Abstract
Background:
Antibiotics remain the most prescribed medicine in children worldwide, but half of the prescriptions are unnecessary or inappropriate, leading to an increase in antibiotic resistance. This study aims to systemically review the effects of different Antimicrobial Stewardship Programmes (ASPs) on reducing the rates of both antibiotic prescriptions and changes in antimicrobial resistance, and on the economic impact in paediatric emergency departments (PED) and primary care settings.
Materials and methods:
Embase, MEDLINE, and Cochrane Library were systematically searched, combining Medical Subject Heading and free-text terms for ‘children’ and ‘antimicrobial’ and ‘stewardship’. The search strategy involved restrictions on dates (from 1 January 2007 to 30 December 2020) but not on language. Randomized controlled trials, controlled and non-controlled before and after studies, controlled and non-controlled interrupted time series, and cohort studies were included for review. The review protocol was registered at the PROSPERO International Prospective Register of Systematic Reviews: Registration Number CRD42021270630.
Results:
Of the 47,158 articles that remained after removing duplicates, 59 were eligible for inclusion. Most of the studies were published after 2015 (37/59, 62.7%) and in high-income countries (51/59, 86.4%). Almost half of the studies described the implementation of an ASP in the primary care setting (28/59, 47.5%), while 15 manuscripts described the implementation of ASPs in EDs (15/59, 25.4%). More than half of the studies (43/59, 72.9%) described the implementation of multiple interventions, whereas few studies considered the implementation of a single intervention. Antibiotic prescriptions and compliance with guidelines were the most frequent outcomes (47/59, 79.7% and 20/59, 33.9%, respectively). Most of the articles reported an improvement in these outcomes after implementing an ASP. Meanwhile, only very few studies focused on health care costs (6/59, 10.2%) and antimicrobial resistance (3/59 5.1%).
Conclusion:
The implementation of ASPs has been proven to be feasible and valuable, even in challenging settings such as Emergency Departments and Primary care.
Keywords
Background
Antibiotics are the most prescribed medicines in children worldwide, especially in outpatient settings.1,2
In the United States, 41% of all outpatient prescriptions were written by family practice doctors, paediatricians, and internal medicine physicians, as reported by Zetts et al. in their review. Of these, at least 30% were considered unnecessary, with the majority prescribed for patients with acute respiratory tract infections. 3
Data from the United States reported that emergency departments (EDs) receive around 30 million paediatric visits annually, with about 7 million associated antibiotic prescriptions; half of these are unnecessary or inappropriate. 4
A cross-sectional observational study in 28 European EDs reported that 19–64% of children receive antibiotics inappropriately, especially broad-spectrum ones. 5 Antibiotic overprescribing and misuse has led to an increase in antibiotic resistance rates. Indeed, although resistance can occur naturally, antibiotic overuse plays a key role in selecting multi-drug-resistant organisms. 6
The concept of Antimicrobial Stewardship Programmes (ASPs) was formally introduced in 2007 by the Infectious Disease Society of America (IDSA). 7 ASP is defined as a set of coordinated interventions designed to improve antimicrobial use in selecting the appropriate agent, dose, route of administration, and therapy duration without compromising patient outcomes.
ASPs are mainly based on two core strategies: ‘prospective audit and feedback’, which involves interaction and feedback between an infectious disease physician and the prescriber, and ‘formulary restriction and preauthorization requirements’ for specific agents.
However, these standard stewardship approaches recommended for the hospital setting are challenging to implement in the ED or primary care settings.
Significant challenges for the implementation of ASP in primary care settings consist in a lack of funding resources to support a programme; identifying a clinical leader who has the time and interest to commit; engaging outpatient prescribers in a quality improvement initiative; obtaining data to identify high impact targets, tracking process improvements; and sustaining these improvements over time. 8
Paediatric EDs (PEDs) are uniquely positioned at the interface of inpatient and outpatient settings and remain a hybrid in which elements of both inpatient and outpatient stewardships are generally merged. Indeed, PED physicians could influence prescribing trends in patients discharged to home and those admitted to the ward.
As reported by Mistry et al., reducing inappropriate antibiotic use in PEDs is not easy because of unique operational, provider-level, and system-level barriers native to paediatric, general, and community ED environments. Challenges in antibiotic prescribing in this setting include high patient and practitioner turnover rates and rapid decision-making. Furthermore, this makes the development of ASP interventions, like prospective audits and feedback or formulary restriction, quite difficult. 9
However, APSs in the outpatient settings are clearly necessary. In 2019, a survey on National Paediatric Antibiotic Stewardship Programmes, Networks and Guidelines was conducted in 23 European Countries. This survey reports a fragmented implementation of Paediatric Antibiotic Stewardship Programmes (PASPs) in Europe, a lack of established PASP competencies, an almost complete lack of national PASP networks, and an absence of dedicated funding to support the implementation of PASPs at a national level. 10
Given the high rates of antimicrobial resistance documented in Europe, developing a robust and committed PASP strategy has become critical. In order to identify the best available strategies, we systematically reviewed published studies that evaluated the different types of ASPs by assessing their impact on three aspects:
The antibiotics prescription rate, especially of broad-spectrum antibiotics.
