Abstract
Background:
Studies comparing the effectiveness of dexamethasone versus methylprednisolone for treating severe-to-critical COVID-19 have produced conflicting results. This study aimed to evaluate the impact of dexamethasone compared with methylprednisolone on in-hospital mortality among patients with severe or critical COVID-19.
Objectives:
The objective of this study was to assess the effectiveness of dexamethasone in comparison to methylprednisolone in reducing in-hospital mortality in patients suffering from severe-to-critical COVID-19 pneumonia.
Design:
This was a retrospective observational study conducted at a tertiary care academic medical center.
Methods:
Clinical data from 706 hospitalized patients with severe or critical COVID-19 in Karachi, Pakistan, were reviewed. Of these patients, 217 received either oral or intravenous dexamethasone, and 393 were treated with methylprednisolone. The primary outcome was in-hospital mortality, while secondary outcomes included the length of hospitalization and the need for mechanical ventilation.
Results:
The methylprednisolone group had a male predominance (74% vs 54%; p < 0.001). However, there was no significant difference in median age between the dexamethasone group (55 years) and the methylprednisolone group (57 years) (p = 0.09). Mortality was significantly higher in the methylprednisolone group compared to the dexamethasone group (13.7% vs 3.2%, p < 0.001). Multivariable analysis showed that dexamethasone was associated with lower in-hospital mortality (adjusted odds ratio (aOR): 0.24; 95% CI: 0.09–0.62; p = 0.003). Furthermore, patients in the dexamethasone group had a shorter length of hospital stay (aOR: 0.87 (95% CI: 0.82–0.92)) compared to the methylprednisolone group. A higher proportion of patients required invasive mechanical ventilation in the methylprednisolone group compared to the dexamethasone group (13.7% vs 3.2%; p < 0.001).
Conclusion:
Dexamethasone was associated with lower mortality and a reduced length of hospital stay and a lower proportion of patients required mechanical ventilation compared to methylprednisolone in patients with severe-to-critical COVID-19.
Plain language summary
This study compared two common drugs, Dexamethasone and Methylprednisolone, to see which one is better for treating severe cases of COVID-19. Previous studies gave mixed results, so this research aimed to find out which drug is more effective in lowering the chances of dying from COVID-19 while in the hospital. The study looked at the medical records of 706 patients with severe or critical COVID-19 who were treated at a hospital in Karachi, Pakistan. Some patients received Dexamethasone, while others got Methylprednisolone. The study checked as to how many patients survived their hospital stay, along with how long they stayed in the hospital and if they needed help breathing with a ventilator. The results showed that the group given Methylprednisolone had more deaths compared to those given Dexamethasone. After taking into account different factors, the researchers found that Dexamethasone was better at lowering the risk of death in the hospital compared to Methylprednisolone. This is important because it helps doctors know which medicine is better for treating severe COVID-19. Using the right medicine can improve the chances of survival and reduce the time spent in the hospital for patients with severe cases of COVID-19.
