Abstract
Objective:
The coronavirus disease 2019 (COVID-19) pandemic reached New York City in March 2020, leading to a state of emergency that affected many lives. Patients who contracted the disease presented with different phenotypes. Multiple reports have described the findings of computed tomography scans of these patients, several with pneumothoraces, pneumomediastinum, and subcutaneous emphysema. Our aim was to describe the incidence and management of pneumothorax, pneumomediastinum, and subcutaneous emphysema related to COVID-19 found on radiologic imaging.
Methods:
A retrospective chart review was conducted of all confirmed COVID-19 patients admitted between early March and mid-May to two hospitals in New York City. Patient demographics, radiological imaging, and clinical courses were documented.
Results:
Between early March and mid-May, a total of 1866 patients were diagnosed with COVID-19 in the two hospitals included in the study, of which 386 were intubated. The majority of these patients were men (1090, 58.4%). The distribution of comorbidities included the following: hypertension (1006, 53.9%), diabetes (544, 29.6%), and underlying lung disease (376, 20.6%). Among the 386 intubated patients, 65 developed study-specific complications, for an overall incidence of 16.8%; 36 developed a pneumothorax, 2 developed pneumomediastinum, 1 had subcutaneous emphysema, and 26 had a combination of both. The mean time of invasive ventilation was 14 days (0–46, interquartile range = 6–19, median 11). The average of highest positive end expiratory pressure within 72 h of study complication was 11 (5–24) cmH20. The average of the highest peak inspiratory pressure within 72 h of complication was 35.3 (17–52) cmH2O. In non-Intubated patients, 9/1480 had spontaneous pneumothorax, for an overall incidence of 0.61 %.
Conclusion:
Intubated patients with COVID-19 pneumonia are at high risk of pneumothorax, pneumomediastinum, and subcutaneous emphysema. These should be considered in differential diagnosis of shortness of breath or hypoxia in a patient with a new diagnosis of COVID-19 or worsening hemodynamics or respiratory failure in an intensive care unit setting.
Introduction
Since the discovery of a novel coronavirus causing severe acute respiratory syndrome, the virus has rapidly spread throughout the world. In early March 2020, the new coronavirus disease 2019 (COVID-19) pandemic reached New York City (NYC). The city became the new epicenter for COVID-19, with almost 32,107 deaths from 11 March to 2 May (CDC). 1 The medical community was witnessing a wide range of clinical presentations, ranging from asymptomatic, to mild hypoxia, to severe acute respiratory distress syndrome requiring intensive care unit (ICU) admission and mechanical ventilation. The reported rate of intubation and invasive mechanical ventilation ranged from 2% to 17%.2–8
Pulmonary barotrauma is a known complication in mechanically ventilated patients.9,10 It is more commonly seen in patients with underlying lung disease than those with normal lungs. Patients with acute respiratory distress syndrome (ARDS) are most at risk of barotrauma, followed by those with primary obstructive lung disease or pneumonia. 11 Predisposing mechanisms of lung injury and bulla rupture include high tidal volumes and high mean airway pressure. 12 Prompt recognition and treatment of pulmonary barotrauma is important to minimize morbidity and mortality. 10 Management includes close monitoring, needle aspiration, or tube thoracostomy with small-bore chest tubes (including pigtail catheters, typically 6–14 French) or large-bore chest tubes (20–32 French).13–16
In non-intubated COVID-19 patients, it has been suggested that increased intrapulmonary pressure, such as coughing, may lead to bullae rupture and secondary pneumothorax. 17 To date, a few case reports and a case series in Europe highlight the incidence of spontaneous pneumothorax and pneumomediastinum as a complication of COVID-19 pneumonia in non-intubated patients.17–26 These spontaneous complications have been reported before in the previous severe acute respiratory syndrome (SARS-CoV-1) outbreak in 2003, more frequently occurring when the lung ground glass opacities (GGOs) and consolidations began to resolve. 27 In the current COVID-19 literature, the prevalence of spontaneous pneumothorax on chest computed tomography (CT) ranges from 0% to 4%.2,3,28–32 Even small pneumothoraces can cause pulmonary compromise or fatal hemodynamic instability in patients with ARDS, where dependent regions of lung are collapsed and poorly compliant, and therefore at risk of overinflation and rupture.33,34 In this study, we present a retrospective cohort study of COVID-19-related pneumothorax, pneumomediastinum, and subcutaneous emphysema and their subsequent clinical management and outcomes in both intubated and non-intubated patients.
