Abstract
Despite the emerging prevalence of left ventricular (LV) thrombus in dilated cardiomyopathy (DCM), clinical characteristics, management, and disease prognosis are poorly studied. We aim to assess the efficacy/safety profile of direct oral anticoagulants (DOACs) compared to warfarin by evaluating thrombus evolution, risk for stroke and systemic embolism (SSE), heart failure (HF) rehospitalization, all-cause mortality, and major adverse cardiovascular events (MACEs), and determine the impact of thrombus evolution on adverse events. We performed a historical cohort study of patients with a primary diagnosis of DCM and LV thrombus. Relationships between anticoagulants and thrombus resolution were analyzed with the Kaplan-Meier method and Cox regression. Associations between longitudinal thrombus evolution and adverse event hazard were measured with joint modeling. Among 122 patients included, 58.0% were prescribed warfarin, and 42.0% DOACs. Complete thrombus resolution at 90-day-after-index and 180-day-after-index was observed in 93 and 111 patients, with no difference in cumulative resolution between DOACs and warfarin. During a median follow-up of 12.5 months, MACE, all-cause death, SSE, and HF rehospitalization occurred in 42.6%, 27.9%, 4.1%, and 13.9% of patients, comparable in warfarin and DOACs groups. Thrombus persistence was associated with a higher risk of HF rehospitalization. Thrombus progression was associated with poor prognosis, with per unit increment in square-root-transformed thrombus-area resulting in a 1.0691-fold increase in MACE risk and a 1.0546-fold increase in death risk. This study suggests that in DCM patients with LV thrombus, DOACs were comparable to warfarin in thrombus resolution and safety profile. Thrombus persistence or progression was associated with an increased risk of HF rehospitalization, MACE, and mortality.
Introduction
Heart failure (HF) is increasing in prevalence and has become the most common etiology of left ventricular (LV) thrombus in the contemporary era, transcending the hazard trend of myocardial infarction.1,2 Characterized by cavity enlargement and contractile function impairment, dilated cardiomyopathy (DCM) accounted for a large proportion of patients presenting with HF and LV thrombus.
Ventricular wall dyskinesia, endothelial injury, hypercoagulable state, and immobility state in DCM potentiate the activation of the coagulation system and triggered fibrin aggregation and intracardiac thrombus formation. Patients with DCM are vulnerable to LV thrombus even in sinus rhythm. 3 Contemporary epidemiological studies suggest that the incidence of LV thrombus in DCM patients may be up to 36% to 44% when detected with optimal imaging modalities.3,4 Despite adequate anticoagulation therapy, LV thrombus is one of the important causes of thromboembolic events in patients with DCM, causing stroke and noncentral systemic embolism, and is closely related to prognosis,3,5 yet the extant guidelines and consensus failed to put forward a uniformed recommendation for the treatment of LV thrombus in the setting of DCM. In recent years, direct oral anticoagulants (DOACs) are being increasingly used for their oral bioavailability, predictable pharmacokinetics, faster onset, ease of administration, and better patient adherence in nonvalvular atrial fibrillation and venous thromboembolism, with the off-label extension to patients with LV thrombus. Due to the lack of evidence, the efficacy and safety profile of DOACs therapy in LV thrombus in the setting of DCM remains largely unknown. 6 What's more, the incidence of LV thrombus, clinical characteristics, subsequent thromboembolic events, and disease prognosis of DCM with LV thrombus are also unclear and require examination. 6
We aimed to characterize patients with LV thrombus in the context of DCM, to quantify the effect of DOACs on LV thrombus evolution compared with warfarin, and to investigate the safety profile of DOACs by assessing the association of anticoagulants with adverse clinical events; Additionally, to determine the impact of thrombus evolution on adverse events in DCM patients.
Methods
Study Design and Population
This is a historical cohort study. From June 2011 to February 2023, all patients aged over 14 years with a primary diagnosis of DCM and LV thrombus in Heart Failure Center, Fuwai Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College (CAMS&PUMC), Beijing, China, were included. Patients who developed cardiomyopathy due to ischemic etiology or severe loading abnormality, patients with thrombus limited to right cavities or left atrium, or patients with baseline thrombus size description absent were not eligible for inclusion.