Background
The WHO declared COVID-19 a global pandemic in March 2020. 1 Since then, the disease has infected over 750 million individuals and caused over 6.9 million deaths. 2 Most patients who are infected with COVID-19 are asymptomatic, or show signs and symptoms resembling a mild upper respiratory tract infection, characterized by fever, headache, cough, dyspnea and occasionally pneumonia. 3 In certain cases, COVID-19 can progress to acute respiratory distress syndrome (ARDS) leading to massive pulmonary damage requiring mechanical ventilation, leading to increased mortality, and burden on the medical system. 4 A total of 15%–30% of patients hospitalized with COVID-19 will develop ARDS. 5 Although the case fatality rate of COVID-19 in the general population is 1.0%, it can be as high as 13% in hospitalized patients, and as high as 37% among patients admitted to the intensive care unit (ICU). 6 The severity of COVID-19 can be classified as mild, moderate, severe, and critical. In mild and moderate forms of the illness, patients manifest with lower respiratory symptoms without any significant change to oxygen saturation. The severe form of the illness is characterized by a decline in the patient’s oxygen saturation necessitating oxygen therapy and/or escalate to non-invasive or invasive mechanical ventilation. 7
ARDS is characterized by elevated levels of tumor necrosis factor-alpha, interleukins, and interferon gamma, which contribute to a dysregulated autoinflammatory response, leading to severe tissue and systemic inflammation, and ultimately high mortality. 8 In June 2020, the first groundbreaking announcement was made regarding the use of dexamethasone to treat COVID-19, and dexamethasone was introduced in the management of severe-to-critical COVID-19 based on the findings from a large randomized controlled trial, which was later published in February 2021 known as the RECOVERY trial. This landmark trial identified that dexamethasone (in comparison to usual care that did not include steroids) leads to a decreased 28-day mortality in patients receiving mechanical ventilation or conventional oxygen therapy.9,10 This evidence has paved the way for the current recommendation (WHO Guideline Therapeutics and COVID-19: living guideline) for the use of dexamethasone in the treatment for critically-ill COVID patients. 11
With these positive strides in treating severe viral lung injury, there is also now growing curiosity in determining the steroid that is most effective in reducing morbidity and mortality in COVID-19 patients. Based on differences in route of administration (oral vs intravenous) and the intensity of anti-inflammatory, immunomodulatory, and mineralocorticoid action, different steroids are available for study and comparison (oral prednisone, oral dexamethasone, intravenous methylprednisolone, and oral or intravenous hydrocortisone). Given the need for quick and potent onset of action (intravenous vs oral route) and a high anti-inflammatory effect, there is growing interest in methylprednisolone, as an alternate to oral dexamethasone, for treating critically-ill COVID-19 patients.
Dexamethasone is long acting whereas methylprednisolone is an intermediate-acting steroid. Dexamethasone is known to have higher potency as compared to other intermediate and short acting steroids but can cause prolonged immunosuppression. 12 Methylprednisolone has greater lung tissue penetration which may lower the severity of ARDS more effectively. Both steroids can dampen the cytokine release syndrome associated with COVID-19. 13 With an increase in cases and increased burden of disease on the medical system, alternative steroids, such as methylprednisolone, have been utilized and have shown positive results in the treatment of severe-to-critical COVID-19.
Studies conducted in China, Iran, and Egypt have demonstrated benefits in severe-to-critical COVID-19 with the use of methylprednisolone. These include a need for reduced need for mechanical ventilation and a decrease in mortality.10,12,13 A systematic review conducted in 2022 by Hong et al. also found that methylprednisolone is effective in treatment of COVID-19. Studies comparing dexamethasone and methylprednisolone, however, are limited and yield conflicting results with some demonstrating equal efficacy, while others demonstrate non-significant differences at equal dosages.14–17 A retrospective (matched pair) clinical trial conducted in 2024 by Kovacevic et al. 18 that looked at moderate to severe COVID-19 with ARDS found that the usage of high-dose dexamethasone had better outcomes for patients when compared to an equipotent dosage of methylprednisolone. In comparison, a retrospective single-center study in Malaysia conducted in 2024 by Soo et al. 19 that also compared high-dose dexamethasone and methylprednisolone was unable to find any significant differences in intubation of ICU admissions.
There does not seem to be any consensus in published data with respect to “a most effective” steroid when it comes to decreasing mortality and improving outcomes of patients who have severe-to-critical COVID-19 pneumonia. Moreover, there are limited data from low-to-middle-income countries (LMICs) where there are resource constraints in terms of the cost and availability of certain steroids. In order to fill this gap in literature, our study aims to compare the effectiveness of dexamethasone with methylprednisolone in preventing in-hospital mortality in patients with severe-to-critical COVID-19 pneumonia. We also aim to determine the differences in length of hospitalization and need for ventilation between the two steroid groups.
