Abstract
Background
Left ventricular thrombus is a frequent complication of acute myocardial infarction and a risk factor for thromboembolic complications. Warfarin has been frequently used, but has some disadvantages that limit its use. Direct oral anticoagulants, in particular Dabigatran and Rivaroxaban have been proved to be effective in preventing thromboembolism among patients with non-valvular atrial fibrillation. However, no randomized clinical trials testing the efficacy and safety of these agents in patients with existing left ventricular thrombus. Furthermore, direct oral anticoagulants are still not approved by the Food and Drug Administration in the management of left ventricular thrombus.
Method
This study was a retrospective cohort assessing the efficacy of direct oral anticoagulants (Dabigatran or Rivaroxaban) on the resolution of left ventricular thrombus in patients taking either of these drugs during the study period from December, 2011 to December, 2016 at King Fahad Medical City. All patients’ records were reviewed and all patients who were diagnosed with left ventricular thrombus were included. Patients without available echocardiogram records were excluded. The study was approved by the institutional review board of King Fahad medical city, Riyadh Saudi Arabia.
Result
During the defined study period we found that 413 and 1218 patients were taking Dabigatran and Rivaroxaban, respectively. After filtering them based on the specialty of the prescriber, we ended up with 299 patients who have been started on Dabigatran and 448 patients who have been started on Rivaroxaban by cardiologists. Moreover, after reviewing echocardiogram reports for all of them (747 patients), we found that 11 patients were diagnosed to have left ventricular thrombus. Among those 11 patients, seven of them were treated with direct oral anticoagulants from the beginning and the remaining four patients were shifted from Warfarin to direct oral anticoagulants. All of them (7 patients) showed left ventricular thrombus resolution on follow-up echocardiogram.
Conclusion
Use of direct oral anticoagulants showed promising results in the resolution of left ventricular thrombus in patients diagnosed with left ventricular thrombus. Further studies at multiple health care centers are needed to further evaluate the efficacy and safety of direct oral anticoagulants as compared to traditional treatment in patients with left ventricular thrombus.
Background
Left ventricular thrombus (LVT) is a common complication of acute myocardial infarction (AMI). 1 The risk of LVT formation increases with infarction size; larger infarction implies a larger area of tissue injury resulting in stagnation of ventricular blood. 1 The most common type of LVT is mural thrombus. 1 Echocardiography (ECHO) is the main diagnostic tool used for LVT, which appears as a visible mass in the left ventricle while computed tomography and magnetic resonance imaging are effective, but less commonly used due to their cost and risks. 1
Mobility and protrusion of the thrombus are two characteristics associated with increased embolic potential. 2 The European Heart Journal guidelines published in 2012 stated that patients with mural thrombi require oral anticoagulant (OAC) with vitamin-K antagonist (VKA) therapy for up to six months, while the American College of Cardiology/American Heart Association (ACC/AHA) 2013 guidelines limited the use of VKA therapy to three months in patients who have LVT or are at risk for LVT, such as patients with antero-apical akinesis or dyskinesis. 1 There is still a controversy regarding stopping treatment after resolution of thrombus or continuing treatment.
Warfarin is an OAC used as standard therapy for LVT. It has debatable safety and many clinical limitations, including multiple foods and drug interactions, a slow onset of action and narrow therapeutic range for preventing thrombosis and minimizing hemorrhagic complications.1,3 These disadvantages increase the need to investigate safer, more practical, and effective alternatives to Warfarin. Although the role of direct oral anticoagulants (DOACs) for the treatment of LVT is not yet established, however DOACs (Dabigatran and Rivaroxaban) have proved their non-inferiority to Warfarin in the prevention of thromboembolism in patients with non-valvular atrial fibrillation with significant superiority to lower rates of intracranial hemorrhage. Although no randomized clinical trial was testing the efficacy and safety of these agents among patients with LVT exists, and DOACs are not approved by the Food and Drug Administration (FDA) in the management of LVT however, there is an increasing body of evidence pointing towards effectiveness of these agents on dissolving LVT.8–10 Herein we present our experience with DOACs in the dissolution of LVT at a tertiary care center in KSA.
