Abstract
Background:
HIV-negative patients have substantial mortality from Pneumocystis jirovecii pneumonia (PJP). We lack predictors of HIV-negative PJP-associated mortality.
Objective:
We aim to characterize the role of prior corticosteroid exposure in PJP-related mortality.
Methods:
We queried a global research network to identify adult HIV-negative patients with PJP with or without corticosteroid exposure in the preceding year before diagnosis (n = 8,021). We performed a propensity score-matched analysis to adjust baseline patient characteristics and analyzed outcomes. We follow-up the results with a multicenter ten years retrospective case-control cohort of HIV-negative patients tested for PJP by PCP Direct Fluorescent Antigen. We used a Cox proportional hazards model for survival analysis.
Results:
1822 HIV-negative propensity-scored matched patients with prior corticosteroid exposure had significantly increased 10 weeks (16% versus 9%, p < 0.0001) and one-year mortality after PJP diagnosis (23% versus 14%, p < 0.0001). (1→3)-β-D-glucan (197.6 ± 155.8 versus 63 ± 0 pg/ml, p = 0.014), ferritin levels (1227 ± 2486 versus 768 ± 1060 mcg/l, p = 0.047), lymphopenia (1.5 ± 1.5 versus 2.0 ± 1.6 103 cells/µl, p < 0.0001) and hypoxia (SatO2: 86.7% versus 91.6%, p < 0.0001) were higher or worse in those with prior steroid use. Patients who died were more likely to have previously received dexamethasone (35% versus 16%, p < 0.001) or prednisone (49% versus 29%, p < 0.001). Adjusted Cox proportional-hazard model validation showed an independently increased mortality at 10 weeks (HR: 3.7, CI: 1.5–9.2, p = 0.004) and 1 year (HR: 4.5, CI: 2.0–10.4, p < 0.0001) among HIV-negative patients with previous corticosteroid exposure.
Conclusion:
Preceding corticosteroids in HIV-negative patients with PJP are associated with higher mortality. A higher fungal burden may influence corticosteroid-mediated mortality. Assessment of PJP prophylaxis must become a standard clinical best practice when instituting corticosteroid therapy courses.
Introduction
Pneumocystis jirovecii pneumonia (PJP) is an opportunistic fungal infection primarily affecting immunocompromised hosts. Epidemiologic and clinical descriptions of PJP were first presented in the 1960s. 1 However, the disease gained widespread relevance following the initial report as an opportunistic infection causing mortality in patients with advanced acquired immunodeficiency syndrome (AIDS). 2 With the advent of antiretroviral therapy in the 1990s and the availability of PJP prophylaxis, PJP-associated mortality among persons with human immunodeficiency virus (HIV) (PWH) has declined substantially. In contrast, the growing non-HIV immunosuppressed population places them at risk of PJP. 3 The primary risk factor for PJP continues to be cell-mediated immunity defects, such as those present in malignancy, hematopoietic stem cell or solid organ transplantation, and those receiving corticosteroids or other immunosuppressive therapies to treat autoimmune diseases. 4
Current mortality assessment in HIV-positive patients with PJP is estimated between 1% and 15%, compared to an estimated 30–40% mortality in HIV-negative patients. 5 Clinical factors associated with increased mortality among HIV-negative patients with PJP are older age, female gender, delayed presentation, respiratory failure, concurrent malignancy, Aspergillus sp. co-infection, and elevated lactate dehydrogenase (LDH) levels at presentation.5,6
Systemic corticosteroid therapy treats several medical conditions, including rejection in solid organ transplant recipients, malignancy, and autoimmune or inflammatory diseases. Corticosteroid exposure is a well-known risk factor for developing PJP.4,7 Still, it remains unclear how prior corticosteroid exposure influences the presentation, severity, and mortality of infection. The preceding corticosteroid exposure – mediating an increase in immunosuppression – may facilitate an increase in fungal burden during infection, blunt patient symptoms contributing to delays in diagnosis and later presentation and serve as a potentially modifiable risk factor for implementing preventive measures. We aim to characterize the role of prior corticosteroid exposure in PJP-mediated mortality among HIV-negative patients.
