Abstract
Coronavirus disease 2019 (COVID-19) is a global pandemic impacting 213 countries/territories and more than 5,934,936 patients worldwide. Cardiac injury has been reported to occur in severe and death cases. This meta-analysis was done to summarize available findings on the association between cardiac injury and severity of COVID-19 infection. Online databases including Scopus, PubMed, Web of Science, Cochrane Library and Google Scholar were searched to detect relevant publications up to 20 May 2020, using relevant keywords. To pool data, a fixed- or random-effects model was used depending on the heterogeneity between studies. In total, 22 studies with 3684 COVID-19 infected patients (severe cases=1095 and death cases=365) were included in this study. Higher serum levels of lactate dehydrogenase (weighted mean difference (WMD) =108.86 U/L, 95% confidence interval (CI)=75.93–141.79, p<0.001) and creatine kinase-MB (WMD=2.60 U/L, 95% CI=1.32–3.88, p<0.001) were associated with a significant increase in the severity of COVID-19 infection. Furthermore, higher serum levels of lactate dehydrogenase (WMD=213.44 U/L, 95% CI=129.97–296.92, p<0.001), cardiac troponin I (WMD=26.35 pg/mL, 95% CI=14.54–38.15, p<0.001), creatine kinase (WMD=48.10 U/L, 95% CI=0.27–95.94, p = 0.049) and myoglobin (WMD=159.77 ng/mL, 95% CI=99.54–220.01, p<0.001) were associated with a significant increase in the mortality of COVID-19 infection. Cardiac injury, as assessed by serum analysis (lactate dehydrogenase, cardiac troponin I, creatine kinase (-MB) and myoglobin), was associated with severe outcome and death from COVID-19 infection.
Introduction
Coronavirus disease 2019 (COVID-19) is a global pandemic impacting 213 countries and territories around the world. As of 31 May 2020, a total of 5,934,936 COVID-19 confirmed cases and 367,166 deaths have been reported worldwide. 1 Full-genome sequencing indicated that the novel coronavirus belongs to the β genus of coronavirus, which also includes severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS). 2
COVID-19 infection is caused by binding of the viral spike protein to the angiotensin-converting enzyme 2 (ACE2) receptor following activation of the viral spike protein by transmembrane protease serine 2. 3 ACE2 is highly expressed in the lung, vascular endothelium, intestinal epithelium, liver and the kidneys, providing a mechanism for the multi-organ failure that can be seen with COVID-19 infection.4,5 ACE2 is also expressed in the heart, counteracting the effects of angiotensin II in states with intense activation of the renin–angiotensin system such as congestive heart failure, hypertension and atherosclerosis. 4
Several studies have reported the clinical and laboratory findings associated with cardiovascular disease in patients with COVID-19 infection.6–27 We are aware of no systematic review and meta-analysis that summarized available findings in this regard. Thus, in the present study, the laboratory findings and mechanism of cardiac dysfunction caused by COVID-19 infection were summarized.
Methods
Study protocol
A systematic literature search and a quantitative meta-analysis were conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. 28
Search strategy
We conducted a literature search using the online databases of Scopus, PubMed, Web of Science, Cochrane Library and Google Scholar for relevant publications up to 20 May 2020. The following medical subject headings (MeSH) and non-MeSH keywords were used: (‘Severe acute respiratory syndrome Coronavirus 2’ OR ‘SARS-CoV-2’ OR ‘COVID-19’ OR ‘Novel Coronavirus’ OR ‘2019-nCoV’) AND (‘Cardiovascular Disease’ OR ‘Heart’ OR ‘Troponin I’ OR ‘Creatine Kinase’ OR ‘Creatine Kinase, MB Form’ OR ‘Myoglobin’ OR ‘Lactate Dehydrogenase’). The literature search was performed by two independent researchers (MP and AS). We also searched the reference lists of the relevant articles to identify missed studies. No restriction was applied to language and time of publication. To facilitate the screening process of articles, all search results were downloaded into an EndNote library (version X8, Thomson Reuters, Philadelphia, USA). The search strategies and keywords are presented in detail in Supplementary Material Table 1 online.
Eligibility criteria
Studies were included in this review if they met the following inclusion criteria: (1) observational studies with retrospective or prospective design; (2) all articles assessing the association between serum levels of cardiac troponin I, creatine kinase, creatine kinase-MB, myoglobin, lactate dehydrogenase and severe outcome or death from COVID-19 infection as the major outcomes of interest and reported median (interquartile range; IQR) or mean (SD) for serum levels of these biomarkers. Review studies, books, expert opinion articles and theses were excluded.
Data extraction and assessment for study quality
Two independent researchers (MP and SY) extracted the following data from the studies: time of publication, first author’s name, age and gender of patients, sample size, study design, serum levels of cardiac troponin I, creatine kinase, creatine kinase-MB, myoglobin and lactate dehydrogenase and outcome assessment methods.
The Newcastle–Ottawa Scale was used for quality assessment of included articles. 29 Based on these criteria, a maximum of nine points can be awarded to each study. In this systematic review and meta-analysis, studies with the Newcastle–Ottawa Scale score of ⩾ 5 were considered as high quality studies.