Methods
Study participants’ inclusion criteria and setting
This retrospective cohort study included all adult patients (18 years and above) admitted to the NewYork-Presbyterian Hospital-Weill Cornell Medical Center (NYP-WCMC) and the NewYork-Presbyterian Lower Manhattan Hospital (NYP-LMH) from 1 March 2020 to 15 May 2020 with confirmed COVID-19 by a positive nasal swab reverse transcription polymerase chain reaction (RT-PCR) for SARS-CoV-2. NYP-WCMC is a tertiary center and NYP-LMH is a community hospital. This project was submitted by our for institutional review board (IRB) committee. Ethics approval was exempted as per the HHS 45 CFR 46.104(d) regulation, and informed consent was waived by the IRB committee in our institution as per the HHS 45 CFR 46.104(d) regulation.
Study flowchart
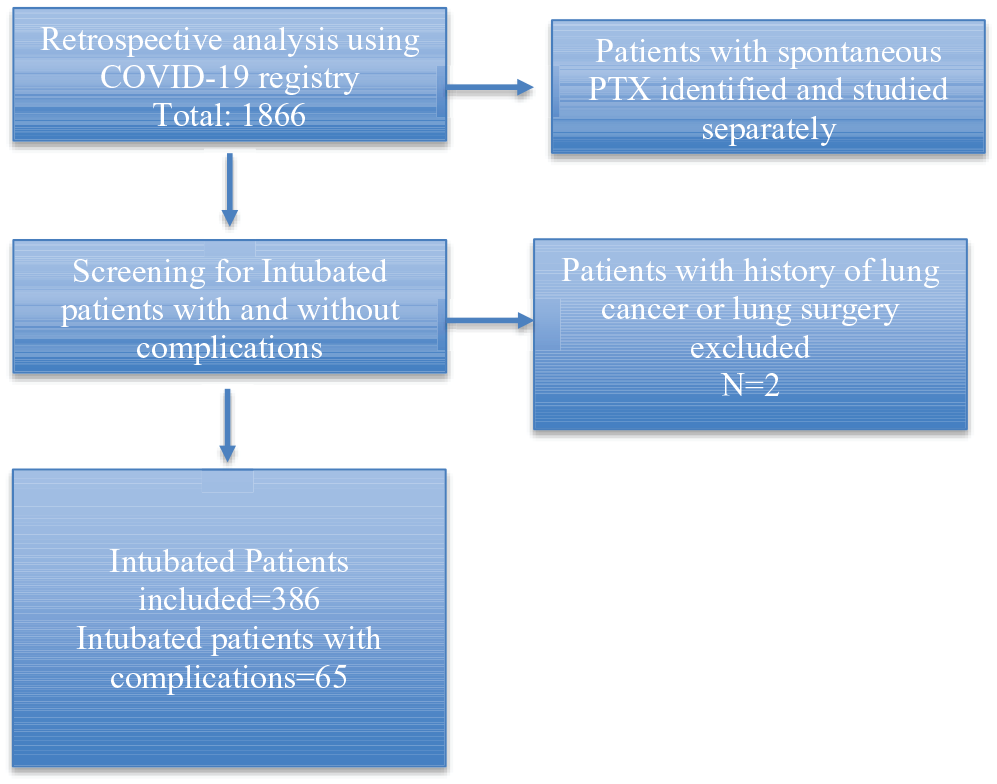
Exclusion criteria
Patients with recent lung surgery or underlying lung malignancy. Patients with a chest X-ray (CXR) or CT radiology read that could not differentiate a pneumatocele from a true pneumothorax.