Materials and methods
Global federated research network study design and population
We used TriNetX, a global federated research network that captures anonymous data from electronic medical records (EMRs) of 66 healthcare organizations. This report described a comparative outcomes analysis generated by the TriNetX platform on July 26, 2022. This analysis compared the outcomes of two cohorts: HIV-negative patients with PJP with prior corticosteroid use (N = 4732) to those without previous corticosteroid use (N = 3289). We also ran a comparative analysis among patients with PJP who died at 1 year (N = 1905) versus survivors (N = 6116). A case of PJP was defined by either a positive specimen for P jirovecii [antigen or polymerase chain reaction (PCR) test] or by the ICD-10 code B59. Corticosteroid use was defined as having received orally or intravenously any of the following within the preceding year: glucocorticoids, prednisolone, dexamethasone, prednisone, and methylprednisolone based on RxNorm codes (Supplementary Table S5). Through TrinetX, the exact duration and doses were not available. The query criteria for each cohort were based on ICD-10-CM codes and laboratory results (Supplementary Table S3). The earliest encounter for PJP was identified as the index encounter in patients with multiple encounters. Available data, including demographic characteristics, diagnoses, procedures, medications, and measurements (e.g. laboratory test results; see Supplementary Data), were captured 3 months before the PJP diagnosis.
Global federated research network outcome measures
The primary outcome was the mortality odds following the diagnosis of PJP among HIV-negative patients at 30 days, 10 weeks, and 1 year. The secondary outcomes included the proportion of patients in each group who required hospitalization, intensive care unit (ICU) admission, or mechanical ventilation (MV).
Multicenter follow-up cohort – data collection
We performed a mortality analysis follow-up in a multicenter cohort from UCHealth, a hospital network that provides care throughout Colorado. We collected data on all patients who underwent testing of respiratory samples for PJP by direct fluorescent antibody (DFA) staining or polymerase chain reaction (PCR) through ARUP laboratories at the UCHealth microbiology laboratory between January 2000 and September 2021. We divided the patients into cases (PJP) and controls (without PJP). We partially matched cases to controls by age and gender. Each case and control had a manual chart review to verify the presence or absence of PJP. We analyzed HIV-negative patients only. A comprehensive review of the medical records was conducted to collect clinical and laboratory data. Detailed variables and definitions were previously published. 8 We followed the mycoses study group consensus that defines corticosteroid exposure based on the receipt of at least two weeks within the 60 days before PJP diagnosis. 9 We calculated the daily prednisone equivalent dose over the 6 months before PJP diagnosis, excluding any burst doses (Supplementary Table S7). We recorded mortality at 30 days, 10 weeks, and 1 year. The Colorado Death Registry was automatically interrogated for the date and cause of death using software supported by the Health Data Compass Data Warehouse project (healthdatacompass.org).
Statistical analysis
Statistical analyses of data obtained from the global federated research network were completed on the TriNetX platform. Descriptive statistics were presented as means and standard deviations for continuous variables and as frequencies and proportions for categorical variables. Continuous data were compared using independent t-tests, whereas categorical data were compared using χ2 or Fisher’s exact test, as appropriate. Outcome analysis was reported after propensity score matching. Propensity score matching was performed to control for differences between groups based on age, gender, ethnicity, comorbidities [systemic connective tissue disorder, neoplasms, transplant organ status, type 2 diabetes mellitus (DM2), systemic lupus erythematosus, and chronic kidney disease (CKD)], additional immunosuppressants, and PJP prophylaxis using a 1:1 greedy nearest-neighbor algorithm. 10 These variables were selected because they are established risk factors for PJP or associated with increased PJP-related mortality. We calculated odds ratios (ORs) with 95% confidence intervals (CIs) for the receipt of dexamethasone, and prednisone, with p < .05 as the cut-off for statistical significance.
