotic prescriptions for UTIs over 8 years in a pediatric general ward of a tertiary center.
Design:
This quasi-experimental study was conducted between 2015 and 2022, with two different implementations, one in 2018 and one in 2021.
Methods:
Demographic, clinical, microbiological, and therapeutic data were retrieved from the electronic clinical records of included patients. The primary outcomes were adherence to local guidelines for empiric therapies and the adequacy of targeted therapy. Secondary outcomes were evaluating antibiotic prescription patterns stratified by antibiotics during hospital stay and at discharge, and assessing the microbiological characteristics of UTI episodes.
Results:
During the study period, 7038 patients were admitted to the pediatric acute care unit (PACU), and 264 (3.7%) were included in this study. Adherence to local guidelines was highest immediately after the interventions, and it slightly decreased thereafter. Use of cephalosporins remained high throughout the 8 years but the changing microbiological scenario observed led to changing recommendations within the study period. An increase in E. coli strains resistant to co-amoxiclav was observed in the last years. Oral second-line agent consumption remained high but was adequate considering the prevalence of resistant bacteria.
Conclusion:
The variability of antimicrobial consumption reflects the changing resistance patterns for UTIs pathogens, underlying the importance of locally adapted, persevering antimicrobial stewardship interventions.
Introduction
Urinary tract infections (UTIs) are the most common bacterial infection in pediatric populations, especially young children, with Escherichia coli being the prevalent pathogen. 1 Resistance of E. coli to commonly used antibiotics, such as aminopenicillin, trimethoprim, cefazolin, and co-amoxiclav, is increasingly reported worldwide.1–4 In the last decades, the emergence of multidrug resistant strains is concerning: Extended-spectrum beta-lactamase (ESBL) producers and carbapenem-resistant Enterobacteriaceae (CRE) pose severe challenges in the choice of antimicrobials, limiting the options for intravenous and oral prescriptions, which are already restricted in pediatrics. The World Health Organization highlighted ESBL-gram-negative bacteria and CRE as important research targets and priorities. 5 This underlines the urge for surveillance and antimicrobial stewardship programs (ASPs) in outpatient and inpatient settings. The concept of ASP was introduced in 2007 by the Infectious Disease Society of America. An ASP is defined as a set of coordinated interventions aimed at optimizing antimicrobial use by selecting the appropriate agent, dose, route of administration, and treatment duration without compromising patient outcomes. 6 The goal is to achieve the best clinical outcomes while minimizing toxicity and other adverse events and reducing the selective pressure on bacterial populations, which can lead to the emergence of antimicrobial-resistant strains. ASPs have been shown to decrease inappropriate antimicrobial utilization and resistance, enhance patient safety, and reduce healthcare costs.7,8
However, considering continuously changing epidemiology, first-line antibiotics for UTIs must be adapted to cover the more probable pathogens guided by local resistance patterns. 9 On the other hand, first-line therapy needs to be as restricted as possible to fight the further emergence of resistance.
This study aims to describe the results of a Pediatric ASP on antibiotic prescriptions for UTIs over a long period in a pediatric general ward of a tertiary center, evaluating its impact on the choice of empiric antibiotic prescription and the switch to oral from intravenous therapy according to guidelines and sensitivity results.
Materials and methods
Study design and setting
This quasi-experimental study was conducted in the pediatric acute care unit (PACU) of the Department of Women’s and Children’s Health of Padua University. This 16-bed unit admits patients from the Pediatric Emergency Department (PED) with infectious or non-infectious conditions. Data were collected retrospectively over an 8 years period, divided into three phases: a pre-implementation phase, from January 2015 to December 2017; a first implementation phase, from January 2018 to December 2020, with the update of UTI local guidelines and educational talks regarding UTI in 2018; a second implementation phase, from January 2021 to December 2022, with a new update of local guidelines, lessons and the development of an app for mobile phones.
Antimicrobial stewardship program
Since 2015, a multistep ASP has been implemented in the Pediatric Department, featuring several key interventions. These include the development of clinical pathways for managing the most common pediatric infectious diseases, the inclusion of infectious disease physicians in the PACU team who participate in ward rounds twice a week, educational sessions led by infectious disease physicians for residents and senior doctors, as well as by residents, involving clinical case scenarios, updates to local guidelines for common childhood infectious diseases, and the introduction of a mobile app to make internal guidelines more accessible. Some of these interventions were generally focused on antibiotic prescription and interpretation of culture results, while others targeted specific infectious diseases.
Considering UTIs, before 2018, no clear indication regarding the type of antibiotics to be used was reported in the local guidelines. Still, physicians could choose between oral or IV therapy with aminopenicillin plus beta-lactamase or a cephalosporin.