Data collection
Data were obtained from the electronic medical records (EMRs) of admitted patients from 1 March 2020 to 15 May 2020. Information was manually abstracted from the EMRs of eligible patients and stored into REDCap, supported by the Weill Cornell Medicine’s Clinical and Translational Science Center (CTSC). Additional data were obtained from the Weill Cornell Medicine COVID Institutional Data Repository (COVID-IDR), which is a database of manually abstracted information from all patients who tested positive for SARS-CoV-2 at NYP-WCMC and NYP-LMH. Data were collected for all admitted patients with COVID-19, with further analysis of intubated patients and patients who developed any of the study complications spontaneously or while intubated. Ventilatory parameters of intubated patients with and without the study complication were collected.
The authors of the article reviewed the database of 1866 patients, including the intubated patients, to identify all patients who developed study complications. Four of the authors (S.M., R.W., K.R., Y.A.) then independently reviewed all charts to abstract the following data: patient characteristics, duration of symptoms prior to complication, comorbidities, official imaging read (which was used to identify the study complication), date of intubation, date of complication, positive end expiratory pressure (PEEP) and peak pressure 72 h prior to complication, duration and type of chest tube, complications, and final outcomes of the patients. We then had two reviewers (Y.A., K.R.) independently review the data. The first author then verified all the data and performed all statistical analyses. Analysis was done to compare ventilator parameters between intubated patients with complications and those without. Two of the five authors (Y.A, R.W.) helped with the initial data collection for the COVID-19 database and four of the five authors (Y.A, R.W., K.R. as Internal Medicine Residents at the time of the study and K.R. as a Pulmonary Critical Care Attending) helped care for most of the intubated patients during their clinical stay.
Primary and secondary outcomes
Our primary objective was to evaluate the incidence and management of pneumothoraces, pneumomediastinum, and subcutaneous emphysema in intubated patients with COVID-19. Our secondary objective was evaluating the incidence and management of spontaneous pneumothoraces, pneumomediastinum, and subcutaneous emphysema in non-intubated patients with COVID-19.
Statistical analyses
We characterized the study population by demographics, comorbidities, oxygen supplementation, imaging findings and patterns, and lung complications related to COVID-19 disease or clinical management. Categorical variables are reported as absolute frequencies and percentages. Continuous variables will be shown with mean and standard deviation or median and interquartile ranges (IQRs) as appropriate.
Results
Incidence and management of pneumothoraces, pneumomediastinum, and subcutaneous emphysema in intubated patients
Between early March and mid-May, a total of 1866 patients were diagnosed with COVID-19 at these two hospitals in NYC. Among the 1866, 386 were intubated. Of the 386 patients, 30% were female and 70% were male. Comorbidities in the intubated population included the following: 30% with active or former history of smoking, 58% with hypertension, 35% with diabetes mellitus, and 22% with obstructive or restrictive lung disease (Table 1).
Demographic and clinical details of intubated and non-intubated patients due to COVID-19 disease.

BMI: body mass index; COPD: chronic obstructive lung disease; OSA: sleep apnea; ILD: interstitial lung disease.
Data available for 371 patients.
Data only available for 258 of 321 patients as some of the patients were transfers.
Average BMI rather than obesity was used in COVID-19 registry, so data not available. Available BMI for all patients was 27.4.
A total of 65 intubated patients (16.8%) developed the study complications defined as pneumothorax, pneumomediastinum, or subcutaneous emphysema. Thirty patients developed a pneumothorax, 7 patients had pneumomediastinum, 1 patient had subcutaneous emphysema, and 26 patients had a combination of these. In intubated patients with complications, 21/65 died, resulting in an overall mortality rate of 32%. All intubated patients met the Berlin ARDS Criteria, with 41% having severe ARDS, 50% having moderate ARDS, and 9% having mild ARDS with mean static compliance of 31 at the time of intubation. On average, study complications developed after being on mechanical ventilation for 14 days (0–46 days, median 11, IQR 6–19). The average of the highest PEEP recorded within 72 h before development of complications was 11 cmH2O (5–24 cmH2O, 95% confidence interval (CI) [10.44–12.35], median 12, IQR 10–14). The average of the highest peak inspiratory pressure in the 72 h prior to complication(s) was 35.3 cmH2O (17–52 mmH2O, 95% CI [33.33–37.63], median 36, IQR 31–40) (Tables 2 and 3).
