Abstract
Proprotein convertase subtilisin/kexin type 9 (PCSK9) is an important and major player in the pathophysiology of hypercholesterolemia and atherosclerosis. Recently, PCSK9 has been implicated in the pathogenesis of inflammatory diseases. Whether PCSK9 is involved in idiopathic pulmonary arterial hypertension (IPAH) remains unclear. This study aimed to investigate the relationship between PCSK9 and IPAH. Serum PCSK9, interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), interleukin-1 β (IL-1β), and monocyte chemotactic protein-1 (MCP-1) were measured by enzyme linked immunosorbent assay. Transthoracic echocardiography was performed among 40 IPAH patients and 20 control subjects. Hemodynamic data were collected via right heart catheterization in patients with IPAH. Serum PCSK9, TNF-α, IL-6, IL-1β, and MCP-1 levels were significantly higher in IPAH patients than in control subjects (p < 0.001). Among enrolled IPAH patients, PCSK9 levels were higher in WHO-FC III/IV patients compared with those in WHO-FC I/II (p < 0.05), and were positively correlated with TNF-α, IL-6, MCP-1, N-Terminal pro-brain natriuretic peptide, pulmonary arterial systolic pressure (r = 0.653, p < 0.001), pulmonary arterial diastolic pressure (r = 0.466, p = 0.002), mean pulmonary arterial pressure (mPAP, r = 0.730, <0.001), pulmonary vascular resistance (r = 0.488, p = 0.001), and right ventricle diameter (r = 0.563, p < 0.001). In multiple regression analysis, mPAP was strongly associated with serum PCSK9 (β = 0.694, p < 0.001), independent of other variables. Receiver operating characteristic curve analysis showed the optimal cutoff value of serum PCSK9 concentration for predicting IPAH was 90.67 ng/ml, with a sensitivity of 90.0% and a specificity of 85.0%. In conclusion, IPAH patients had elevated serum PCSK9 levels which correlated the presence and severity of pulmonary hypertension. PCSK9 may be a novel potential therapeutic target.
Keywords
Introduction
Proprotein convertase subtilisin/kexin Type 9 (PCSK9) has been identified as an important player in the pathophysiology of hypercholesterolemia and atherosclerosis, and gained much attention due to its ability to degrade low-density lipoprotein cholesterol receptors (LDL-R) and subsequently raise low-density lipoprotein cholesterol (LDL-C) levels.1,2 Recently, accumulating data suggest that PCSK9 has pleiotropic effects beyond cholesterol metabolism, including adipogenesis modulation, 3 neurogenesis regulation during neuronal development, 4 as well as platelet activation, thrombosis, and myocardial infarct expansion. 5 Most interestingly, mounting evidence indicate PCSK9 is closely associated with inflammation and widely involved in inflammatory diseases, such as atherosclerosis, sepsis, psoriasis, rheumatoid arthritis, periodontitis, and Graves' ophthalmopathy.6–11 Proinflammatory factors such as tumor necrosis factor (TNF-α), lipopolysaccharide, and oxidized-LDL could upregulate PCSK9 expression, which itself positively regulated inflammation. 12 Additionally, plasma PCSK9 levels were positively and independently correlated with white blood cell count, lymphocyte count and neutrophil count, 13 as well as fibrinogen and high-sensitivity C-reactive protein levels in patients with stable coronary artery disease. 6 Plasma levels of pro-inflammatory cytokines TNF-α, interleukin (IL)-6, IL-8, and monocyte chemoattractant protein (MCP)-1 were significantly lower in septic shock patients with PCSK9 loss-of-function alleles compared with those with gain-of-function alleles. 7 Moreover, PCSK9 inhibitors or antagonists exerted anti-inflammatory effects, e.g., PCSK9 antibody alirocumab could attenuate arterial wall inflammation, 14 while AT04A vaccine against PCSK9 reduced systemic and vascular inflammation and atherosclerotic lesions in APOE*3Leiden.CETP mice. 15 Briefly, these studies suggest PCSK9 can act as a pro-inflammatory mediator.
Idiopathic pulmonary arterial hypertension (IPAH) is a progressive cardiopulmonary disease with high morbidity and mortality, and is characterized by vasculopathy in which vascular cell overgrowth and inflammation-driven structural changes play vital roles.16,17 In addition to increased perivascular immune cell accumulation and intravascular infiltration, circulating levels of certain cytokines and chemokines, such as IL-1β, IL-6, MCP-1, and TNF-α, were abnormally elevated.18–20 IL-6 was reported to induce proliferation of pulmonary artery smooth muscle cells, 21 and TNF-α played pivotal roles in endothelial cell apoptosis and smooth muscle cell proliferation in many experimental models of pulmonary hypertension, and its inhibition could prevent the pathological progression of this deadly disease. 22
In light of above findings, we hypothesized that PCSK9 was involved in the pathogenesis of IPAH, thus the purpose of this study was to first investigate among IPAH patients the serum levels of PCSK9 and its association with hemodynamic and structural parameters, and especially multiple inflammatory cytokines that have previously been identified to play important roles in the pathogeneses of pulmonary arterial hypertension (PAH).
Materials and methods
Study participants
Forty IPAH patients and 20 healthy controls were enrolled in this cross-sectional study, and IPAH patients were diagnosed from December 2019 to November 2020 during their visit to Ward 38 of our Hospital, according to the ESC/ERS guideline. 23 Patients with one or more of the following conditions were excluded: (1) WHO group 2–5 pulmonary hypertension; (2) previous targeted therapy; (3) hyperlipemia, including those taking lipid-lowering drugs or not; (4) diabetes mellitus, impaired fasting glucose or glucose tolerance; (5) other heart disease, such as coronary artery disease, acute heart failure, and sustained arrhythmia; (6) any other confirmed inflammatory diseases, such as pneumonia, arthritis, periodontal infection; (7) renal failure; and (8) hepatic failure.