Thrombus Evaluation
Transthoracic echocardiography (TTE) is the standard imaging technique for LV thrombus detection. 6 Ultrasound-enhancing agents during echocardiography and late gadolinium enhancement (LGE) imaging on cardiac magnetic resonance (CMR) could improve the sensitivity. So, for patients diagnosed with LV thrombus, baseline and follow-up TTE, CMR, and ultrasound contrast reports were screened. All imaging reports were confirmed by 2 independent experts, and discrepancies were solved by consensus.
LV thrombus was defined as a discrete mass within the LV cavity with margins distinct from the endocardium and present on 2 or more views. 7 The sizes of the thrombus were measured as the longest diameter (mm) and area (mm2). Mobility and location of LV thrombus were also defined, LV thrombus was considered mobile if thrombus had a pedunculated nonmural component.
Baseline Data Collection
All patient baseline characteristics including demographics, comorbidities, smoking and alcohol state, disease course of DCM, and past medical history of ischemic events including history of stroke, transient ischemic attack (TIA), and noncentral nervous system thromboembolism were collected. Type (vitamin K antagonist [VKA], DOAC) and timing of anticoagulant treatment, detailed data on laboratory investigations, and imaging data were also collected. The medical prescription was collected from medical reports. Comorbidities (eg, diabetes mellitus, hypertension, atrial fibrillation, and atrial flutter) were diagnosed according to the World Health Organization (WHO) International Classification of Disease (ICD). Imaging data included the morphologic characteristics, number, location, and mobility of thrombi, LV ejection fraction (LVEF), left ventricle end-diastolic diameter (LVEDD), left atrial diameter (LAD), LV posterior wall thickness (LVPWT), interventricular septal thickness (IVST), and LV aneurysms.
Outcomes
Predefined thrombus outcomes included complete LV thrombus resolution and LV thrombus persistence. Complete LV thrombus resolution was defined by the disappearance of LV thrombus on all imaging views; LV thrombus persistence was defined as an increased thrombus dimension, a stable thrombus, or a partial thrombus resolution. Since all the anticoagulant agents were prescribed for 3 to 6 months according to extant guidelines on LV thrombus,8,9 and all patients were told to undergo an outpatient visit within 3 months, thrombus outcomes were determined at 90 days and 180 days separately after the index imaging.
Predefined clinical outcomes included: (1) all-cause death (including heart transplantation [HTx] and left ventricular assist devices [LVAD] requirement); (2) all-cause death (excluding HTx and LVAD); (3) HF rehospitalization; (4) stroke and systemic embolism (SSE) defined as the composite of stroke/TIA and systemic embolism; (5) major adverse cardiovascular events (MACE) defined as the composite of all-cause death (including HTx and LVAD), SSE, or HF rehospitalization.
Study Definitions
DCM is defined as LV or biventricular systolic dysfunction and dilatation in the absence of abnormal loading conditions or coronary artery disease (by coronary angiography or computed tomography), 10 systolic dysfunction is defined by abnormal LVEF measured using any modality.
Statistical Analysis
Continuous variables are expressed as means and standard deviations (normal distribution) or medians and interquartile ranges (skewed distribution), Student's t-test (Mann-Whitney test for non-Gaussian variables) were applied for comparison; categorical variables were presented as frequencies and percentages, chi-square test or Fisher's exact test was used for frequency comparisons. N-terminal pro-B-type natriuretic peptide (NT-proBNP) was transformed with the natural logarithm, given the right-skewed distribution of the data, and the thrombus area was square root transformed.
The cumulative incidence of complete LV thrombus resolution was estimated by the Kaplan-Meier method according to anticoagulant agents and compared with the log-rank test. Subject-specific longitudinal trajectories of thrombus size according to anticoagulant agents were depicted.
To measure the effect of thrombus evolution on adverse events, the cumulative incidence of adverse events was estimated by the Kaplan-Meier method according to LV thrombus resolution, and assessed with the log-rank test. Subject-specific longitudinal trajectories of thrombus size with and without events were depicted.
