Abstract
Background:
Current guidelines recommend vitamin K antagonists (VKAs) for left ventricular thrombus (LVT) resolution. Direct oral anticoagulants (DOACs) are increasingly evaluated as alternatives to the standard of care in anticoagulation.
Methods:
We performed a systematic review and meta-analysis to assess the use of DOACs vs VKAs for LVT treatment. The occurrence of LVT resolution, systemic embolism (SE) or stroke, and bleeding events were compared during follow-up using random-effects analysis.
Results:
The 5 included studies were all observational (a total of 828 patients). Of these, 284 patients (34%) were treated with DOACs, and 544 (66%) treated with VKAs. Thrombus resolution was similar for both methods (pooled odds ratio [OR], 0.91; 95% CI, 0.47-1.75; I 2 = 63%; P = .78). The incidence of SE or stroke was also similar (pooled OR, 1.59; 95% CI, 0.85-2.97; I 2 = 0%; P = .14). Clinically relevant bleeding incidence was similar for both groups (pooled OR, 0.66; 95% CI, 0.31-1.40; I 2 = 0%; P = .28), although all bleeding events were less frequent in the DOAC group (pooled OR, 0.49; 95% CI, 0.26-0.90; I 2 = 0%; P = .02).
Conclusion:
Our systematic review and meta-analysis suggests DOACs were as effective as VKAs for LVT resolution, with a similar risk of systemic embolism/stroke and clinically relevant bleeding. These results, obtained from observational studies, are not definitive and hence randomized controlled trials are needed. Nevertheless, our analysis identifies key experimental features required in future studies.
Background
The reported incidence of left ventricular thrombus (LVT) ranges from 15% to 25% in patients with anterior myocardial infarction (MI) 1 and as high as 36% with dilated cardiomyopathy when optimal imaging modalities are used. 2,3 LVT is associated with increased risk of cerebral and peripheral ischemic events and subsequent increased mortality. 4,5
Current guidelines for LVT resolution recommend anticoagulant treatment with vitamin K antagonists (VKAs) for 3 months 6 or 6 months. 7 Direct oral anticoagulants (DOACs) have emerged as alternatives to the standard of care in anticoagulation for the prevention of SE in atrial fibrillation (AF) 8 -11 and for prevention and treatment of venous thromboembolism. 12 -15 Compared with VKA treatment, DOACs offer the advantage of providing a stable and predictable anticoagulant effect with no need for monitoring. DOACs are also associated with higher medication adherence. 16 In patients with AF, DOACs have a favorable risk-benefit profile; they provide a significant reduction in the risk of intracranial hemorrhage and all-cause mortality. 17
Although there are no randomized clinical trials to assess DOAC treatment of LVT, observational studies have been published. 5,18 -23 In addition, several case reports evaluated off-label use of DOACs for LVT resolution. 24 -31
We performed a systematic review and meta-analysis to assess the effect of different anticoagulation strategies (DOACs versus VKAs) on the resolution of LVT and the prevention of related ischemic and bleeding events.
Methods
We searched MEDLINE and the Cochrane Library databases using the terms “left ventricular thrombus” or “left ventricular thrombi” and “anticoagulation” or “anticoagulants” or “treatment” or “direct oral anticoagulants” or “vitamin K antagonists,” without language or date restrictions. Also, we manually reviewed references cited in the retrieved articles. This meta-analysis of data was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. The selection of papers to include followed a 3-step methodology: (1) reading the title and evaluation of its relevance, (2) reading the abstract, and (3) reading the full text.
Articles were considered for inclusion if they described comparison of DOACs and VKAs for LVT treatment and assessed LVT resolution and recorded occurrence of SE and stroke. Observational studies were eligible for inclusion. Exclusion criteria included duplicate publications and studies published only in abstract format.
A total of 269 studies were identified. Of these, 262 were excluded after title and abstract evaluation indicated they did not fulfill the inclusion criteria. Two studies were excluded after full-text reading because, although they compared anticoagulant strategies, adequate data for analysis were not presented. 5,23 Figure 1 shows the selection diagram.