The following clinical data and characteristics were obtained from electronic medical records: sex, age, height, body weight, echocardiographic parameters, medical history, and inspection reports. Body mass index (BMI) was calculated as weight/(height) 2 in kilogram per square meter. The protocol was performed in accordance with the principles of the Declaration of Helsinki and was approved by the local Research and Ethics Committee of our Hospital. Written informed consent was obtained from all participants.
Blood collection and biochemical measurements
Peripheral venous blood samples were collected into evacuated and promoting coagulating tubes after an overnight fasting. Individual samples were separated by centrifugation at 3000 r/min for 10 min at 4°C, and the serum was then frozen and stored in aliquots at –80°C until assayed. Serum PCSK9, TNF-α, IL-6, IL-1β, and MCP-1 were measured by enzyme linked immunosorbent assay (ELISA) according to the manufacturer’s instructions. Human PCSK9 ELISA kit was obtained from Abcam, and TNF-α, IL-6, IL-1β, and MCP-1 ELISA kits were purchased from Westang Biological Technology Co., Ltd. (Shanghai, China). Total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C, and triglyceride (TG) were assayed at the Central Clinical Laboratory of our Hospital.
Right heart catheterization
Right heart catheterization was performed on each patient by experienced cardiologists with a Swan–Ganz balloon-tipped catheter in the cardiac catheterization room. All subjects were examined in the supine position. Pulmonary arterial systolic pressure (PASP), pulmonary arterial diastolic pressure (PADP), mean pulmonary arterial pressure (mPAP), pulmonary artery wedge pressure (PAWP), and cardiac output (CO) were obtained during catheterization. Cardiac index (CI) was calculated by dividing CO by body surface area. Pulmonary vascular resistance (PVR) was calculated as (mPAP-PAWP)/CO. To ensure the accuracy of data, all subjects did not inhale oxygen during the examination.
Statistical analysis
Continuous variables were presented as means ± SD and categorical variables as percentages. Statistical differences between two groups were analyzed with unpaired Student’s t test and χ2 test for categorical variables. Relationships between variables were explored using Pearson’s coefficient. Multiple linear regression analysis was performed to assess the relative strength of the association. Receiver operating characteristic (ROC) curve analysis was used to evaluate the sensitivity, specificity, Youden index, and areas under the ROC curve (AUC) with 95% confidence interval (CI). A p value of <0.05 was considered statistically significant. All analyses were performed using SPSS 26.0 (SPSS Inc., Chicago, IL, USA) or GraphPad Prism 8 (GraphPad Software Inc., San Diego, CA, USA)
