Abstract
Background:
Corticosteroids (CSs), specifically dexamethasone (DEX), are the treatment of choice for severe acute respiratory distress syndrome (ARDS) due to COVID-19 pneumonia (CARDS). However, data from both ARDS and relatively small CARDS clinical trials have suggested improved outcomes with methylprednisolone (MP) versus DEX. The objective of this retrospective cohort study was to compare the safety and effectiveness of MP and DEX in critically ill CARDS patients.
Methods:
The study cohort included CARDS patients admitted to a tertiary referral intensive care unit (ICU) between April and September 2020 who received at least 5 days of CSs for CARDS.
Results:
The cohort was notable for a high severity of illness (overall, 88.5% of patients required mechanical ventilation and 16% required vasopressors on admission). The DEX group (n = 62) was significantly older with a higher illness severity [Sequential Organ Failure Assessment (SOFA) 6 (4.75–8) versus 4.5 (3–7), p = 0.008], while the MP group (n = 51) received significantly more loading doses [19 (37.3%) versus 4 (6.5%), p < 0.0001]. MP was associated with a shorter time to intubation and more rapid progression to mortality [days to death: 18 (15–23) versus 27 (15–34), p = 0.026]. After correction for baseline imbalances in age and SOFA score, DEX was associated with improved mortality at 90 days compared with MP [hazard ratio (HR) = 0.43, 95% confidence interval (CI) = 0.23–0.80, p = 0.008]. However, there were no differences between rates of secondary infections during hospitalization or insulin requirements at 7 and 14 days.
Conclusion:
In this cohort of critically ill CARDS, choice of CS was associated with mortality but not adverse event profile, and thus warrants further investigation.
Introduction
Background
The coronavirus disease 2019 (COVID-19) pandemic continues to strain intensive care units (ICUs) across the world years after the first case was reported. Corticosteroids (CSs) have become the standard of care for patients with acute respiratory distress syndrome (ARDS) due to COVID-19 pneumonia (CARDS). 1 The immunosuppressive effects of CSs are well described and have been effectively used for a wide range of inflammatory conditions, but there is a relative paucity of data on drug-specific effects of methylprednisolone (MP) versus dexamethasone (DEX) in critically ill CARDS.
DEX was associated with improved outcomes in non-COVID-19 ARDS but was not recognized as standard of care in CARDS until the RECOVERY Trial; DEX is now the most widely studied CS and is associated with improved mortality and ventilator-free days in patients with CARDS.2–6 Both the Infectious Disease Society of America and National Institutes of Health guidelines recommend DEX as first line (strong recommendation) in CARDS, but consider MP or prednisone as alternatives when DEX is not available.1,7 MP is an intermediate-acting CS that improves mortality and morbidity in moderate to severe ARDS. 8 MP has also been shown to be beneficial in the treatment of CARDS.9–12 Moreover, preclinical pharmacologic data have supported the use of MP in treating CARDS. 13 Despite emerging data supporting efficacy of MP in CARDS, DEX remains standard of care due to a relative paucity of comparative literature. The available studies comparing CS options in CARDS have mixed results, suggesting the need for further studies.14–19
Common concerns with use of CS in critically ill patients include increased risk of secondary infections and potential for complications associated with hyperglycemia. Secondary infections are common in CARDS, occurring in up to 46–63% of patients.20,21 Hospital- and ventilator-acquired pneumonia (HAP and VAP) are major sources of mortality in the ICU and significant concerns in CARDS patients receiving high-dose CSs and prolonged mechanical ventilation (MV). Some studies have reported no significant increase in secondary infections due to CSs, 21 while others reported an increased risk as well as delayed viral clearance with CSs.22,23 The discrepancies between results may be due to differing properties between CS agents. 24 Furthermore, steroid-induced hyperglycemia (SIHG) is a common adverse event (AE) and has been associated with death in severe COVID-19 patients, but is not well described in studies of critically ill COVID-19 patients. 25
Objectives
The objectives of this study were to characterize the implications of CS choice on mortality and AEs, namely, secondary infections and hyperglycemia, in critically ill patients with CARDS.
Methods
Study design
We performed a single-center, retrospective cohort study at a large academic referral center in San Antonio, Texas, comparing use of MP versus DEX in critically ill patients with CARDS. Management decisions were made by treating providers based on clinical judgment.