The antimicrobials resistance rate.
The health care costs in the PED and primary care settings.
Materials and methods
Search strategies and study design
This systematic review is based on the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. 11 A systematic search was conducted in Embase, MEDLINE, and Cochrane Library database. The search strategy combined Medical Subject Heading (MeSH) and free-text terms for ‘children’ and ‘antimicrobial’ and ‘stewardship’. To identify all possible interventions recognized as stewardship, we expanded the search strategy to also include specific intervention MeSH terms.
The search strategy involved restrictions on dates (from 1 January 2007 to 30 December 2020) but not on language. Additional studies were identified through reference checking. The full search strategy is provided in the Supplementary Material.
The review protocol was registered at the PROSPERO International Prospective Register of Systematic Reviews: Registration Number CRD42021270630.
Inclusion and exclusion criteria
Studies were eligible for full-text review if they included patients younger than 18 years of age with suspected infections attending a PED or primary care where an ASP was implemented.
Randomized controlled trials (RCTs), controlled and uncontrolled before and after studies, interrupted time series, and cohort studies were included for review.
Studies about children admitted to a paediatric hospital ward where an ASP was implemented were excluded.
Review articles, case series, notes and letters, conference abstracts, opinion articles, and studies from which it was not possible to extract paediatric data were also excluded. Studies published before 2007 were excluded because the concept of ASP was formally introduced that year. Studies about ASP on malaria, HIV, viral, and fungal treatment were also excluded.
Studies selection
Identified references were downloaded into Rayyan software for further assessment and handling. In line with the PRISMA guidelines for systematic reviews, titles, and abstracts, identified through an electronic database, were independently screened by two investigators (GB and SR), and any references which did not meet the inclusion criteria were excluded. For all remaining references, full-text copies were obtained and were examined in detail to determine whether they met all the inclusion criteria for the review. Discussion with a third reviewer (DD) resolved any disagreement regarding selection of studies.
Data collection
Data were extracted using a standardized data collection form which summarized information about the study characteristics (authors, year of publication, study design, study location, and country), the patient characteristics, the type of ASP, and the main results with accuracy measures (health outcomes – e.g. rate of prescription, days of therapy – and economic outcomes).
Studies were grouped by setting (PED and primary care) and intervention type.
Each country’s income level was defined according to the World Bank List of economies published in July 2021. 12
Quality assessment (risk of bias)
The quality of each eligible study was evaluated depending on the study design. Clinical trials were assessed using the Revised Cochrane risk-of-bias tool for randomized trials (RoB 2), 13 while non-randomized studies were assessed using the Risk of Bias in Non-randomized studies – of Interventions (ROBINS-I) assessment tool. 14 The overall risk of bias judgements of each study did not affect the inclusion in this review. It was not possible to conduct a meta-analysis as the disparity in types of interventions and study setting prevented the combining of data from different studies.
Results
After removing duplicates, a total of 47,158 articles remained for the screening. Of these, 59 were eligible for inclusion in this review. The selection process is summarized in Figure 1.

Flowchart of the study selection process (PRISMA).
Authors, title, publication year, study design, country, study period, setting, type of ASP, and quality assessment are summarized in Table 1.
Characteristic of studies included in the review.

ASP, Antimicrobial Stewardship Programme; AMS, Antimicrobial Stewardship Programme; CAP, community-acquired pneumonia; CDS, clinical decision support; CG, clinical guideline; ED, emergency department; Edu, Education; EMR, Electronic Medical Record; FLHC, First level health care; GAS, Group A Streptococcal; IDSA, Infectious Disease Society of America; ITS, Interrupted Time Series; MC, multicenter; OP, observational Prospective; OPAT, Outpatient Parenteral Antibiotic Therapy; PCT, Procalcitonin; PDSA, Plan-Do-Study-Act; PIDS, Paediatric Infectious Diseases Society; RCT, randomized controlled trial; RT, Randomized Trial; SCM, Standard Care Management; UTI, urinary tract infection; URTI, upper respiratory tract infections.