Results
Basic characteristics
A total of 60 subjects were included in this study, with 40 in IPAH group and 20 in control group. Their demographic, biochemical, hemodynamic, and echocardiographic data are summarized in Table 1. There were no statistically significant differences between the two groups in terms of mean age, sex, BMI, ejection fraction (EF), HDL-C, and TG. Obviously, right ventricle diameter (RVD), right ventricular anterior wall (RVAW), pulmonary artery trunk diameter (PAD), and N-Terminal pro-brain natriuretic peptide (NT-proBNP) were significantly higher, but tricuspid annular plane systolic excursion (TAPSE), TC, and LDL-C were lower in the IPAH group compared with the control group.
Characteristics of the subjects included in the study.
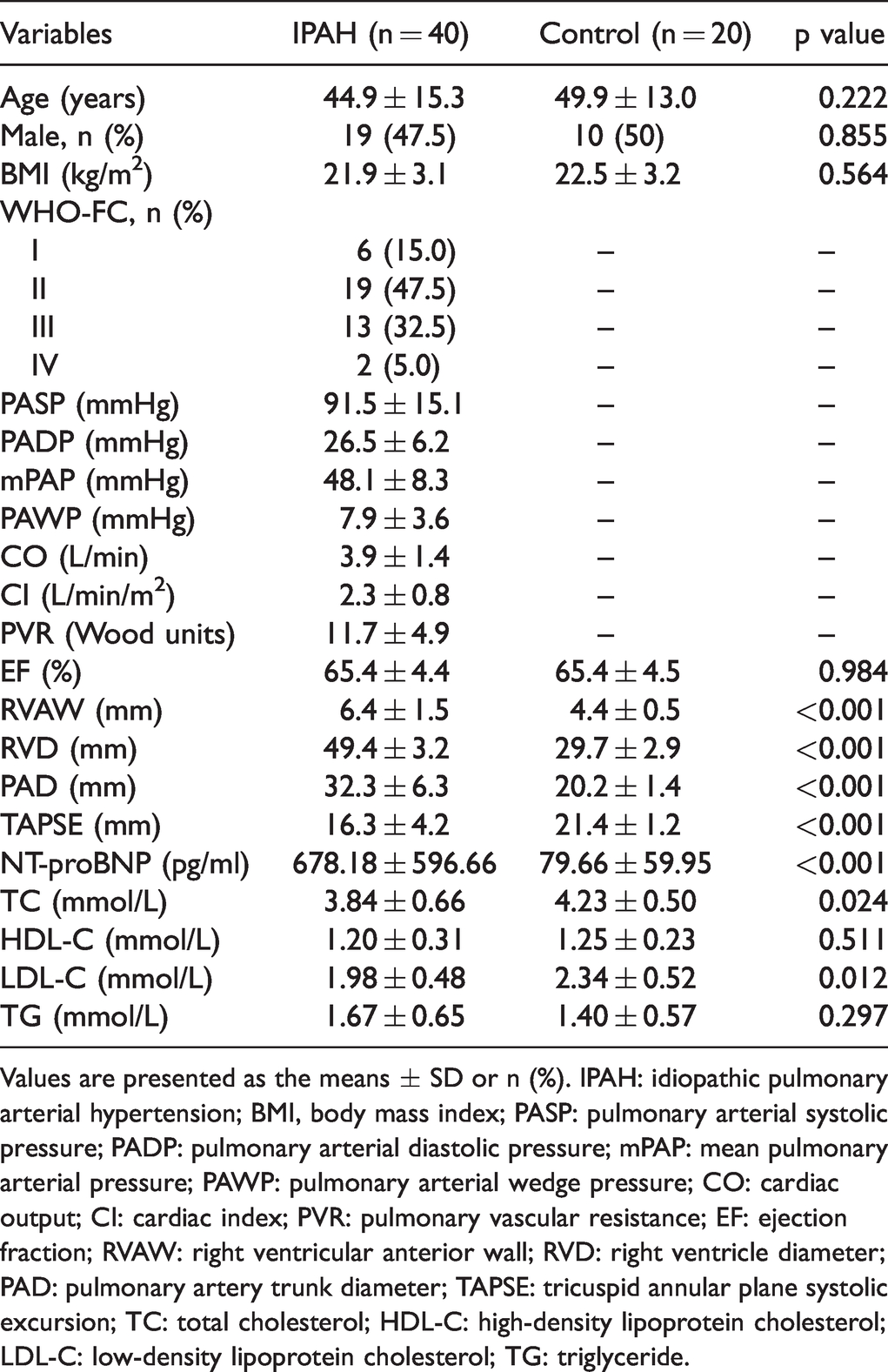
Values are presented as the means ± SD or n (%). IPAH: idiopathic pulmonary arterial hypertension; BMI, body mass index; PASP: pulmonary arterial systolic pressure; PADP: pulmonary arterial diastolic pressure; mPAP: mean pulmonary arterial pressure; PAWP: pulmonary arterial wedge pressure; CO: cardiac output; CI: cardiac index; PVR: pulmonary vascular resistance; EF: ejection fraction; RVAW: right ventricular anterior wall; RVD: right ventricle diameter; PAD: pulmonary artery trunk diameter; TAPSE: tricuspid annular plane systolic excursion; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TG: triglyceride.
Serum PCSK9 and cytokines levels between groups
As displayed in Table 2 and Fig. 1, serum PCSK9 levels were significantly higher in IPAH patients than in controls (126.87 ± 36.29 vs. 77.26 ± 19.39 ng/ml, p < 0.001). The levels of four inflammatory cytokines, namely, TNF-α, IL-6, IL-1β, and MCP-1, were also significantly elevated in IPAH compared with healthy control subjects (p < 0.001). Furthermore, we divided all patients into two groups (WHO-FC I/II group and WHO-FC III/IV group) according to cardiac function assessment. Serum PCSK9 levels were significantly increased in WHO-FC III/IV group compared to the WHO-FC I/II group (146.85 ± 44.10 vs. 114.88 ± 24.62 ng/ml, p < 0.05). However, although the average levels TNF-α, IL-6, IL-1β, and MCP-1 were slightly higher in the WHO-FC III/IV group, the differences between the two groups were not statistically significant (Fig. 2).
Serum PCSK9 and cytokine levels in IPAH patient versus healthy controls.
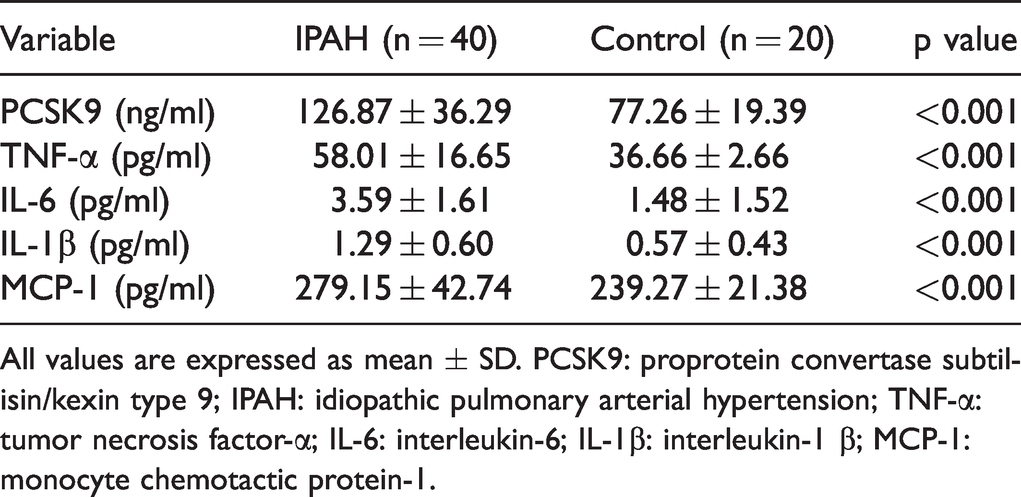
All values are expressed as mean ± SD. PCSK9: proprotein convertase subtilisin/kexin type 9; IPAH: idiopathic pulmonary arterial hypertension; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; IL-1β: interleukin-1 β; MCP-1: monocyte chemotactic protein-1.