In January 2018, UTI local guidelines were updated based on the updated evidence and presented to residents and senior doctors during educational talks. The pathway favored co-amoxiclav or ampicillin-sulbactam as the first-line agent in well-appearing children older than 1 month of age, while for neonate indications did not change (i.e., ampicillin combined with netilmicin or gentamicin). Third-generation cephalosporins were reserved for ill-appearing cases or as targeted therapy according to antibiotic sensitivity.
In April 2021, a mobile application called “Firstline®” became accessible to all residents and senior doctors at the hospital. This app provided easy access to all updated guidelines directly from the physicians’ mobile phones, eliminating the need to search for them on the hospital’s intranet. The major update of the 2021 guidelines, as well as their method of distribution, focused on the recommendation of ceftriaxone as the first-line treatment for infants between 1 and 3 months of age due to increased E. coli strains resistant to co-amoxiclav (probably acquired during birth from maternal flora) observed in this age group in the Veneto region. 9 Furthermore, it was recommended to change the antibiotic to a sensitive one in case of a resistant strain despite a clinical improvement. These new recommendations and the mobile app were presented to all physicians during an educational talk.
Inclusion criteria
The study included pediatric patients admitted to the PACU from PED with an ICD-9 or descriptive diagnosis of UTI between January 1st, 2015, and December 31st, 2022. The list of ICD-9 and descriptive diagnoses used to identify patients is reported in Supplemental Table S1. Children admitted from other hospitals with an ongoing antibiotic prescription were excluded. Data regarding patients who were transferred to another ward/hospital were limited only to their admission to PACU.
Children with comorbidities such as nephron-urological malformations or immunocompromising conditions or with a concomitant diagnosis that could have changed antibiotic choice were included in the descriptive analysis but excluded from the analysis regarding adherence to local guidelines, while children transferred in other wards were excluded from the analysis regarding targeted therapy if the transfer happened before the cultures were available.
Outcomes
Primary outcomes were the evaluation of adherence to local guidelines regarding the choice of empiric therapy and the adequacy of targeted oral therapy based on sensitivity results.
Adherence to empiric treatment recommended by local guidelines was evaluated from 2018 to 2022, excluding years before the first update, due to the absence of precise first-line empiric treatment. The evaluation of empiric first-line choices took into account the age of patients, the clinical appearance at the initial assessment in the PED, and the approved guidelines at the moment of the evaluation (see Supplemental Figure 1).
Based on sensitivity results, adherence to guidelines regarding the selection of oral target therapy was evaluated for all patients discharged with oral antibiotic therapy after IV treatment. A therapy was considered adequate if the antibiotic with the narrowest spectra among those tested in sensitivity results. The use of co-amoxiclav in patients with clinical improvement but resistant E. coli was considered appropriate between 2018 and 2020, in line with internal and Italian guidelines. 10
Secondary outcomes were the evaluation of antibiotic prescription patterns stratified by type of antibiotic during hospital stay and at discharge, and the assessment of the microbiological characteristics of UTI episodes, considering the susceptibility profiles of E. coli (resistance to co-amoxiclav, ESBL producers, and other types of resistance).
Data collection
Demographic, clinical, microbiological, and therapeutic data were retrieved from the electronic clinical records of included patients. For antibiotic prescriptions, type of drug and route of administration were collected. Data were manually collected from electronic medical records using a password-protected REDCap data collection form. All data were stored in the secured server at Padua University. To guarantee anonymity and privacy, a unique study-specific number was assigned to each patient.
Statistical analysis
The analysis had a mainly descriptive nature. Categorical variables were expressed using numbers and percentages, and continuous variables were expressed as medians with interquartile ranges (IQR). Categorical variables were compared with a chi-squared test, while continuous variables were compared by the non-parametric Kruskal–Wallis rank sum test. A two-sided p < 0.05 was considered statistically significant. Analysis was performed using R statistical software–2023.12.1 (R Foundation for Statistical Computing, Vienna, Austria). 11
Results
Between 2015 and 2022, 7038 patients were admitted to the PACU, and 264 (3.7%) were included in this study: 111 (111/2638, 4.2%) in the pre-implementation phase, (84/2676, 3.1%) in the first implementation phase, and 69 (69/1724, 4%) in the second implementation phase. Median age was similar between the 8 years of the study (median age 79.0 days, IQR 31.8–311.3), with a higher prevalence of children under 3 months of age (more than 50% in almost all years, except 2018) and of male patients (168 patients, 63.6%). There was no difference in the presence and type of comorbidities throughout the 8 years of the study, with nephron-urologic disease being the most common comorbidities (13.6%). The rate of patients admitted with ongoing antibiotic therapy was similar throughout the periods. More than 90% of patients in almost all years were admitted for upper UTIs, and only in two cases for renal abscess. Lower UTIs were less frequent. Table 1 summarizes the demographic and clinical characteristics of included patients.
Characteristics of the included population.

IQR, interquartile range; UTI, urinary tract infection.