To measure the association between the longitudinal evolution of the thrombus area over time and the hazard of the event of interest unbiasedly, we performed joint modeling. Unlike classical models such as the linear mixed model for longitudinal data and the Cox proportional hazards model, joint models bring longitudinal and time-to-event data together into a single model so that one can infer the dependence and association between the longitudinal thrombus area and time-to-event.11–13 A mixed-effects model is used to analyze the longitudinal thrombus area (square root transformed) over time, which combines fixed and random effects. In our study, the fixed effects modeled the thrombus area trajectory during follow-up, the random effects were estimated to account for the within-subject correlations between repeated measurements at the subject level. To allow for flexibility in the specification of these profiles, we include natural cubic splines to adjust the nonlinear relationship of square-root-transformed thrombus area with follow-up year in the fixed effects parts of the longitudinal submodel. In the survival submodel, age, sex, and body mass index (BMI) were included as covariates. The joint models were fitted under a Bayesian approach using Markov chain Monte Carlo algorithms by R package JMbayes. 14
All statistical tests were two-tailed, and P < .05 was considered to be statistically significant unless otherwise stated. All analyses were performed using R 4.2.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Characteristics of the Patients and Thrombus Description
A total of 122 patients with DCM and LV thrombus diagnosis were included in the analysis from June 2011 to February 2023. The median length of follow-up was 12.5 months (interquartile range [IQR]: 3.6-40.9 months). Most LV thrombi are detected early (1.0 years [IQR: 0.14-3.88 years] delay) after DCM diagnosis.
The baseline characteristics of the patients with confirmed LV thrombus were described in Table 1. The mean age was 40 years and 79.5% (n = 97) were male. A total of 8.2% (n = 10), 23.0% (n = 28), and 13.9% (n = 17) of patients were complicated with atrial fibrillation or atrial flutter, hypertension, and diabetes, respectively. Additionally, 14.8% (n = 18) of patients had a history of stroke/TIA, and 4.1% (n = 5) of patients had a history of noncentral system thromboembolism. 104 (86.7%) patients were in New York Heart Association (NYHA) functional class III or IV, median NT-proBNP was 4083 (IQR: 1807-7240) pg/mL. Background guideline-directed medical therapy (GDMT) including beta-blockers, diuretics, renin-angiotensin II-aldosterone system (RAAS) inhibitors, and aldosterone antagonists were extensively prescribed.