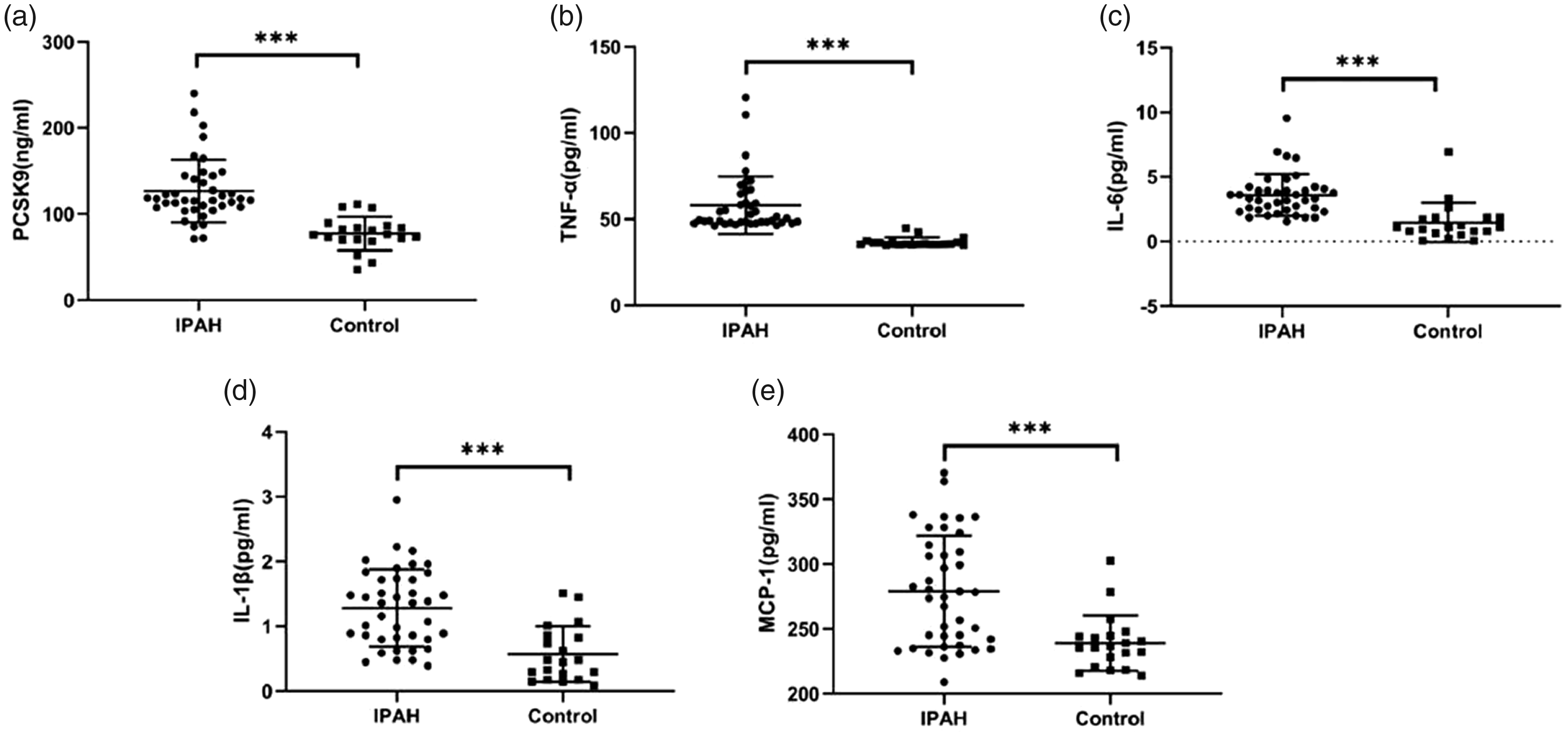
Serum PCSK9 (a), TNF-α (b), IL-6 (c), IL-1β (d), and MCP-1 (e) levels in control and IPAH group. ***p < 0.001, compared with control group. PCSK9: proprotein convertase subtilisin/kexin type 9; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; IL-1β: interleukin-1 β; m MCP-1: monocyte chemotactic protein-1; IPAH: idiopathic pulmonary arterial hypertension.
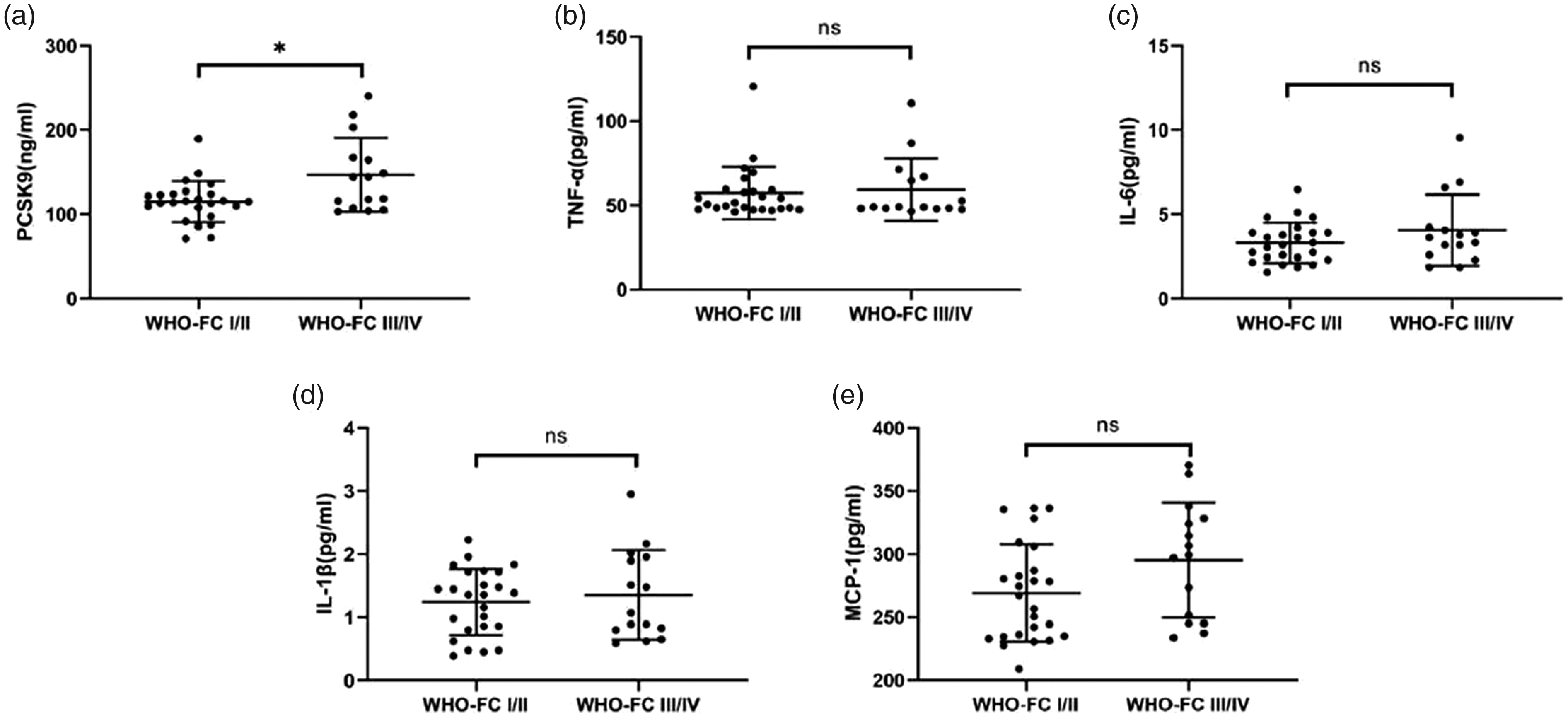
Serum PCSK9 (a), TNF-α (b), IL-6 (c), IL-1β (d), and MCP-1 (e) levels in WHO-FC I/II group and WHO-FC III/IV group. *p < 0.05, compared with WHO-FC I/II group. PCSK9: proprotein convertase subtilisin/kexin type 9; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; IL-1β: interleukin-1 β; MCP-1: monocyte chemotactic protein-1.