Methods
This study was a retrospective cohort assessing the efficacy of DOACs (Dabigatran or Rivaroxaban) on the resolution of LVT in patients taking either of these drugs during the study period from December, 2011 to December, 2016 at King Fahad Medical City. Data were collected from the hospital medical records for patients taking either of the two drugs. All records were reviewed, and all patients who were Diagnosed or Rivaroxaban with LVT were included. Patients without available echocardiogram (ECHO) records were excluded. Follow-up was done for all cases until June, 2017. The study was approved by the institutional review board (IRB) of King Fahad medical city, Riyadh Saudi Arabia.
Results
During the defined study period we found that 413 and 1218 patients were taking Dabigatran and Rivaroxaban, respectively. After filtering them based on the specialty of the prescriber, we ended up with 299 patients started on Dabigatran and 448 patients started on Rivaroxaban that prescribed by cardiologists. Moreover, after reviewing ECHO reports for all of them (747 patients), we found that 11 patients were diagnosed to have LVT. Among those 11 patients, seven of them were treated with DOACs from the beginning and the remaining four patients were shifted from Warfarin to DOACs. Those four patients who were treated first with Warfarin were excluded, and we did further data collection for the seven patients who started on DOACs, all of them are male, with mean ages 49 ± 18.3 years; one patient has acute coronary syndrome that required cardiac catheterization and stent implementation, this patient was kept on low dose aspirin plus clopidogrel, another patient has atrial fibrillation (AF) in addition to the LVT and six out of seven patients have heart failure with reducing ejection fractions (Figure 1).

Patients distribution.
Interestingly, follow-up ECHO for all of them showed LVT resolution, but we found variation in documented time for follow-up ECHO without clear reasons that have a clear impact on the anticoagulant duration, means ECHO follow-up was 10.3 month and median 4 month. Tables 1, 2, and 3 summarized all cases.
Summary of baseline patients’ cases.

R: Rivaroxaban; D: Dabigatran; DOAC: direct oral anticoagulant; EF: ejection fraction
Summary of patients’ labe.

ALT: alanine aminotransferase; aPTT: activated partial thromboplastin time; BMI: body mass index; INR: international normalized ratio
Medical History of Patients.
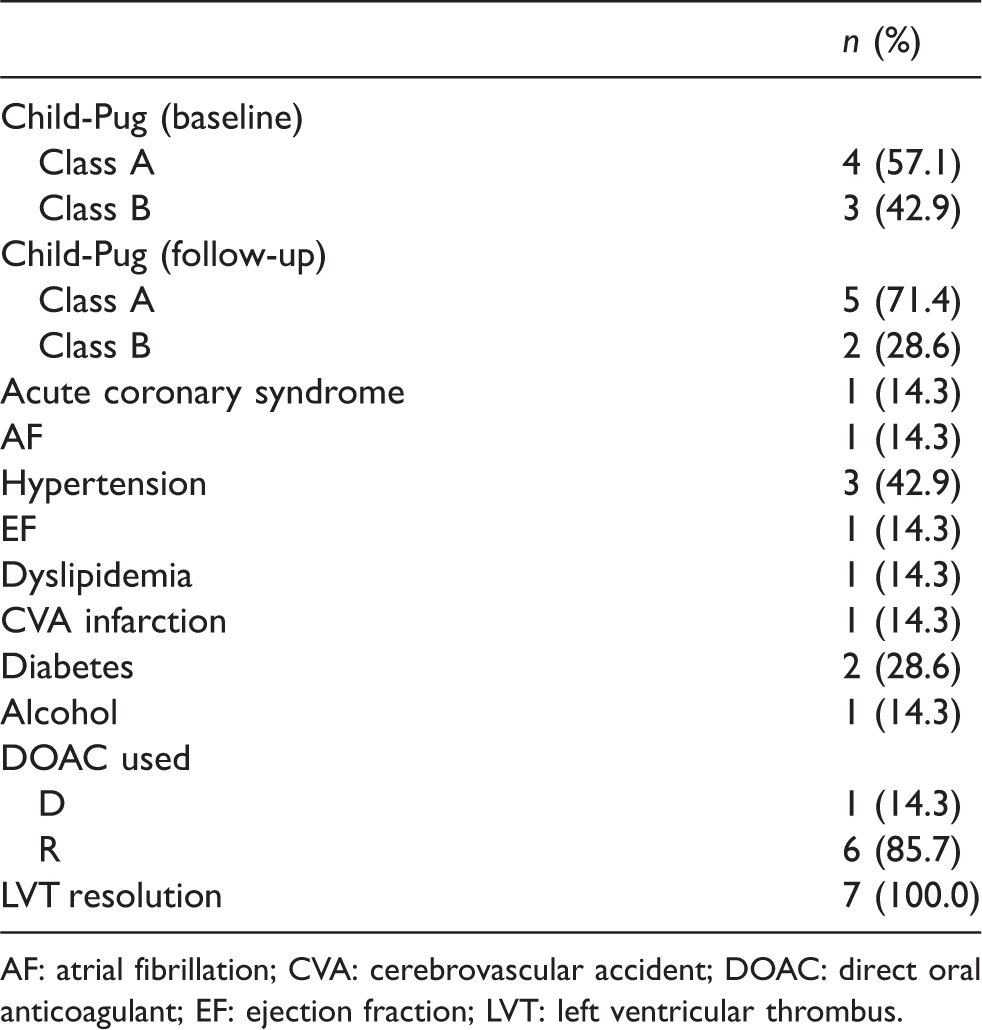