Statistical analysis
Median (IQR) or mean (SD) for serum levels of cardiac troponin I, creatine kinase, creatine kinase-MB, myoglobin and lactate dehydrogenase were used to estimate the effect size. The fixed- or random-effect model was used based on the heterogeneity between studies. Heterogeneity of the studies was assessed using the Cochrane Q test and I2 statistics. 30 The publication bias was evaluated by the Egger’s regression tests and visual inspection of funnel plot. 31 The sensitivity analysis was performed to evaluate the effect of each study on the pooled effect size. All statistical analyses were done using the Stata 14 software package (Stata Corp., College Station, Texas, USA).
Results
Search results
Overall, 239 publications were identified in our literature search. Of these, 54 duplicates, 24 non-English, 15 reviews and 110 publications that did not fulfill our eligibility criteria were excluded, leaving 36 articles for further assessment. Out of remaining 36 articles, 14 were excluded because of the following reason: did not report median (IQR) or mean (SD). Finally, we included 22 articles in the present systematic review and meta-analysis (Figure 1).
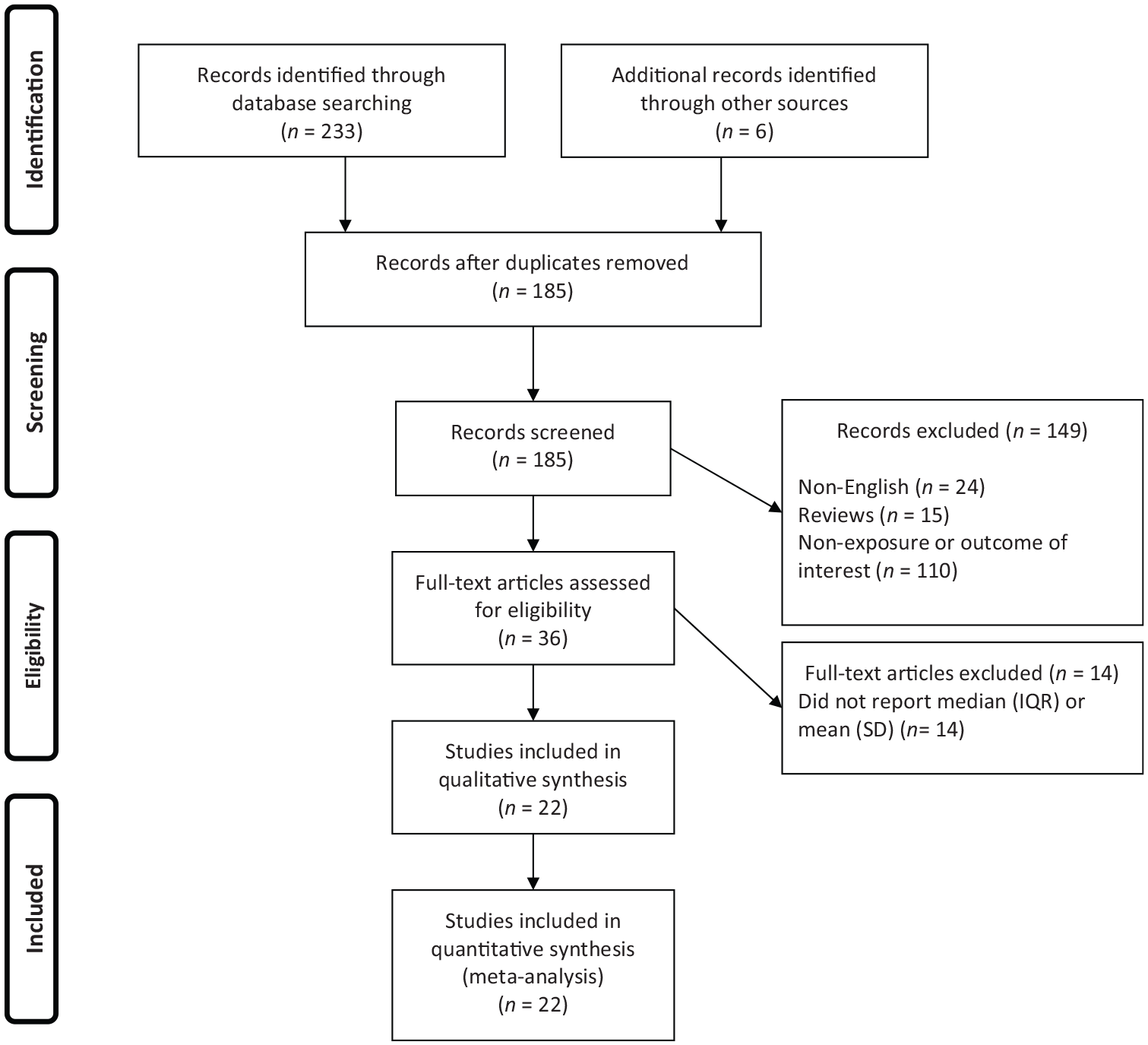
Flow chart of study selection.
Study characteristics
All studies were conducted in China. Twenty studies used retrospective design6,7,9–18,20–27 and two studies used prospective design.8,19 The sample size of studies ranged from 10 to 645 patients (mean age in severe patients: 60.95 years, mean age in non-severe patients: 46.95 years). All studies used real-time reverse transcriptase–polymerase chain reaction to confirm COVID-19 infection. The characteristics of the included studies are presented in Table 1.
Characteristics of studies included in the meta-analysis.