Statistical analyses for the follow-up cohort (UCHealth) were performed using Stata software, version 16.0 (StataCorp, College Station, TX, USA). We had crude mortality as our primary outcome at 30-days, 10 weeks, and 1 year. Kaplan–Meier survival curves were constructed to show cumulative mortality over the study period for HIV-negative PJP patients by prior corticosteroid administration status. Multivariable Cox regression was used to estimate the mortality rate ratio among HIV-negative PJP patients with or without previous corticosteroid use. Selected variables associated with increased mortality added to the model included age, gender, race, comorbidities (liver disease, lung disease, cancer, inflammatory disease, solid organ transplant, immunosuppressive medications, smoking, and hematologic malignancy), PJP prophylaxis, and case-control status. A receiver operating characteristic curve (ROC) was constructed for prednisone equivalent daily dose in UCHealth patients to classify patients who died at ten weeks, and areas under the ROC curves were calculated.
Results
Global research network clinical characteristics of PJP HIV-negative patients on previous corticosteroids
We identified a total of 8021 HIV-negative patients with PJP: 4732 with prior corticosteroid exposure within the preceding year and 3289 without corticosteroid exposure (Table 1). Patients with preceding corticosteroid exposure were slightly younger, with a higher proportion of white individuals and Hispanic persons. All comorbidities were more frequent in the group receiving corticosteroids – primarily malignancy, transplant organ recipient status, DM2, CKD, connective tissue disorders, and chronic respiratory diseases. HIV-negative patients with PJP and prior corticosteroid exposure showed lower leukocytes, lymphocytes (Figure 1(a)), platelet counts, and hemoglobin.
Characteristics for HIV-negative patients with PJP with or without previous corticosteroid exposure, (Global health network database).

BMI, body mass index; CS, corticosteroids; HIV, human immunodeficiency virus; MKS, musculoskeletal; PJP, Pneumocystis jirovecii pneumonia; UC, ulcerative colitis.
Data was captured within the previous 3 months from reaching PJP diagnosis.