Clinical summary of management of intubated patients due to COVID-19 who developed pneumothorax, pneumomediastinum, or subcutaneous emphysema and their management.

PTX: pneumothorax; PM: pneumomediastinum; SQE: subcutaneous emphysema; CXR: chest X-ray; PEEP: positive end expiratory pressure.
Ventilatory parameter in intubated patients.

P:F: PaO2:FiO2; PEEP: positive end expiratory pressure; PIP: peak inspiratory pressure; Cstat = static compliance; PBW: predicted body weight; TV: tidal volume.
Data available for 371 patients, P:F available for 365 patients.
Data only available for 258 of these patients. P:F ratio not available for 11 patients.
P:F available for 50 patients.
Of the 386 intubated patients, 321 patients did not develop complications; of those, we had the ventilatory parameters of 257 patients. Using this group as a control to our patients with study complications, we were able to compare ventilator parameters between both groups to look for any differences that may be linked to study complications. The average of the highest recorded PEEP in our control group in the first 72 h of intubation was 10 versus 11 cmH2O in the group with complications (p = 0.001, 5–22 cmH2O, 95% CI [9.72–10.44], median 10, IQR 8–12) (Table 3). The average of the highest peak inspiratory pressure in this group in the first 72 h of intubation was 30.8 versus 35 in the group with complications (p < 0.0001, 17–60 cmH2O, 95% CI [29.99–31.58], median 31, ICR 26–35) (Table 2). Other parameters including oxygenation protective ventilation were also assessed. Patients without complications had a higher average PaO2 of 127 in comparison with 49 in patients with complications; in contrast, despite having slightly high predicted body weight/tidal volume (PBW/TV) average in patients with complications, the range was wide and protective lung ventilation did not correlate with the complications, which was also shown in a prior study. 35
Fifty-one patients had a pneumothorax (alone or in combination with other complications). Thirteen of these 51 had a unilateral small pneumothorax with no hemodynamic instability. They were managed with 100% oxygen and serial chest radiography. All these patients had a resolution of their pneumothorax. One patient had extensive subcutaneous emphysema, pneumomediastinum, and a small unilateral pneumothorax that was managed by decreasing the PEEP with eventual resolution of all complications. Of the remaining 38 patients, 24/38 were managed with pigtail catheters and 14 were managed with large-bore chest tubes. The choice of catheter was operator-dependent, but most providers used pigtail catheters initially. Five patients needed an upgrade to a large-bore chest tube given there was no resolution of pneumothorax with the pigtail catheter. One patient had a Heimlich valve attached to his tube for a persistent air leak that was removed prior to discharge. Two of the pigtail catheters were complicated by hemothorax; one patient was a Jehovah’s Witness and unfortunately died due to this complication. In addition, one pigtail catheter was complicated by perforation of the main stem bronchus. The pigtail was removed and another tube was to be placed; however, the patient rapidly decompensated and unfortunately passed away due to a tension pneumothorax (Table 2). Similarly, two of the large-bore chest tubes were complicated by a hemothorax and one patient had a hemothorax after chest tube removal while on moderate-dose enoxaparin prophylaxis (0.5 mg/kg BID as per hospital policy at the time). The average duration of pigtail and large-bore chest tube was 16 days (1–134 days). The average number of chest tubes per patient was 2. Two patients needed five chest tubes and one needed six. Out of these three patients, two had all chest tubes removed and one passed away in the hospital. Patients with pneumomediastinum and subcutaneous emphysema with no pneumothorax were mostly managed by PEEP down-titration. None of the patients were hemodynamically unstable enough to need catheter placement.
Incidence and management of spontaneous pneumothoraces, pneumomediastinum, and subcutaneous emphysema in non-intubated patients
Among the 1866 patients admitted between mid-March and mid-May, 1480 were not intubated. Of the 1480 patients, 44.7% were female and 55.3% were male. Comorbidities in the intubated population included the following: 24.9% with active or former history of smoking, 53% with hypertension, 27.8% with diabetes mellitus, and 19.7% with obstructive or restrictive lung disease.