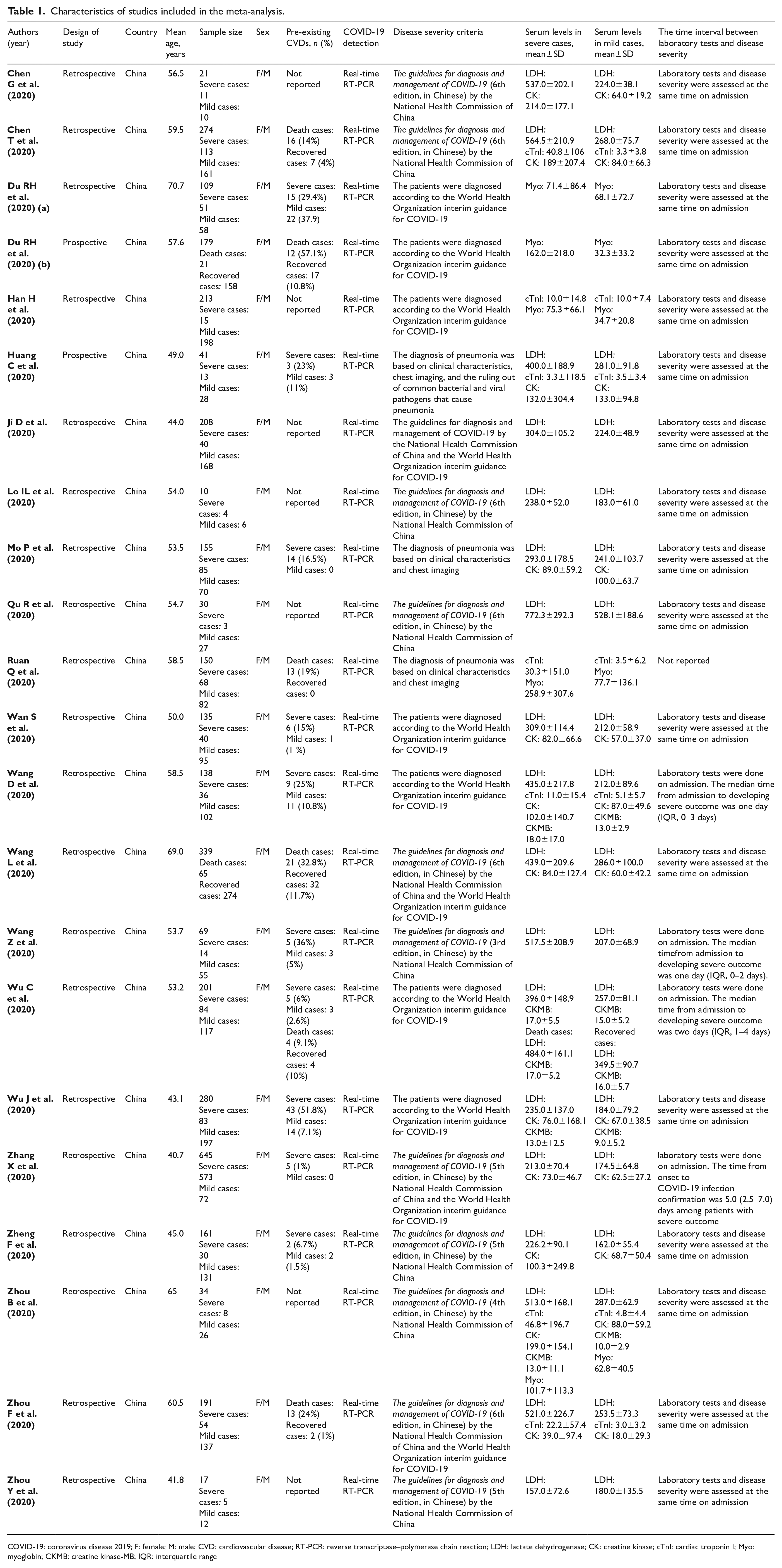
COVID-19: coronavirus disease 2019; F: female; M: male; CVD: cardiovascular disease; RT-PCR: reverse transcriptase–polymerase chain reaction; LDH: lactate dehydrogenase; CK: creatine kinase; cTnI: cardiac troponin I; Myo: myoglobin; CKMB: creatine kinase-MB; IQR: interquartile range
Serum levels of lactate dehydrogenase, creatine kinase-MB, creatine kinase, cardiac troponin I, myoglobin and severity of COVID-19 infection
In the pooled estimate of 17 studies6,8–16,18,20–22,24–26 with 2467 COVID-19 infected patients (severe patients = 1095 and non-severe patients = 1372), it was shown that higher serum levels of lactate dehydrogenase (weighted mean difference = 108.86 U/L, 95% confidence interval (CI) = 75.93 to 141.79, p<0.001, I2 = 85.4%, pheterogeneity <0.001) (Figure 2) and creatine kinase-MB (weighted mean difference = 2.60 U/L, 95% CI = 1.32 to 3.88, p<0.001, I2 = 0.0%, pheterogeneity = 0.517) (Figure 3) were associated with a significant increase in the severity of COVID-19 infection. Combined results showed that serum levels of creatine kinase (weighted mean difference = 15.10 U/L, 95% CI = ‒0.93 to 31.12, p = 0.065, I2 = 46.9%, pheterogeneity = 0.058) (Figure 4), cardiac troponin I (weighted mean difference = 4.05 pg/mL, 95% CI = ‒0.20 to 8.30, p = 0.062, I2 = 0.0%, pheterogeneity = 0.591) (Figure 5) and myoglobin (weighted mean difference = 21.40 ng/mL, 95% CI = ‒0.22 to 43.02, p = 0.052, I2 = 29.3%, pheterogeneity = 0.243) (Figure 6) had no significant association with severity of the disease.