AF: atrial fibrillation; CVA: cerebrovascular accident; DOAC: direct oral anticoagulant; EF: ejection fraction; LVT: left ventricular thrombus.
Discussion
This study shows successful resolution of LVT in all patients treated with DOACs, despite the variation in documented time for follow-up ECHO is one of the main limitations as it is high, inconsistent, and not adhering to internationally accepted recommendations.
The successful resolution of LVT among all patients entered on this cohort is consistent with the result of nine internationally published case repots between 2013 and 2017 described patients whose LVT was successfully resolved on DOACs (Appendix 1, Table 4). One patient was on Apixaban, three patients were on Rivaroxaban, and five patients were on Dabigatran. Patients were managed with anticoagulation therapy for a period ranging from seven days to four months. 1 None of the patients exhibited any bleeding incident due to use of DOACs. Moreover, our result shows the successful result of LV thrombus resolution which was previously achieved with Warfarin therapy only. 9
Nevertheless, our results support the AHA/American Stroke Association guidelines on stroke prevention, 2014 that introduced a new recommendation advising that Low Molecular Weight Heparin, Dabigatran, Rivaroxaban, or Apixaban may be considered as an alternative to VKAs for post-MI LVT for patients not tolerating VKAs due to of non-hemorrhagic adverse events.
Conclusion
Use of direct oral anticoagulants showed promising results in the resolution of LVT in patients diagnosed with LVT. Further studies at multiple health care centers are needed to further evaluate the efficacy and safety of DOACs as compared to traditional treatment in patients with LVT.
Summary of current cases
Case I: A 21-year-old male with a history of labile international normalized ratio (INR) was admitted to the hospital in January 2015 with the diagnosis of LVT. Upon admission patient’s baseline laboratory investigations were normal with normal sinus rhythm on ECG. On the second day of admission, transthoracic echocardiography (TTE) was performed which revealed an apical thrombus with severe global LV systolic dysfunction, moderate to severe tricuspid regurgitation and severe pulmonary hypertension. The patient was started on warfarin to achieve a target INR of 2–3 and discharged in a stable condition. At five months follow-up, TTE showed persistent LVT, and warfarin was replaced by rivaroxaban 20 mg daily for a year at the end of which the follow-up transesophageal echocardiography (TEE) showed complete resolution of LVT.