Case identification and study period
Patients were identified through an internal list of all patients admitted for COVID-19 from April to September 2020 and cross-referenced with a list extracted from the electronic medical record of all patients with a diagnosis of COVID-19 (International Classification of Diseases, 10th revision codes, U07.1 and U07.2). Data were obtained using a structured data collection form electronically generated.
Study population
Inclusion criteria were an ICU admission for at least 24 h with documented severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, ARDS according to the Berlin definition, 26 and treatment for at least 24 h of MP or DEX. Exclusion criteria included (1) any chronic outpatient steroids, (2) hydrocortisone as an initial steroid regimen in lieu of MP or DEX, (3) simultaneous use of both DEX and MP, and (4) greater than 80 mg per day of MP. Patients receiving more than 80 mg MP were excluded based on CS equivalency studies and the theoretically increased risk of greater likelihood of AEs. 27 From March through June 2020, the institutional protocol for management of patients with COVID-19 pneumonia requiring supplemental oxygen support recommended MP 0.5 mg/kg intravenous (IV) twice daily (maximum dose of 80 mg daily) for 5–7 days, with an optional initial bolus dose of 125 mg. After publication of the RECOVERY Trial in June 2020, the institutional protocol was updated to recommend DEX 6 mg IV or oral daily for 5–10 days with an optional loading dose of 10 mg. 3 All patients received routine infection prevention measures, including IV line care, enhanced contact precautions, daily chlorhexidine bath, head of the bed at elevation at 30°, and oral care with chlorhexidine. All patients received care consistent with institutional protocols that included prone positioning, lung protective ventilation as well as deep venous thrombosis prophylaxis. Protective lung ventilation was employed using a low tidal volume ventilation strategy (6 ml/kg) and titration of the fraction of inspired oxygen and positive end expiratory pressure as per the National Heart, Lung, and Blood Institute Acute Respiratory ARDS Network protocol.
Outcomes
The primary outcome was mortality at 28 and 90 days. Secondary outcomes were days alive and free of MV at days 28 and 60 after ICU admission, hospital and ICU lengths of stay, duration of MV, acute renal insufficiency (using the Kidney Disease: Improving Global Outcomes[KDIGO] criteria: increase in serum Cr > 0.3 or 1.5× baseline), renal replacement therapy, new arrythmia (other than sinus tachycardia), new systolic heart failure (left ventricular ejection fraction < 40% assessed by echocardiogram), and need for tracheostomy. Safety outcomes of interest were hyperglycemia and secondary infections, including bacterial respiratory, blood and urine infections as well as fungal infections. Respiratory infection was defined as a pathogen not considered to be normal respiratory flora isolated from the lower respiratory tract. Urinary tract infection was defined as > 100K CFU of a pathogen not considered to be commensal flora. Uncontrolled hyperglycemia was determined by requirement of continuous IV insulin versus subcutaneous insulin (long- and short-acting) and was assessed at hospitalization day 7 after completion of CS and day 14 after clearance of DEX, based on its longer half-life.
Other variables
Demographics and baseline comorbidities were collected, in addition to onset and symptoms of SARS-CoV-2 infection. Vital signs, respiratory support, vasopressor use, SOFA score, and laboratory values were obtained at the time of ICU admission. Microbiological data were recorded at admission and throughout hospitalization and followed up to 90 days from admission to the ICU.
Data processing and statistical analysis
Patient demographics, clinical characteristics, and outcomes were summarized with descriptive statistics. Categorical data were analyzed using chi-square test or Fisher’s exact, as appropriate. Continuous data were assessed for normality using the Shapiro–Wilk W test. Normally distributed data were reported as mean and standard deviation and analyzed with the Student’s t test. Non-normal continuous data were reported as median and interquartile range and analyzed with the Wilcoxon rank-sum test. Kaplan–Meier curves for time to secondary infection and survival to 90 days were analyzed using log rank. Survival to 90 days was also analyzed with Cox proportional hazards regression, adjusted for age and baseline SOFA score. Model selection was guided by the results of bivariate analyses. A p value less than 0.05 defined significance. A convenience sample of all patients meeting inclusion criteria at the institution was included in the cohort. All statistical analyses were performed using SPSS Version 28 (IBM Corporation, Chicago, IL, USA).