Primary outcomes
Adherence to recommendations for empiric therapy
In 2018, immediately after the local guidelines update, the choice of empiric antibiotic therapy was adherent to guidelines in more than 90% of cases. This adherence decreased in the following years and reached 75% in 2020. In 2021, after the second local guidelines update and the app’s release, the empiric therapy adherence to guidelines increased again by over 80%, with a new decrease the year after (Figure 1(a)). The number of access to the app increased during the study period, from less than 100 per month in the first months to more than 200 per month in the last months. UTIs were the second most commonly viewed guidelines.
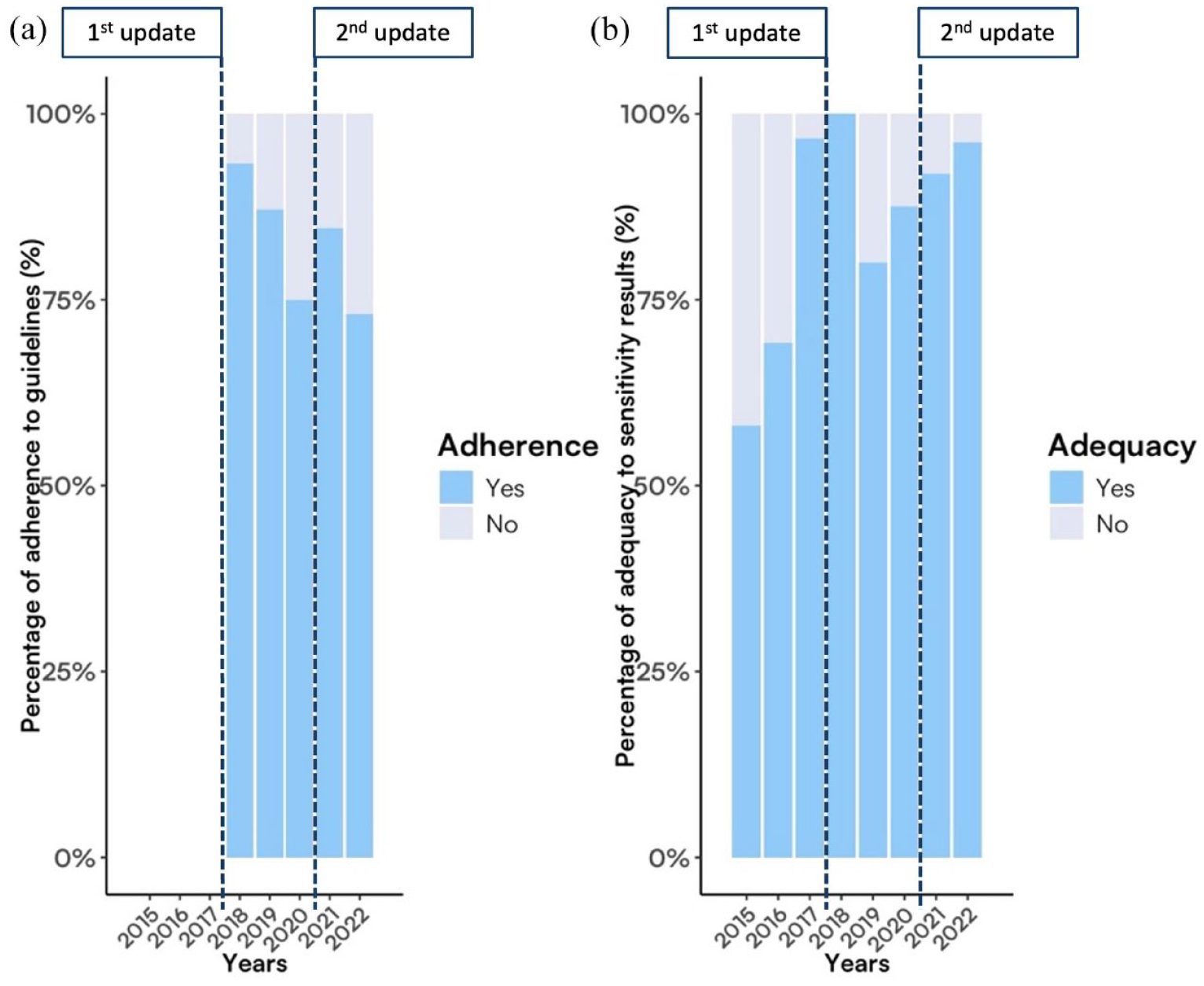
(a) Adherence to the choice of empiric antibiotic therapy based on the internal guidelines for patients with UTI and (b) Adequacy of IV to oral switch based on the sensitivity results of urine culture.
Adequacy to sensitivity results for target therapy
In the first 2 years of the pre-implementation period, more than 30% of antibiotics had a spectrum broader than needed (42% in 2016 and 31% in 2016). Since 2017, the choice of the antibiotic with the narrowest spectra among those sensitive to antibiograms increased and reached complete adequacy in 2018. After a drop in 2019 (75%), a progressive increase in adequacy was observed, reaching more than 90% in 2022 (Figure 1(b)).