Baseline Characteristics of the Study Participants According to Anticoagulant Therapy.
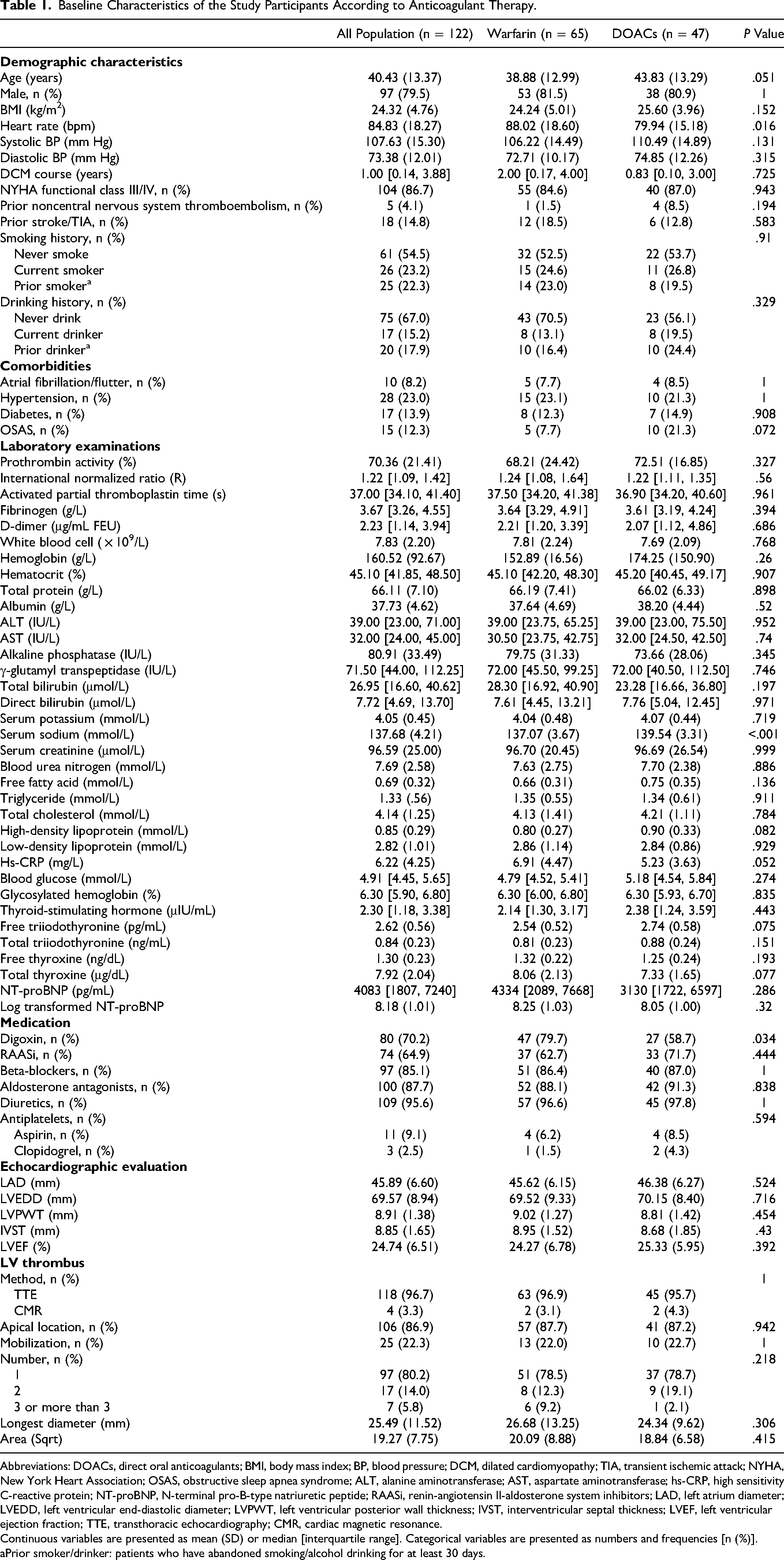
Abbreviations: DOACs, direct oral anticoagulants; BMI, body mass index; BP, blood pressure; DCM, dilated cardiomyopathy; TIA, transient ischemic attack; NYHA, New York Heart Association; OSAS, obstructive sleep apnea syndrome; ALT, alanine aminotransferase; AST, aspartate aminotransferase; hs-CRP, high sensitivity C-reactive protein; NT-proBNP, N-terminal pro-B-type natriuretic peptide; RAASi, renin-angiotensin II-aldosterone system inhibitors; LAD, left atrium diameter; LVEDD, left ventricular end-diastolic diameter; LVPWT, left ventricular posterior wall thickness; IVST, interventricular septal thickness; LVEF, left ventricular ejection fraction; TTE, transthoracic echocardiography; CMR, cardiac magnetic resonance.
Continuous variables are presented as mean (SD) or median [interquartile range]. Categorical variables are presented as numbers and frequencies [n (%)].
Prior smoker/drinker: patients who have abandoned smoking/alcohol drinking for at least 30 days.
The mean LVEF was 24.74%. The most frequent location of the thrombus was the left ventricular apex (86.9%, n = 106). LV thrombi were classified as mobile in 22.3% (n = 25) of patients. The mean baseline thrombus longest diameter (mm) and baseline thrombus surface area (square root transformed) were 25.49 and 19.27. In terms of imaging modalities, among LV thrombus reported on the baseline, 96.7% (n = 118) were observed by TTE, and 3.3% (n = 4) were observed by CMR (Table 1).
Thrombus Evolution
Complete LV thrombus resolution from the baseline to 90-day and 180-day after the baseline imaging was observed in 90.3% (n = 93) and 98.2% (n = 111) of the patients among whom follow-up data was available. Nineteen patients were censored regarding 90-day resolution due to the following reasons: (i) performed HTx surgery soon (< 90 days) after thrombus detection (n = 3); (ii) thrombectomy (n = 1); (iii) unclear thrombus status (unresolved thrombus in a follow-up test prior to day-90, and resolved in a follow-up test later than day-90; n = 15). Nine patients were censored regarding 180-day resolution due to the following reasons: (i) performed HTx surgery soon (< 90 days) after thrombus detection (n = 3); (ii) thrombectomy (n = 1); (iii) unclear thrombus status (unresolved thrombus in a follow-up test prior to day-180, and resolved in a follow-up test later than day-180; n = 5).