Methods
Study design and population
A retrospective observational study was conducted at a 700-bed academic medical center in Karachi, Pakistan. The Aga Khan University Hospital received the first patient with COVID-19 in February 2020. It is one of the few Joint Commission International accredited tertiary care centers in the country and was at the forefront of Pakistan's COVID-19 response with the facility of testing for COVID-19, dedicated COVID-19 patient care areas, and strict infection prevention measures. 20 Clinical data were collected on all adult patients aged 18 years or older diagnosed with severe or critical COVID-19, hospitalized in special care and intensive care COVID-19 units between May 2020 to April 2021 from Hospital Information Management Systems. The diagnosis of COVID-19 was established using polymerase chain reaction (PCR) for SARS-CoV2 in all hospitalized patients. Patients with a high clinical suspicion of COVID-19 but negative PCR results, pregnant women, and patients with chronic conditions requiring long-term steroid treatment were excluded in order to minimize confounding by indication because these patients exhibit altered immune responses, increased susceptibility to infections, and higher baseline risks of comorbidities like osteoporosis, diabetes, and cardiovascular diseases. Severe COVID-19 was defined as clinical signs of pneumonia (fever/cough), in addition to any of the following (respiratory rate > 30 breaths per minute, severe respiratory distress, SpO2 ⩽ 90% on room air, or chest X-ray involving > 50% of the lung fields). Critical COVD-19 included any of the following manifestations: ARDS, Multiorgan Dysfunction, or Septic Shock. 11 Management of patients at the hospital was in accordance with WHO guidelines. However, the hospital did not have extracorporeal membrane oxygenation, high flow nasal oxygen, or transplant facility during the study period and did not have monoclonal antibody treatment or antivirals other than remdesivir for COVID-19. The most frequent ventilatory support apart from mechanical ventilation was “non-invasive positive pressure ventilation,” in the modes of continuous positive airway pressure (CPAP), bilevel positive airway pressure (BiPAP), or Average volume-assured pressure support (AVAPS) modes. Initiation of mechanical ventilation was always primarily based on clinical judgment as determined by the treating physician (in most cases a certified Internal Medicine attending) and was also assessed by a Rapid Response Team at the hospital (composed of critical care nursing and physician team). Indications included standard parameters for mechanical ventilation: overt respiratory failure, muscular fatigue, altered mentation, acute lung injury or ARDS (PF ratio < 300 or 200 mmHg respectively), severe or refractory hypoxemia and/or hypercapnia, failure as defined by the respiratory rate-oxygenation (ROX) index (in some cases), pH < 7.30, pCO2 > 50 mmHg, SpO2 < 85% etc.
The primary endpoint was in-hospital mortality, defined as death during hospitalization. The secondary end-points included length of hospitalization and need for mechanical ventilation. Participants were grouped into three cohorts based on one of the following treatments: intravenous methylprednisolone (n = 393), oral/intravenous dexamethasone (n = 217), or no steroids (n = 96). Patients’ baseline characteristics such as age, sex, demographics, date of admission, comorbid conditions, presenting symptoms, vitals, systemic examination, lab workup, along with the concomitant treatment provided and outcomes (e.g. in-hospital mortality and length of hospital stay) were recorded in a structured case-report form. Patients with incomplete data were excluded from the study and this constituted 3% of records. Complete case analysis remained unbiased since the missingness was unrelated to any observed or unobserved variables (Missing Completely at Random).
Sample size
A total of 615 patients with severe COVID-19 were estimated to be required to achieve 80% power to detect a mortality reduction of no more than 20% in the dexamethasone group as the primary outcome. To account for potential exclusions due to missing information and unavailability of patient files, an additional 15% was included. Therefore, the final sample size was adjusted to be approximately 707 patients to ensure that the study’s statistical power is maintained.
Statistical analysis
All analyses were performed with SPSS (Statistical Package of Social Sciences) version 19 Armonk, NY: IBM Corp. Descriptive statistics included percentages for categorical variables and medians with interquartile ranges (IQRs) for continuous variables. For categorical variables, comparisons were determined by χ2 test or Fisher’s exact test as appropriate. Univariate logistic regression was performed to explore the association of all variables with mortality keeping treatment with either dexamethasone or methylprednisolone as main exposure variable. Variables found to be significant at p = 0.1 on univariate analysis were further evaluated in the multivariable model. Confounding due to other concomitant treatments, such as remdesivir and tocilizumab, was addressed by including these variables in the multivariable regression analysis. Adjusted odds ratio (aOR) and 95% confidence intervals were computed. We constructed the Kaplan–Meier survival curves to compare the cumulative survival between the two treatment groups: methylprednisolone and dexamethasone between different age groups and also stratified by diabetes as comorbid condition over the length of hospital stay, and the log-rank (Mantel–Cox) test was used for the comparison between the survival curves of the two groups. Cox-proportional hazards regression analysis was also performed to determine predictors of in-hospital mortality during hospitalization with severe-to-critical COVID-19. All p-values were two sided and a p-value of ⩽0.05 was considered as significant.