Case II: A 74-year-old male with a history of hypertension was admitted in August 2015 as a case of acute coronary syndrome. All pertinent baseline laboratory investigations were normal. ECG on admission showed: Q-waves in V1 and V2, deep T-wave inversion in V2, V3, V4, T-wave inversion in inferior leads V5 and V6. Furthermore, TTE revealed that the left ventricle was mildly dilated. Left ventricular ejection fraction was 35%, and there was a moderate size apical thrombus, and an abnormality in regional wall motion and mild mitral regurgitation. Consequently, coronary cardiography was done with percutaneous coronary intervention (PCI) and the patient was started on acute coronary syndrome treatment regimen including aspirin 81 mg and clopidogrel 75 mg daily, and rivaroxaban 15 mg daily for three months for management of LVT. On November 2015, TTE showed the resolution of thrombus.
Case III: A 51-year-old male was diagnosed with LVT in September 2015, based on the TTE result which showed normal-sized left ventricle, mildly reduced systolic function, 50% ejection fraction with dyskinetic apex and apical thrombus. The right ventricle was normal in size and function, and there were no hemodynamically significant valvular disease and no pericardial effusion. All pertinent baseline laboratory investigations were normal, and ECG showed normal sinus rhythm. The patient was started on rivaroxaban 20 mg daily and the follow-up TTE at three months showed apical dyskinesia with spontaneous echo-contrast but no obvious thrombus as before.
Case IV: A 50-year-old male with a history of ischemic heart disease, hypertension, and diabetes mellitus was diagnosed with LVT in December 2014. TEE showed moderate size apical thrombus, moderately dilated left ventricle, ejection fraction of 25–30%, and regional wall motion abnormalities. All pertinent baseline laboratory investigations were normal. ECG showed normal sinus rhythm and possible left atrial enlargement. He was started on rivaroxaban 20 mg daily in addition to ischemic heart disease regimen. After two years, TEE showed resolution of the LV apical thrombus.
Case V: A 45-year-old male with a history of AF was diagnosed with LVT in June 2015. TTE done at that time showed severely dilated left ventricle, large apical thrombus, left ventricular ejection fraction of less than 20%, severe global hypokinesis of the left ventricle, moderately dilated right ventricle, moderate to severely reduced right ventricular systolic function, moderate mitral regurgitation, and moderate tricuspid regurgitation. All other baseline laboratory investigations were normal. ECG showed multiple premature ventricular contractions with underlying sinus rhythm. Dabigatran 110 mg twice daily (BID) was started. The follow-up TTE done at two months showed resolution of LVT.
Case VI: A 68-year-old male was diagnosed with LVT in April 2014, and TTE showed a large apical thrombus, moderately reduced left the ventricular systolic function, ejection fraction of 35–45%, regional wall motion abnormalities, and a large apical aneurysm. His laboratory investigations were normal, and ECG showed sinus bradycardia. The patient was started on rivaroxaban 15 mg daily for two years. Follow-up TTE done at two years follow-up showed no apical LV thrombus.
Case VII: A 34-year-old male with a history of hypertension, heart failure with reduced ejection fraction, dyslipidemia, ischemic heart disease, diabetes mellitus, and cerebrovascular accident infarction was diagnosed with LVT in September 2016. TTE showed a moderate size apical thrombus, left ventricular ejection fraction of 40–45%, and hypokinetic left ventricular apex. All baseline laboratory investigations and ECG were normal. Rivaroxaban 15 mg was given daily for three months. In the next follow-up, TTE showed no thrombus in LV apex.
Highlights
Using DOAC for dissolving LVT has a great advantage over using warfarin which requires frequent laboratory monitoring at least every six weeks with a steady range. DOAC are not being studied in LVT resolution; therefore, this report may shed light on this topic. There is a need for conducting randomized clinical trials testing the safety and efficacy of DOAC in resolving LVT .
Footnotes
Contributorship
Reem : reviweing data and proposal , Writing manuscript, edit manuscript, submetted to the journal
Shroq and MAha: wtiring proposal,collecting data and clean data
ASoofi: reviwe study proposalHassan:reiwed study proposal
Madona: reviweing proposal and mauscript
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by KFMC IRB office with IRB no. 17–178.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
Reem Bahmaid