Diagram of study selection.
Study design and characteristics were collected from each included study. Data on age, sex, and the incidence of hypertension, diabetes mellitus, ischemic cardiomyopathy history, AF diagnosis, LV ejection fraction, and concomitant antiplatelet therapy were also collected when available.
The coprimary endpoints were (1) thrombus resolution and (2) SE or stroke events. The secondary endpoint was clinically relevant bleeding and all bleeding events. The definition of bleeding events varied between studies. Therefore, we employed 2 categories: (1) “clinically relevant bleeding events,” defined as BARC ≥2 (for Bleeding Academic Research Consortium) bleedings or bleeding events requiring cessation in anticoagulation or with need for hospitalization; and (2) all bleeding events. Variable definition by study is shown in Table 1.
Outcomes Definition by Study Included in the Meta-Analysis.

BARC, Bleeding Academic Research Consortium.
Risk of Bias Assessment
We assessed the risk of bias in the studies, following the Newcastle-Ottawa Scale. 32 Three quality parameters were evaluated: selection, comparability, and outcome. The maximum score for each study was 9 points; studies with a score of less than 5 were considered to have a high risk of bias.
Statistical Analysis
Continuous variables were expressed as mean ± SD or median and interquartile range (IQR), and categorical variables were expressed as frequencies or percentages. Pooled odds ratios (ORs) and 95% CIs were estimated based on a random-effects meta-analysis and were obtained from the pooled adjusted ORs of the primary studies. The I 2 statistic was used to assess statistical heterogeneity across studies (moderate heterogeneity was considered present for values between 30% and 60%). Sensitivity analysis for all outcomes was conducted excluding data from the study with the highest risk of bias. Statistical analyses were performed using RevMan Review Manager, version 5.2 (Nordic Cochrane Centre).
Results
Studies Included and Baseline Characteristics
Five studies were included with a total of 828 patients. The baseline characteristics of the patients stratified by study are shown in Table 2. A total of 284 patients (34%) were treated with DOACs, and 544 (66%) were treated with VKAs. The mean age of the study patients was similar (DOAC, 65 ± 15 years vs VKA, 65 ± 14 years). Sixty-four patients (8%) switched treatment between treatment groups, and the analysis was conducted so that an individual event was counted for each group according to the patient’s treatment at the time of the event. Among DOAC-treated patients, 65% received apixaban, 30% rivaroxaban, 4% dabigatran, and 1% edoxaban. All the studies had observational designs. 18 -22 Table 3 summarizes the studies.
Baseline Population Characteristics by Study.

HTN, hypertension; Hyperlip., hyperlipidemia; DM, diabetes mellitus; DVT/PE, deep venous thromboembolism/pulmonary embolism; AF, atrial fibrillation; LVEF, left ventricular ejection fraction; CM, cardiomyopathy; AAS, acetylsalicylic acid; DOAC, direct oral anticoagulants; VKA, vitamin-K antagonists; N.A., not applicable.
Study Designs and Main Characteristics.