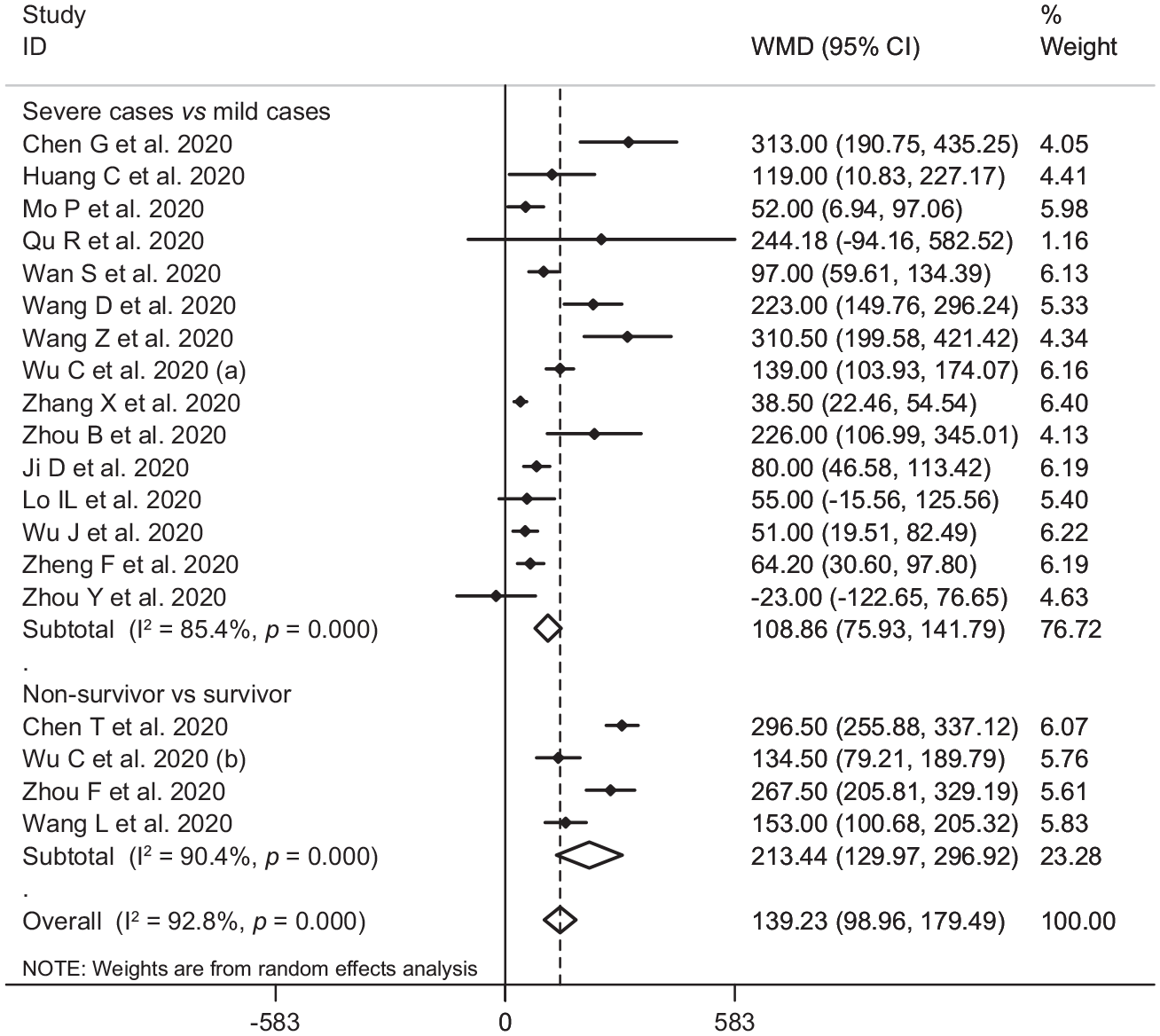
Forest plot for the association between serum levels of lactate dehydrogenase and severe outcome or death from COVID-19 infection using random-effects model.
