Abstract
Background:
Urinary tract infection (UTI) is one of the most common bacterial infections in clinical practice. Given the rapid increase in antimicrobial resistance and the scarcity of new antibiotics, along with the absence of individual antibiogram testing in some countries, there is an urgent need for robust surveillance systems.
Objective:
This study aimed to provide evidence for the surveillance of resistance, a crucial component in developing national UTI treatment guidelines and guiding empirical therapy decisions.
Design:
This study utilized a retrospective, serial cross-sectional design.
Methods:
Antimicrobial surveillance was conducted using data collected from January 1, 2017 to December 31, 2021. A total of 2595 patients with UTIs were recruited for this study. From these patients, 2004 bacterial isolates were identified and subjected to epidemiological and antibiotic resistance analyses.
Results:
Escherichia coli (E. coli, 42.7%), Pseudomonas aeruginosa (P. aeruginosa, 11.9%), and Klebsiella pneumoniae (K. pneumoniae, 10.9%) were identified as the predominant causes of UTIs. E. coli isolates demonstrated a high level of sensitivity (80%–90%) to carbapenems (imipenem, ertapenem, and meropenem), aminoglycosides (amikacin), piperacillin/tazobactam, cefoperazone/sulbactam, and fosfomycin. The antibiotic resistance rates of K. pneumoniae strains consistently exceeded 50%, except for amikacin, ertapenem, imipenem, meropenem, and fosfomycin. Notably, all K. pneumoniae strains isolated from patients with UTIs were resistant to ampicillin. During the coronavirus disease pandemic, the E. coli and K. pneumoniae isolates exhibited reduced antibiotic resistance compared to the pre-pandemic period. The resistance rate of P. aeruginosa isolates remained consistently high (60%–70%).
Conclusion:
Amikacin, ertapenem, imipenem, meropenem, and fosfomycin are promising treatment options for enterobacterial UTIs. However, their efficacy against P. aeruginosa is limited. This study revealed alarmingly high rates of primary etiological pathogen resistance to commonly prescribed empirical therapies for UTIs. These findings provide crucial data for optimizing national guidelines and implementing personalized treatment strategies to enhance the effectiveness of UTI treatments.
Introduction
Urinary tract infections (UTIs) are among the most common bacterial infections in various parts of the urinary tract, including the kidneys, ureters, bladder, and urethra.1,2 Although men and women of all ages can be affected, the incidence is higher in women.3,4 UTIs can affect both outpatients and hospitalized patients, especially those with immunosuppression. 2 Escherichia coli (E. coli) is the predominant pathogen responsible for 80%–90% of community-acquired UTIs. 5 However, other bacteria, such as Klebsiella spp., Proteus spp., Enterococcus spp., and Staphylococcus saprophyticus can also contribute to UTIs. 3
In the United States, UTIs account for more than 10 million ambulatory visits annually. 6 UTIs primarily involve the invasion and proliferation of microorganisms within the urinary tract. 6 In Vietnam, as in most low- and middle-income countries, data on antimicrobial resistance are limited. A report from the Vietnam National Antimicrobial Resistance Surveillance Network, including the antimicrobial sensitivity testing results of bacterial isolates from clinical specimens, revealed that E. coli (18%) and Klebsiella spp. (13%), including Klebsiella pneumoniae (K. pneumoniae, 9%), are the most frequently isolated microorganisms in Vietnam. 7 The antimicrobial resistance levels are high in Vietnam, with more than 50% of Enterobacteriaceae isolates showing resistance to third-generation cephalosporins and fluoroquinolones. 8 However, the long-term monitoring of antimicrobial resistance in UTIs is limited.
UTIs occasionally occur without noticeable urinary symptoms, leading to delayed diagnosis and treatment. 2 Obtaining conclusive results from urine culture and antibiotic susceptibility testing typically takes at least 3 days. 2 Moreover, antibiogram testing is not routinely performed in Vietnam and other countries. For example, data extracted from a large urban healthcare system in the United States revealed that urine cultures were only ordered in 26.8% of the patients. 9 Hence, doctors in emergency departments face significant pressure to promptly assess the appropriateness of empirical antibiotics administered to patients with UTIs. 10 To assist clinicians in addressing gaps in empiric antibiotic regimens for treating UTIs and help policymakers understand the burden of UTIs in Vietnam, we analyzed the microbial profiles and antimicrobial resistance rates in both inpatients and outpatients at a Class I hospital over 5 years.