Results
Cohort description
Two hundred ninety-one patients were screened for inclusion in the study, of which 113 met criteria for inclusion. Of the 158 ICU patients, 16 did not have CARDS, 20 patients received less than 24 h of CS, resulting in 122 patients, of which 9 were excluded (Figure 1). In total, 113 patients were included in analysis, where 51 patients received MP and 62 patients received DEX. The MP group was significantly younger with a lower severity of illness at inclusion [age 53.3 ± 14.2 versus 59.9 ± 13.0 years (p = 0.01); SOFA 4.5 (3–7) versus 6 (4.8–8) (p = 0.008), Table 1]. The overall cohort was notable for a predominantly Hispanic male population with a significant, but equally distributed burden of metabolic disease between MP versus DEX [male 66.7% versus 64.5%; hypertension 52.9% versus 48.4%; diabetes 49% versus 51.6%; body mass index (BMI) 32.2 versus 30.0 kg/m2 (all ps > 0.2)]. The temporal distribution of corticosteroid use is shown on Figure 2. Clinical presentation and comorbidities are summarized in Tables S1 and S2 in the Supplemental material, respectively.

Study flow diagram.
Medical comorbidities and illness characterization.
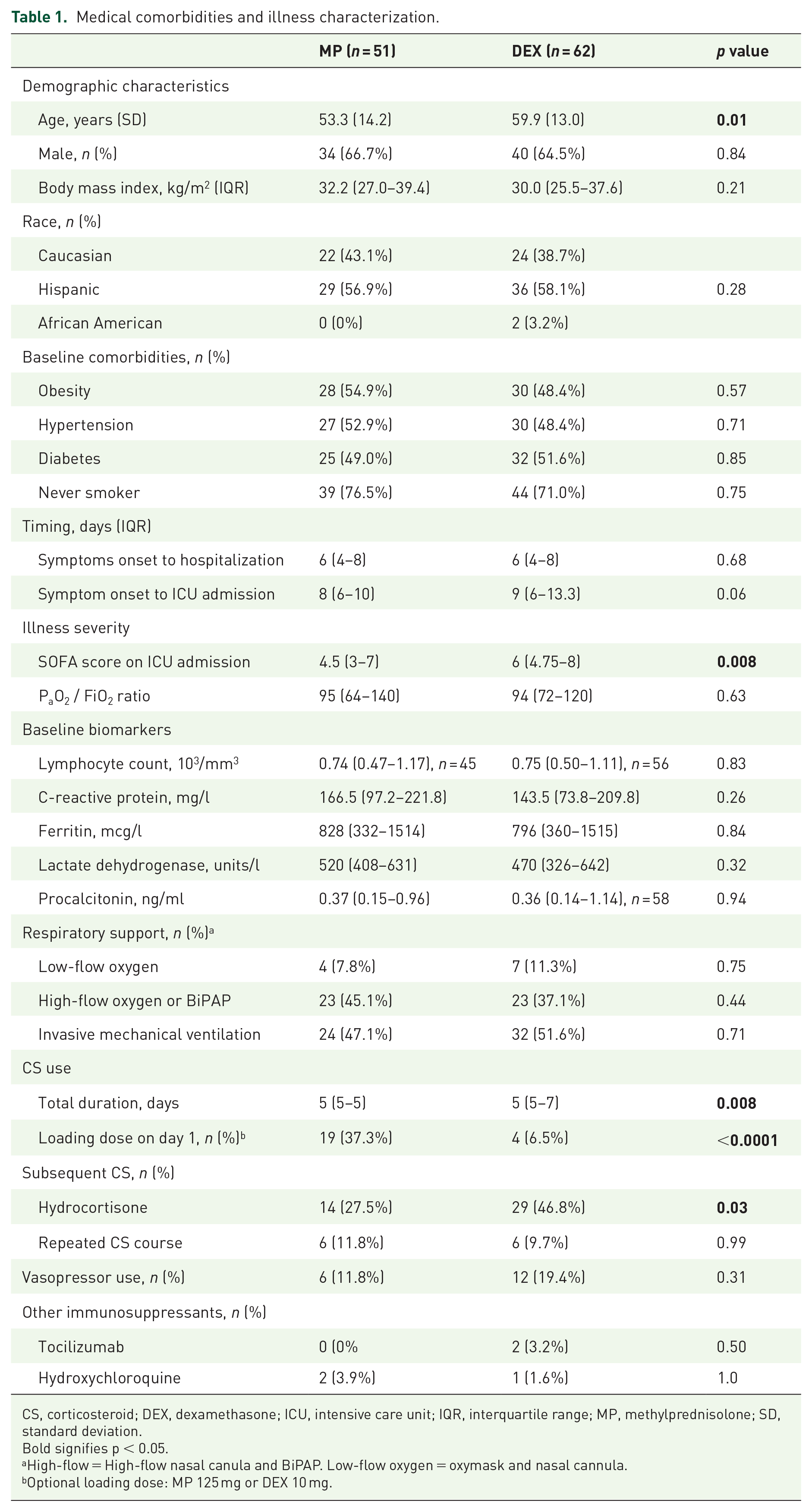
CS, corticosteroid; DEX, dexamethasone; ICU, intensive care unit; IQR, interquartile range; MP, methylprednisolone; SD, standard deviation.