DOAC, direct oral anticoagulants; VKA, vitamin-K antagonists; SE, systemic embolism; USA, United States of America; UK, United Kingdom; OR, odds ratio; IQ, interquartile range; HR, hazard ratio; RED VELVT, Retrospective Evaluation of DOACs and Vascular Endpoints of Left Ventricular Thrombi.
Risk of Bias
The quality assessment for each study is presented in the Newcastle-Ottawa Scale summary (Figure 2). The study by Robinson et al was judged to have the highest risk of bias (7 points). Therefore, sensitivity analysis for all outcomes was conducted excluding that study from the analysis.
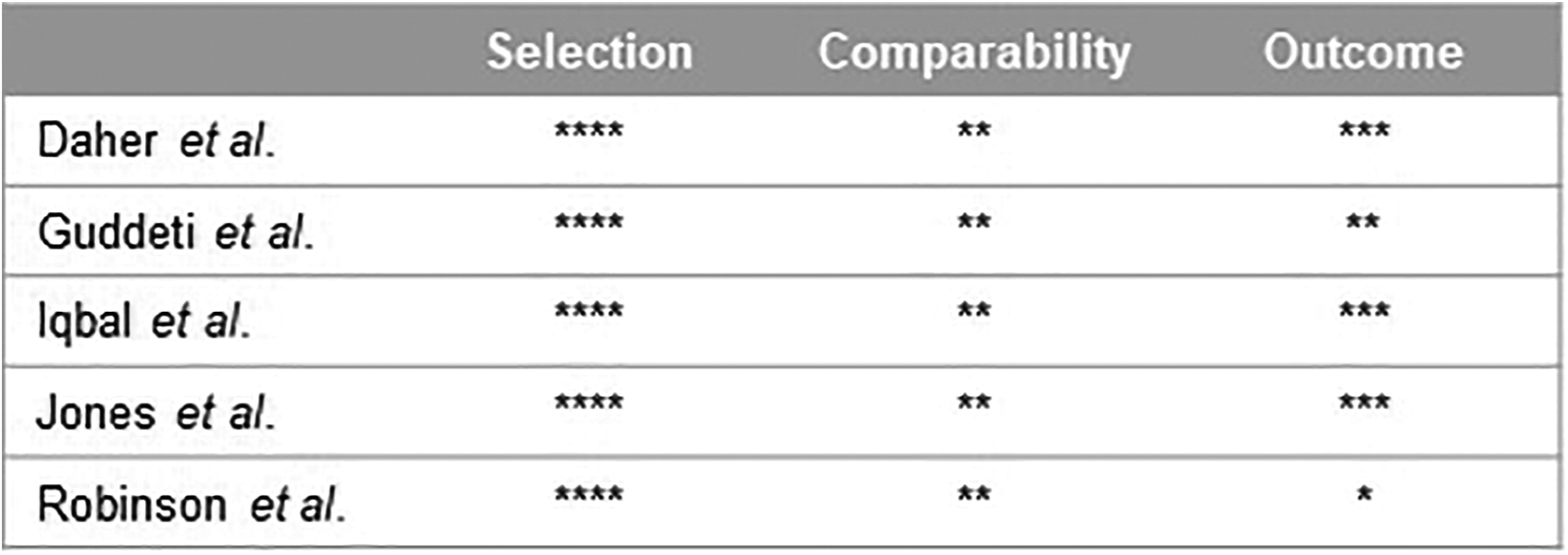
Newcastle-Ottawa scale summary. *Represents one point for each topic.
Thrombus Resolution
All studies reported the LVT resolution, although the timing of echocardiographic evaluation varied, and follow-up echocardiography was not performed for every patient. The pooled analysis comprised 819 patients with a similar proportion of thrombus resolution for patients taking DOACs versus those with VKA treatment (pooled OR, 0.91, 95% CI, 0.47-1.75; I 2= 63%; P = .78; Figure 3). In a sensitivity analysis, including the 3 studies that reported the total number of patients with follow-up echocardiography (n = 235), 19,21,22 there was also no inter-group difference in thrombus resolution (pooled OR, 1.20, 95% CI, 0.47-3.10; I 2 = 59%; P = .70; Figure 4).

Pooled analysis of thrombus resolution comparing patients receiving DOACs with patients receiving VKAs. Numbers displayed represent ORs with 95% CIs. DOAC, direct oral anticoagulant; OR, odds ratio; VKA, vitamin K antagonist. * Please note that for easy of understanding, VKA is represented “left” and DOAC is presented “right” in the forest plot.

Sensitivity analysis of thrombus resolution comparing patients receiving DOACs with patients receiving VKAs. Numbers displayed represent ORs with 95% CIs. DOAC, direct oral anticoagulant; OR, odds ratio; VKA, vitamin K antagonist. * Please note that for easy of understanding, VKA is represented “left” and DOAC is presented “right” in the forest plot.
Systemic Embolism
All included studies reported SE or stroke; 828 patients and 45 pooled events. There was a similar incidence of SE or stroke during follow-up (pooled OR, 1.59; 95% CI, 0.85-2.97; I 2 = 0%; P = .14; Figure 5). Sensitivity analysis (removal of data from Robinson et al) showed similar results (pooled OR, 0.80; 95% CI, 0.24-2.63; I 2 = 0%; P = .72). A subgroup analysis that focused only on stroke (284 patients and 7 pooled events), also revealed no difference between groups (pooled OR, 0.64; 95% CI, 0.13-3.18; I 2 = 0%; P = .59; Figure 6).

Pooled analysis of systemic embolism or stroke comparing patients receiving DOACs with patients receiving VKAs. Numbers represent ORs with 95% CIs. DOAC, direct oral anticoagulant; OR, odds ratio; VKA, vitamin K antagonist.