Correlations between various parameters
Pearson correlation results for each parameter are shown in Table 3, Fig. 3 and Supplementary Fig. 1. Serum PCSK9 levels were positively correlated with TNF-α (r = 0.327, p = 0.040), IL-6 (r = 0.355, p = 0.025), MCP-1 (r = 0.322 p = 0.043), NT-proBNP (r = 0.330, p = 0.037), PASP (r = 0.653, p < 0.001), PADP (r = 0.466, p = 0.002), mPAP (r = 0.730, p < 0.001), PVR (r = 0.488, p = 0.001), and RVD (r = 0.563, p < 0.001). However, there were no significant correlations between serum PCSK9 and BMI, PAWP, CO, CI, EF, RVAW, PAD, TAPSE, TC, HDL-C, LDL-C, and TG. None of the four cytokines (TNF-α, IL-6, IL-1β, and MCP-1) showed significant correlations with hemodynamic parameters (PASP, PADP, mPAP, PAWP, CO, CI, and PVR) and echocardiographic variables (EF, RVAW, RVD, PAD, and TAPSE).
Correlations of PCSK9, TNF-α, IL-6, IL-1β, and MCP-1 with various parameters in IPAH patients.
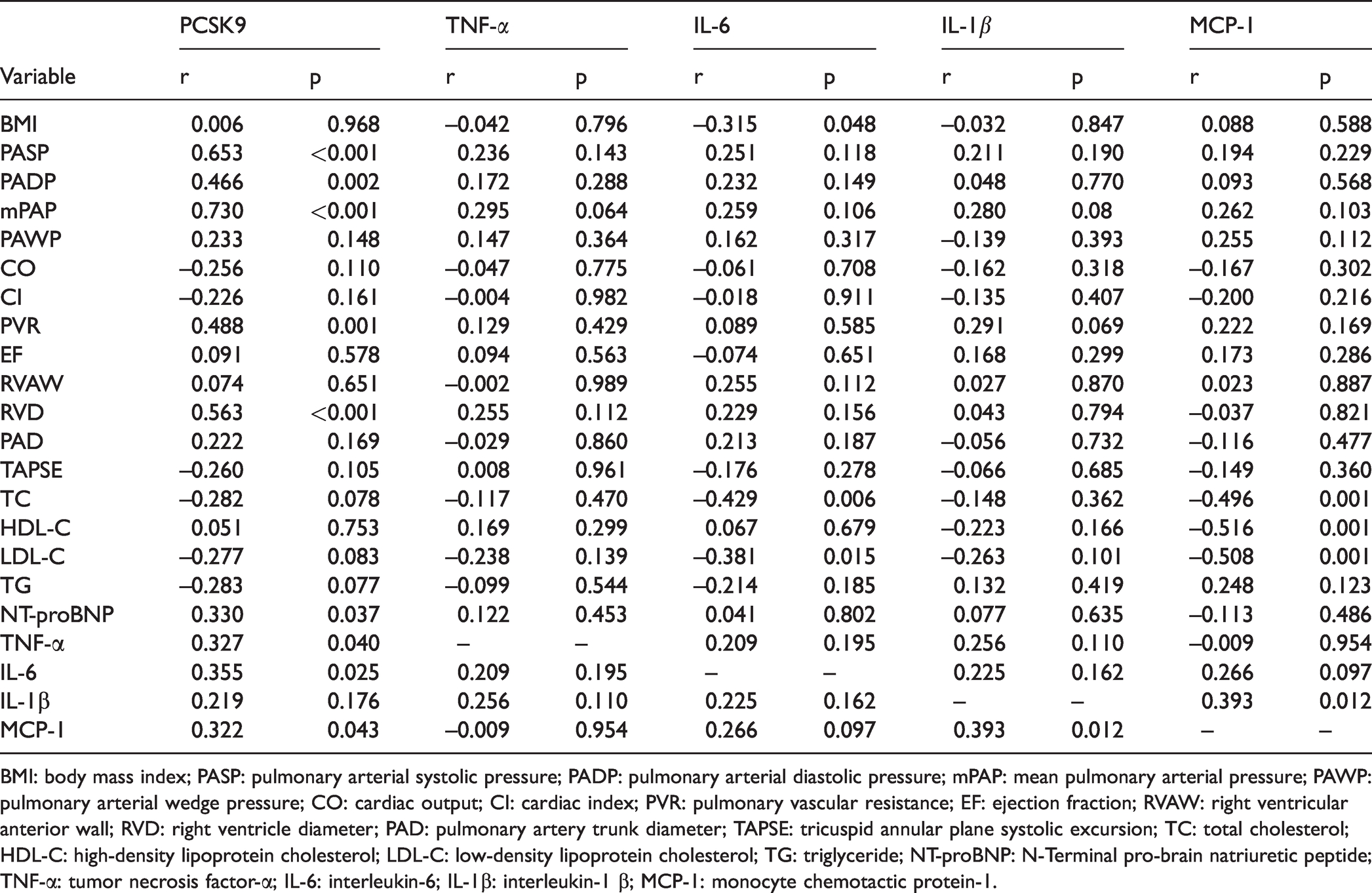
BMI: body mass index; PASP: pulmonary arterial systolic pressure; PADP: pulmonary arterial diastolic pressure; mPAP: mean pulmonary arterial pressure; PAWP: pulmonary arterial wedge pressure; CO: cardiac output; CI: cardiac index; PVR: pulmonary vascular resistance; EF: ejection fraction; RVAW: right ventricular anterior wall; RVD: right ventricle diameter; PAD: pulmonary artery trunk diameter; TAPSE: tricuspid annular plane systolic excursion; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TG: triglyceride; NT-proBNP: N-Terminal pro-brain natriuretic peptide; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; IL-1β: interleukin-1 β; MCP-1: monocyte chemotactic protein-1.