Methods
Study design
This serial cross-sectional study examined the patient data over 5 years (January 2017 to December 2021) at Nghe An Friendship General Hospital, a Class I provincial hospital located in central Vietnam. This study was conducted and reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement guidelines. 11
Data collection with inclusion and exclusion criteria
The data collection process is outlined in the flowchart (Figure 1). Briefly, the information regarding UTIs was extracted from computerized medical records using a standardized data collection form. The dataset comprises various details, including demographic characteristics (age and sex), admission unit during infection, identified causative microorganisms, specimen type, and antimicrobial susceptibility testing data. The data from confirmed positive urinary cultures, including pathogen identification and antibiotic susceptibility testing, were collected. A positive culture was defined as the growth of no more than two urinary tract pathogens, with a minimum of 1 × 105 colony-forming units per milliliter (CFU/mL) in urine specimens containing an increased white blood cell count, bacteria, proteins, or positive nitrites upon testing. 12 For instance, involving repetitive isolates from the same patient or contamination (i.e., the growth of three or more types of bacteria in a single specimen), and the fungal species were excluded from the study.
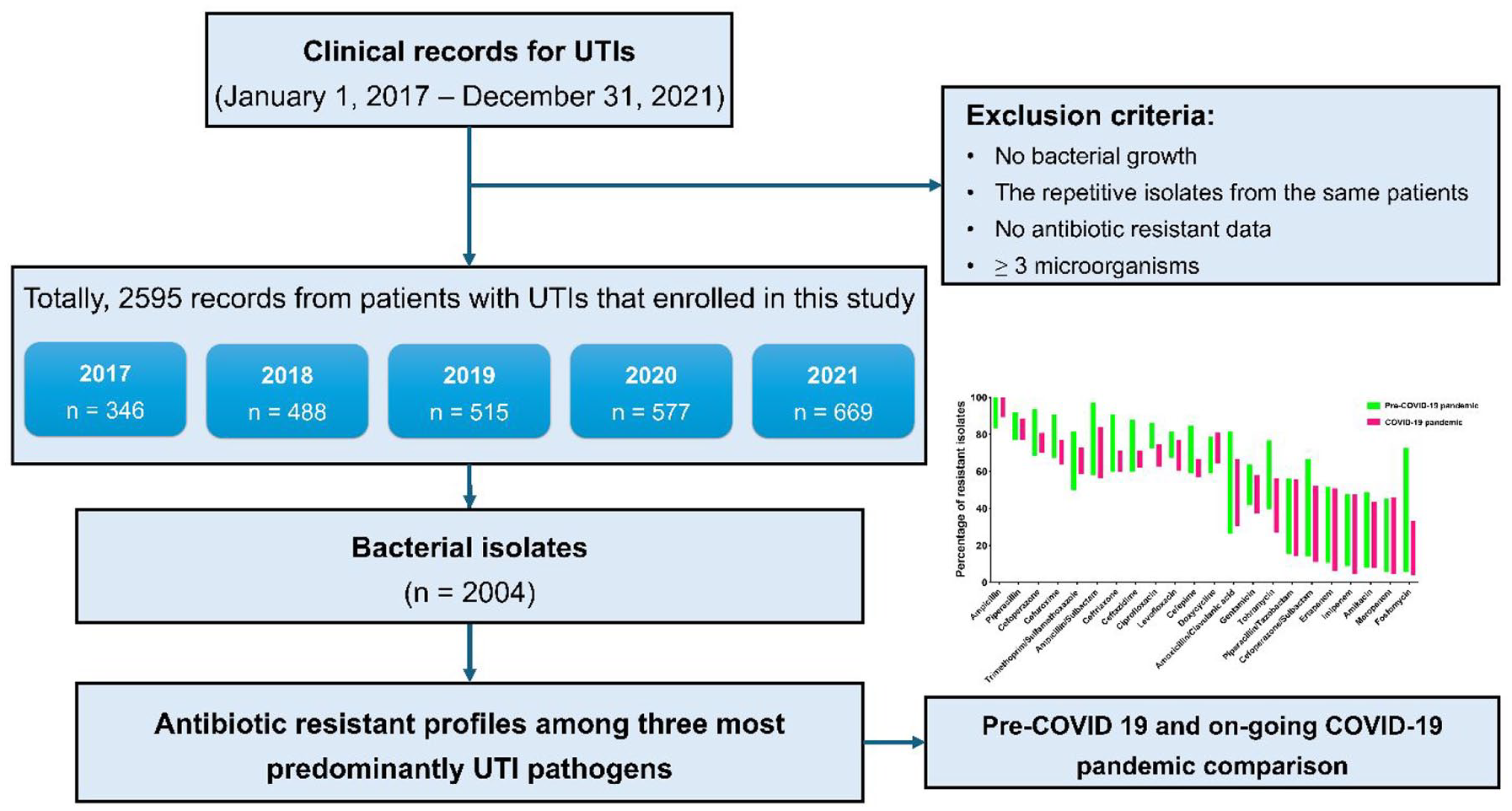
The flowchart depicts the procedure for collecting samples and data throughout the 5-year study period, spanning from January 1, 2017 to December 31, 2021.