Secondary outcomes
Antibiotic prescription patterns
During 2018 and 2019, there was a high increase in co-amoxiclav prescriptions, with a concomitant reduction in the prescription of third-generation cephalosporins, especially in 2018. Nevertheless, during the last 3 years, the number of prescriptions for third-generation cephalosporins and broad-spectrum antibiotics (meropenem, piperacillin-tazobactam, cefepime) increased again (Figure 2(a)).

Antibiotic prescription pattern: (a) antibiotic prescription prevalence rate stratified by years considering hospital antibiotic prescription (IV and oral) and (b) antibiotic prescription prevalence rate stratified by years considering oral antibiotic prescription after IV therapy.
Considering the switch from IV to oral, the number of prescriptions for second- and third-generation cephalosporins decreased over the 8 years with some variability (Figure 2(b)).
Evaluating the choice of antibiotic (empiric or target therapy), the most common antibiotics prescribed as empiric were third-generation cephalosporins (ceftriaxone and cefotaxime), the combination of ampicillin and aminoglycosides (standard treatment for neonates with fever) and the combination of aminopenicillin with beta-lactams inhibitor (co-amoxiclav and ampicillin-sulbactam). Other broad-spectrum antibiotics such as meropenem and piperacillin-tazobactam were prescribed less frequently as first-line (empiric) treatment, while they were more commonly prescribed as second-line (target) treatment. Most patients received an oral antibiotic as a second treatment, such as co-amoxiclav or second- or third-generation cephalosporins. These antibiotics were the most commonly prescribed as a third treatment as well.
Overall, the antibiotic that was switched the most was third-generation IV cephalosporins, especially second- or third-generation oral cephalosporins and oral co-amoxiclav. The second most frequent class of antibiotics switched was IV aminopenicillin associated with beta-lactamase inhibitor, to oral co-amoxiclav and second- or third-generation oral cephalosporins (Figure 3).
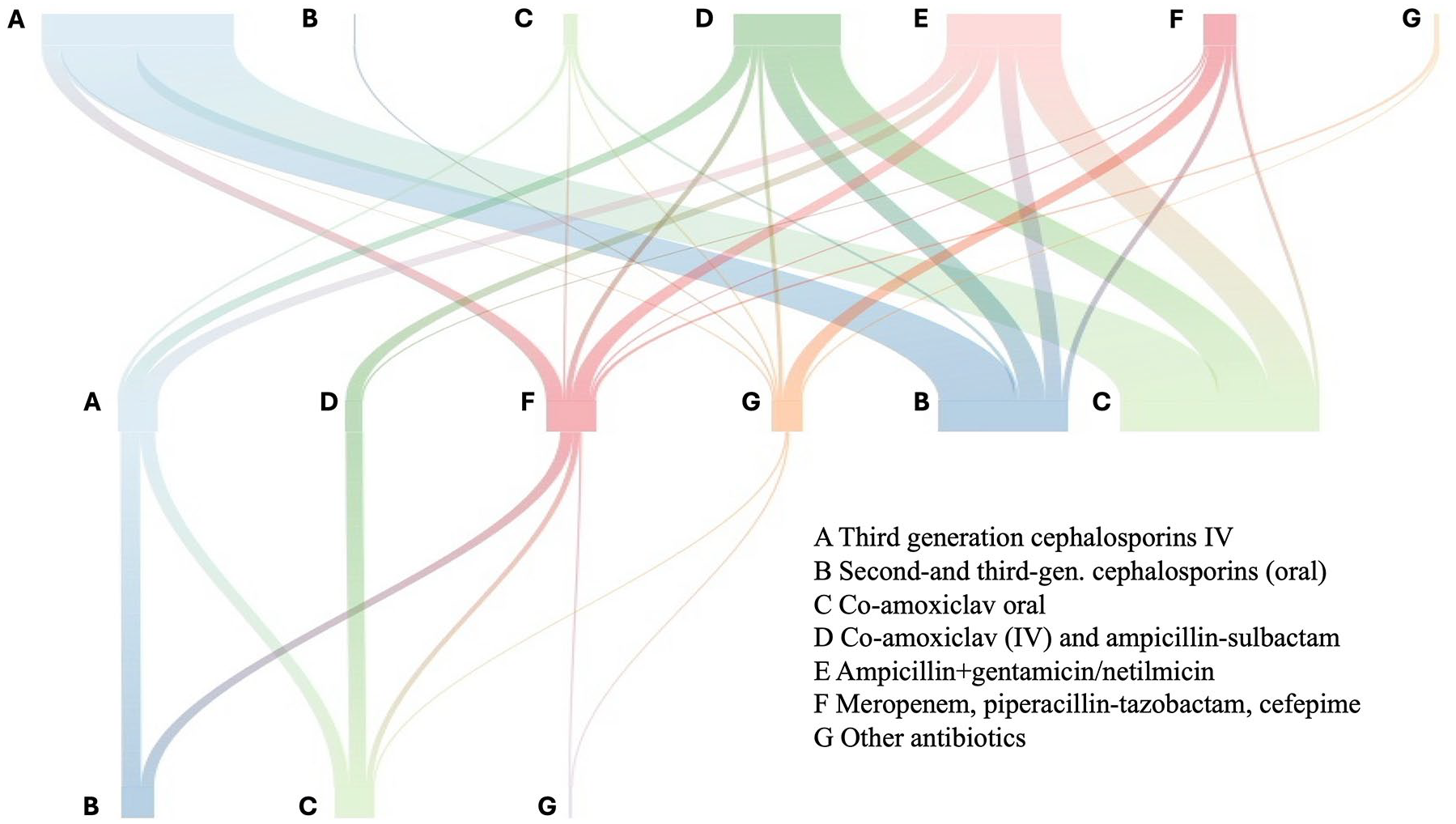
River plot of antibiotic treatment switching. The rows of letters represent the prescriptions grouped by antibiotic classes/molecules in timely order from the upper part (first prescription) to the bottom part (last prescription, oral), and the colored lines show the connection with the following prescriptions in the same treatment episode.