Includes tacrolimus, mycophenolate mofetil, mycophenolic acid, cyclosporine, azathioprine, sirolimus, infliximab, basiliximab, belatacept, omalizumab, siltuximab, belumosudil, and ustekinumab.
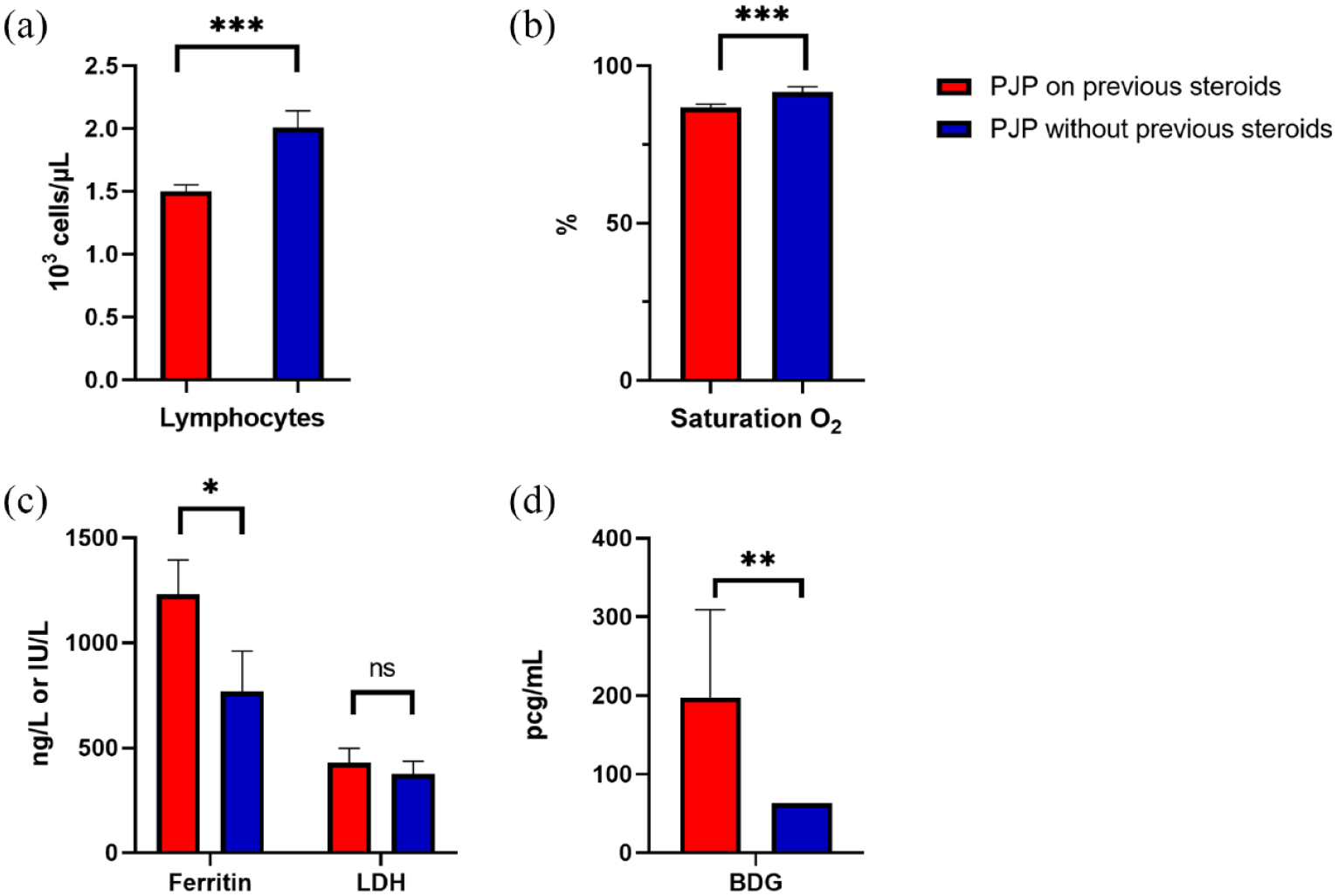
Levels of different biomarkers between HIV-negative patients with PJP with or without previous steroids prior to diagnosis (Global health network database). (a) Absolute lymphocyte count. (b) Oxygen saturation. (c) Levels of ferritin and lactate dehydrogenase. (d) Levels of (1,3)-β-D-glucan (BDG).
Conversely, lung tissue injury markers such as LDH and ferritin levels were higher in those previously on corticosteroids (Figure 1(c)). (1→3)-β-D-glucan was also higher in patients with prior corticosteroid exposure (Figure 1(d)). Differences in hemoglobin A1c concentrations were not statistically significant between both groups. Patients on previous corticosteroids were also more likely to be receiving additional immunosuppressant medications, including tacrolimus and rituximab, to be on PJP chemoprophylaxis, and to have lower oxygen saturation (Table 1 and Figure 1(b))
Global research network outcomes measures
There were 1822 patients in each group after the completion of propensity matching. HIV-negative PJP patients on previous corticosteroids experienced adjusted worse outcomes, including death, need for MV, ICU admission, and hospitalization at three time points (Table 2). Mortality almost doubled at 30 days, 10 weeks, and 1 year among patients on previous corticosteroids (Figure 2). White race, sarcoidosis, pulmonary fibrosis, use of rituximab or tacrolimus, lymphopenia, hypoxia, and elevated (1,3)-β-D-glucan remained independently associated with HIV-negative PJP patients on previous corticosteroids after propensity score balance.
Propensity score matching outcomes differences among HIV-negative patients with PJP and prior corticosteroid exposure compared to those HIV-negative with PJP and no corticosteroid exposure (Global health network database).

CI, confidence interval; ICU, intensive care unit; HIV, human immunodeficiency virus; MV, mechanical ventilation; OR, odds ratio; PJP, Pneumocystis jirovecii pneumonia.
Data are presented as n (%) unless otherwise noted.