Nine patients out of 1480 were found to have a spontaneous pneumothorax for an incidence of 0.61%. None of the patients were smokers, and 70% were men. The average age was 55.4 years; the average duration of symptoms was 9.4 (2–14) days. Interestingly, three of these patients reported hearing “a popping sound” before developing shortness of breath.
Five of nine patients were managed conservatively with a cough suppressant, 100% FiO2, and serial CXRs. All five had resolution of their pneumothorax. The other four were managed with pigtail catheters. One patient had a right pigtail followed by a left pigtail catheter, which were both removed. He then had a recurrence on the right side and needed another pigtail for 2 days. He was subsequently discharged with no recurrence on 1-month follow-up. One patient was discharged with a left pigtail catheter with a Heimlich valve, which was subsequently removed by thoracic surgery as an outpatient with no recurrence. The third patient needed bilateral pigtail catheters, but ended up intubated given worsening respiratory status. The pigtail catheters were removed 28 days later. A fourth patient received bilateral pigtail catheters for extensive pneumomediastinum and subcutaneous emphysema. Given worsening respiratory status, he was intubated. He remained on a PEEP of 10 cmH2O, but unfortunately 6 days later upon removal of pigtail catheter, the patient developed a hemothorax on the right side. He subsequently died from a tension pneumothorax on the left side.
Discussion
Our study looked at 1866 patients admitted with COVID-19 infection, of which 386 were intubated. Among those 386 patients, 65 developed either of the three complications for an incidence of 16.8%. Among the remaining 1480 patients, 9 presented with spontaneous pneumothorax for an incidence of 0.61%
We know from previous data that spontaneous pneumothorax is well described as a complication in patients with underlying pneumonia. Patients with acquired immunodeficiency syndrome with Pneumocystis jirovecii pneumonia have a reported incidence of these complications of 2% to 6%. 36 Studies attributed this to increased lung inflammation and peripheral tissue necrosis predisposing patients to these complications. 37 To put our study numbers into perspective, a previous study in 2004 showed an incidence of 1.7% of spontaneous pneumothorax in SARS-CoV-1—6 out of 265 non-intubated patients. 37 Another study in 2003 looking at 75 patients with SARS-CoV-1 population showed a 12% (9/75) incidence of pneumomediastinum at 3 weeks after onset infection. 27 In addition, multiple smaller studies, mostly case reports from China of patients with COVID-19, showed a prevalence of spontaneous pneumothorax on chest CT ranging from 0% to 4%.2,3,28–32 A case series in Europe described nine cases of spontaneous pneumothorax on admission and 14 spontaneous pneumothoraces that occurred during admission. The same series reported 38 cases of pneumothorax in intubated patients with COVID-19. The study has an approximate aggregate of 0.91% in all groups, calculated by approximation of total COVID-19 admissions. 26 Another study in Amsterdam done in March–April 2020 found a 1% (6/542) incidence of pneumothorax in intubated patients in the first 4 days of intubation as compared to our 16.8%. 38 The mean duration of intubation of our patients ranged from 14 to 46 days, which was similar to the reported duration of mechanical ventilation of patients with SARS CoV-1 (14–37 days). 37 Furthermore, multiple recent systematic reviews have shown similar incidence rates to ours, overall averaging from 12% to 24%.35,39,40 The study from 2022 shows a pneumothorax and pneumomediasitum incidence of 10.7% and 11.2%, which was lower than ours likely due to the difference in sample size. 35
Regarding our patients with study complications occurring spontaneously, as opposed to our intubated population, there were no identifiable risk factors in this group. All were non-smokers, and the level of oxygen requirement when the complication developed did not predict the likelihood of eventual progression to intubation. The four patients who eventually got intubated had signs of more diffuse infiltrates on their imaging, making it likely that the severity of the disease itself was a predictor of development of complications and progression to mechanical ventilation. A study from 2021 and case report from 2020 described similar findings in 44 patients with spontaneous pneumothorax, with the majority having resolution of their pneumothorax by conservative or surgical management.41,42
This study is one of the largest cohorts describing the incidence and management of spontaneous and positive pressure–related pneumothorax, pneumomediastinum, and subcutaneous emphysema. It provides deeper insight into the incidence of these complications during the first wave of the pandemic and different approaches to management, but these data have the following limitations. The overall small numbers of subjects with complications limited our ability to identify specific risk factors associated with the development of these complications. In addition, we did not conduct a sample size/power analysis given uncertainty in the beginning of the pandemic while gathering the data and doubt for full extent of these complications. Furthermore, there were some missing data for some of the patients admitted during the peak of the pandemic as indicated in the tables. This study only reviewed patients from the first wave, and further studies are recommended to reassess that incidence in the following waves and now with newer variants especially after the introduction of various therapeutics. Further analysis with comparison to a control group would help deepen our understanding of the main differences between both groups and risk factors contributing to the development of these complications.