Microbiology results
The most common pathogen identified in urine was E. coli, representing 82.6% of all pathogens in the 8 years. Other bacteria identified were Klebsiella spp. and P. aeruginosa (Table 2).
Microbiological results of patients admitted with UTI.

CSF, Cerebrospinal Fluid; IQR, interquartile range; UTI, urinary tract infection.
Fourteen patients had positive blood cultures: eight for E. coli, two for Klebsiella spp., one for Citrobacter spp. Only one patient had a positive bacterial CSF culture, with Citrobacter spp. isolated.
Looking at the resistance pattern of E. coli, an increase in E. coli strains resistant to co-amoxiclav was observed in the 8 years, increasing from 13.9% in 2015 to 48.1% in 2022, with a peak of almost 70% in 2021. ESBL strains were almost always lower than 10%, except in 2018 when they reached a peak of nearly 30% (Figure 4).
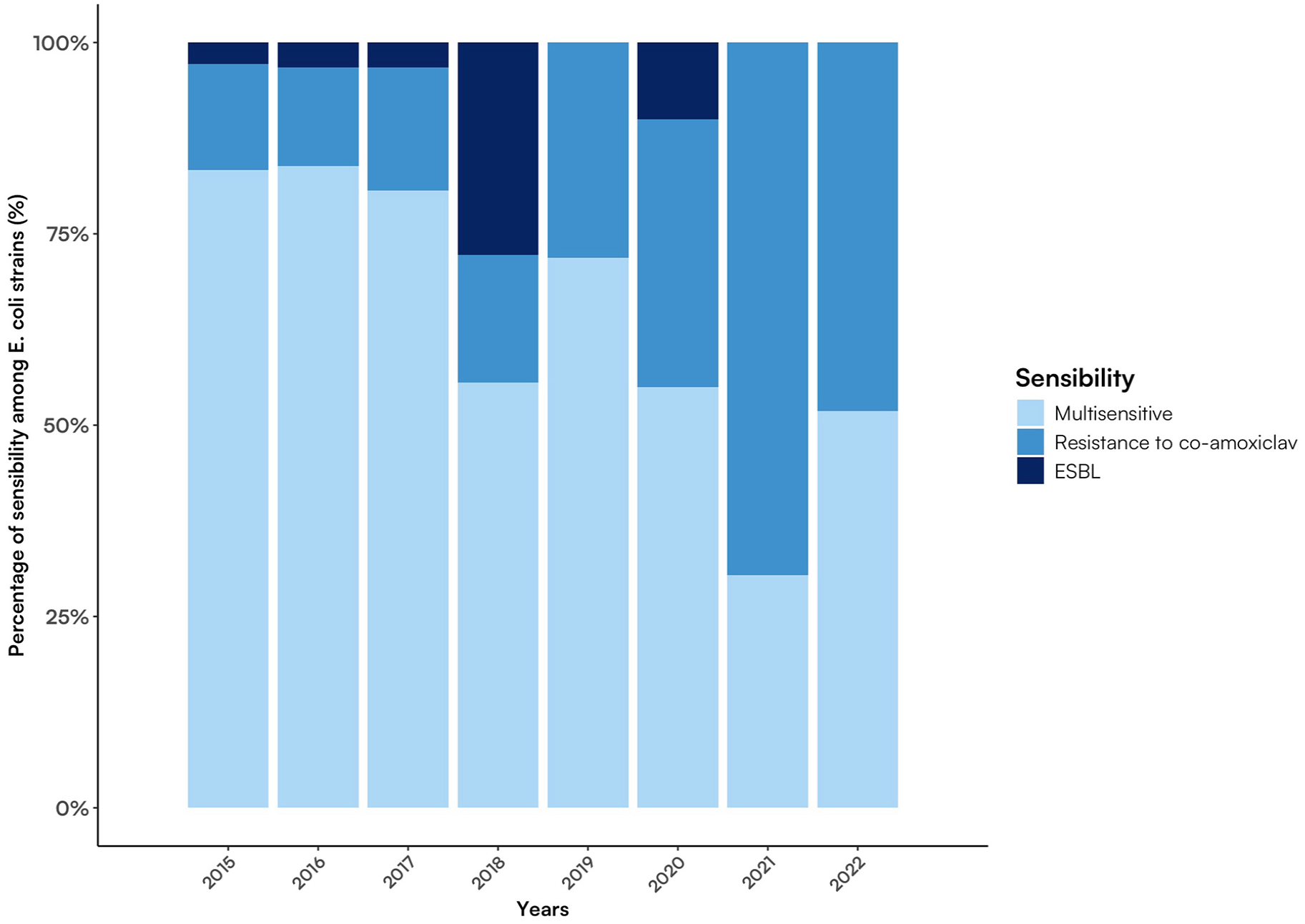
Sensibility to antibiotics among E. coli strains isolated in patients admitted with UTI.