Pooled analysis of stroke comparing patients receiving DOACs with patients receiving VKAs. Numbers represent ORs with 95% CIs. DOAC, direct oral anticoagulant; OR, odds ratio; VKA, vitamin K antagonist.
Bleeding Events
Bleeding events were reported in 4 studies, involving a total of 769 patients (with 267 [35%] treated with DOACs) and 66 pooled events. In an analysis for clinically relevant bleedings (36 pooled events), the anticoagulation strategy did not differ in the odds for bleeding (pooled OR, 0.66; 95% CI, 0.31-1.40; I 2 = 0%; P = .28; Figure 7). Sensitivity analysis showed similar results (pooled OR, 0.64; 95% CI, 0.13-3.09; I 2 = 0%; P = .58). The risk of all bleeding events was halved in the DOAC group compared with the VKA group (pooled OR, 0.49; 95% CI, 0.26-0.90; I 2 = 0%; P = .02; Figure 8). Sensitivity analysis showed similar results (pooled OR, 0.35; 95% CI, 0.14-0.83; I 2 = 0%; P = .02).

Pooled analysis of relevant bleeding events comparing patients receiving DOACs with patients receiving VKAs. Numbers displayed represent ORs with 95% CIs. DOAC, direct oral anticoagulant; OR, odds ratio; VKA, vitamin K antagonist.