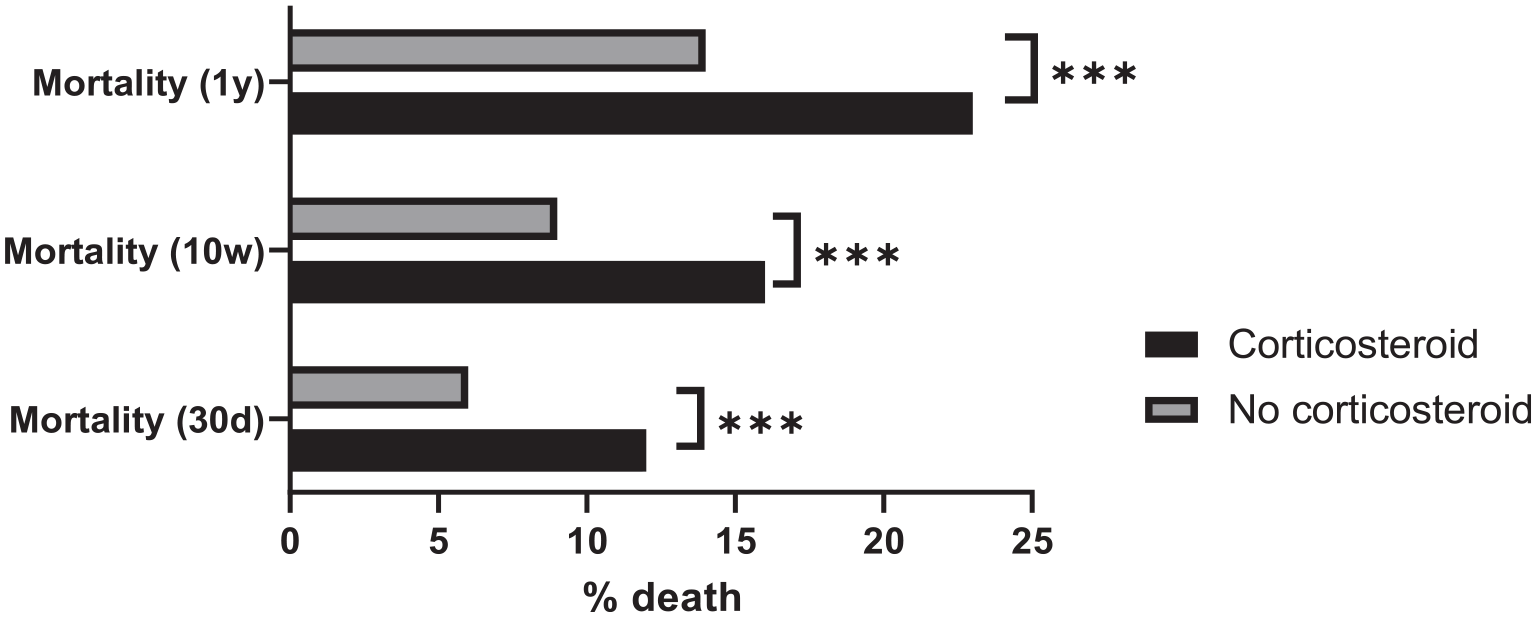
Adjusted mortality rates after propensity score matching among HIV-negative patients with PJP by prior corticosteroid exposure (Global health network database).
Global research network clinical features associated with 1-year mortality
We identified 1905 HIV-negative patients with PJP who died within 1 year of diagnosis and 6116 HIV-negative patients with PJP who survived 1 year. Patients who died were older (68 versus 63 years of age, p < 0.001) and more likely to be white. Patients who died were more likely to have a diagnosis of systemic connective tissue disorders (10% versus 6%, p < 0.001), neoplasms (63% versus 32%, p < 0.001), DM2 (29% versus 17%, p < 0.001), CKD (29% versus 16%, p < 0.001), or pulmonary fibrosis (10% versus 5%, p < 0.001). Patients who died were more likely to have received dexamethasone [35% versus 16%, p < 0.001; OR: 2.2 (2.0–2.5, p < 0.0001)] or prednisone [49% versus 29%, p < 0.001; OR: 1.7 (1.5–1.8, p < 0.0001)] within 3 months before diagnosing PJP. LDH (643.6 ± 878.5 versus 388.3 ± 1595.5 U/l, p < 0.001) and ferritin levels (1976.4 ± 3446.2 versus 951.1 ± 1783.0 mcg/l, p < 0.001) were higher in those who died. After matching by age, gender, ethnicity, comorbidities [systemic connective tissue disorders, neoplasms, transplanted organ status, DM2, systemic lupus erythematosus (SLE), CKD], and other immunosuppressant medications; pulmonary fibrosis, ferritin levels, and corticosteroids use remained higher in the nonsurvivors group.
