Abstract
In coronavirus disease 2019 pneumonia, a cytokine storm resulting from an excessive inflammatory response to the viral infection is thought to play a role in the exacerbation of the pneumonia and its prognosis. Favipiravir and ciclesonide are not effective in the inhibition of the cytokine storm. In this case report, we describe the experience of tocilizumab administration and polymyxin B immobilized fiber direct hemoperfusion in severe coronavirus disease 2019 pneumonia patient. A 52-year-old man presented with fever and dyspnea and was diagnosed with coronavirus disease 2019 pneumonia based on a polymerase chain reaction test. Mechanical ventilation and favipiravir administration were started for respiratory failure. However, favipiravir could not be continued due to hepatic dysfunction. Consequently, tocilizumab was administered, and continuous hemodiafiltration and endotoxin adsorption therapy (polymyxin B immobilized fiber direct hemoperfusion) were performed for acute renal failure. C-reactive protein decreased from 44 to 3.52 mg/dL, and the patient’s respiratory status improved over time, enabling mechanical ventilation to be withdrawn. This case indicates that adding polymyxin B immobilized fiber direct hemoperfusion to tocilizumab administration may further increase efficacy in coronavirus disease 2019 treatment; however, more case–control studies are needed.
Introduction
Although inhalation of favipiravir and ciclesonide is currently being used to treat coronavirus disease 2019 (COVID-19) infection, these treatments have not shown convincing efficacy in severe cases.1,2 In COVID-19 pneumonia, a cytokine storm resulting from an excessive inflammatory response to the viral infection is thought to play a pivotal role in the exacerbation of the pneumonia and its prognosis. 3 Favipiravir and ciclesonide are not effective in the inhibition of the cytokine storm. Tocilizumab (Actemra, Chugai pharmaceutical, Japan), a humanized anti-human interleukin (IL) 6-receptor monoclonal antibody, is a drug than can be used in rheumatoid arthritis and cytokine release syndrome, 4 and it is considered effective against cytokine storm resulting from COVID-19 pneumonia. 5 Tocilizumab and endotoxin absorption therapy (polymyxin B immobilized fiber direct hemoperfusion: PMX-DHP) were used in a patient with acute respiratory distress syndrome (ARDS) caused by COVID-19, and mechanical ventilation could be withdrawn. The case is described in this report.
Case report
A 52-year-old man presented to our hospital with fever and a sore throat. Acute laryngitis was diagnosed, acetaminophen prescribed, and the patient’s clinical course was observed. However, the fever persisted, and the patient was examined in our department. A nasopharyngeal specimen was collected, a polymerase chain reaction test was positive for COVID-19, and the patient was found to be hypoxemic. He was therefore admitted. On arrival to our hospital, body temperature was 38.3°C; blood pressure, 144/82 mmHg; pulse rate, 94/min; and percutaneous oxygen saturation, 89% (10 L by mask). Breath sounds were clean, with no difference between left and right; no adventitious sounds or cardiac murmurs were heard. There was no pedal edema. Laboratory findings at admission are shown in Table 1 and Figure 1: blood tests showed white blood cells in the normal range, but an elevated neutrophil fraction of 83% and a severely elevated C-reactive protein (CRP) level of 19.97 mg/dL. Simple thoracic computed tomography (CT) showed diffuse ground-glass opacities in both lungs, some of which had mixed infiltrative shadows.
Laboratory results before treatment.

RBC: red blood cells; Hb: hemoglobin; WBC: white blood cells; PLTS: platelets; TP: total protein; Alb: albumin; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; CK: creatine kinase; BUN: blood urea nitrogen; Cr: creatine; CRP: C-reactive protein; KL-6: sialylated carbohydrate antigen KL-6; IgG: immunoglobulin G; IgM: immunoglobulin M; PCT: procalcitonin; LDH: lactate dehydrogenase; IL-6: interleukin-6.