Intervention legend:  = Guidelines,
= Guidelines,  =Audit and feedback,
=Audit and feedback,  = Physicians education,
= Physicians education,  = Parents education,
= Parents education,  = Pre-authorization,
= Pre-authorization,  = CDS tool,
= CDS tool,  = CP,
= CP,  = Other ASP intervention
= Other ASP intervention
Most of these studies (51/59, 86.4%) were conducted in high-income countries. Twenty-three articles (39.0%) described ASPs implemented in Europe (5 in Italy,24,32,39,43,53 4 in Spain,16,40,41,55 3 in France,20,21,44 2 in the Netherlands,19,25 United Kingdom,59,61 and Switzerland18,62 respectively, one each in Sweden, 26 Belgium, 45 Cyprus, 68 Ireland 23 and the Czech Republic 64 ); 24 studies were set in North America (40.7%, 23 in the United States,15,17,22,27–31,33,36,37,42,48,49,52,54,56–58,60,65,66,70 1 in Canada, 35 8 studies (13.6%) were set in Asia [3 in China,71–73 1 in Saudi Arabia, 51 1 in Israel, 69 1 in Pakistan, 50 1 in Thailand, 38 1 in Bangladesh 47 ), and two studies (3.4%) in Australia.34,63 Only one study was conducted in South America (Argentina 46 ) and one in Africa (Benin 67 ). Figure 2 shows the geographical distribution of articles.

Geographical distribution of articles included in this review.
Thirty-seven studies were published between 2015 and 2020 [37/59, 62.7%;15,19,22–24,26–30,32–34,36–39,41–45,47,49,51–56,61–63,66,71–73 15/37 (40.5%) from the United States, 14/37 (37.8%) from Europe, 6/37 (16.2%) from Asia, 2/37 (5.4%) from Australia], four times more than during the 2007–2010 period [9/59, 15.2% in total,16,35,40,48,57,59,64,67,70 4/9 (44.4%) from Europe, 3 (33.3%) from United States, 1 (11.1%) from Canada, 1 (11.1%) from Benin].
Thirty-six (36/59, 61.0%) were multicentre studies: 15/36 (41.7%) were set in North America,15,17,30,36,37,42,48,52,54,56–58,60,65,70 14/36 (38.9%) in Europe,9,18–20,41,43,44,53,55,59,61,62,64,68 6/36 in Asia (16.6%)47,50,69,71–73 and 1/6 (2.8%) in Africa. 67
More than a half of the studies were before-and-after studies (34/59, 57.6%),16,17,20–22,24–43,47,49,52,53,55,64–66,68 and more than a quarter of the total studies were randomized control studies (19/59, 32.2%).18,19,45,46,48,54,58–62,67,69–73 The remaining were se studies [4 observational prospective studies (6.8%),23,50,51,63 as well as 1 retrospective study (1.7%), 15 and 1 was an interrupted time series study (1.7%) 44 ].
ASPs setting and type of intervention
More than half of the studies (43/59, 72.9%) described the implementation of multiple interventions, like clinical pathways or clinical guidelines, combined with education or rapid test, or a combination of prescribers’ or parents’ education, clinical guideline, or clinical pathways, audit, and rapid test [Figure 3(a)].20–32,34,36,38,40,41,47,50–73

(a) Different type of ASP implemented as single intervention and multiple intervention and (b) number of studies for each different settings.
More than half of the studies included in this review (38/59, 64.4%) regarded the implementation of multiple interventions for managing respiratory tract infections, both upper and lower.18–21,24,26,30–35,37–39,43,44,46–48,50–52,54–56,58,59,61,62,65–68,70–73 Of these, eleven articles regarded the implementation of intervention for community-acquired pneumonia (CAP),30–33,35,37,39,46,50,54,67 two for acute otitis media (AOM),56,58 two for pharyngitis,43,66 two for both AOM and pharyngitis,24,26 and two for bronchiolitis. The other articles considered, in general, upper and lower respiratory tract infections. Ten studies (10/59, 17.0%) regarded the implementation of interventions for the management of children with fever both in ED or primary care,16,23,40,41,53,57,60,64,69,74 while three articles (3/59, 5.1%) considered specific interventions for febrile infants with less than 56 days of age.15,22,27 Four studies (4/59, 6.8%) considered the implementation of intervention for the management of urinary tract infection (UTI),25,29,36,42 and two articles are about parenteral antibiotic therapy in outpatients.49,63 One article considered the implementation of educational interventions for both UTIs, skin and soft-tissue infections, and upper and lower respiratory tract infections. 17 All the specific syndromes targeted by each intervention are specified in the table in the Supplementary material.