Bold signifies p < 0.05.
High-flow = High-flow nasal canula and BiPAP. Low-flow oxygen = oxymask and nasal cannula.
Optional loading dose: MP 125 mg or DEX 10 mg.
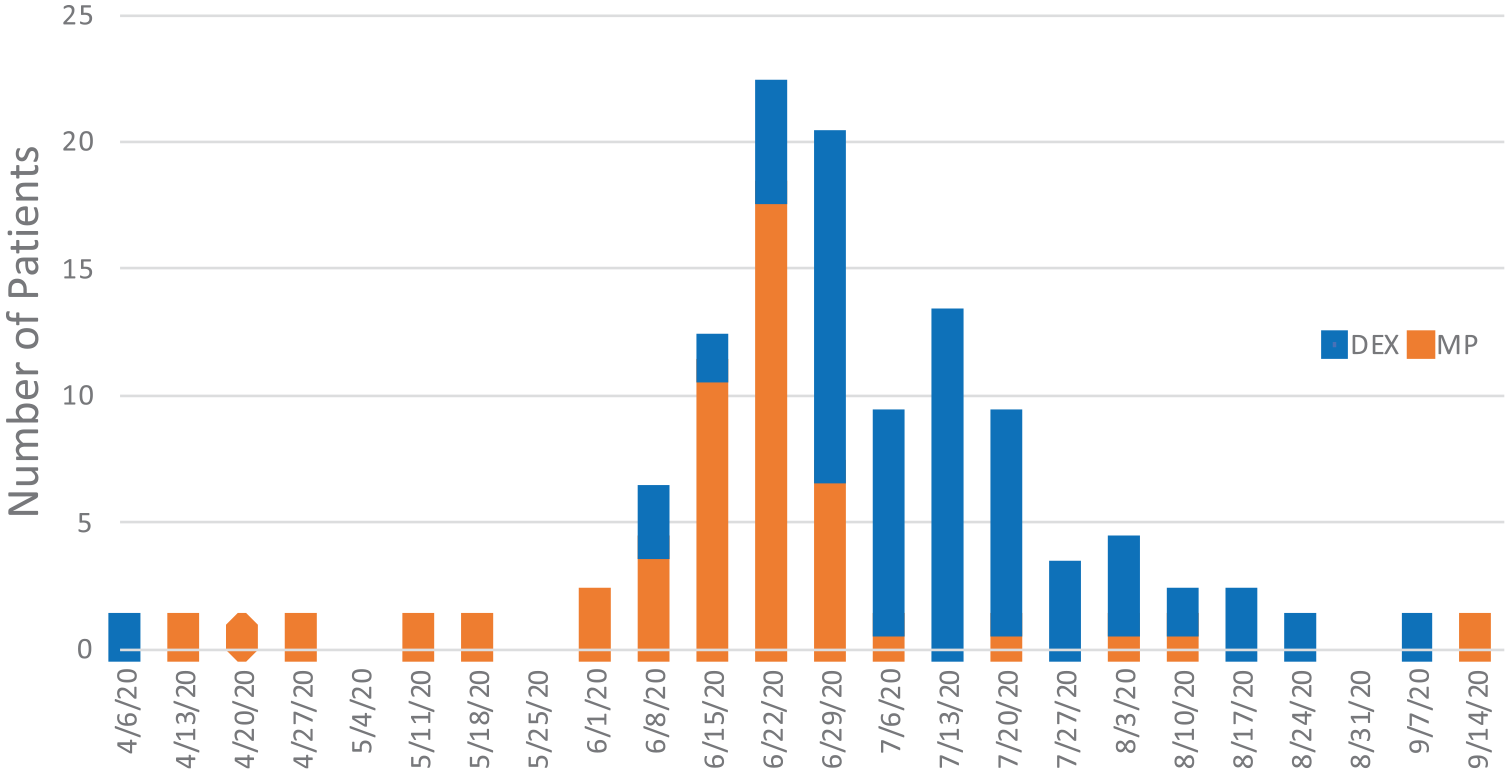
Temporal distribution of corticosteroid use during data collection period from April to September 2020. RECOVERY Trial was initially published on 16 June 2020.