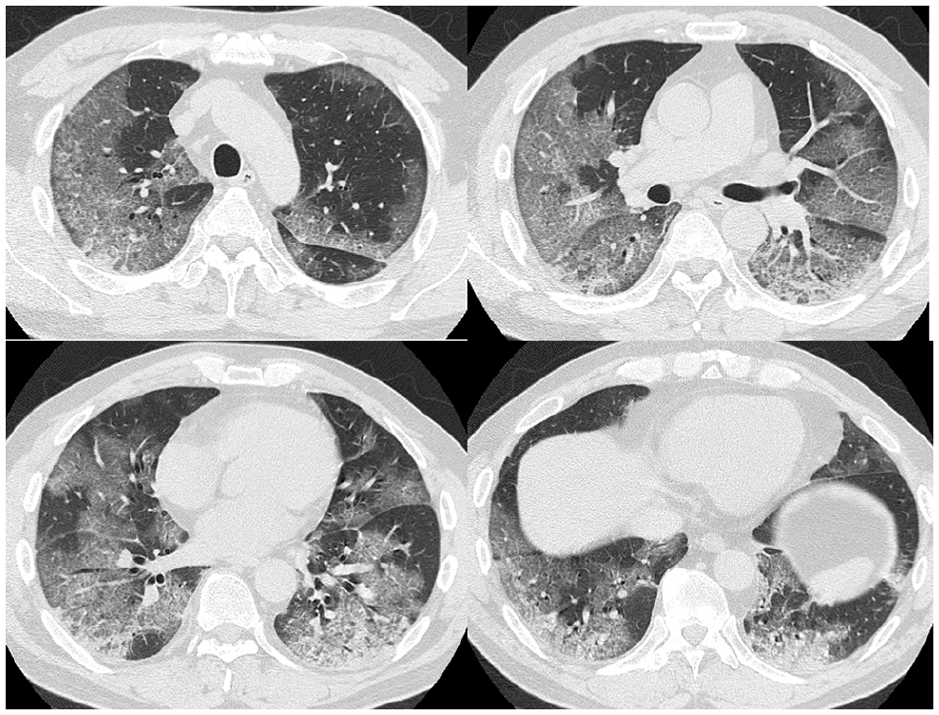
Lung CT on day 1 (before treatment). Ground-glass opacity was observed in both lungs, and some infiltrated shadows were also observed in lower lung.
Clinical course is shown in Figure 2. Because the respiratory failure was severe, management with mechanical ventilation was started immediately after admission. Favipiravir was administered by nasogastric tube at a dose of 1800 mg on the first day and 800 mg on subsequent days. Although the patient’s respiratory status did not worsen, bilirubin and transaminase elevations were seen. The elevations were attributed to drug-induced liver injury caused by favipiravir, which was therefore discontinued on day 4. However, because the CRP level had worsened, tocilizumab administration (8 mg/kg) was started on day 5. Blood tests on the following day also showed CRP to be elevated, and tocilizumab was therefore administered a second time. CRP subsequently decreased over time. At the same time, deterioration of kidney function and decreased urine output were seen after admission. Continuous dialysis was therefore started on day 5, and PMX-DHP was performed on days 7 and 8. The creatine level decreased, and sufficient urine output was obtained after the start of continuous dialysis. Consequently, continuous dialysis was withdrawn on day 16. Although the respiratory failure improved little following tocilizumab administration, improved oxygenation was seen with the improvement in CRP, and the patient was extubated on day 27. Lung CT performed after extubation (Figure 3) and at the time of his discharge (Figure 4) showed extensive residual infiltrative shadows. However, the patient’s condition stabilized with administration of 3 L of oxygen by nasal cannula, and he was discharged on day 54 after rehabilitation under home oxygen therapy.
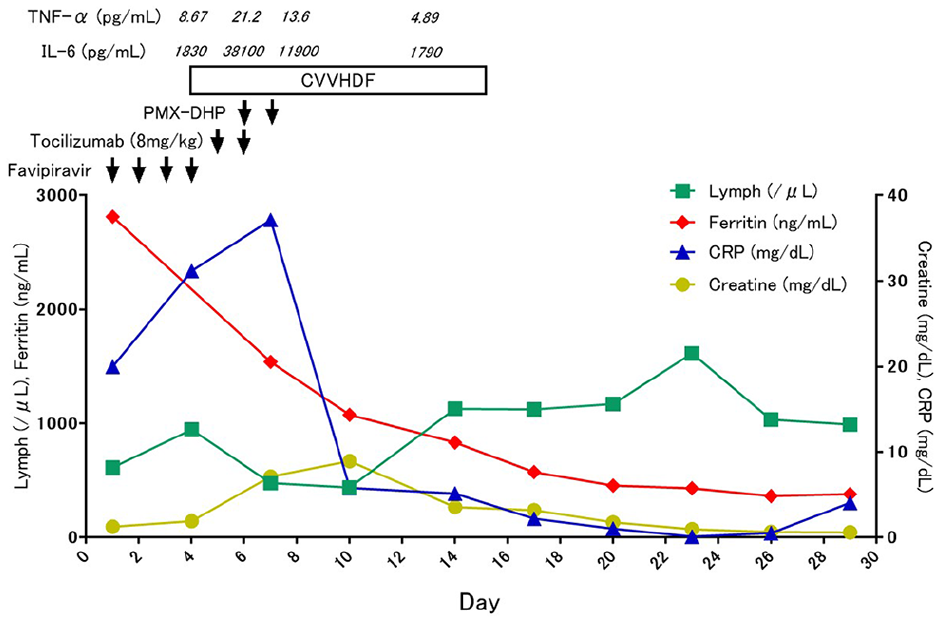
Clinical course.

Lung CT on day 30 (after extubation).

Lung CT on day 51 (before discharge).