Almost half of the studies described the implementation of an ASP in the primary care setting (28/59, 47.5%) [Figure 3(b)]. Ten of these studies were conducted in Europe (10/28, 35.7%),43–45,53,55,59,61,62,64,68 11 in the United States (11/28, 39.2%),48,49,52,54,56–58,60,65,66,70 and the remaining seven studies in South America (1/28, 3.6%), 46 Australia (1/28, 3.6%), 63 Africa (1/28, 3.6%), 67 and Asia (4/28, 14.3%).47,50,51,69 Most of the ASP implemented were multiple interventions (22/28, 78.6%).47,50–67,69,70,74 Single interventions were constituted by the implementation of clinical practice guidelines (2/28, 7.1%),43,44 audit (1/28, 3.6%), 49 clinical decision support (CDS; 1/28, 3.6%) 48 and other ASP (2/28, 7.1%, rapid C-reactive protein test and prediction rule for bacteria pneumonia).45,46
Fifteen manuscripts described the implementation of ASPs in ED (15/59, 25.4%) and were conducted in the United States (6/15, 40.0%)15,17,22,27–29 and Europe (9/15, 60.0%).16,18–21,23–26 Among these, 10 were focused on multiple interventions like clinical practice guidelines or clinical pathways combined with education (10/15, 66.7%).20–29 Single interventions implemented were educational interventions (2/15, 13.3%),16,17 other types of ASP (2/15, 13.3%, rapid procalcitonin and validated clinical prediction model for pneumonia),18,19 and clinical practice guidelines (1/15, 6.7%). 15
The remaining 16 studies were conducted in multiple settings simultaneously (16/59, 27.1%), 7 both in the ED and primary care setting (7/16, 43.8%),36–42 6 both in the ED and inpatient setting (37.5%),30–32,34,35,75 and 3 both in primary care and inpatient settings (18.8%).71–73 Eleven of these studies described the implementation of multiple interventions like clinical pathways or clinical practice guidelines combined with education and sometimes audit (11/16, 68.8%).30–32,34,36,38,40,41,71–73 Single interventions implemented were clinical practice guidelines (3/16, 18.8%),35,37,42 educational sessions [1/16, 6.3% 39 ], and clinical pathways [1/16, 6.3% 33 ].
ASPs outcomes and types of intervention
The different outcomes considered in this review are reported in Figure 4, stratified by the successful or unsuccessful intervention [Figure 4(a)] as reported in each article and by the country’s income level [Figure 4(b)].

Different type of outcomes stratified by (a) the successful or unsuccessful of the intervention and (b) by the country’s income level.
Forty-seven studies reported changes in antibiotic prescriptions rate (47/59, 79.7%) as their main outcome. These studies were mainly conducted in Europe (20/47, 42.6%)16,18–21,24,26,32,39–41,43–45,53,55,59,61,62,68 and North America [19/47, 40.4%, 18 in the United States,15,17,22,27–31,33,36,37,42,48,49,57,60,65,70 1 in Canada 35 ]. Of the eight remaining studies, six were conducted in Asia (12.8%),38,47,51,69,71,72 one in Australia (2.1%) 34 and one in South America (Argentina, 2.1%). 46 Thirty-one of these (31/47, 66.0%)20–22,24,26–32,34,36,38,40,41,47,51,53,55,57,59–62,65,68–72 were studies focused on multiple interventions, above all on the implementation of clinical practice guidelines or clinical pathways combined with educational talks and sometimes audit. The remaining 16 studies (16/47, 34.0%) focused on the implementation of a single intervention (which differed as follows: six articles on clinical guidelines,15,35,37,42–44 three on educational sessions,16,17,39 one on audits, 49 one on clinical pathways, 33 one on CDS tools, 48 and four on other types of ASP, like clinical prediction models or rapid tests).18,19,45,46
Forty articles (40/47, 85.1%)15–17,20–22,24,26,27,29–33,35,36,38–49,51,53,55,57,59,60,65,68–72 described a statistically significant reduction of inappropriate prescribing that could regard both the unnecessary administration of antibiotics and the selection of the narrow-spectrum instead of the broad-spectrum ones. Regarding the prescription of broad-spectrum antibiotics, only 14 studies reported the variation in total antibiotics prescription rates and the difference between narrow-spectrum and broad-spectrum, usually identified as the second- and third-generation cephalosporins and macrolides.20,21,24,32,36,39,40,44,48,53,60,65,69,72 Of these 14 studies, 13 reported a significant reduction in prescribing these antibiotics after the different interventions were analysed.20,24,32,36,39,40,44,48,53,60,65,69,72 The remaining seven articles did not show any statistically significant change in antibiotic prescribing after implementing an ASP.18,19,28,34,37,61,62
Twenty articles (20/59, 33.9%) considered the main outcome of compliance to clinical guidelines or clinical pathways. More than half of these studies are conducted in North America [11/20, 55.0%, 10 in the United States,27,29,30,37,52,54,56,58,60,66 1 in Canada 35 ], 5 in Europe (5/20, 25%),23,25,26,40,41 and the remaining four in Asia (2/20, 10%),38,50 Africa (1/20, 5%), 67 and Australia (1/20, 5%). 63 Of these studies, 18 articles described increased compliance among prescribers (18/20, 90%).23,25–27,29,30,35,38,40,41,50,52,54,56,58,60,63,67 Most of these studies (18/20, 90.0%) focused on implementing clinical practice guidelines or clinical pathways alone or in combination with educational sessions or audit and feedback.23,25–27,29,30,38,40,41,50,52,54,56,58,60,63,66,67 Some of these studies have also evaluated the sustainability of these interventions over time. Two of these studies reported using CDS tools in combination with audit and feedback.