Compliance with reporting guidelines
This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist, ensuring transparency and adherence to standardized reporting practices for observational research. 21
Ethics approval
The study received ethical approval from Aga Khan University, Ethics Review Committee (AKU-ERC) (ERC Reference number: 5249), and the requirement of informed consent was waived as this was a retrospective study that used anonymized data from hospital records.
Results
Out of 706 patients with PCR confirmed severe-to-critical COVID-19, 477 (67.6%) were male and median age was 55 years (IQR: 45–65). Of the 706 patients, n = 217 (30.7%) received dexamethasone, n = 393 (55.7%) received methylprednisolone and n = 96 (13.6%) did not receive any steroids (Figure 1). Patients were also given concomitant medications for the treatment of COVID-19 based on treatment guidelines during that period. A total of 361 (51.1%) patients received azithromycin, n = 158 (22.4%) patients received hydroxychloroquine, n = 65 (9.2%) patients received remdesivir, and n = 53 (7.5%) patients received tocilizumab. Table 1 summarizes key demographic, laboratory parameters, treatment, and outcome characteristics of all three groups: dexamethasone, methylprednisolone, and no steroid group.

Flowchart of study participants.
Characteristics of the study population.

Data were shown as median (q1–q3), mean ± standard deviation or n (%).
Kruskal–Wallis H test.
DM, diabetes mellitus; HTN, hypertension; IHD, ischemic heart disease; IQR, interquartile range; LAMA, leave against medical advice; LDH, lactate dehydrogenase; LoS, length of hospital stay; WBC, white blood cells count.
Both the treatment groups, dexamethasone and methylprednisolone, were comparable for age and comorbid conditions. However, there was a higher proportion of males (n = 290 (74%)) in the methylprednisolone group versus n = 116 (53.5%) in the dexamethasone group. Table 2 compares the characteristics of both treatment cohorts, dexamethasone and methylprednisolone. In the univariate analysis, there was a statistically significant difference in mortality, indicating a higher rate in the methylprednisolone group 13.7% (n = 54 deaths) when compared to dexamethasone 5.1% (n = 11 deaths) with a p-value of 0.001. Patients in the methylprednisolone group also had a longer length of hospital stay (n = 7 days) compared to those who had received dexamethasone (n = 4 days) (p-value: <0.001). In the multivariable logistic regression analysis, treatment with dexamethasone was found to be protective for in-hospital mortality (aOR: 0.24 (95% CI: 0.09–0.62) when compared to treatment with methylprednisolone in severe-to-critical COVID-19. The secondary outcomes included length of stay and the need for mechanical ventilation. Patients in the dexamethasone group had a shorter length of hospital stay (aOR: 0.87 (95% CI: 0.82–0.92)) compared to the methylprednisolone group. The analysis adjusted for age, sex, number of drugs used, and all concomitant drug usage, including azithromycin, remdesivir, and tocilizumab, and among lab parameters, d-dimer > 0.5, and Cr > 1.5, which were significant on univariable analysis at p-value of >0.1. Furthermore, n = 7 (3.2%) patients in the dexamethasone group required mechanical ventilation compared to n = 54 (13.7%) in the methylprednisolone group (p < 0.001) (Supplemental Table 1).
Comparison of severe-to-critical COVID-19 patients who received dexamethasone versus methylprednisolone.