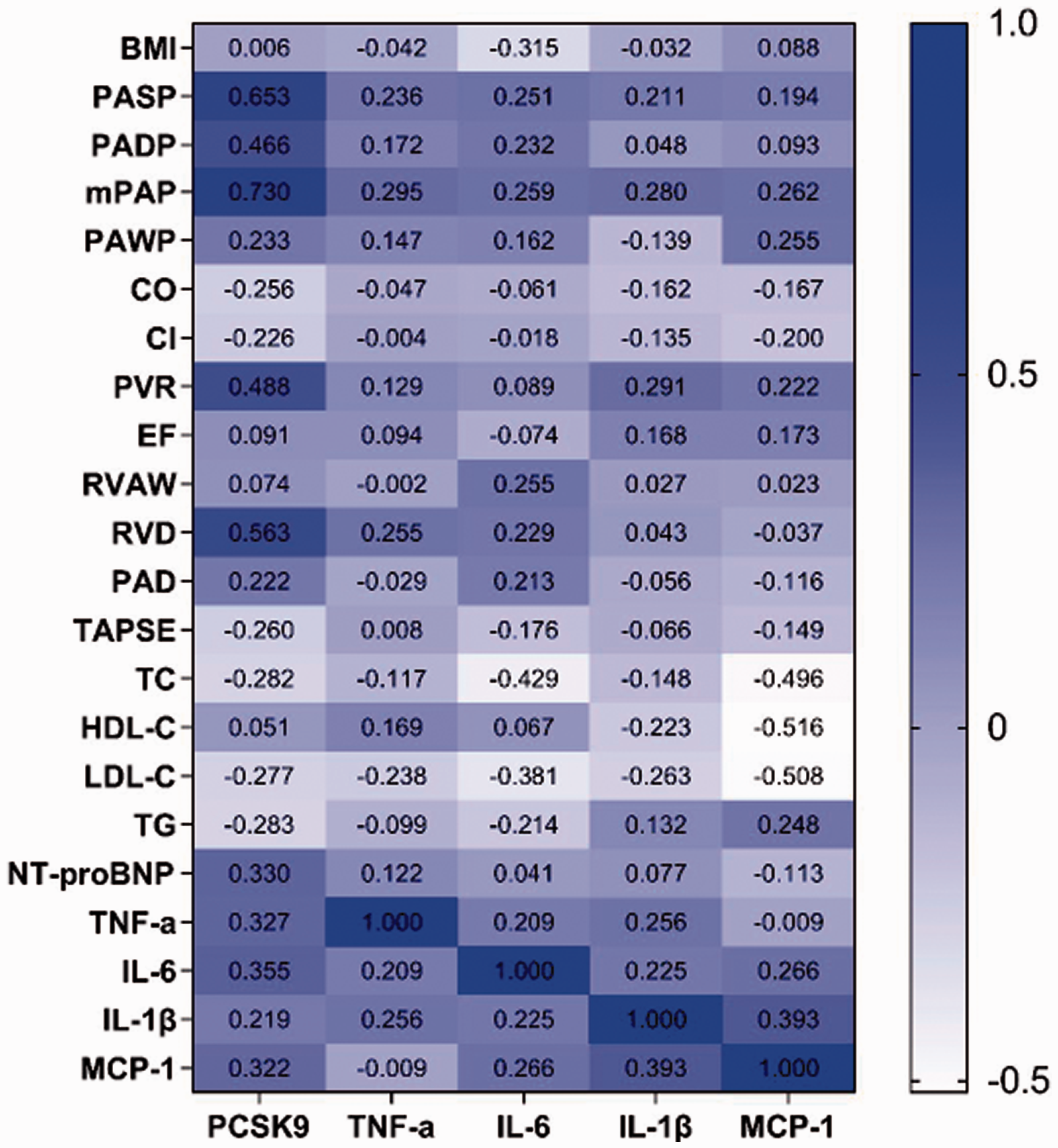
Correlations of PCSK9 and cytokines with various parameters. PCSK9: proprotein convertase subtilisin/kexin type 9.
Association of serum PCSK9 levels with IPAH
Linear regression analyses were utilized to explore the association between various parameters and IPAH (Table 4). Single-variable linear regression showed that TC, TG, and PCSK9 levels were significantly associated with decreased or increased risk of PAH. In further multiple linear regression analysis, mPAP were strongly associated with PCSK9 levels (β = 0.694, p < 0.001), independent of other variables, including TC, NT-proBNP, and IL-6.
Single-variable and multi-variable linear regression analyses of association between variables and mPAP.