Only six of the included studies (6/59, 10.2%) described the change in health care cost associated with the intervention; three were conducted in the United States (3/6, 50.0%),15,30,33 two in Europe (2/6, 33.3%),23,24 and 1 in Asia (1/6, 16.7%). 73 Three [3/6, 50.0%, 1 in the United States 30 and two in Europe23,24] reported a statistically significant reduction in health care costs, mainly due to the reduction in prescription, above all in broad-spectrum antibiotics. Half of these papers (3/6, 50.0%24,33) were focused on implementing clinical pathways alone or in combination with educational sessions, one on implementing a clinical practice guideline, 15 and the remaining two studies on the implementation of multiple interventions.23,73 Five of these studies (5/6, 83.3%) were set in ED (three only in ED,15,23,24 2 both in ED and inpatient settings30,33).
Only three articles described the change in drug resistance (3/59 5.1%, 1 in the United States, 36 and 2 in Europe43,64). Two out of three articles reported a reduction in erythromycin resistance after the fall of the use of macrolides.43,64
All the details regarding study outcomes are reported in Table S1 in the Supplementary material.
Risk of bias assessment for Randomized Controlled Trial
The risk of bias assessment for RCT is summarized in Figure 5(a). Selection, detection, performance, attrition, and other bias have been assessed for all the studies. The assessment of each specific bias is reported in Table 2 in the Supplementary Material. Most of them (13/19) presented some concerns as overall judgements.19,45,46,57–59,61,62,67,69–73 Five studies were considered at overall high risk of bias.48,54,56,60,67 Only one study has been considered overall at low risk of bias. 18
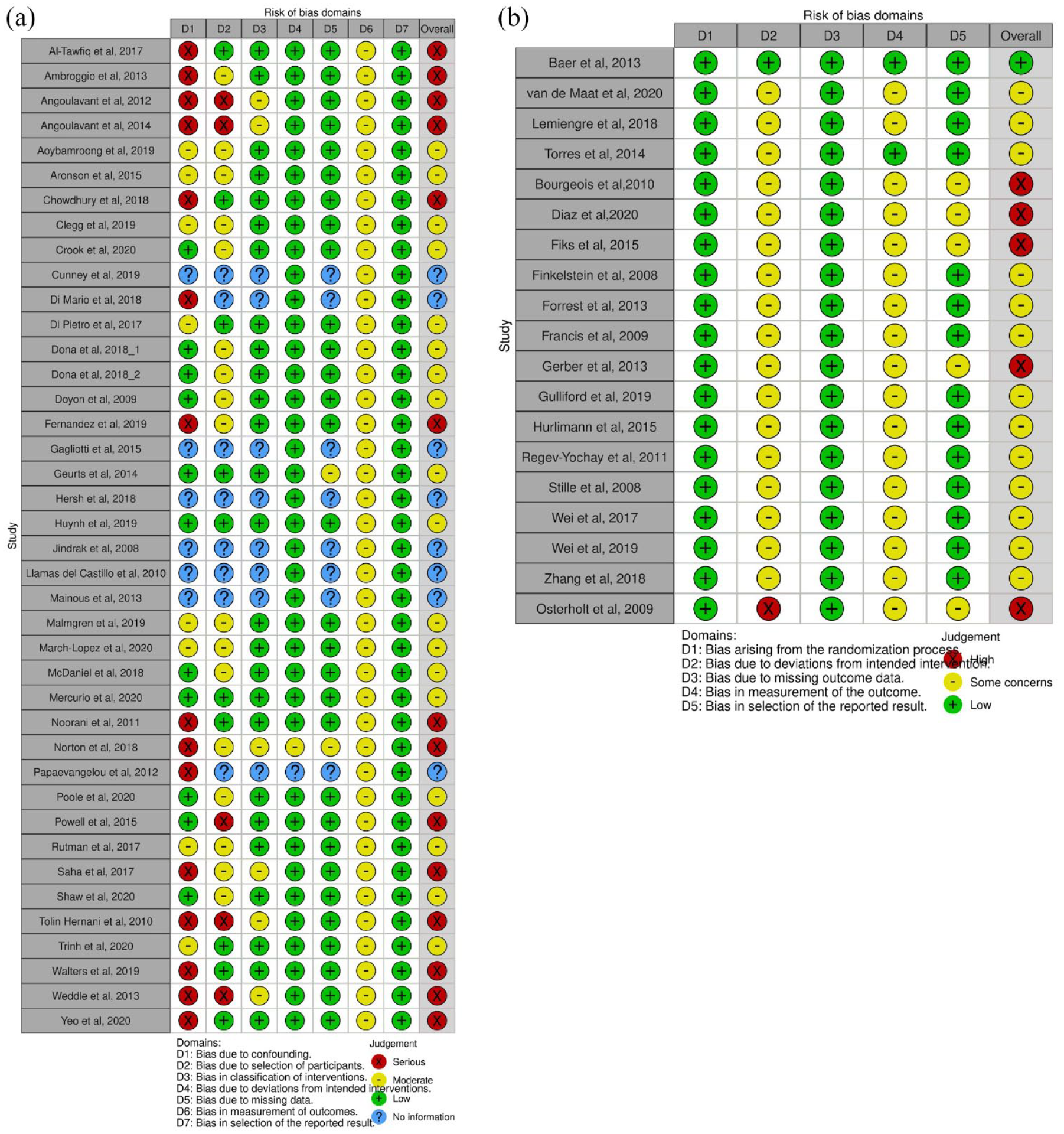
Traffic light plot of the domain-level judgements of (a) observational studies and (b) randomized controlled trials.