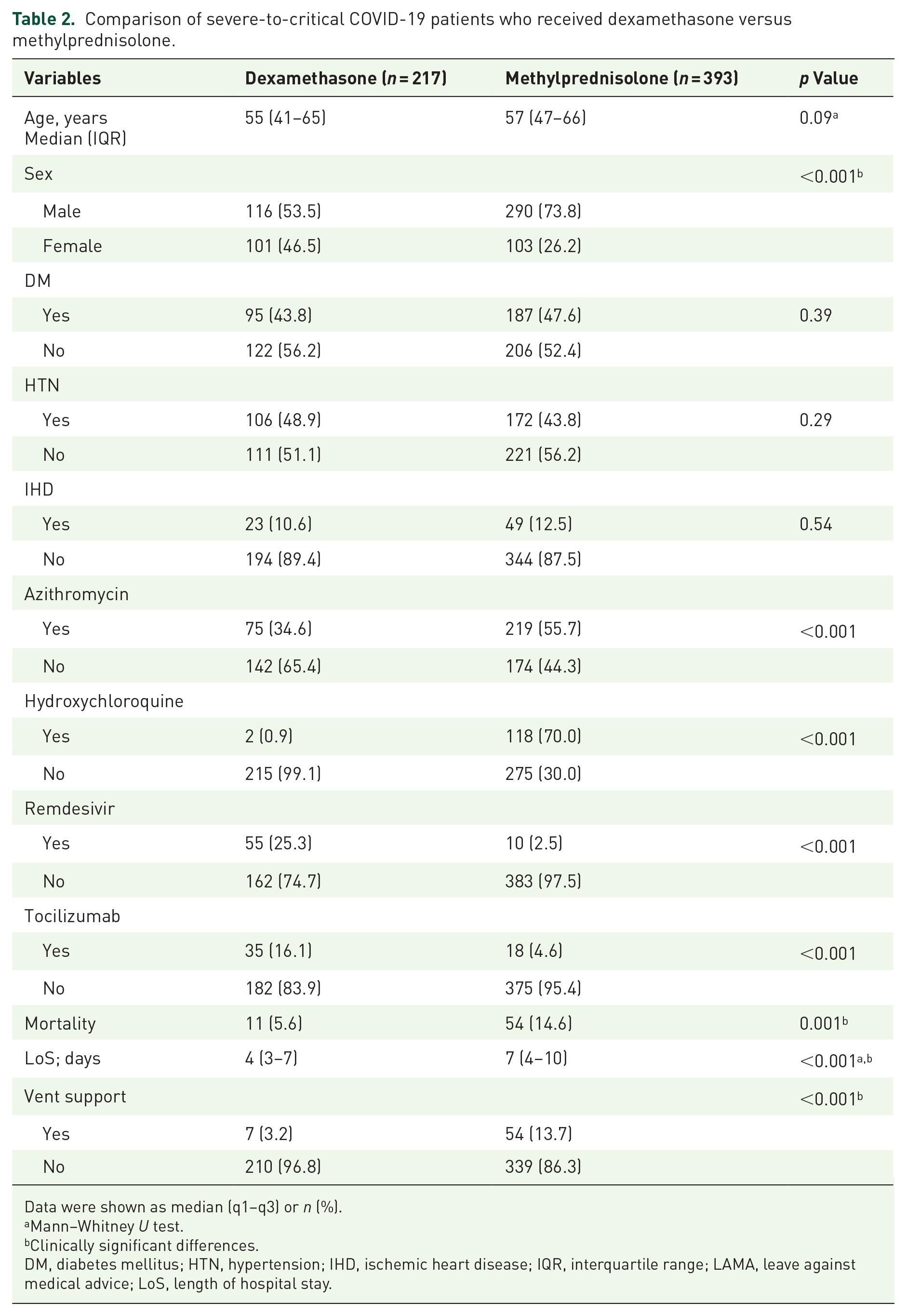
Data were shown as median (q1–q3) or n (%).
Mann–Whitney U test.
Clinically significant differences.
DM, diabetes mellitus; HTN, hypertension; IHD, ischemic heart disease; IQR, interquartile range; LAMA, leave against medical advice; LoS, length of hospital stay.