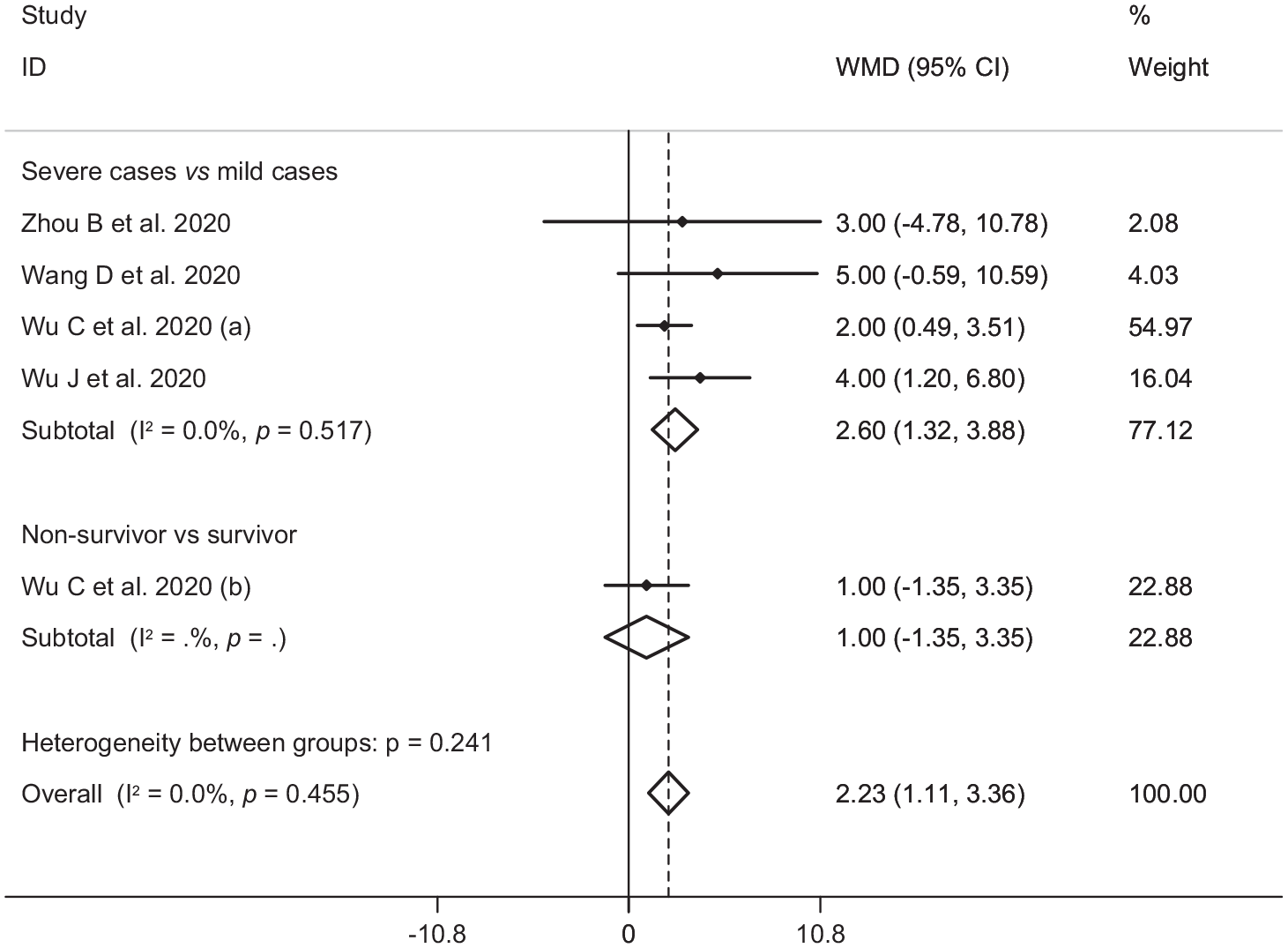
Forest plot for the association between serum levels of creatine kinase-MB and severe outcome or death from COVID-19 infection using fixed-effects model.
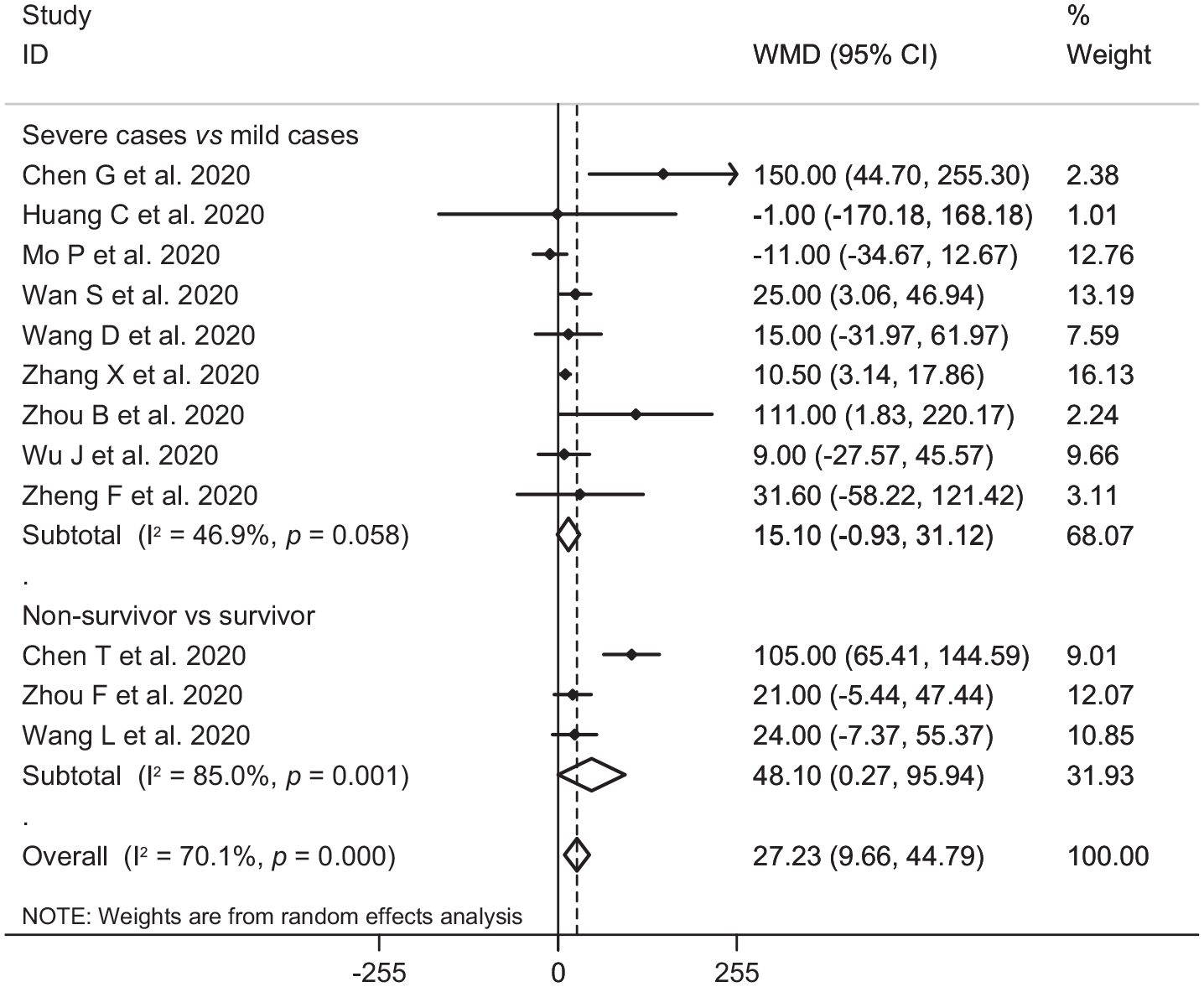
Forest plot for the association between serum levels of creatine kinase and severe outcome or death from COVID-19 infection using random-effects model.