The cohort had a high severity illness, with 88.5% ultimately requiring invasive MV and 16% requiring vasopressors at baseline. There were no differences between groups in rates of MV [MP 44 (86.3%) versus DEX 56 (90.3%), p = 0.56], vasopressor requirement [MP 6 (11.8%) versus DEX 12 (19.4%), p = 0.32], nor vasopressor regimens, which were mostly norepinephrine monotherapy other than two patients requiring addition of vasopressin upon ICU admission. It was characterized by lymphopenic, hyperinflammatory severe CARDS, with similar biochemical characteristics between MP and DEX groups [partial pressure of arterial oxygen to fraction of inspired oxygen [P/F] ratio 95 (64–140) versus 94 (72–210); C-reactive protein (CRP) 166.5 versus 143.5 mg/l; ferritin 828 versus 796 mg/l; lymphocyte count 740 versus 750/mm3 (all ps > 0.2)]. Acute renal insufficiency was common and similar between MP and DEX groups (41.2% versus 45.9%). Other baseline laboratory biomarkers were similar between groups (Table S2 in the Supplemental material).
Time to the first CS dose after hospital admission was similar between groups (1.35 versus 1.37 days, p = 0.52). The most common regimens were MP 40 mg IV twice daily (88.2%) and DEX 6 mg PO daily (93.5%). The total duration of MP therapy was significantly shorter with 12/51 (23.5%) receiving MP for greater than 5 days versus 30/62 (48.4%) in the DEX group, and use of loading doses was more common with MP (37.3% versus 6.5%, p < 0.0001, Table 1). Patients in both groups commonly received concomitant antibiotics, with more antibiotics upon ICU admission in the MP group (96.1% versus 82.2%, p = 0.04). Other CS were also used throughout hospitalization after completion of CARDS-specific treatment with MP or DEX, and hydrocortisone was used more frequently in the DEX group (46.8% versus 25.5%, p = 0.03, Table 1).
Mortality and MV
Days from hospital admission to death were lower in patients on MP versus DEX (19.9 ± 7.8 versus 26.7 ± 13.8 days, p = 0.025), yet the unadjusted mortality rate at 90 days was similar in both groups (53.0% versus 56.5%, p = 0.85, Table 2). Figure 3(a) illustrates the disease progression in the MP group and a similar survival at 90 days. However, given the unequal baseline illness severity based on bivariate analyses, we performed a Cox proportional hazards regression analysis. After adjusting for age and SOFA score at ICU admission, DEX was associated with decreased mortality through 90 days [hazard ratio = 0.43, 95% confidence interval (CI) = 0.23–0.80, p = 0.008] (Figure 3(b)).
Mortality and life-threatening complications.