A subgroup analysis was performed comparing severe-to-critical COVID-19 patients who received systemic steroids versus those who did not receive them, and it was found that mortality was comparatively higher in patients who did not receive steroids (n = 18 (19% )versus n = 65 (11%,) p-value = 0.02) (Supplemental Table 2).
The Kaplan–Meier curves showed that methylprednisolone had a more gradual decline, while dexamethasone showed an early plateau (Figure 2). Dexamethasone offered longer event-free survival (mean = 36 days; 95% CI: (26.17, 45.57)) compared to methylprednisolone (mean = 25 days; 95% CI: (21.62, 28.29)); however, the log-rank test showed no statistically significant difference between the two groups (p = 0.716). This suggests that the longer mean survival time with dexamethasone may be possibly due to a small sample size or censoring. Kaplan–Meier survival curves were also constructed stratified by age groups and showed that age significantly impacts survival probability, with younger patients having better survival outcomes over time compared to older patients (p = 0.004) (Figure 3). However, survival probability was not impacted by the presence of diabetes as a comorbid (Figure 4).

Kaplan–Meier survival curves that represent the general survival probability during hospitalization when using dexamethasone compared to methylprednisolone in severe-to-critical COVID-19 patients.

Kaplan–Meier survival curves that represent the general survival probability during hospitalization when stratified by age groups in severe-to-critical COVID-19 patients.

Kaplan–Meier survival curves that represent the general survival probability during hospitalization when stratified by presence of diabetes as a comorbid condition in severe-to-critical COVID-19 patients.
In the Cox proportional hazards analysis, only age was found to be significantly associated with in-hospital mortality (HR: 1.033; 95% CI: 1.017–1.051; p-value < 0.001), suggesting that with each 1-year increase in age, the hazard of death during hospital stay increased by 3.3%. None of the other variables were found to be significant, including treatment with dexamethasone versus methylprednisolone. Although the hazard ratio of 0.885 suggests that patients on dexamethasone have an 11.5% lower risk of in-hospital mortality compared to those on methylprednisolone, this reduction was not statistically significant (HR: 0.885; 95% CI: 0.454–1.723; p-value = 0.718).
Discussion
Our study found that patients who received dexamethasone had lower mortality rate and were less likely to require mechanical ventilation when compared to those who received methylprednisolone. We also found that patients in the dexamethasone group had a significantly shorter hospital stay compared to those in the methylprednisolone group.
Studies have variably reported clinical outcomes with different steroids with some favoring the use of dexamethasone, while others favoring the use of methylprednisolone in COVID-19 pneumonia with no clear consensus. A multicenter cohort study conducted in 2023 by Watanabe et al. 22 compared methylprednisolone and dexamethasone and found that COVID-19 patients on non-invasive mechanical ventilation had a higher mortality with methylprednisolone. Our study shows similar outcomes with the addition of patients who were on invasive mechanical ventilation, thereby further expanding on previously published literature and providing a broader perspective on the effect of steroid across various intensities of respiratory support. These findings could inform clinical decision making and in determining the choice of steroids based on ventilatory status.
A randomized controlled trial conducted in Iran in 2022 that looked at the length of hospital stay and compared dexamethasone and methylprednisolone came to the same conclusion that dexamethasone was more effective as it had a reduced mean length of hospital stay. 23 Our study also found a significant difference between length of hospital stay when comparing dexamethasone to methylprednisolone (4 vs 7 days; p < 0.001), reaffirming in the real-world conclusions from randomized clinical trials. This strengthens the argument for dexamethasone as a preferred steroid in managing COVID-19, while also demonstrating the importance of validating randomized trial outcomes in broader patient populations.