Time-dependent cumulative complete LV thrombus resolution is presented in Figure 1A and B showing a progressive decrease in thrombus longest diameter and thrombus area.

Time-dependent progressive left ventricular (LV) thrombus resolution: (A) thrombus area; (B) thrombus longest diameter
Efficacy and Safety of Anticoagulant Therapy
Most of the study population (58.0%) were treated with anticoagulation with VKA (n = 65), all of whom received warfarin; and 42.0% were treated with DOACs (n = 47), among which 89.4% (n = 42) were treated with rivaroxaban, 10.6% (n = 5) were treated with dabigatran. A detailed description of anticoagulant dosage was depicted in Supplemental Table 1. Oral anticoagulation was not prescribed in 10 patients, among which 5 patients were prescribed heparin, 2 patients were at an extremely high risk of bleeding (INR = 4.1 and 3.41, respectively), and 3 patients for unknown reasons.
Baseline characteristics of patients with VKA (warfarin) and DOACs prescription were described in Table 1. Two groups were comparable with regard to demographics, comorbid diseases, and laboratory and imaging examinations.
Descriptive plots for anticoagulant therapy and longitudinal outcomes were presented in Figures 2A and B and 3A and B, which depicted the Kaplan-Meier estimate of cumulative complete LV thrombus resolution to day-90 and day-180 for the 2 anticoagulant groups, and the subject-specific longitudinal thrombus size trajectories for patients with VKA and DOACs prescription, respectively. We observed that patients showed similar variability in longitudinal thombus profiles in VKA and DOACs groups. Kaplan-Meier survival analysis did not yield diverging curves, with the 90-day and 180-day thrombus resolution comparison between VKA and DOACs showing unpronounced risk stratification (log-rank test P = .76 and .19, respectively). A comparison between thrombus evolution in patients treated with VKA (warfarin) and DOACs was presented in Table 2, significant differences between DOACs and VKA in terms of LV thrombus evolution were not discerned. Regarding warfarin and DOACs on adverse events, warfarin was similar in risk of SSE as compared to DOACs (odds ratio [OR], 0.904; 95% confidence interval [CI], 0.145-5.635) (Table 2).

Cumulative incidence of left ventricular (LV) thrombus resolution according to anticoagulant therapy: (A) 90-day resolution; (B) 180-day resolution.

Subject-specific longitudinal trajectories of thrombus size for patients treated with VKA (warfarin) and DOACs (A) thrombus area; (B) thrombus longest diameter
Thrombus Evolution According to Anticoagulant Therapy.

Abbreviations: OR, odds ratio; CI, confidence interval; DOACs, direct oral anticoagulants; SSE, stroke, and systemic embolism; NA, OR, and 95% CI not applicable.
Characteristics are presented as n (%).
Nineteen censorships due to the following reasons: (i) performed HTx surgery soon (< 90 days) after thrombus detection (n = 3); (ii) thrombectomy (n = 1); (iii) unclear thrombus status (unresolved thrombus in a follow-up test prior to day-90, and resolved in a follow-up test later than day-90; n = 15).
Nine censorships due to the following reasons: (i) performed HTx surgery soon (< 90 days) after thrombus detection (n = 3); (ii) thrombectomy (n = 1); (iii) unclear thrombus status (unresolved thrombus in a follow-up test prior to day-180, and resolved in a follow-up test later than day-180; n = 5).