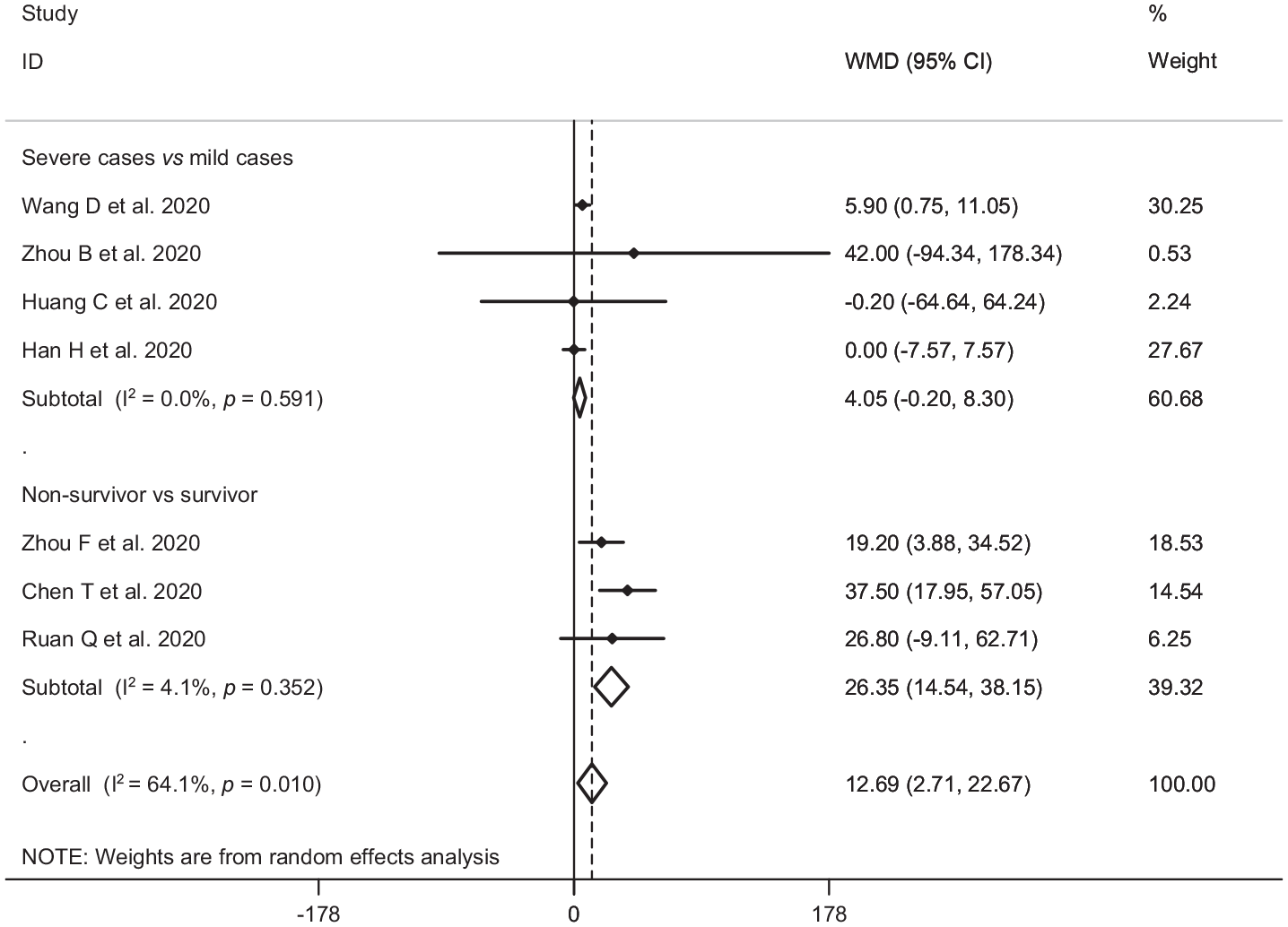
Forest plot for the association between serum levels of cardiac troponin I and severe outcome or death from COVID-19 infection using random-effects model.