Multicenter UCHealth HIV-negative PJP case-control clinical characteristics
Next, we followed-up on the mortality findings in our UCHealth-based replication cohort. We used the partially matched case-control cohort of HIV-negative patients with PJP (n = 105) or without it (n = 71) (Supplementary Table S1). PJP cases were older and less likely to have a tobacco or lung disease history or receive PJP chemoprophylaxis. Still, they were more likely to have end-stage renal disease, metastatic disease, inflammatory disease, or to be on immunosuppressive medications or prior corticosteroids. Steroids may have been used as part of the rejection prevention regimen in transplant recipients. HIV-negative PJP patients had a median prednisone equivalent dose higher in patients who died at seven weeks (33.7 versus 21.5 mg/d) and one year (33.7 versus 20 mg/d), although not statistically significant. PJP cases were also more likely to die during hospitalization. Similarly, patients with steroid exposure in the previous six months were more likely to have a history of malignancy (26.5% versus 11.9%, p = 0.026), solid organ transplant (14.8% versus 3.4%, p = 0.023), inflammatory disease (27.6% versus 8.5%, p = 0.003), or use of other immunosuppressive drugs (83.8% versus 13.6%, p < 0.001).
Multicenter UCHealth clinical factors associated with an increase in HIV-negative PJP mortality
The 30-days, 10-weeks, 1-year, and overall mortality among HIV-negative patients with PJP were 37%, 38%, 51%, and 61%, respectively (Supplementary Table S1). The adjusted Cox proportional-hazard model found an increased rate of death at 10 weeks (HR: 3.7, CI: 1.5–9.2, p = 0.004) (Figure 3) and one year (HR: 4.5, CI: 2.0–10.4, p < 0.0001) among patients previously on corticosteroids. Liver disease was also associated with an increase in 10-week mortality (Table 3). A prior empirical prednisone equivalent dose cut-off point of ⩾25 mg/day generates a sensitivity of 67.4% and a specificity of 57.1% to correctly classify a mortality case at 10 weeks among HIV-negative patients. The prednisone equivalent dose had low diagnostic accuracy (AOC = 0.6) in predicting 10-week mortality (Supplement Figure S1).
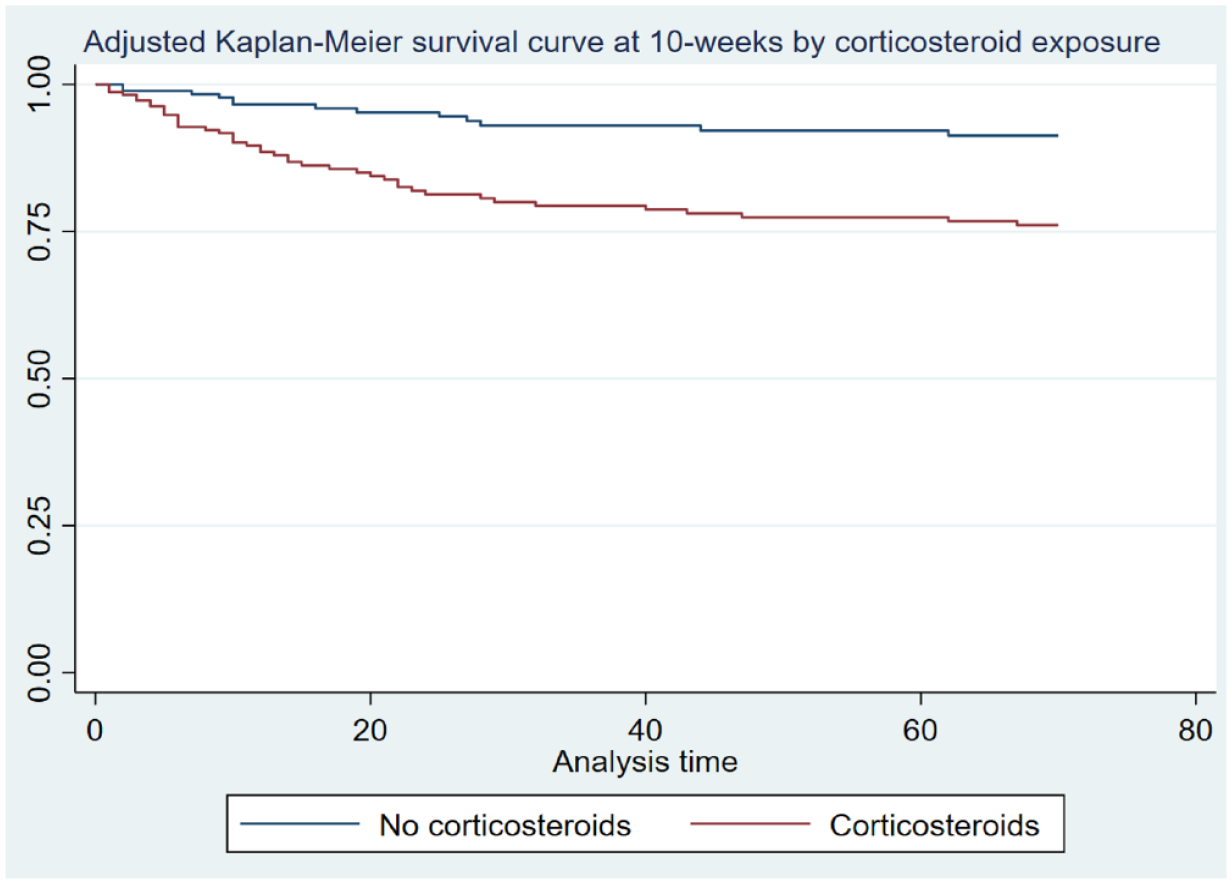
Adjusted Cox model 10-week mortality among HIV-negative patients with PJP by corticosteroid exposure in the follow-up UCHealth case-control study.
Multivariable Cox proportional hazards model for HIV-negative PJP associated 10-week mortality (follow-up UCHealth case-control dataset).

CI, confidence interval; HIV, human immunodeficiency virus; PJP, Pneumocystis jirovecii pneumonia.
Discussion