Urine culture
Urine culture and subculture procedures were conducted in accordance with standard experimental guidelines established by the Department of Clinical Laboratory at Nghe An Friendship General Hospital. Briefly, urine samples were collected from patients suspected of UTIs using either midstream clean-catch or urethral catheterization. Samples were promptly delivered to the laboratory within 2 h for processing. Using a calibrated loop, 1 µL of urine was streaked onto both MELAB Columbia Agar containing 5% sheep blood (MELAB Diagnostics, Vinh Phuc, Vietnam) and MELAB Chromogenic UTI agar (MELAB Diagnostics). 13 The plates were then incubated overnight at 35°C. Subsequently, the resulting colonies calculated were counted, and a semi-quantitative assessment of bacterial concentration was in CFU/mL.
Microorganism identification and antimicrobial resistance determination
Microorganism identification and assessment of antimicrobial resistance were performed utilizing a combination of automated (VITEK 2 Compact System, bioMérieux, France) and conventional methods. The susceptibility of each antibiotic was determined through minimal inhibitory concentration (MIC) assessment and categorized as susceptible, intermediate, or resistant, based on the annual breakpoint updates from the Clinical and Laboratory Standards Institute (CLSI) M100 guidelines. 14
Statistical analysis
The breakpoints for each antibiotic were referenced from the CLSI M100 document and analyzed using WHONET 5.6 software (Boston, MA). Subsequently, the recorded data were analyzed utilizing Prism v9.0 software (Boston, MA).
Results
E. coli dominance with a rising prevalence of other UTI pathogens
A total of 2595 UTI patients were eligible for this study, from whom 2004 bacterial isolates were obtained (Figure 1). Most isolates (96.7%) were from inpatients, with only 3.3% from outpatients (Table S1). Of these isolates, 86% were associated with cases of simple cystitis, and 14% were identified as catheter-associated UTIs (Table S2). The most common etiological bacteria isolated in the 5-year study were E. coli (856 isolates, 42.7%), P. aeruginosa (239 isolates, 11.9%), and K. pneumoniae (219 isolates, 10.9%). Throughout the 5-year study period, the number of individuals who developed UTIs increased annually, with E. coli being the dominant pathogen. Among the known Enterococcus species responsible for most cases of UTIs, the prevalence of Enterococcus faecium exhibited a steady increase from approximately 1.5% (5 of 273 in 2017) to over 7.5% (37 of 492 in 2021), while that of Enterococcus faecalis fluctuated at around 5.7% (Table 1).
The distribution of microorganisms isolated from patients with UTIs, 2017–2021.

UTI, urinary tract infection.
Gender-associated disparities in UTI pathogen distribution
A comparison of the four predominant UTI pathogens by sex over 5 years revealed a higher prevalence of E. coli in women (62.3%, 531 out of 853; p < 0.05) (Figure 2). Conversely, the prevalence of male patients infected with Pseudomonas aeruginosa (P. aeruginosa) was 76.9% (183 out of 238), which was significantly higher than that of women (p < 0.05). Similarly, K. pneumoniae infection was more common in male patients than in female patients (62.1%, 136 out of 219; p = 0.07).