Other studies comparing methylprednisolone and dexamethasone have found contrary results. A triple blinded randomized control trial conducted by Ranjbar et al. compared outcomes in patients with methylprednisolone and dexamethasone and found a significant improvement in the clinical status, length of hospital stay, and a lower need for mechanical ventilation (18.2% vs 38.1%, p = 0.04) in patients on methylprednisolone. 14 Although the dosage of methylprednisolone in the intervention arm was higher and any differences in outcome could be a result of the difference in potency. A recent large systematic review and network meta-analysis comparing different steroids found that in addition to usual care, methylprednisolone treatment led to better outcomes in COVID-19 pneumonia and ARDS, 24 whereas another systematic review did not find any significant differences in outcomes when comparing dexamethasone with methylprednisolone. 25 Another randomized controlled trial conducted by Pinzon et al. found that high-dose methylprednisolone for 3 days followed by oral prednisolone for 14 days was more effective in treating severe COVID-19 pneumonia than 6-mg intravenous dexamethasone for 7–10 days. This intervention led to a decrease in recovery time, transfer to intensive care, and reduced markers. 26
These differing observations continue to lend to the debate surrounding finding an ideal steroid for the treatment of severe or critically-ill COVID-19 patients. While our findings suggest better outcomes with dexamethasone, particularly in reducing hospital stays and mortality, the results of Ranjbar et al. and Pinzon et al. suggest otherwise, emphasizing the need for more rigorous work. Studying the specific subtype of patient (i.e. biomarkers to identify intensity of inflammation, radiographic findings, timing of steroids with respect to onset of symptoms, and clinical presentation) are further needed to determine the optimal steroid type, dose, and regimen.
We also highlight other important observations from this data. Patients treated with methylprednisolone were more likely to receive azithromycin (55.7% vs 34.6%, p < 0.001) and hydroxychloroquine (70% vs 0.9%, p < 0.001) as compared to those treated with dexamethasone who were more likely to receive remdesivir (25.3% vs 2.5%, p < 0.001) and tocilizumab (16.1% vs 4.6%, p < 0.001). These differences can be explained by the study period as it was conducted during a time when the understanding and optimal treatment strategies for COVID-pneumonia were continually evolving. The results of this study should be viewed with the lens of a constantly changing real-world landscape.
Our study also found that mortality in severe-to-critical COVID-19 patients not treated with steroids was higher compared with those treated with steroids (18.8% vs 10.7%, p = 0.02). The odds of mortality in patients treated with steroids was 0.49 (95% confidence interval 0.27–0.87), suggesting that steroids have a protective effect in patients with severe COVID-19. While the Kaplan–Meier survival analysis suggests differential survival patterns between the two treatment groups, this was not statistically significant (smaller sample size or short duration of follow-up). Although some studies had cautioned against the use of steroids in COVID-pneumonia,27,28 the overwhelming majority of evidence now affirms their benefits, especially the RECOVERY trial that demonstrates a decreased mortality rate in patients treated with steroids. 29 Our findings add to this body of evidence by demonstrating mortality benefits in a real-world clinical setting.
Furthermore, the length of hospital stay was longer in patients who were treated with steroids (6 vs 4 days, p = 0.003). Patients treated with steroids had a higher mean age (56 vs 52, p = 0.005) compared to those who were not treated with steroids. This can be explained by the fact that patients who were treated with steroids most likely had a more severe form of disease, thus leading to increased length of hospitalization, further illustrating the decreased mortality associated with steroid usage and demonstrating their beneficial effects. Moreover, there was also a statistically significant increase in adverse outcomes (need for ventilatory support, patient discharge from the hospital, and death) in the patients who did not receive steroids. These findings have been similarly reported in a meta-analysis of 21 studies comprising 9922 patients that steroids reduced mortality in hospitalized patients in COVID-19 and highlight the protective effect of steroid use on adverse outcomes, which has important implications in the clinical management of hospitalized COVID-19 patients. 30
Despite a high male-to-female ratio of severe-to-critical COVID-19 patients in both treatment groups, our study did not find a statistically significant association of biological sex with mortality in the multivariable analysis in either treatment group. Studies during the early phase of the pandemic have shown that male sex is associated with a higher risk of complications and mortality. Moreover, it has been hypothesized that corticosteroid efficacy may be influenced by biological sex because of differences in sex hormones and immune responses. 31 However, a 2022 Cochrane systematic review did not find any difference in the treatment efficacy of corticosteroids in COVID-19 with estimated effects of both males and females favoring corticosteroid use, which is similar to the findings in our study. 32