Pooled analysis of all bleeding events comparing patients receiving DOACs with patients receiving VKAs. Numbers displayed represent ORs with 95% CIs. DOAC, direct oral anticoagulant; OR, odds ratio; VKA, vitamin K antagonist.
Discussion
Our systematic review and meta-analysis suggest a similar proportion of complete LVT resolution in patients treated with DOACs and those treated with VKAs. The ischemic risk was also similar for both treatments. Clinically relevant bleeding events were similar between the 2 groups of patients; however, the risk for all bleeding events was lower with DOAC treatment.
The clinical prognosis of patients with LVT is considered poor with a high risk of major cardiovascular events and mortality. 23 Thrombus resolution with anticoagulation is a predictor of a good prognosis. Nevertheless, a large proportion of patients, as assessed by our pooled analysis, failed to achieve total thrombus resolution and hence remained at high risk of adverse events.
Although evidence is limited, the standard of care for LVT treatment has been VKAs. However, 3 important factors may significantly influence the success of VKA treatment. The first is treatment duration. A recent study reported a lower occurrence of major adverse cardiovascular events in patients receiving anticoagulants for at least 3 months versus patients receiving anticoagulants for a shorter time. The second factor is the intensification of the antithrombotic treatment. 23 In a subgroup of patients initially treated with DOACs without thrombus resolution at 3 months, treatment was changed to VKA anticoagulation with a target INR of 3-4; these patients subsequently obtained resolution of their thrombus. This intensification of treatment may nevertheless be compromised by more frequent bleeding complications. The third, and perhaps most important factor in VKA therapy, is the time in therapeutic range (TTR). Clinical benefit is associated with high TTR 33 and, conversely, risk of bleeding is increased when TTR is low. 34 It is, therefore, clear that there is scope for improvement in VKA anticoagulation management of LVT.
The efficacy of DOACs for LVT resolution was initially described in case reports with varying regimens and durations of treatment. A recent case-report meta-analysis reported successful LVT resolution after 6 months of DOAC therapy varying from 81% to 100%, with different DOACs. 35 Other series reported a success of 83%, 83% and 100%, respectively. 29 -31 The incidence of SE or stroke was 2%, 0%, 0%, and 2% respectively. 29 -31,35 The reported proportion of bleeding events were 8%, 13%, 10% and 2%, respectively. 29 -31,35 However, it is possible that publication bias occurred because negative results might not have been reported.
Each individual study (except for Jones et al) included in our pooled analysis found no differences between the incidence of thrombus resolution for DOACs vs VKAs. 18 -22 Jones and colleagues reported greater and earlier LVT resolution in the DOAC group. 21 Two studies not included in the analysis showed divergent results. 5,23 Although it was not powered for direct comparison of anticoagulant strategies, one of these studies suggested a similar incidence of thrombus resolution, major adverse cardiovascular events, and bleeding events. 23 The other study showed thrombus resolution in all patients treated with DOACs versus 75% resolution in VKA-treated patients. 5 In acute MI, LVTs can be caused by blood stasis and endocardial changes, or both. 36 DOACs were first approved for prevention of embolism in AF, where thrombosis is primarily the result of stasis. Thus, the presence of endocardial damage may be less amenable to DOAC therapy. The same argument applies to VKAs.
Most studies used a target international normalized ratio (INR) of 2 to 3 in patients treated with VKAs. 19,21,22 TTR was reported only by Jones et al, 21 which revealed almost half (47%) of the patients in the VKA group had suboptimal INR control (TTR < 65%). As mentioned earlier, TTR is crucial in determining clinical efficacy and bleeding risk. Another issue that could influence anticoagulation response is the concomitant use of antiplatelet therapy. However, this was similar in both treatment groups and therefore probably not responsible for any difference in efficacy.
In AF, studies have consistently shown that not only was there a similar or even reduced bleeding incidence with DOACs versus VKAs. Furthermore, patients experiencing major bleeding with DOACs were also shown to have a more favorable outcome than those receiving VKA treatment. 37 -40 Most patients with an LVT present to their clinician after an acute MI. The recent evidence of DOAC use after percutaneous coronary intervention in combination with a P2Y12 inhibitor has also revealed a noninferior or even superior safety profile compared with VKA-based regimens in patients with AF, and this is without increasing the risk of ischemic or embolic events. 41 -44 For LVT management in patients with concomitant acute MI, there is certainly a need for an individualized approach that considers anticoagulation duration, intensification regimes, the need for antiplatelet therapy, and bleeding risk.
Although DOACs confer several advantages over VKAs, the off-label use of DOACs may be problematic. There is evidence that DOACs may not be equivalent to VKAs; for example, for the thromboprophylaxis of mechanical heart valves 45 or for patients with antiphospholipid syndrome. 46 In contrast, encouraging evidence has emerged for the use of DOACs in conditions where they were previously contraindicated, such as rheumatic mitral stenosis and late-stage chronic renal failure. 47,48
Limitations
All the studies included in this analysis had a nonrandomized observational design and are thereby prone to bias. For example, the off-label use of DOAC may result in selection bias; i.e. the selection of a specific type of patient—one with less probability for adverse outcomes. Follow-up time for thrombus resolution and embolic events varied considerably between studies. Therefore, comparison is compromised. Only one study reported TTR. Consequently, we have no idea of TTR in the majority of patients; the greater the TTR, the greater the likelihood of clinical success. Not all the patients included had a follow-up echocardiogram. Patients without imaging re-evaluation in the studies by Robinson et al and Guddeti et al were included as if the thrombus had not resolved. Therefore, the real proportion of thrombus resolution might have been higher than the values used in our analysis. Because we were unable to determine how many patients had undergone re-evaluation echocardiography within each treatment group, we decided that our designation was the best way to conduct the analysis. However, the results might lead to bias in the follow-up, because patients receiving off-label DOACs would not be denied a follow-up echocardiogram. This possibly accounts for the high heterogeneity for this outcome. The criteria for reporting bleeding events varied (see Table 1). Finally, the included studies employed different DOACs and so the results should be interpreted with caution because efficacy may be agent dependent.
Future Directions
Clearly, randomized controlled trials (RCTs) are needed to remove potential bias that may have been present in the observational studies. In fact, several RCTs are underway. A multicenter randomized trial (EARLY-MYO-LVT) is currently underway to investigate the efficacy and safety of rivaroxaban (15 mg daily) vs warfarin (INR, 2-2.5) plus antiplatelet therapy for the prevention of LVT after ST-segment elevation acute MI. 49 Similarly, 2 single-center randomized trials comparing apixaban (5 or 2.5 mg twice a day) with warfarin (INR, 2-3) regarding the size reduction or resolution of LVTs are also under way. 50,51 These RCTs should have well-defined follow-up times with appropriate echocardiography imagine and should report TTR for warfarin-treated patients. Furthermore, cost-effectiveness should also be determined if DOACs are to become standard therapy for LVT.
Conclusion
The results of this systematic review and meta-analysis suggest that DOAC use was as effective as VKA use for the treatment of patients with LVT, with a similar risk of systemic embolism/stroke and clinically relevant bleeding. Nonetheless, the limitations of the observational studies revealed by our meta-analysis emphasize the need for RCTs designed to determine the best anticoagulant strategy for LVT treatment.
Footnotes
Authors’ Note
All authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Author Contributions
CS and RT designed the work, made the acquisition, analysis, and interpretation of the data and draft of the work. JL, DC, and LP designed the work and revised it. MC and LN revised the work. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