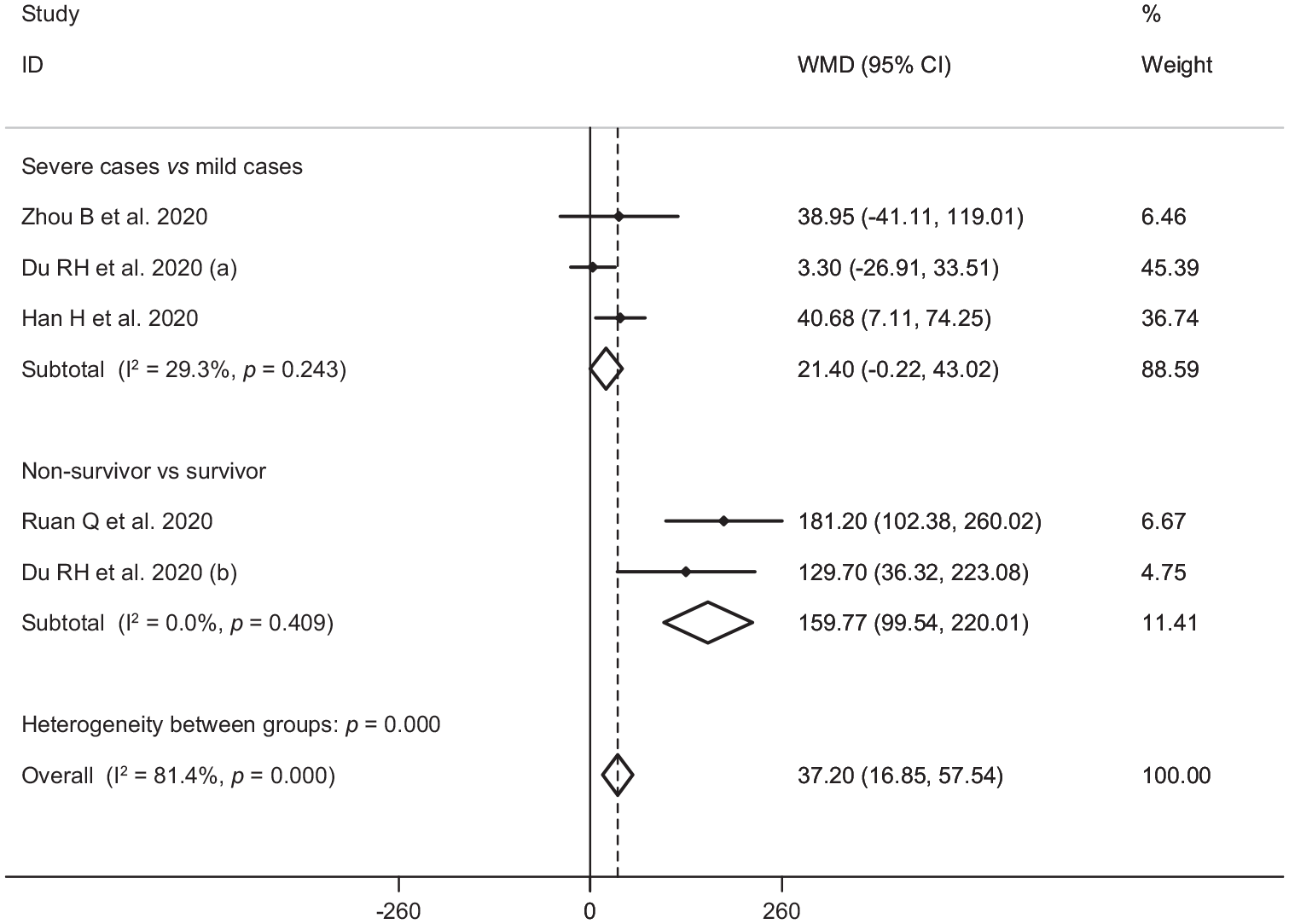
Forest plot for the association between serum levels of myoglobin and severe outcome or death from COVID-19 infection using fixed-effects model.
Serum levels of lactate dehydrogenase, creatine kinase-MB, creatine kinase, cardiac troponin I, myoglobin and mortality from COVID-19 infection
Six studies7,14,17,19,23,27 including a total of 1217 patients with COVID-19 infection (non-survivor = 365 and survivor = 852) reported mortality as an outcome measure. Combined results showed that higher serum levels of lactate dehydrogenase (weighted mean difference = 213.44 U/L, 95% CI = 129.97 to 296.92, p<0.001, I2 = 90.4%, pheterogeneity <0.001) (Figure 2), creatine kinase (weighted mean difference = 48.10 U/L, 95% CI = 0.27 to 95.94, p = 0.049, I2 = 85.0%, pheterogeneity = 0.001) (Figure 4), cardiac troponin I (weighted mean difference = 26.35 pg/mL, 95% CI = 14.54 to 38.15, p<0.001, I2 = 4.1%, pheterogeneity = 0.352) (Figure 5) and myoglobin (weighted mean difference = 159.77 ng/mL, 95% CI = 99.54 to 220.01, p<0.001, I2 = 0.0%, pheterogeneity = 0.409) (Figure 6) were associated with a significant increase in the mortality of COVID-19 infection.