Discussion
This study describes the implementation of ASP UTI-focused interventions into a multistep, multi-sided stewardship program over 8 years. This program started at the end of 2015 and was conducted as a clinical practice intervention to improve empirical antibiotic prescriptions. It was composed of a combination of actions pointing to the same objectives: providing clear and updated information and guidelines to all prescribing physicians rounding in the emergency department and pediatric wards, with regular frontal lessons, and with the presence of the ID specialist in the ward twice a week. The introduction of the app Firstline® in 2021 represented a step forward to improve the accessibility of our local guidelines. The three phases identified in this study served as a cut-off between the two updates of the guideline regarding UTI.
In 2018, the year in which UTI local guidelines were updated for the first time, the proportion of empiric therapy adherent to guidelines was very high, over 90%. At the beginning of that year, the new updates were presented to all physicians, residents, and senior doctors as part of the ASP. In 2019, adherence to guidelines decreased, reaching 75% of adherence in 2020. No more lessons were taken in these 2 years until the new guideline update in 2021 when the latest recommendations and the mobile app were presented again to all physicians. Therefore, adherence to empiric therapy increased again. This trend of adherence is in line with the literature. Numerous studies have reported that educational sessions and continuous recaps with lessons are an essential part of ASP, even more so in settings with a high turnover of physicians, such as in emergency departments, where empiric therapies are usually started. 12 In the second phase of the implementation, adherence to empiric therapy decreased more quickly than in the period before, dropping under 75% of adherence in 2022. However, in 2021, the number of E. coli resistant to co-amoxiclav increased, reaching almost 75% of strains isolated on the urine culture of children admitted to PACU. It is possible that considering these data, physicians decided to start with third-generation cephalosporins instead of co-amoxiclav in children older than 3 months and good clinical conditions despite the recommendations, increasing the number of non-adherent treatments.
The choice of the more adequate antibiotic based on sensitivity results increased the year before the first guidelines update. However, these updates and lessons were part of a bigger ASP implemented since 2015. Although no specific interventions for UTIs were done in the years before, it is possible that physicians were sensitized to choose the adequate therapy in each situation. Adequacy of target therapy followed a similar pattern as adherence, with a drop in 2019 and a subsequent new increase, remaining over 90% after the second educational talks.
The increase in guideline adherence observed in our study aligns with findings from other published studies. For instance, Metz et al. found that implementing an ASP in a secondary care children’s hospital significantly increased guideline adherence from 33% to 63%, improving the choice of antibiotics by reducing cephalosporin use and increasing aminopenicillin use. 13 Similarly, Walters et al. reported an increase in guideline adherence for both the diagnosis and treatment of UTIs following ASP implementation. 14 On the other hand, some studies have reported no difference in guideline compliance. For example, Shaw et al. found no difference in adherence to pneumonia treatment recommendations, despite physicians being aware of the guidelines, concluding that knowledge does not always translate into clinical practice. 15 Furthemore, to sustain results over time, repeat educational talks are crucial, as demonstrated by a recent study published by our team in 2020. This paper compared the implementation of clinical pathways (CPs) for the most common infections in children in three different settings with different approaches: dissemination of CPs without educational talks, dissemination of CPs with one educational talk, and dissemination of CPs with continuous educational talks. Although good results were immediately achieved in all the centers, only the latter center maintained the good results achieved, whereas the other centers went to the prescription pattern pre-implementation. Lessons and educational talks are indeed essential to maintain the results achieved. 12
Considering the type of antibiotic prescribed as empiric therapy, in the pre-implementation period, the existing UTI guideline leaves the choice to prescribers between aminopenicillin plus beta-lactam inhibitor or cephalosporins. According to most guidelines at that period, the choice of antimicrobial for UTIs should be guided by local epidemiological background.16–18 Almost half of the prescribed drugs in this period were third-generation cephalosporins and broad-spectrum antibiotics such as meropenem and piperacillin-tazobactam. This reflects the tendency of physicians to prescribe broader-spectrum antibiotics in the lack of a first-line therapy indication. After the first update of the guidelines, the use of co-amoxiclav started to increase, reaching its peak in 2019.