As it can be seen from Figure 1A in the Supplementary Material that shows the different types of bias, all the studies are at low risk of the bias arising from the randomization process and the bias due to missing outcome data.
Almost all the studies have been considered as having a risk of attrition bias. One study has been considered at high risk of bias because most of its analysis was based on the per-protocol analysis instead of the intention to treat analysis; the other studies have been considered as having some concerns. More than three-quarters of the studies have been classified as having some concerns in the section of bias in measuring the outcome due to the absence of the blinding. A quarter of the studies have been classified as having some transparency bias concerns because the published analyses were not always pre-planned.
Risk of bias assessment for observational studies
The risk of bias assessment for non-RCT is summarized in Figure 5(b). Bias due to confounding, selection of participants, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes, and selection of the reported result were assessed. The assessment of each specific bias is reported in Table 3 in the Supplementary Material. All of them were considered in the overall judgement as moderate risk of bias (18/40, 45.0%)15,22,24–27,30,32,33,35–39,41,44,52,63 or high risk of bias (14/40, 35.0%).16,17,20,21,28,29,31,34,42,47,50,51,55,66 No studies were considered at low risk of bias. Eight studies23,40,43,49,53,64,65,68 were classified as ‘No information’ because there were no described characteristics of the population of interest.
Figure 1B in the Supplementary Material shows the different types of bias. All or nearly all the studies were considered at low risk of bias due to deviations from the intended interventions and the selection of the reported results. All the studies were considered at moderate risk of bias in the measurement of the outcomes because, in all these studies, the outcome assessors were aware of the intervention received by study participants. Most of the studies were considered at moderate or high risk of bias due to confounding or selection of participants because many studies did not consider a lot of the confounding domain that we considered necessary. In almost a quarter of the studies, we did not find any information about the confounding domains that authors could have considered about selecting participants and, potentially, the missing data.
Discussion
Although IDSA has recommended implementing ASPs since 2007, the effort to implement ASPs in paediatric settings is still limited.
Looking at PASP’s literature evidence, most studies focused on the implementation of ASP in inpatient settings (92/113 studies), with more than a quarter focused on audit and feedback as a single intervention. 76 However, the core strategies proposed by IDSA are challenging to implement in the outpatient settings because of logistical and level barriers. For example, the audit and feedback strategies can be time-consuming and, differently from the inpatient setting, not as useful because of the rapid turnaround time. Nevertheless, despite these difficulties, implementing ASPs in the outpatient setting is necessary, and it is essential to find and adopt the best type of ASP.
To our knowledge, this is the first systematic review that has focused on the implementation of ASPs in paediatrics globally, both in the ED and primary care. In the last 2 years, other systematic reviews about the implementation of PASPs were published, one in premature infants, 77 and the other in PED, but only for respiratory infections. 78 In addition, another recently published review focused on PASP implementation in low- and middle-income countries. 79
Only 59 articles out of 47,158 were eligible for our review. Our search strategy, also based on the scoping review of Donà et al., 76 was extensive because it was not limited to the term ‘stewardship’, but was also widened to the general terms ‘antimicrobial’ and ‘children’. We decided to expand our search strategy because the interventions we wanted to consider in our review are not always labelled as ‘stewardship’. With this comprehensive search strategy, we found and excluded many articles about the use of antibiotics in children but without any interventions.
Due to the high variability of the type of ASPs implemented, it was impossible to conduct a meta-analysis. Only 16 out of 59 focused on a single intervention, such as implementing clinical pathways or clinical guidelines or educational sessions for prescribers and parents.