Our study addresses a clinically relevant unresolved question in COVID-19 corticosteroid management utilizing a robust retrospective observational design with a relatively large sample size and employing multivariable regression model to control for confounding factors, providing a nuanced understanding of the data. However, the study has several limitations. First, this was a single-center study, and the findings are generalizable to tertiary care centers in LMICs with dedicated COVID-19 facilities. We excluded patients on long-term steroid use to avoid confounding by indication, but this limits the applicability of our findings in long-term steroid users, who may experience diminished therapeutic effects or exacerbated adverse events due to prior steroid exposure. Due to the retrospective nature of the study, data lacked completeness in terms of transfer-ins from other facilities and long-term outcomes and recovery times after hospital discharge. Moreover, our study could not capture the dosage, frequency of administration, and time interval in relation to symptoms and initiation of methylprednisolone and dexamethasone and other residual confounders. If patients in more severe conditions received either of the steroids earlier or at higher doses, this could have biased our findings, potentially impacting their true effect on survival. Future studies with detailed treatment records are needed to address this limitation. Furthermore, data on secondary bacterial infections and/or coinfections that could have affected the outcome were also not available, and fewer patients received remdesivir and tocilizumab because of non-availability in our center during the specified time-period. We recommend larger, multicenter observational studies for the determination of treatment effectiveness with multiple dosages and formulations as availability of certain steroids can be a limiting issue in LMICs. Furthermore, adverse event data need to be captured in subsequent studies particularly since LMICs including Pakistan witnessed a surge in invasive fungal infections and secondary bacterial infections, which were linked to excessive utilization of steroids in COVID-19 patients.
Conclusion
Our study found that the treatment of severe-to-critical COVID-19 with dexamethasone is associated with reduced in-hospital mortality when compared to methylprednisolone. The use of steroids is associated with improved outcomes in this population. These findings could have important implications for better resource management (e.g. costs, ease of administration, etc.) and improved patient outcomes when dexamethasone is used as the preferred steroid for severe and critical COVID-19 in resource-limited settings.
However, rigorous research, including randomized controlled trials, is still needed to provide head-to-head comparisons of dosages, timing of administration, and patient selection to find the ideal steroid regimen for our patients. Studies to effect on long-term outcomes and the occurrence of secondary bacterial or fungal infections are also crucially needed.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251328824 – Supplemental material for The comparative effectiveness of methylprednisolone versus dexamethasone on in-hospital mortality in patients with severe or critical COVID-19: a retrospective observational study
Supplemental material, sj-docx-1-tai-10.1177_20499361251328824 for The comparative effectiveness of methylprednisolone versus dexamethasone on in-hospital mortality in patients with severe or critical COVID-19: a retrospective observational study by Abdullah Wagley, Samar Fatima, Safia Awan, Maheen Zakaria, Aleena Arshad, Farah Khalid, Noreen Nasir, Masooma Aqeel and Nosheen Nasir in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251328824 – Supplemental material for The comparative effectiveness of methylprednisolone versus dexamethasone on in-hospital mortality in patients with severe or critical COVID-19: a retrospective observational study
Supplemental material, sj-docx-2-tai-10.1177_20499361251328824 for The comparative effectiveness of methylprednisolone versus dexamethasone on in-hospital mortality in patients with severe or critical COVID-19: a retrospective observational study by Abdullah Wagley, Samar Fatima, Safia Awan, Maheen Zakaria, Aleena Arshad, Farah Khalid, Noreen Nasir, Masooma Aqeel and Nosheen Nasir in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-3-tai-10.1177_20499361251328824 – Supplemental material for The comparative effectiveness of methylprednisolone versus dexamethasone on in-hospital mortality in patients with severe or critical COVID-19: a retrospective observational study
Supplemental material, sj-docx-3-tai-10.1177_20499361251328824 for The comparative effectiveness of methylprednisolone versus dexamethasone on in-hospital mortality in patients with severe or critical COVID-19: a retrospective observational study by Abdullah Wagley, Samar Fatima, Safia Awan, Maheen Zakaria, Aleena Arshad, Farah Khalid, Noreen Nasir, Masooma Aqeel and Nosheen Nasir in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