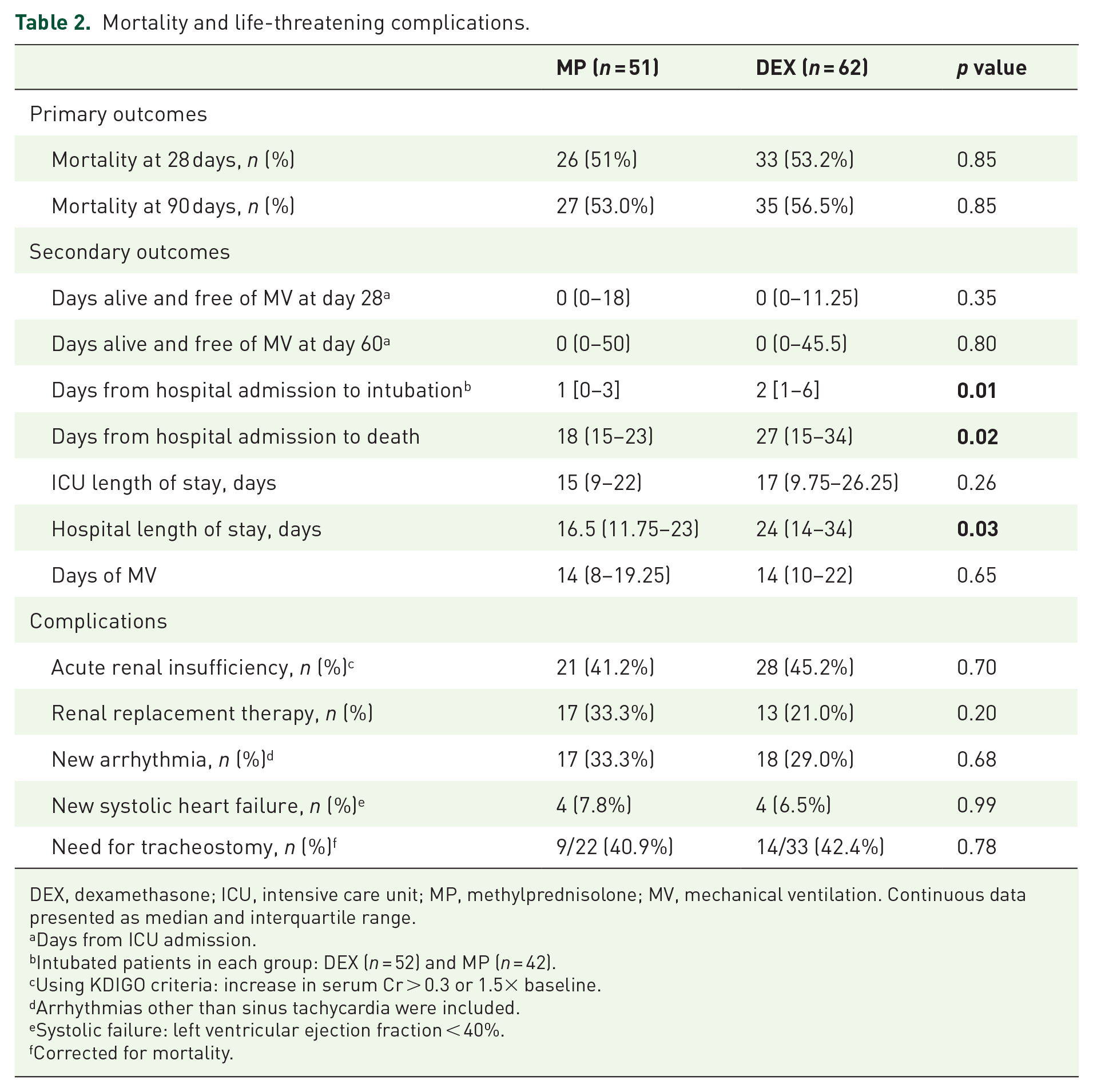
DEX, dexamethasone; ICU, intensive care unit; MP, methylprednisolone; MV, mechanical ventilation. Continuous data presented as median and interquartile range.
Days from ICU admission.
Intubated patients in each group: DEX (n = 52) and MP (n = 42).
Using KDIGO criteria: increase in serum Cr > 0.3 or 1.5× baseline.
Arrhythmias other than sinus tachycardia were included.
Systolic failure: left ventricular ejection fraction < 40%.
Corrected for mortality.

Mortality over time: (a) Kaplan–Meier curve for mortality during hospitalization and (b) Cox proportional hazard regression of mortality by steroid group, adjusted for baseline SOFA score and age, both of which were associated with mortality (HR = 1.04 (1.02–1.06), p < 0.001, and HR = 1.20 (1.10–1.31), p < 0.001, respectively).
There was no difference in days alive and free of MV at day 28: MP 5.5 (1–20.5) versus DEX 8 (1–18), p = 0.87). This finding was the same at day 60 as well as in subgroups of patients with known risk factors for progression of CARDS (diabetes, age over 65 years, BMI over 30 kg/m2, and SOFA > 6).