Discussion
Although a cytokine storm resulting from viral infection is thought to play a role in the exacerbation of COVID-19 pneumonia, there are few treatments that control a cytokine storm. Many reports have been published in other countries on the use of tocilizumab in COVID-19 pneumonia, but there have been few such reports in Japan.6,7 IL-6 is thought to play a critical role in pneumonia and airway inflammation resulting from viral infection, and there is evidence suggesting that it may be a prognostic factor in COVID-19 pneumonia. 8 Consequently, controlling IL-6 may inhibit the worsening of viral infections. A study in patients with juvenile idiopathic arthritis who contracted influenza found that, in the patients given tocilizumab, fever and CRP were lower before treatment, whereas there was no difference in worsening of the influenza virus infection or in the risk of worsening. 9 In the present case, CRP decreased and the fever subsided beginning from day 3 of tocilizumab administration. However, it was not until 1 week after the start of administration that the oxygen concentration used in mechanical ventilation could be reduced and approximately 28 days until mechanical ventilation was withdrawn. This may have been because the delay in tocilizumab administration prevented inhibition of cytokine storm development. Although there are data that corticosteroids prevent the aggravation of COVID-19 pneumonia, we did not consider steroid use due to lack of evidence when we experienced this case. 10
Continuous hemodiafiltration was performed in the present case because of concomitant acute renal failure. In addition, PMX-DHP using a Toraymyxin (TORAY, Japan) and adsorption column was performed at the same time following the start of tocilizumab administration. Although the use of PMX-DHP to adsorb activated neutrophils and reduce hypercytokinemia, including inflammatory mediators, has been found effective in ARDS, 11 there have been no reports of the effectiveness of PMX-DHP in COVID-19 pneumonia, and its effectiveness is unknown. However, Kudo et al. reported that PMX-DHP was effective against ARDS caused by influenza virus 12 and PMX-DHP has been found to significantly reduce IL-6. 13 Although the blood IL-6 level increased, to 38,000 pg/mL, following tocilizumab administration in the present case, it decreased to 11,900 pg/mL early after PMX-DHP was performed and decreased further, to 1790 pg/mL, after 5 days. Han et al. reported that IL-6 and IL-10 are disease severity predictors. We consider that cytokine storm mainly occurred by IL-6 is the cause of COVID-19 pneumonia, since the tocilizumab had efficacy in present case. 14 It is clinically normal situation for serum IL-6 to elevate with the tocilizumab administration. The tocilizumab blocks the IL-6 receptor rather than IL-6 itself, so negative feedback is applied and serum IL-6 was elevated.15,16 These findings suggest that there is a strong possibility that PMX-DHP would also be effective against ARDS caused by COVID-19. Lin et al. 17 performed continuous dialysis and plasma exchange to treat cytokine storm and reported that high-volume hemofiltration was effective in removing cytokines and stabilizing hemodynamics. In the present case, continuous dialysis after PMX-DHP was performed using a sepXiris (Baxter, Japan) filter, which is a continuous dialysis cytokine adsorption filter consisting of an AN69ST membrane. This filter can not only regulate uremic substances and fluids, but it can also remove cytokines by adsorption. Matsuda et al. 18 examined the effectiveness of a filter consisting of polymethylmethacrylate (PMMA) that was capable of cytokine adsorption and reported it to be effective in ARDS patients. Moreover, Kobashi et al. 19 compared the 28-day survival rates in patients with sepsis after treatment using two types of hemofilters, one consisting of AN69ST and the other of PMMA, and they reported that the AN69ST filter, the type used in the present case, was more useful. However, a characteristic of COVID-19 is thrombus formation resulting from severe inflammation, hypoxemia, bed rest, and disseminated intravascular coagulation. 20 Although nafamostat has been used at high doses, dialysis circuits have been clogged by thrombi, often necessitating circuit replacement.
Even at 1 month after onset, thoracic CT performed after mechanical ventilation had been withdrawn showed residual diffuse ground-glass opacities, and the sialylated carbohydrate antigen (KL-6) level had increased to ⩾2000 U/mL (normal: <499 U/L), indicating fibrogenesis of lung tissues. With regard to the effect of fibrogenesis of lung tissues, although there is a report suggesting that it indicates a good prognosis in COVID-19 infection, 21 there is another report indicating that lung fibrosis results in a poor clinical outcome.22,23
Conclusion
It was possible to save a patient with ARDS resulting from COVID-19 by administering tocilizumab and performing PMX-DHP. Although tocilizumab appears to be useful against COVID-19 pneumonia, caution must be exercised with regard to secondary infection when using it. We believe adding PMX-DHP to tocilizumab administration may further increase efficacy in COVID-19 treatment; however, more case–control studies are needed.
Footnotes
Author contributions
S.S., K.N., A.F., Y.T., H.A., M.O., Y.I., H.Y., T.I., and S.M. contributed to the clinical patient care and management. S.S. and S.M. contributed to the manuscript preparation. All authors have read and approved the final manuscript.
Consent for publication
Written, informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