Publication bias and sensitivity analysis
Based on the results of Egger’s test, we found no evidence of publication bias for lactate dehydrogenase (p=0.454), creatine kinase-MB (p=0.367), creatine kinase (p=0.220), cardiac troponin I (p=0.961) and myoglobin (p=0.748). Furthermore, findings from sensitivity analysis indicated that overall estimates did not depend on a single publication (Supplementary Figures 1–5).
Discussion
Findings from this review supported the hypothesis that heart injury is associated with severe outcome and death in patients with COVID-19 infection. To our knowledge, this study is the first meta-analysis to assess the association between serum levels of cardiac biomarkers and severity of COVID-19 infection.
Our results are partially in line with previous narrative reviews.32–36 Previously, cardiac lesion has been reported as a risk factor for severe outcome and death in SARS and MERS.37–40
Older age (⩾65 years), male gender and presence of comorbidities such as hypertension, diabetes, chronic obstructive pulmonary disease and cancer are known to be the major risk factors for COVID-19 mortality. 41 Presence of myocarditis and cardiac injury (defined by elevated cardiac troponin I levels greater than the 99th percentile upper limit) are other independent risk factors associated with mortality.27,42
COVID-19 may either exacerbate underlying cardiovascular diseases and/or induce new cardiac pathologies. Previous studies have shown that the incidence of acute cardiac injury in severe COVID-19 patients and death cases ranged from 5% to 31% and 59% to 77%, respectively.7,8,11,17 Contributory mechanisms include hemodynamic changes, induction of pro-coagulant factors and systemic inflammatory responses that are mediators of atherosclerosis directly contributing to plaque rupture through local inflammation, which predispose to thrombosis and ischemia.43–45
In addition, ACE2, the receptor for COVID-19, is expressed on vascular endothelial cells and myocytes,46,47 so there is at least theoretical potential possibility of direct cardiovascular involvement by the virus. In theory this could have a potential impact on patients taking angiotensin-converting enzyme inhibitors, resulting in greater risk of acquiring COVID-19 infection and increased severity of the disease. 33
Other suggested mechanisms of COVID-19 related heart injury include cytokine storm, mediated by increased pro-inflammatory cytokine production by innate immunity after COVID-19 infection, and hypoxia induced excessive intracellular calcium leading to myocyte apoptosis.17,36
COVID-19 appears to affect the myocardium and cause myocarditis. 48 Interstitial mononuclear inflammatory infiltrates in myocardium has been documented in death cases of COVID-19. 48 Furthermore, cases of myocarditis with reduced systolic function have been reported after COVID-19 infection. 32 Cardiac injury is likely associated with ischemia and/or infection-related myocarditis and is an important prognostic factor in patients with COVID-19 infection.
Cardiac biomarker studies suggest a high prevalence of heart injury in death cases of COVID-19 infection.48,42 Mortality was significantly higher in patients with high serum levels of lactate dehydrogenase, cardiac troponin I, creatine kinase and myoglobin. The mechanism of cardiac biomarker elevation in COVID-19 infection is not fully understood. The underlying pathophysiology is suggestive of a cardio-inflammatory response as many severe COVID-19 infected patients demonstrate concomitant elevations in cardiac biomarkers and acute phase reactants such as C-reactive protein. 33 The rise in cardiac biomarkers with other inflammatory biomarkers raises the possibility that this reflects cytokine storm and may present clinically as fulminant myocarditis. 33
Until effective and specific antiviral therapies against COVID-19 become available, the treatment of COVID-19 infection will be primarily based on the treatment of complications and supportive care. Treatment of cardiovascular complications should be based on optimal use of guideline-based therapies. As with other triggers for cardiovascular events, the use of β-blockers, statins and antiplatelet agents are recommended per practice guidelines.
The present study has some limitations. First, interpretation of findings might be limited by the small sample size. Second, this study did not include data such as body weight, body mass index and smoking history, which are potential risk factors for disease severity.
Conclusion
In this meta-analysis of 3534 patients with confirmed COVID-19, cardiac injury as assessed by serum analysis (lactate dehydrogenase, cardiac troponin I, creatine kinase (-MB) and myoglobin) was associated with severe outcome and death from COVID-19 infection. With the fast-moving development of COVID-19 across the globe and with better understanding of the mechanisms of cardiac involvement in patients with COVID-19 infection, cardiac biomarkers can be utilized as an indicator of improving response due to cardioprotective intervention or as a metric of a worsening clinical scenario.
Supplemental Material
Supplemental_Material – Supplemental material for Cardiac injury is associated with severe outcome and death in patients with Coronavirus disease 2019 (COVID-19) infection: A systematic review and meta-analysis of observational studies
Supplemental material, Supplemental_Material for Cardiac injury is associated with severe outcome and death in patients with Coronavirus disease 2019 (COVID-19) infection: A systematic review and meta-analysis of observational studies by Mohammad Parohan, Sajad Yaghoubi and Asal Seraji in European Heart Journal: Acute Cardiovascular Care
Footnotes
Acknowledgements
Author contribution: MP and SY had the idea for the article, MP, SY and AS performed the literature search and data analysis, and MP and SY drafted and critically revised the work. Availability of data and materials: all data generated or analyzed during this study are included in this manuscript. Supplemental material: supplemental material for this article is available online.
Conflict of interest
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