Articles on a single implementation of clinical pathways and clinical guidelines have proved to change antibiotic prescriptions, reducing and improving the antibiotic prescriptions rate, with a fall in broad-spectrum prescriptions and a concomitant increase in narrow-spectrum ones. Only one article described unchanged antibiotic prescription rates after implementing a clinical practice guideline. 37 In this before and after trial, the new local clinical guideline was distributed by email without any educational sessions. The authors supposed that combining the in-person training and real-time feedback with the implementation of clinical practice guideline would have had better outcomes, improving adherence to clinical guidelines, as demonstrated by other studies.25,40
Forty-three studies focused on multiple interventions in different combinations.
The most widespread, valuable, and feasible type of ASP was represented by educational sessions, both to prescribers and parents or guardians, combined with other types of intervention.
Implementation of PASP must consider many more factors compared to the adult settings. Indeed, it is important to highlight how, above all, the parents’ pressure on prescribing is very high in outpatient settings. In addition, prescribers may be conditioned by emotional factors based on fear, uncertainty in the diagnosis, anxiety, risk perception, and difficulty in the doctor–parent communication. 80 Therefore, an important part of ASP interventions in this setting undoubtedly concerns education of the parents.
The second most chosen type of ASP in multiple intervention studies was the implementation of clinical practice guidelines or clinical pathways, combined with educational sessions, audit and feedback or point of care tests.
Studies that described the implementation of audit and feedback were conducted mainly in primary care, with only one study reporting this core strategy in both the ED and primary care. Audit and feedback were often combined with the implementation of clinical practice guidelines, educational sessions, or CDS tools and were proven to reduce unnecessary antibiotic prescriptions. Only one study describing the implementation of multiple interventions (educational sessions, CDS tools and monthly feedback) both in adult and paediatric populations did not show any change in prescribing practice in children. 61 However, it showed a statistically significant change in the adult population. Authors hypothesized that this could be due to the greater difficulty in changing antibiotic prescription behaviour when dealing with children because of higher concerns for the youngest.
Most of the articles included in this review were conducted in high-income countries, with only a few studies in South America and Africa. This is in line with our previous findings 76 and the survey conducted in 2020 both in high- and low-income countries about the implementation of ASPs and infection prevention and control activities. 81 It could be assumed that this is because ASPs are considered part of the new standard of care, and difficult to implement in low-income countries; nevertheless, a Global Action Plan is urgently needed to make ASP feasible and sustainable in resource-limited settings. A recent systematic review on the impact of PASP in low- and middle-income countries found that more than half of the 34 studies included were published after 2015, showing increasing efforts in the implementation of PASPs. 79 In addition, all the articles in this review described a positive impact on antibiotic prescribing and an important reduction of multidrug-resistant organism (MDRO)-related morbidity and mortality in children.
The implementation of ASPs should improve antimicrobial use, reducing antimicrobial resistance, adverse drug events, and health care costs. The most common outcome was a variation in antibiotic prescriptions, meant as both a reduction in total antibiotic prescriptions and a reduction of broad-spectrum antibiotics combined with an increase in narrow-spectrum ones if indicated as first-line therapy. Most of the studies in our review described a statistically significant change in antibiotic prescribing, confirming the results of previous reviews.
A third of the studies focused on compliance and adherence to the implemented guideline. Indeed, an excessively complicated or time-consuming intervention could have a negative impact on the main outcome, leading to an increase in unnecessary antibiotic prescriptions. Therefore, the prescribers must perceive the implemented intervention as a useful tool.
It has been reported that the effect of the ASP implementation and guidelines adherence declined over time in the absence of repeated educational sessions. A study conducted in a PED in Ireland, implementing a multiple intervention ASP 23 reported that the guideline compliance increased from a median of 30% to 100% after 4 months. The increase was sustained at 100% after 6 months and 90% 1 year after the start of the intervention. Over time, other studies about sustainability are needed to understand how to maintain the positive results obtained after the ASP implementation.
The evaluation of the reduction of antimicrobial resistance is described only in a few studies. This is probably due to the difficulties related to the surveillance of antimicrobial resistance in outpatient settings; indeed, microbiological culture tests, like throat swabs or urine culture, are less frequently prescribed than in the inpatient setting.
A few studies only considered the assessment of the variation of health care costs, and only half of these showed a reduction in costs. Most of the studies focused only on the variation in antibiotic costs due to the reduction of antibiotic prescriptions, without considering the cost of ASPs implementation and the other healthcare costs that could be reduced, such as ancillary tests that could be requested in the ED and infections from multi-drug-resistant bacteria.
As already shown in the literature, the quality of the studies is often very low. 82 Most of the studies included in our review are ‘before and after’ with a moderate-high risk of bias. Only one of the randomized controlled trials was considered as having a low risk of bias; the others were classified as presenting some concerns or at having of a high risk of bias. In order to support investigations in the design and implementation of the ASP, in 2020, Schweitzer et al. 83 published a consensus recommendation about the optimization of the design of studies in this field, with the hope of improving the quality and the impact of the research in this area.