Thrombus Evolution and Clinical Outcomes
All-cause death (including HTx and LVAD) occurred in 27.9% (n = 34) of patients during the follow-up. In the nonsurvivor group, a median follow-up period of 3.1 months (IQR: 1.0-8.7 months) was found, whereas in the survivor group, the median follow-up period was 18.5 months (IQR: 7.2-50.0 months). During the follow-up, HF rehospitalization occurred in 13.9% (n = 17) of patients. There was a total of 5 SSE events, consisting of 4 episodes of stroke/TIA, which occurred 40-, 91-, 339-, and 3377-days following diagnosis of LV thrombus, and 1 episode of systemic embolism (pulmonary thromboembolism), which occurred 1638-days following LV thrombus diagnosis. MACE occurred in 42.6% (n = 52) of patients during follow-up.
Descriptive plots are presented in Supplemental Figures 1A to D and 2A to H, which depict the Kaplan-Meier estimate of time-to-event for patients with and without 90-day LV thrombus resolution, and the subject-specific longitudinal thrombus area and thrombus longest diameter profiles for patients with and without adverse event occurrence, respectively. We observed that in all groups, patients free from adverse events showed steadily low thrombus burden. Kaplan-Meier survival analysis for all-cause death (including HTx/LVAD), all-cause death (excluding HTx/LVAD), and MACE did not yield diverging curves, with the comparison showing unpronounced risk stratification (log-rank test P = .59, .60, and .13, respectively), whereas persistent thrombus group has lower HF rehospitalization events-free probability than the resolved thrombus group (log-rank test P = .0039).
The joint model for longitudinal and time-to-event data is a powerful method that assesses the 2 data types simultaneously so that one can infer the dependence and association between the longitudinal data and time-to-event. The results of the joint model suggest that the association between thrombus enlargement and risk of MACE is statistically significant (P = .001), with per unit increment in square-root-transformed thrombus area, resulting in a 1.0691-fold (95% CI, 1.0216-1.1769) increase in the risk. Similarly, a 1.0546-fold (95% CI, 1.0096-1.1156) increase in the all-cause death risk was observed for per unit increase in square-root-transformed thrombus area (P = .019) (Table 3).
Relationship Between Longitudinal Thrombus Area and Clinical Events.

Abbreviations: HR, hazard ratio; CI, confidence interval; HF, heart failure; MACE, major adverse cardiovascular events.
Discussion
The present historical cohort study is the largest to date describing the clinical profile, anticoagulant response, and clinical outcomes of DCM patients with LV thrombus, to our knowledge. Among the key findings were: (1) DOACs were comparable to VKA in terms of thrombus resolution; (2) DOACs were not associated with a higher risk of adverse events compared with warfarin; (3) Thrombus persistence was significantly associated with increased HF rehospitalization risk; (4) Increase in LV thrombus size by visits was associated with a higher risk of MACE and mortality in DCM patients.
Pathophysiology and Risk Factors of LV Thrombogenesis in DCM
Virchow's triad includes blood stasis, endothelial abnormality, and a hypercoagulability state. Being generally acknowledged as the cornerstone of thrombogenesis, Virchow's triad also contributes to LV thrombus formation in DCM. First, aberrant blood flow and stasis attributable to globally reduced ventricular systolic function and dilated cardiac cavity are the key factors of predisposition towards thrombogenesis. 15 In fact, decreased LVEF and enlarged LV have been proven to be the risk factors of LV thrombus formation in DCM. 16 What's more, defective endothelial function and hypercoagulability have been demonstrated in HF, 15 they could act as pathophysiological mediators that predispose to intracardiac thrombus formation in DCM-caused HF patients. In addition, myocardial inflammation is associated with a significant increase in platelet activation. 17 In DCM of autoimmune, toxic, and infectious causes (also known as inflammatory cardiomyopathies), inflammation plays the primary and persistent pathologic role, 18 and several studies have observed a high prevalence of intracardiac thrombus in inflammatory cardiomyopathy populations.19,20
To summarize, 3 factors of Virchow's triad of thrombogenesis collectively contributed to and are risks factors of LV thrombus formation in DCM: (1) intracardiac blood stasis attributable to globally reduced ventricular systolic function, (2) endothelium abnormalities, and (3) inflammation and hypercoagulable state.