Secondary infections
Pathogenic bacteria were commonly isolated but not significantly different between MP and DEX (61.2% versus 60.1%, p = 0.99) (Table 3). The distribution of pathogens was notable for a high prevalence of Pseudomonas aeruginosa and methicillin-susceptible Staphylococcus aureus (MRSA) in both groups (Table S4 in the Supplemental material). There was a trend toward more Klebsiella aerogenes infection with DEX (11.3% versus 2.0%, p = 0.07). Fungal pathogens were less common, with a similar incidence in both groups (7.8% versus 4.8%, p = 0.70). The median time to secondary infection was similar between MP and DEX (13.2 ± 9.2 and 14.9 ± 9.1 days, p = 0.53) (Figure S1 in the Supplemental material).
Safety outcomes of super infection and steroid-induced hyperglycemia.

Hyperglycemia
Prevalence of hyperglycemia requiring long-acting insulin was high and similar between MP and DEX at hospital days 7 and 14, whereas uncontrolled hyperglycemia requiring an IV insulin infusion was rare (Table 3). Patient was prescribed sliding scale insulin as needed, as recommended by institutional protocol. Need for insulin regimens (sliding scale versus scheduled long-acting versus infusion) were not significantly different at days 7 and 14. Nursing protocols recommended intermittent subcutaneous over continuous IV insulin to mitigate exposure to SARS-CoV-2, which may account for the higher use of long-acting versus IV insulin.
Discussion
Despite the strong guideline recommendations for CS therapy in CARDS, there is uncertainty whether MP or DEX is superior and safer in critically ill CARDS. 1 The cohort is notable for a higher rate of MV compared with other studies of CS in CARDS, which typically include non-ICU patients. In this study, MP was associated with a shorter time to intubation, more rapid death, and higher adjusted 90-day mortality but had similar safety profiles compared with DEX. Secondary infection rates, time to positive culture, and insulin requirements were not different. Overall, our study suggests that there is not a class effect of CS and potentially an effect on drug-specific mortality in critically ill patients with CARDS.
SOFA scores have consistently been associated with ICU mortality and were associated with mortality in our cohort. However, despite relatively low SOFA scores in the overall cohort, mortality was significantly higher than expected as predicted by SOFA scores, which is consistent with prior studies suggesting limited predictive value of SOFA scores in CARDS. 28 While the overall mortality was higher than other studies, 29 differences may reflect the substantial illness severity of our cohort. Future studies may consider evaluating the prognostic value of the highest or change in SOFA score in the first 48 h after admission, as these variables have been closely correlated with mortality of ICU patients. 30
While our findings are consistent with a previous small study comparing these agents, 17 prior trial results have been mixed with relatively smaller trials demonstrating a mortality benefit of using MP and DEX in CARDS.6,14,15 The discordance may be partially explained by large differences in disease severities and patient populations. Almost 90% of our study population required invasive MV, compared with rates of approximately 30–40% in previous studies evaluating DEX versus MP in CARDS.14,15 For example, in the study by Ranjbar et al., the mean World Health Organisation (WHO) ordinal clinical score at baseline was between 4 and 5, reflective of a hospitalized population not requiring oxygen therapy. Our cohort also had a higher prevalence of chronic comorbidities, particularly cardiovascular disease and diabetes, which are associated with worse outcomes in COVID-19. 31 The efficacy of CS in CARDS is context specific, and further research should evaluate the impacts of timing of treatment, severity of hypoxemia, and phase of illness (inflammatory infiltrates versus fibroproliferative).
The dosing of CSs may have significant mortality implications. Higher doses of steroids (i.e. DEX 10–20 mg or MP 80–100 mg) are associated with rapid-onset, anti-inflammatory, non-genomic effects. 32 However, variable benefits have been seen with higher dose CS in patients with CARDS.33–35 A large recent randomized trial did not demonstrate a difference in days alive without life support when comparing patients given 12 mg with those treated with 6 mg of DEX. 33 Although the recommended CS regimens suggested equivalent doses of MP and DEX, the average effective CS dose of the MP group was more than twice that of DEX, which could have affected our observations. The group receiving MP may have benefited from both non-genomic and genomic effects through receipt of a loading dose and higher daily dose, 13 yet this did not translate to improved clinical outcomes. A recent large CARDS study of predominantly MP found an initial protective effect but no overall mortality benefit of treatment with CS. 5 Hence, additional studies are required to clarify the optimal agent, dosing, and length of CS therapy in CARDS.