We found a 2-fold increase in mortality risk among HIV-negative patients with PJP who previously received corticosteroids when adjusted for other key risk factors. The mortality risk increased by 6%, 7%, and 9% at 30 days, 10 weeks, and 1 year respectively, in the TriNetX data. Our local network cohort corroborated these findings with a 4- and a 5-fold increase in death rates at 10 weeks and 1 year among HIV-negative patients with preceding corticosteroid therapy. Higher mortality rates within our local network may be attributable to a referral bias with sicker patients cared for in a tertiary care network and a lower number overall of patients studied. In addition, most of the patients identified in our cohort were diagnosed with PJP in a hospital setting, portending higher acuity. In contrast, the TriNetX database does not exclude testing done in ambulatory settings and could capture subclinical disease or colonization cases.
We also found a meager rate of PJP prophylaxis among patients at risk. Less than a quarter of patients on previous corticosteroids were on PJP prophylaxis, and less so in our health system. Patients on previous corticosteroids can have an increased risk of severe PJP based on (1) impaired cell-mediated immunity and (2) an increased fungal burden at presentation. Chronic administration of corticosteroids decreases lymphocyte counts, which correlates with HIV-negative PJP-related mortality,11,12 as seen in our study. Antibiotic prophylaxis has proven effective in preventing PJP.13–15 The precise dose and duration of corticosteroid use that increases the risk for PJP have been long debated and are variable between studies.7,16 In our cohort of patients tested for PJP, cases were more likely to have received equivalent prednisone doses ⩾ 25 mg/day in the previous 6 months. More patients that developed PJP had received steroid tapers, defined as a gradual decrease in steroids spanning more than 2 weeks. This highlighted the need to recognize additional patient groups that will benefit from PJP prophylaxis and may not fall within consensus recommendations based on historical data.7,17
The effects of corticosteroids on cellular immunity may be difficult to assess precisely based on quantitative assays of immune function, such as total lymphocyte count or hypogammaglobulinemia, and if these effects extend beyond the development of PJP. Although not statistically significant, CD4 counts were much lower in patients with PJP without previous corticosteroids (97.6 cells/µl± 137 versus 334.3 cells/µl± 494.9), also seen in other reports in HIV-negative patients 18 and may provide additional information about the net immunosuppression status to guide chemoprophylaxis among patients regardless of corticosteroid administration.