Thrombus Burden in DCM
Attention has long been drawn to atrial thrombus in atrial fibrillation patients and LV thrombus in myocardial infarction patients. LV thrombus in exclusively DCM, which accounted for a considerable proportion, remained largely uncharacterized.5,6 As stated in the present study, the incidence of LV thrombus in DCM is relatively high, with 122 patients diagnosed within 11 years span in a single center, the incidence is comparable to, or even exceeds, the incidence of LV thrombus in HF of ischemic etiology or mixed etiology reported in some studies.21,22 Accounting for invisible thrombus formation in imaging, the actual thrombus burden in DCM could be even higher.
Anticoagulation for LV Thrombus
Clinical practice guidelines offer little discussion on anticoagulation for the treatment of LV thrombus in the setting of DCM as stated in a 2022 Scientific Statement from the American Heart Association (AHA). 6 Conventional VKA therapy is limited by several disadvantages, for example, various food/drug interactions, variable effectiveness due to genetic polymorphisms, and the need for regular anticoagulation monitoring, resulting in unsatisfying clinical compliance. Increasingly used in clinical, DOACs are advocated for similar or even better effectiveness, more rapid onset and offset of action, fewer drug–drug interactions, and convenience to use. There have been increasing retrospective studies and metaanalysis of cases investigating DOACs’ role in LV thrombus since 2020 in postmyocardial-infarction patients and ischemic HF patients. By and large, most of them concluded DOACs showed equal efficaciousness and safety compared to VKA in LV thrombus resolution,21,23–26 albeit several studies drew contrary or less-than-satisfying conclusions and questioned the safety of off-label use of DOACs in LV thrombus. For example, in the RED-VELVET study, 27 DOACs treatment was associated with a higher risk of SSE risk in LV thrombus; in other scenarios, 28 warfarin remains the standard of care. What's more, heterogeneity exists in pathophysiological mechanisms between thrombus in LV and atrium, 23 and LV thrombus in the setting of ischemic and nonischemic cardiomyopathy, which makes it hard to interchange experience. Given the discrepancies in existing evidence and vacancy of studies on LV thrombus in DCM, relevant investigations are warranted.
In the present study, we did not observe significant differences regarding thrombus resolution in DOACs and the warfarin group, and DOACs were associated with similar rates of adverse events compared with warfarin in patients with DCM. It seems that DOACs are a steadfast choice and are promising to be the first-line therapy of LV thrombus resolution in DCM patients.
Our study is the largest in size evaluating off-label therapeutic use of DOACs in LV thrombus in DCM, and it further adds to the growing body of evidence. Future prospective, randomized studies are warranted in the establishment of LV thrombus management.
Persistent Hypercoagulable State in DCM Patients Presenting With LV Thrombus
Most of the existing guidelines recommended at least 3 months of anticoagulation in the treatment of postmyocardial infarction patients with intracardiac thrombus. The scenario is slightly different in DCM. LV thrombus in DCM might be a marker of increased long-term thrombotic risk as suggested by previous studies.2,29 In the present study, the interval from LV thrombus diagnosis to systemic embolic events varies widely, ranging from several weeks to 9 years after LV thrombus resolution. Though our study was under-powered to attest the direct association of embolic events with LV thrombus, we inferred that LV thrombus might be a sign of a persistent hypercoagulable state in DCM patients, prolonging anticoagulation in patients with DCM and LV thrombus could be reasonable. This is consistent with the study by Lattuca et al, 26 in which authors observed a lower occurrence of MACE among patients who were anticoagulated for a longer duration than 3 months and hypothesized that a longer anticoagulation duration could lead to better prevention of embolic complications. However, the intensification of anticoagulation will be confronted by a bewildering array of complications. Given the vacancy of relevant studies, necessity and optimal anticoagulation duration are yet to be determined.
Thrombus Evolution and Clinical Outcomes
At 11 years, the HTx/LVAD-free survival rate in the present study population is 72.1% despite the extensive prescription of background GDMT prescription, the rate was much lower than the figure (86%) reported in a study on idiopathic DCM patients with evidence-based integrated therapeutic strategies in 1998 to 2007 (nearly one decade earlier than present study). 30 It is reasonable to extrapolate that LV thrombus and its evolution are associated with prognosis in DCM.