In 2018, higher use of broad-spectrum antibiotics such as meropenem and piperacillin-tazobactam was observed compared to the other years. However, looking at the microbiological results, 2018 was characterized by a high percentage of ESBL E. coli strains, which could justify the increase in the use of antibiotics such as meropenem and piperacillin-tazobactam. In 2020, the use of co-amoxiclav decreased, with an increase in IV and oral cephalosporins prescriptions. Although this result could be solely linked to the increase in E. coli strains resistant to co-amoxiclav, it is essential to highlight that the 2020 COVID-19 pandemic changed healthcare organizations worldwide. As reported by numerous studies, the COVID-19 pandemic undermined the results achieved up to that point with the antibiotic stewardship program worldwide.19,20 Due to lockdown and restrictions to avoid the spread of the novel Coronavirus, many antimicrobial stewardship activities were reduced, especially audits, quality improvement initiatives, education, training, ward rounds, and multidisciplinary meetings. In our department, at the beginning of the COVID-19 pandemic, all the lessons and updates of guidelines were suspended, and the ID physician could not regularly participate in the ward rounds due to the restriction rules. These changes could have increased the difference in prescriptions and adherence to empiric therapy observed in 2020 compared to other years.
In 2021, the recommendations regarding empiric treatment for children younger than 3 months of age changed. From 2016 to 2018, a microbiological survey study for UTI was conducted as part of the stewardship program. Urine culture results were collected from outpatients evaluated in PED of our hospital and from another territory net center, providing an epidemiology scenario specific to our setting and the population and syndrome of interest. It was estimated that co-amoxiclav provided an overall 80% coverage, decreasing in children with renal/urological comorbidities and neonates. For these reasons, starting in 2021, with the second update of local guidelines and the dissemination of the electronic app, besides complicated cases, children younger than 3 months (considered possibly colonized with maternal pathogens) were recommended to be empirically treated with a cephalosporin, pending from cultures results. Cephalosporins are an acceptable option because of their antimicrobial spectrum, with data showing efficacy as empiric therapy even in the case of pathogens producing ESBL in children, 21 although trials are missing and strong recommendations cannot be made. As a result, in 2021 and 2022, the use of cephalosporins increased. During these 2 years, our study concomitantly observed an increase in co-amoxiclav-resistant E. coli compared to the pre-implementation phase. The containment measures due to the COVID-19 pandemic limited the access of patients to healthcare services; this resulted in a tendency to prescribe, from primary care services, broad-spectrum antibiotics for uncomplicated respiratory infections (pneumonia, pharyngitis) or to prescribe antibiotics as cautioned behavior in the impossibility of face-to-face visit. 22 The exposure of children to antibiotics has been shown to increase the possibility of urinary tract infections caused by resistant pathogens.23,24 The magnitude of this effect decreases with time since exposure. 25 This could explain the microbiological features of UTI during the second implementation phase, assuming that containment measures did not impact UTI occurrence and did not decrease during the SARS-CoV-2 pandemic, as shown in this study. The lack of stewardship programs in outpatient settings, even impaired during a pandemic, contributes to unnecessary or inappropriate prescriptions, increasing the rate of multi-drug-resistant bacteria.
The use of oral cephalosporins as target therapy after IV treatment was very high in the first year of the pre-implementation period, and the choice of antibiotics needed to be more adequate compared to sensitivity results. Since 2016, oral co-amoxiclav use has increased, probably due to the physicians’ sensibilization to choose adequate antibiotics based on sensitivity results. Furthermore, according to 2019 national UTI guidelines, the possibility of leaving co-amoxiclav in the case of resistant pathogen with clinically improving patients was provided by ID consultations, 10 according to case-by-case evaluation. This was consistent with a study showing that 83.5% of discordant therapy in third-generation-resistant UTIs experienced clinical improvement and urine sterilization (65%). 26 However, in the years after the first and the second implementation, the number of oral cephalosporins prescribed started to increase again due to the increase in E. coli strains resistant to co-amoxiclav, especially in the last 2 years, being almost always adequate compared to the sensitivity results.