(1,3) β-D-glucan is a cell wall component of P jirovecii and has been found to have a heightened sensitivity to diagnose PJP and can be used as an initial screening method. 19 Retrospective studies did not find it a valuable prognostic factor for diagnosis or respiratory burden.20,21 However, it correlated with bronchoalveolar lavage fungal load in earlier studies.22,23 Our findings of higher (1,3)-β-D-glucan levels among patients with PJP previously on corticosteroids suggest a higher fungal burden on those individuals. 24
LDH and ferritin levels can be markers of lung injury during infection. 25 LDH levels are usually elevated in PWH and PJP.26,27 Severe PJP causes surfactant insufficiency leading to alveolar collapse and acute respiratory distress syndrome (ARDS). 28 Intracellular components – such as LDH and Ferritin – get released into the circulation during pulmonary injury.29,30 These findings together suggest patients on previous corticosteroids can present with a higher fungal burden leading to a more severe PJP-mediated lung injury and respiratory failure. We demonstrated worse hypoxemia at presentation among those patients previously on corticosteroids.
The benefit of adjuvant corticosteroid therapy during PJP treatment – although clear for people with HIV – is less clear among HIV-negative patients. A meta-analysis performed among HIV-negative patients, and previous work from our group suggest corticosteroids may benefit as adjuvant therapy, especially in hypoxemic respiratory failure (9, 10). However, the benefit in patients already receiving corticosteroids is unknown.
Mortality due to PJP among HIV-negative patients remains exceptionally high. Numbers from our hospital network cohort are similar to those reported previously: 30–40% mortality.5,31,32 The overall lower mortality found with the global network implies potentially lower mortality in a more community-based setting or capture of Pneumocystis colonization cases. Although nonsignificant, this study of HIV-negative patients found a higher proportion of ongoing corticosteroid therapy (57% versus 39%, p = 0.083) among patients with severe PJP at presentation. Given the significant mortality risk with steroids, this might argue for alternate immunosuppression and steroid-sparing regimens as potential mechanisms for reducing an individual patient’s risk. This can become a critical consideration when managing severe COVID-19 in critically ill patients, where high-dose steroids are widely implemented. 33
Our study has several limitations. The retrospective nature of the follow-up cohort can introduce some selection bias. Isolating the effect of steroids is complex, and differences may be confounded by indication. Also, some missing data can impair the association’s strengths. We captured the global network data mainly through ICD-10-CM codes that may be unreliable or not fully meet diagnostic criteria for PJP. The platform also limited us from running a subgroup analysis by participating centers. Corticosteroid patients may have additional unaccounted comorbidities that could confound the mortality findings. However, this is one of the most extensive PJP studies ever analyzed, with close to 8000 subjects, specific adjustment of comorbidities and other mortality risk factors, and robust validation analysis.
Conclusion
HIV-negative patients on previous corticosteroids carry a significant risk of developing PJP, with an observed two-fold increase in PJP mortality compared to those without corticosteroid use. Possible mechanisms contributing to mortality may include increased fungal load, lung endothelial injury, ARDS, and worsening hypoxemic respiratory failure. We urgently need widely based implementation of PJP prophylaxis in these patients. Further studies can help stratify patients most likely to benefit from PJP prophylaxis based on previous corticosteroid doses. We recommend clinicians have a low threshold to initiate PJP chemoprophylaxis in HIV-negative patients receiving corticosteroids.
Supplemental Material
sj-docx-1-tai-10.1177_20499361231159481 – Supplemental material for Previous corticosteroid exposure associates with an increased Pneumocystis jirovecii pneumonia mortality among HIV-negative patients: a global research network with a follow-up multicenter case-control study
Supplemental material, sj-docx-1-tai-10.1177_20499361231159481 for Previous corticosteroid exposure associates with an increased Pneumocystis jirovecii pneumonia mortality among HIV-negative patients: a global research network with a follow-up multicenter case-control study by Lilian Vargas Barahona, Kyle C. Molina, Laura C. Pedraza-Arévalo, Stefan Sillau, Alex Tagawa, Sias Scherger, Daniel B. Chastain, Leland Shapiro, Jose Tuells, Carlos Franco-Paredes, Kellie L. Hawkins, James P. Maloney, George R. Thompson and Andrés F. Henao-Martínez in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