One of the major findings of the present study is that LV thrombus persistence and progression portend a poor prognosis in DCM patients. Kaplan-Meier estimate of time-to-event for patients with and without 90-day LV thrombus resolution yielded obvious diverging curves with regard to HF rehospitalization, persistent thrombus was associated with significantly higher HF rehospitalization risk, and the results of the joint models suggested that thrombus enlargement was associated with adverse events of MACE and mortality, both underpinning the adverse role LV thrombus persistence or progression played in DCM prognosis and importance of timely and sustained anticoagulation. To reverse poor prognosis brought about by thrombus persistence or enlargement, more efficient and practical anticoagulant regimens, and monitoring schemes should be developed, taking into account patients’ initiative and clinical evidence.
Limitation
Repeat imaging was performed at the discretion of the treating cardiologist, and the standardized interval of repeat imaging is not defined, which leads to information censorship of 90-day and 180-day thrombus outcomes, and the conclusion in our study might be biased. However, because all patients were told that he/she must undergo an outpatient visit within 3 months, data censorship only occurred in a few patients. In addition, this study is limited by its retrospective, nonrandomized approach, and single-center setting, it was not powered for a direct comparison between the 2 anticoagulant regimens. So, the efficacy equivalence of DOACs compared with VKA in LV thrombus resolution should be extrapolated cautiously. The optimal anticoagulant regimen should be tested in prospective, randomized trials. What's more, though the present study is among the largest size to date investigating longitudinal thrombus evolution and adverse events in DCM, a larger scale prospective study is needed to derive a more robust and confirmative association.
Conclusions
LV thrombus burden in DCM is relatively high, with 122 DCM patients diagnosed with LV thrombus in a single center over an 11-year span. With regards to anticoagulants, DOACs were comparable to VKA in terms of thrombus evolution, and were not evident to be associated with a higher risk of adverse events compared with warfarin. In terms of the association between LV thrombus evolution and outcomes, thrombus persistence was associated with an increased risk of HF rehospitalization, and thrombus progression was associated with an increased risk of MACE and mortality in DCM patients.
Highlights
LV thrombus is highly prevalent in DCM and is associated with poor prognosis, while characteristics and therapeutic regimens of LV thrombus in DCM patients are poorly determined due to the lack of relevant research.
The present study is the first systemic and largest to date research describing the clinical profile, anticoagulant response, and clinical outcomes of DCM patients with LV thrombus.
The present study reveals that DOACs are comparable to VKA in thrombus resolution in DCM, and it first demonstrates that LV thrombus persistence or progression portends a high risk of adverse events in DCM using joint modeling.
Randomized trials are needed to assess the efficacy/safety profile of DOACs in intracardiac thrombus resolution in DCM and explore the optimal anticoagulation duration.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231179683 - Supplemental material for Clinical Profile, Treatment, and Prognosis of Left Ventricular Thrombus in Dilated Cardiomyopathy
Supplemental material, sj-docx-1-cat-10.1177_10760296231179683 for Clinical Profile, Treatment, and Prognosis of Left Ventricular Thrombus in Dilated Cardiomyopathy by Liyan Huang, Xuemei Zhao, Jing Wang, Lin Liang, Pengchao Tian, Yuyi Chen, Mei Zhai, Yan Huang, Qiong Zhou, Anran Xin, Yuhui Zhang and Jian Zhang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
We are grateful to all members who contributed to the study.
Author Contributions
Conception and design of the study: Liyan Huang, Yuhui Zhang, Jian Zhang. Acquisition of data: Xuemei Zhao, Jing Wang, Lin Liang, Pengchao Tian, Yuyi Chen, Mei Zhai, Yan Huang, Qiong Zhou, Anran Xin, Liyan Huang. Analysis and interpretation of data: Liyan Huang. Drafting the article: Liyan Huang. Revising the manuscript critically for important intellectual content: Yuhui Zhang, Jian Zhang.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval was obtained from the institutional Ethics Committee of Fuwai Hospital (Approval number: 2014-501).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Projects in the National Science and Technology Pillar Program of the 13th Five-Year Plan Period, National Natural Science Foundation of China, the CAMS Innovation Fund for Medical Science (grant numbers 2017YFC1308300, 81873472, 2020-I2M-1-002).
Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