Although the high use of cephalosporins as empiric therapy was not always adherent to guidelines, the switch to oral therapy was more often adequate to sensitivity results. However, infants younger than 3 months could appear more ill than older children, leading physicians to prescribe broad-spectrum antibiotics more often. The choice of targeted therapy was based on microbiological results and usually decided when the child is in good clinical condition, leading to a more adequate choice in line with the sensitivity results. In our study, infants younger than 3 months of age represent more than 50% of the total population annually and could represent one of the reasons for the persistent high use of cephalosporins. Besides, we considered only children admitted to PACU, who probably have a more severe disease than children discharged home and who could have already had ongoing antibiotic therapy, leading to the decision to prescribe antibiotics as cephalosporins.
The strength of this study lies in its multi-approach, dynamic, and evolving intervention. PEDs and PACUs typically experience a high turnover of patients, with limited time available for each patient, and a high turnover of residents and senior doctors. This makes it challenging to ensure consistent alignment of knowledge among all healthcare providers. Our interventions were designed to be simple and practical for physicians to integrate into their daily routines with minimal effort, sometimes even simplifying their normal workflow. This approach ensured little disruption to their established routines, allowing them to incorporate our intervention into their clinical practices seamlessly.
Pediatric ASP UTI-focused interventions have been described. Most studies had diagnostic accuracy as their main aim, to avoid unnecessary treatment courses for children with negative urine cultures.27–29 This is out of the present study’s scope; however, it remains a fundamental step in managing fever without source in pediatric Emergency Department (ED). This study is in line with a previous by Walters in 2019, describing a similar intervention in a pediatric ED, including developing a UTI algorithm, feedback, and peer-coaching strategies during the implementation phase. The main aims were to achieve 100% urine collection before starting antimicrobials and 80% therapies prescribed according to the provided algorithm; both confirmed the validity of a constant, dynamic, and locally tailored set of interventions. 14
As the epidemiology of pathogens considerably changed during the study period, the variability of results is related to indirect effects, confirming the adherence to guidelines, which have changed over time. In our study, the pandemic led to the interruption of face-to-face consultation. On the other hand, providing an electronic tool like the app was primarily appreciated and used, and it remains essential nowadays.
The study’s limitations include its retrospective nature, the missed opportunity to catch prescriptions from transferred patients, and the lack of clinical outcomes. Furthermore, we collected data only on hospitalized patients and not on patients evaluated in the emergency department and discharged home with oral antibiotic therapy. Indeed, it is possible that we considered patients with more severe disease and a previous failure of oral antibiotics at home, which can justify the use of broader-spectrum antibiotics and the high prevalence of E. coli resistant to co-amoxiclav that we found.
Conclusion
ASP based on guidelines and education for pediatric UTIs needs to be locally adapted to follow microbiological changes. Susceptibilities surveillance is paramount in the ASP initiative to decide on empiric therapy, especially when clear and robust recommendations are missing. These interventions have proven feasible and low-cost, with efficient use of resources, in improving physicians’ adherence to empirical and targeted antibiotic therapy, especially in complex populations such as infants under 3 months old. However, these efforts cannot be sustained, and sometimes not even achieved, without intermittent and frequent educational talks and reminders of the guidelines, as adherence tends to decrease following the initial implementation. This underscores the critical importance of education in maintaining and enhancing the effectiveness of ASP initiatives.
Supplemental Material
sj-docx-1-tai-10.1177_20499361241282824 – Supplemental material for Impact of an antibiotic stewardship program on adherence to antibiotic prescription in children admitted with urinary tract infection
Supplemental material, sj-docx-1-tai-10.1177_20499361241282824 for Impact of an antibiotic stewardship program on adherence to antibiotic prescription in children admitted with urinary tract infection by Giulia Brigadoi, Cecilia Liberati, Emelyne Gres, Elisa Barbieri, Elena Boreggio, Sara Rossin, Francesca Tirelli, Francesca Tesser, Lorenzo Chiusaroli, Giulia Camilla Demarin, Linda Maestri, Carlo Giaquinto, Liviana Da Dalt, Silvia Bressan and Daniele Donà in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-jpg-2-tai-10.1177_20499361241282824 – Supplemental material for Impact of an antibiotic stewardship program on adherence to antibiotic prescription in children admitted with urinary tract infection
Supplemental material, sj-jpg-2-tai-10.1177_20499361241282824 for Impact of an antibiotic stewardship program on adherence to antibiotic prescription in children admitted with urinary tract infection by Giulia Brigadoi, Cecilia Liberati, Emelyne Gres, Elisa Barbieri, Elena Boreggio, Sara Rossin, Francesca Tirelli, Francesca Tesser, Lorenzo Chiusaroli, Giulia Camilla Demarin, Linda Maestri, Carlo Giaquinto, Liviana Da Dalt, Silvia Bressan and Daniele Donà in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
Elisa Barbieri’s position is currently supported by the European Union – Next Generation EU under the National Recovery and Resilience Plan (NRRP) (project INF-ACT – One Health Basic and Translational Research Actions addressing Unmet Needs on Emerging Infectious Diseases).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