Secondary infections are common in hospitalized CARDS patients, reflecting the high prevalence of comorbid conditions and immunosuppression and the long duration of hospitalization.36–38 The rate of secondary infections in our study was approximately 60% – similar to previous studies of patients with COVID-19.20,21,39 This rate is notably higher than the general critically ill population. Significant immune derangements occur in COVID-19 which predispose patients to super-infections, including both bacterial and fungal infections.38,40 The link between CS and risk of infection appears stronger with fungal infections, such as COVID-associated pulmonary aspergillosis (CAPA).38,41,42 However, fungal infections were rare in this cohort and similar between groups, so we are unable to discern an effect related to CS agent used. Many studies assessing differences between MP and DEX in CARDS do not report secondary infection rates.6,14,15 Other results have been mixed, with some reporting an association between secondary infections and DEX, 43 and others reporting no effect on the risk of superinfection.16,17 The risk of secondary infections does not appear to be affected by steroid choice based on our study.
While hyperglycemia has been associated with ICU mortality, there was not a significant difference in insulin therapy between MP and DEX to account for the different mortality. The RECOVERY trial reported only two cases of SIHG out of 2343 patients, and other large trials with DEX did not report hyperglycemia endpoints. However, approximately half of our cohort required scheduled long-acting insulin, reflecting a significant portion with uncontrolled hyperglycemia despite the institutional protocol for sliding scale insulin. During the study period, there was also an institutional recommendation to avoid continuous IV insulin to mitigate exposure to nursing staff, which likely artificially increased the number of patients on scheduled long-acting insulin. In our cohort, hyperglycemia affected both diabetic and non-diabetic patients as evidenced by 16 non-diabetic patients requiring scheduled insulin at day 14.
Limitations
Our study has several limitations, including the retrospective design and relatively small sample size. Our definition of secondary infection was based on a positive culture or fungal seroconversion, but did not consider other clinical parameters, such as imaging or biomarkers, which may have affected the diagnostic accuracy. We also did not evaluate other common CS adverse effects such as steroid-induced myopathy or gastrointestinal bleeding. However, need for tracheostomy may be reflective of muscle weakness and was not significantly different between MP and DEX after accounting for differences in mortality. While the secondary outcomes were not significantly different, there may have been a non-quantifiable time effect as DEX was predominantly used during the second half of the COVID surge and growing experience with CARDS likely improved outcomes. CS selection was based on physician discretion and could have affected the results. Institutional protocols recommended prone positioning if P/F ratio was less than 150 as well as lung protective ventilation – however, actual implementation was not consistently documented, thus not assessed. These protocols did not change throughout the study period but may have affected mortality. Finally, although patients treated with hydrocortisone initially were excluded, some patients with refractory shock were changed to hydrocortisone after receiving at least 24 h of DEX or MP. The incidence of vasopressor use in both groups was similar, but we cannot fully account for the effects of this adjustment in the patient population.
Conclusion
This study further contributes to the knowledge of the implications of CS choice in critically ill patients with CARDS by comparing MP and DEX mortality and AEs up to 90 days after treatment. We found a benefit in adjusted 90-day mortality with DEX, but comparable rates of secondary infections and insulin requirements among critically ill COVID patients treated with DEX and those treated with MP. Further studies are needed to validate our findings.
Supplemental Material
sj-docx-1-tai-10.1177_20499361231153546 – Supplemental material for Safety and efficacy of methylprednisolone versus dexamethasone in critically ill patients with COVID-19 acute respiratory distress syndrome: a retrospective study
Supplemental material, sj-docx-1-tai-10.1177_20499361231153546 for Safety and efficacy of methylprednisolone versus dexamethasone in critically ill patients with COVID-19 acute respiratory distress syndrome: a retrospective study by Dean Kellogg, G. Christina Gutierrez, Clay E. Small, Benjamin Stephens, Paloma Sanchez, Moezzullah Beg, Holly L. Keyt, Marcos Restrepo, Rebecca L. Attridge and Diego J. Maselli in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