Comparing the top three most prevalent microorganisms causing UTIs by gender, with the percentage of isolates as follows: (a) E. coli, (b) K. pneumoniae, and (c) P. aeruginosa.
Antimicrobial resistance trends among bacterial isolates
E. coli and K. pneumoniae are the two most common enterobacterial isolates that cause UTIs. The annual rates of extended-spectrum β-lactamase (ESBL) positivity and antibiotic resistance among E. coli (Figure 3(a)) and K. pneumoniae (Figure 3(b)) isolates within 5 years were investigated. Although the prevalence of ESBL-producing E. coli was higher than that of ESBL-producing K. pneumoniae, a higher rate of antibiotic resistance was observed in the latter. For E. coli, more than 50% of the isolates were identified as ESBL producers during the study period. The ESBL-positive rate peaked in 2018, accounting for approximately 79.2%, and then eventually decreased by approximately 49.4% in 2019 and 2020. The ESBL-positive rate then rose to 58.4% in 2021. Following the fluctuations in ESBL-positive rates, the resistance rates of ESBL producers against other antimicrobials showed similar trends, including those for cefoperazone and trimethoprim-sulfamethoxazole. Notably, almost all cephalosporins and fluoroquinolones exhibited a resistance rate of > 50%, as indicated by the dashed red line, which denotes the 50% threshold.

The antimicrobial resistance profiles of the three most prevalent UTI pathogens isolated from 2017 to 2021. This includes the annual rates of ESBL-positive strains and antibiotic resistance for (a) E. coli, (b) K. pneumoniae, and (c) P. aeruginosa. The red and gray dashed lines denote the cutoff points at 50% and 20% for resistant strains, respectively.
The rates of resistance to amoxicillin/clavulanic acid, gentamicin, and tobramycin fluctuated erratically, ranging from 30% to 50%. Notably, seven antimicrobial agents, including carbapenems (imipenem, ertapenem, and meropenem), aminoglycosides (amikacin), piperacillin/tazobactam, cefoperazone/sulbactam, and fosfomycin, exhibited high sensitivity to over 80%–90% of E. coli isolates throughout the 5-year study period (Figure 3(a)). Additionally, the resistance rates of K. pneumoniae isolates exceeded 50%, except for amikacin, ertapenem, imipenem, meropenem, and fosfomycin, which showed resistance rates of <50% (Figure 3(b)). Notably, the proportion of ESBL-positive K. pneumoniae strains fluctuated between 20% and 50%. Additionally, all K. pneumoniae strains isolated from patients with UTIs were resistant to ampicillin. In summary, almost all enterobacterial isolates were non-susceptible to cephalosporins, fluoroquinolones, and aminoglycosides, except for amikacin. Conversely, carbapenems, amikacin, and fosfomycin displayed the highest sensitivity among the Enterobacteriaceae isolates.
P. aeruginosa has emerged as the second most common bacterial pathogen in UTIs within this study. The resistance rate of P. aeruginosa isolates remained constantly high, ranging from 60% to 70% across the 12 different antimicrobial agents tracked throughout the study period (Figure 3(c)). Notably, the susceptibility of P. aeruginosa isolates to carbapenems (imipenem and meropenem) and amikacin, which typically exhibit sensitivities exceeding 80%–90% in enterobacterial isolates, was <30%.
Changes in the antibiotic resistance rates during the coronavirus disease 2019 pandemic
The coronavirus disease 2019 (COVID-19) outbreak has presented significant challenges to all aspects of the healthcare system, including infectious disease prevention and control, diagnostics, and antibiotic usage. 15 The CDC has reported an increase in the incidence of hospital-acquired antibiotic-resistant infections in the United States during the pandemic. 16 To assess the changes in the antibiotic resistance profile of Enterobacteriaceae isolates in our hospital, we compared the proportion before and after the COVID-19 pandemic. As shown in Figure 4(a), the proportion of resistant E. coli following the post-COVID-19 pandemic (2020–2021) slightly decreased compared with that in the pre-COVID-19 period (2017–2019). Notably, the rates of resistance to doxycycline and tobramycin were significantly decreased. A similar downward trend was observed in the rates of K. pneumoniae resistance to the aforementioned antibiotics (Figure 4(b)).