The primary limitation of our review is that only three databases were searched, and it is possible that not all PASP manuscripts were identified. In addition, most of the studies included in our review showed a positive impact on antibiotic prescribing, but we cannot exclude those other studies describing a negative impact that were conducted and not published.
Due to the low quality of the studies and the high variability among the considered interventions, which made the comparison difficult, it is challenging to reach major conclusions.
Many of the interventions included in our review have been proven valuable and feasible, but more studies with better quality are needed to identify the best intervention in each setting.
To help prescribers and researchers, Mistry et al. 9 reported in their papers some strategies to enhance the implementation of generalizable, ED-based ASPs.
First, ‘collaboration and engagement’ with the involvement of frontline providers is crucial for success. To be generalizable, ASPs require input and effort from key stakeholders in paediatric, general, and community EDs. Second, the ‘dissemination of best practices’: through adapted clinical pathways with locally tailored recommendations for antibiotic prescribing will likely increase guideline uptake. Finally, using ‘effort-independent mechanisms methods’ to prevent the need for active provider solicitations has been proven effective at curbing inappropriate prescribing in outpatient settings.
To highlight the importance of the ASPs, different organizations both in Europe and in North America have released specific guidelines about the implementation of ASP: the National Institute for Health and Care Excellence (NICE) guideline in 2015 84 and the Centre for Disease Control and Prevention (CDC) guideline in 2016. 85 Four core elements were identified for optimizing outpatient antimicrobial stewardship. First, ‘commitment’: outpatient prescribers must be engaged to prescribe antibiotics appropriately. Second, ‘action for policy and practice’: outpatient prescribers have to implement at least one policy or practice to improve antibiotic prescription. Third, ‘tracking and reporting’: prescribers and leaders of outpatient clinics have to track antibiotic prescriptions and regularly report these data back to prescribers (i.e. audit and feedback) to guide change in practice and assess progress in improving antibiotic prescribing. Finally, ‘education and expertise’: education on appropriate antibiotic prescription has to involve not only prescribers but patients and caregivers as well in order to improve antibiotic use further.
Conclusion
Implementing ASPs has been proven to positively impact antimicrobial use, healthcare costs, and antimicrobial resistance in inpatient and outpatient settings. Even if the implementation of ASP in the outpatients setting could be more challenging than in inpatient settings, some interventions have shown promising results. Multiple interventions, combining clinical pathways or clinical guidelines with education, audit, and feedback, have been proven valuable and feasible in this specific setting. However, it is difficult to draw strong conclusions from these studies because of their poor/moderate quality and the outcomes’ heterogeneity that precludes a meta-analysis. More well-designed studies are needed to reliably assess the implementation of ASPs in paediatric settings, both in high- and low- to middle-income countries.
The implementation in the paediatric settings may be more complicated, but hopefully, it could be improved in the following years. There remains a critical need for National PASP networks to develop and perform uniform interventions. Nowadays, to our knowledge, only two countries have a national PASP network established, Germany and United Kingdom, both of which are not formally funded. It is desirable that all the European countries develop a similar National PASP network, formally funded, to share results intending to promote the more judicious use of antibiotics in children.
Supplemental Material
sj-docx-2-tai-10.1177_20499361221141771 – Supplemental material for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review
Supplemental material, sj-docx-2-tai-10.1177_20499361221141771 for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review by Giulia Brigadoi, Sara Rossin, Davide Visentin, Elisa Barbieri, Carlo Giaquinto, Liviana Da Dalt and Daniele Donà in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-3-tai-10.1177_20499361221141771 – Supplemental material for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review
Supplemental material, sj-docx-3-tai-10.1177_20499361221141771 for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review by Giulia Brigadoi, Sara Rossin, Davide Visentin, Elisa Barbieri, Carlo Giaquinto, Liviana Da Dalt and Daniele Donà in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-4-tai-10.1177_20499361221141771 – Supplemental material for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review
Supplemental material, sj-docx-4-tai-10.1177_20499361221141771 for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review by Giulia Brigadoi, Sara Rossin, Davide Visentin, Elisa Barbieri, Carlo Giaquinto, Liviana Da Dalt and Daniele Donà in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-5-tai-10.1177_20499361221141771 – Supplemental material for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review
Supplemental material, sj-docx-5-tai-10.1177_20499361221141771 for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review by Giulia Brigadoi, Sara Rossin, Davide Visentin, Elisa Barbieri, Carlo Giaquinto, Liviana Da Dalt and Daniele Donà in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-jpg-1-tai-10.1177_20499361221141771 – Supplemental material for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review
Supplemental material, sj-jpg-1-tai-10.1177_20499361221141771 for The impact of Antimicrobial Stewardship Programmes in paediatric emergency department and primary care: a systematic review by Giulia Brigadoi, Sara Rossin, Davide Visentin, Elisa Barbieri, Carlo Giaquinto, Liviana Da Dalt and Daniele Donà in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