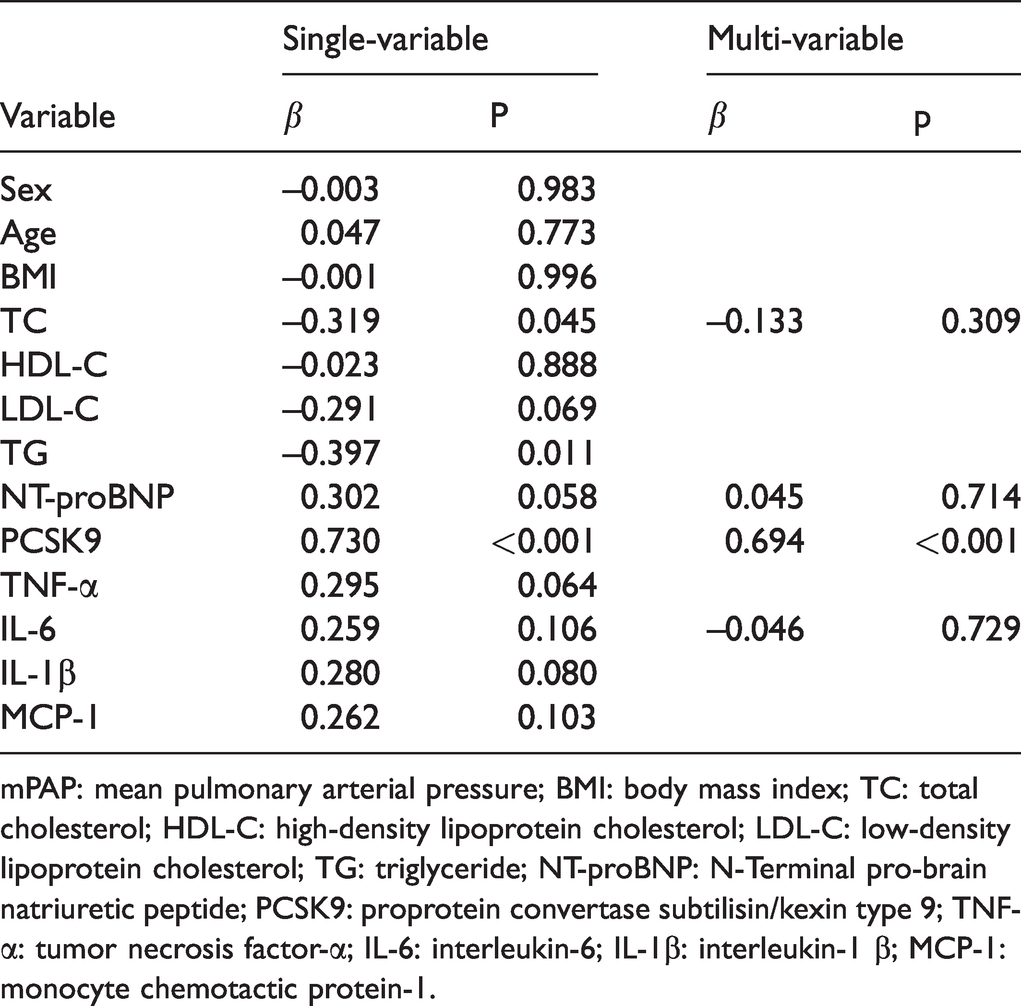
mPAP: mean pulmonary arterial pressure; BMI: body mass index; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TG: triglyceride; NT-proBNP: N-Terminal pro-brain natriuretic peptide; PCSK9: proprotein convertase subtilisin/kexin type 9; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; IL-1β: interleukin-1 β; MCP-1: monocyte chemotactic protein-1.
ROC curve analyses of serum PCSK9 and cytokines for predicting IPAH
As shown in Table 5 and Fig. 4, a PCSK9 level of 90.67ng/ml discriminated IPAH patients from healthy controls with a sensitivity of 90% and a specificity of 85% (AUC 0.9288, 95% CI 0.8652–0.9923, p < 0.001). The optimal cut-off value of IL-6 was 1.92 pg/ml with a sensitivity of 90.0%, a specificity of 85.0%, and an AUC of 0.9013 (95% CI 0.7962–1.000, p < 0.001). The optimal cut-off value of IL-1β was 0.770 pg/ml, with a sensitivity of 80.0%, a specificity of 70.0%, and an AUC of 0.84 (95% CI 0.7336–0.9464, p < 0.001). As for MCP-1, a cut-off value of 244.9 pg/ml harbored a sensitivity of 70.0% and a specificity of 80.0% with an AUC of 0.7856 (95% CI 0.6705–0.9008, p < 0.001). A NT-proBNP level of 206.5 pg/ml discriminated IPAH subjects from healthy controls with a sensitivity of 80% and a specificity of 100% (AUC 0.9138, 95% CI 0.8424–0.9851, p < 0.001).
Receiver operating characteristic curve analyses of serum PCSK9 and cytokines for predicting IPAH
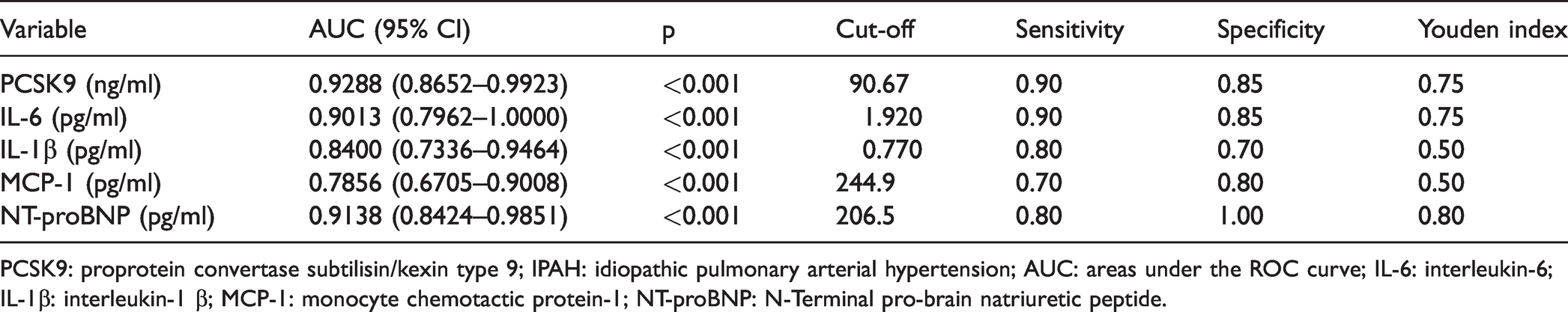
PCSK9: proprotein convertase subtilisin/kexin type 9; IPAH: idiopathic pulmonary arterial hypertension; AUC: areas under the ROC curve; IL-6: interleukin-6; IL-1β: interleukin-1 β; MCP-1: monocyte chemotactic protein-1; NT-proBNP: N-Terminal pro-brain natriuretic peptide.