Antimicrobial resistance patterns of E. coli and K. pneumoniae isolates from UTIs spanning 2017 to 2021 are depicted. The bar graph showcases the prevalence of ESBL-positive strains and their resistance profiles to various antimicrobial agents for both (a) E. coli and (b) K. pneumoniae isolates. A comparison is drawn between surveillance before (2017–2019) and after (2020–2021) the COVID-19 pandemic. The dashed lines denote the cutoff points at 20% (blue) and 50% (red) for resistant strains.
Discussion
UTI is the third most prevalent category of infection in human medicine worldwide. Our study, which included 2595 patients with UTIs, observed a nearly equal male-to-female ratio. However, the distribution of the four most prominent pathogens causing UTIs varies. E. coli infections were predominantly isolated from female patients, while K. pneumoniae and P. aeruginosa infections were more frequently detected in male patients. These findings suggest that women may be more susceptible to E. coli infections, while men may be more prone to infections caused by other opportunistic pathogens. Moreover, Malmros et al. reported 6–15 national guidelines for the empirical antibiotic treatment of UTIs that provide specific recommendations for female patients. 17 Therefore, the significant sex differences among UTI pathogen distributions may contribute to the development of effective treatment guidelines.
Data analysis revealed that E. coli, K. pneumoniae, and P. aeruginosa were the three most common bacterial pathogens isolated from patients with UTIs. This finding was consistent with those of other studies that have documented a high risk of UTI associated with these pathogens.10,18 In the present study, most E. coli strains were resistant to commonly prescribed antibiotics such as ampicillin, cephalosporin, and fluoroquinolone (>50% threshold). Notably, E. coli isolates displayed high susceptibility, with rates ranging from 80% to 90%, to seven antimicrobial drugs analyzed in the study, including three carbapenems (imipenem, ertapenem, and meropenem), amikacin, piperacillin/tazobactam, cefoperazone/sulbactam, and fosfomycin. Consistent with several national guidelines, fosfomycin is highly recommended as a first-line treatment for UTIs.12,17,19 By contrast, K. pneumoniae exhibited high-resistance rates across all classes of antibiotics, with most antibiotics showing resistance rates exceeding 50%. Only five antibiotics (amikacin, ertapenem, imipenem, meropenem, and fosfomycin) exhibited < 50% resistance rates during the study period. Previous and more recent studies have shown that enterobacterial isolates are highly sensitive to amikacin, imipenem, and meropenem,20–22 suggesting that these antibiotics can serve as effective treatment options for UTIs caused by Enterobacteriaceae. Besides, ampicillin has been suggested in numerous national guidelines for pregnant patients owing to its safety,12,17 However, our antibiotic profile of enterobacterial UTIs indicated that ampicillin is not effective for the treatment of enterobacterial UTIs in our country.
Meanwhile, the sensitivity of P. aeruginosa UTIs to carbapenems and amikacin was significantly low, falling below 30%. Of particular concern is the limited susceptibility of P. aeruginosa isolates to carbapenems (imipenem and meropenem) and amikacin, which are crucial therapeutic agents for the treatment of multidrug-resistant infections. This result underscores the need for developing alternative treatment strategies and personalized testing to address multidrug resistance.
The increasing prevalence of ESBL-producing bacteria is a global concern. In our study, the prevalence of ESBL-producing E. coli was relatively high, and more than half of the isolates exhibited ESBL activity. Conversely, the prevalence of ESBL-producing K. pneumoniae fluctuated between 20% and 50%. In previous studies, the proportions of ESBL-producing E. coli and K. pneumoniae isolated from urine were approximately 48%–65%23,24 and 37.0%–44.0%,25,26 respectively. Consistent with the findings of other studies conducted in Vietnam, the prevalence of ESBL producers was approximately 50%. 27 The acquisition of ESBLs renders these bacteria resistant to several commonly used antibiotics.20,28 The molecular characteristics of β-lactamases, including ESBL, AmpC β-lactamases, and carbapenemases, among Enterobacteriaceae have been previously reported to be highly prevalent in Vietnam.27,29 Since the use of ESBL detection data from automated systems has limitations in identifying these enzymes, 30 further studies on the phenotypic and genotypic distribution of ESBL, AmpC β-lactamase and carbapenemase-producing Enterobacteriaceae would help elucidate the detailed mechanisms of antimicrobial resistance.