Conclusion
In summary, intubated patients with COVID-19 pneumonia are at high risk of pneumothorax, pneumomediastinum, and subcutaneous emphysema. These complications should be considered in differential diagnosis of shortness of breath or hypoxia in a patient with a new diagnosis COVID-19 or with worsening hemodynamics or respiratory failure in an ICU setting.
Footnotes
Acknowledgements
We would like to thank all members of the medical team who relentlessly worked during this pandemic. This work was made possible through data provided by the Cornell COVID-19 Registry, led by Parag G Goyal, MD; Justin Choi, MD; Laura Pinheiro, PhD; and Monika Safford, MD of Weill Cornell Medicine and intubated cohort data from Dr Edward Schenck and Katherine Hoffman. We would also like to thank the contributions of the Architecture for Research Computing in Health team to this work. In addition, we would like to acknowledge Dr Mark Pecker, Dr Anthony Ogedegbe, and Dr Gregory Mints for sharing cases with us.
Author contributions
Y.A., K.R., S.A., and R.W. carried out data collection. Y.A., K.R., S.A., and K.R. contributed to conception and design, acquisition, analysis, and interpretation of data. Y.A., K.R., S.A., R.W., and K.R. wrote the article. K.R. provided critical analysis and discussion. All authors reviewed and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was obtained as follows:
16 June 2020
Principal Investigator: Kapil Rajwani MD
Department: Medicine-Pulmonary Medicine and Critical Care
Protocol Number: 20-05022035-02
Protocol Title: Incidence and management of pre- and post-intubation pneumothorax, pneumomediastinum, and subcutaneous emphysema in admitted patients with COVID-19.
This notice certifies that the research project listed above has been determined to meet exemption requirements at HHS 45 CFR 46.104(d) because it includes only the following research procedures: Exemption Categories: Secondary research for which consent is not required: Secondary research uses identifiable private information or identifiable biospecimens, if at least one of the following criteria is met: (1) the identifiable private information or identifiable biospecimens are publicly available; (2) information, which may include information about biospecimens, is recorded by the investigator in such a manner that the identity of the human subjects cannot readily be ascertained directly or through identifiers linked to the subjects, the investigator does not contact the subjects, and the investigator will not re-identify subjects; (3) the research involves only information collection and analysis involving the investigator’s use of identifiable health information when that use is regulated under 45 CFR parts 160 and 164, subparts A and E, for the purposes of “health care operations” or “research” as those terms are defined at 45 CFR 164.501 or for “public health activities and purposes” as described under 45 CFR 164.512(b); or (4) the research is conducted by, or on behalf of, a Federal department or agency using government-generated or government-collected information obtained for non-research activities, if the research generates identifiable private information that is or will be maintained on information technology that is subject to and in compliance with applicable federal privacy standards found in the E-Government Act, Privacy Act, and the Paperwork Reduction Act. Documents reviewed and approved:
Protocol V 1.0 (8 May 2020)—Clean and Tracked.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CTSC Grant UL1 TR002384.
Informed consent
Informed consent was waived by the IRB committee in our institution as per the HHS 45 CFR 46.104(d) regulation.