Globally, our results on antimicrobial resistance address a significant data gap by identifying urgent trends in UTI resistance. These findings provide essential surveillance data on antibiotic resistance profiles, helping to prioritize the development of new antibiotics for high-resistance pathogens, redirect research focus, and shape global treatment guidelines, particularly for countries facing similar resistance challenges. This approach ensures that empirical therapies remain effective, even in regions with limited local data.
Additionally, during the COVID-19 pandemic, several factors, such as hand hygiene, mask usage, social distancing, and the widespread dissemination of infection prevention and control guidelines,31,32 may have contributed to the occurrence of antibiotic resistance. When comparing the periods before and after the COVID-19 outbreak, a declining trend was observed in the antibiotic resistance rates of E. coli and K. pneumoniae. This trend aligns with the report of recent studies, which indicated a significant reduction in antimicrobial resistance during the pandemic. 33 However, various studies yielded conflicting findings regarding the correlation between COVID-19 and antimicrobial drug resistance. Although some studies have suggested a non-statistically significant increase,34,35 others have observed an increase in the prevalence of multidrug-resistant infections during the pandemic.36–38 These results highlight the importance of implementing effective infection prevention and control guidelines as an additional strategy to manage antibiotic-resistant UTIs.
This study has several limitations. The retrospective study design introduced inherent biases, as data on the severity of illness and duration of hospital stay for these isolates were not collected. The sample size was based on all available UTI samples from both inpatient and outpatient cases during the study period. Additionally, this study lacked access to individual patient records, limiting the ability to evaluate correlations with clinically relevant risk factors. Further investigations, incorporating a broader range of diagnostic methods and larger sample sizes, may provide a more comprehensive understanding of the microbial landscape of UTIs.
Conclusion
This study investigated the epidemiology and antimicrobial patterns of UTIs in Vietnam in recent years. Two-thirds of UTI cases were caused by enterobacterial pathogens, including E. coli and K. pneumoniae, which exhibit high rates of antibiotic resistance. Amikacin, ertapenem, imipenem, meropenem, and fosfomycin are promising treatment options for UTIs caused by Enterobacteriaceae. However, their effectiveness against P. aeruginosa, which accounts for 11.9% of all UTI cases, is limited. These findings underscore the importance of optimizing national guidelines and implementing personalized treatment strategies to effectively manage UTIs amidst the increasing prevalence of antimicrobial resistance.
Supplemental Material
sj-doc-1-tai-10.1177_20499361251315346 – Supplemental material for Epidemiology and antimicrobial resistance patterns of urinary tract infection: insights and strategies from a 5-year serial cross-sectional study in Vietnam
Supplemental material, sj-doc-1-tai-10.1177_20499361251315346 for Epidemiology and antimicrobial resistance patterns of urinary tract infection: insights and strategies from a 5-year serial cross-sectional study in Vietnam by Anh Tram Que, Anh Dao Tran, Thi Hong Nhung Trang, Thi Nhu Le Tran, Ngoc-Niem Bui and Chih-Ho Lai in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251315346 – Supplemental material for Epidemiology and antimicrobial resistance patterns of urinary tract infection: insights and strategies from a 5-year serial cross-sectional study in Vietnam
Supplemental material, sj-docx-2-tai-10.1177_20499361251315346 for Epidemiology and antimicrobial resistance patterns of urinary tract infection: insights and strategies from a 5-year serial cross-sectional study in Vietnam by Anh Tram Que, Anh Dao Tran, Thi Hong Nhung Trang, Thi Nhu Le Tran, Ngoc-Niem Bui and Chih-Ho Lai in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
The authors thank the editor and the reviewers for their editorial assistance and valuable comments. The authors gratefully acknowledge the support from Nghe An Friendship General Hospital, Tra Vinh University, and Can Tho University of Medicine and Pharmacy, Vietnam.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