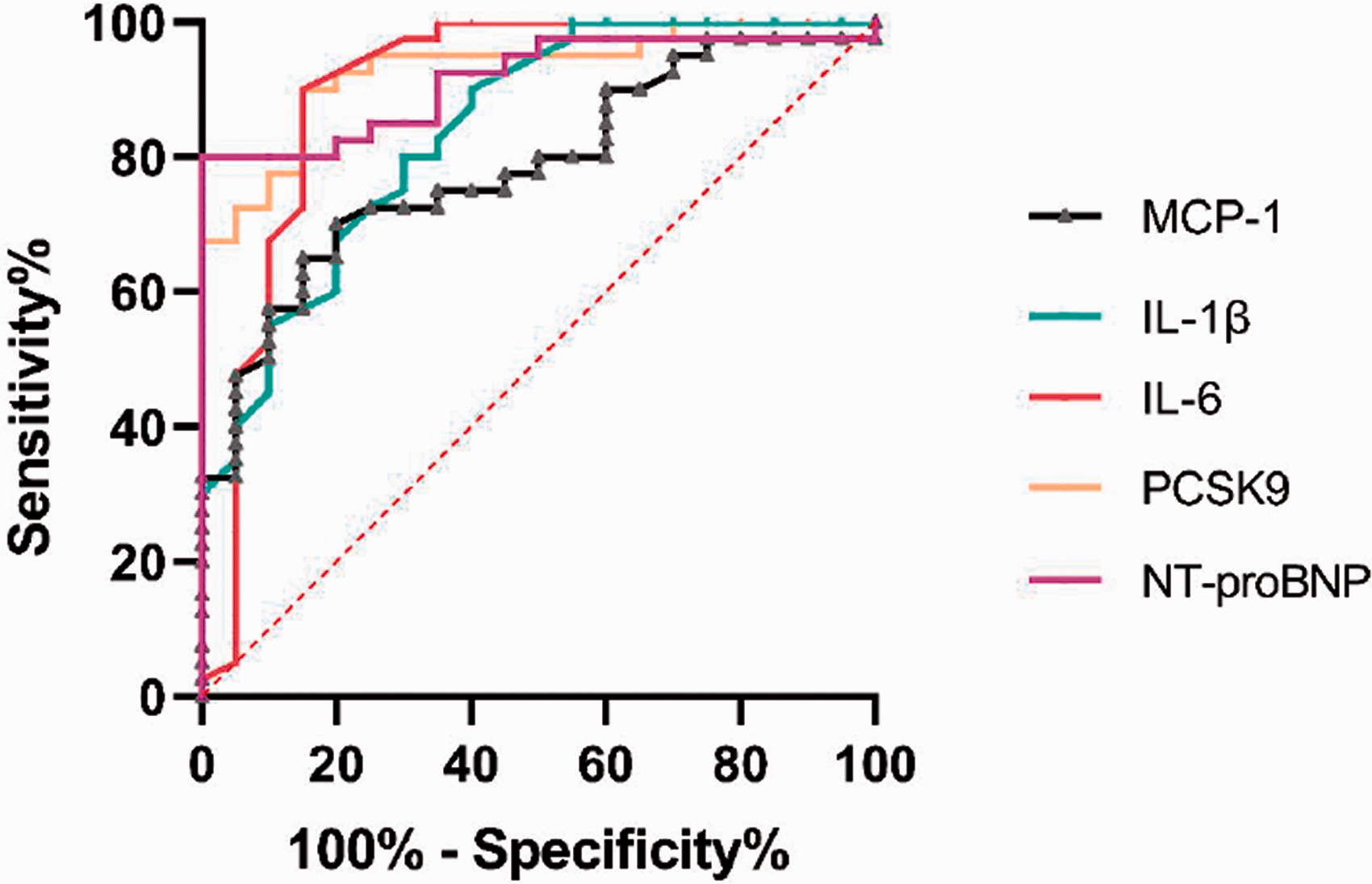
ROC analyses of serum PCSK9, cytokines, and NT-proBNP for predicting IPAH. ROC: receiver operating characteristic; PCSK9: proprotein convertase subtilisin/kexin type 9; NT-proBNP: N-Terminal pro-brain natriuretic peptide; IPAH: idiopathic pulmonary arterial hypertension; MCP-1: monocyte chemotactic protein-1; IL-1β: interleukin-1 β; IL-6: interleukin-6.
Discussion
To the best of our knowledge, this is the first study to demonstrate that serum PCSK9 levels were elevated in IPAH patients, and were positively correlated with inflammatory cytokines (TNF-α, IL-6, and MCP-1), cardiac function (NT-proBNP) as well as hemodynamic and echocardiographic parameters (PASP, PADP, mPAP, PVR, and RVD). Moreover, increased PCSK9 was a strong predictor for IPAH.
Substantial evidence supports that inflammation is a feature of PAH and inflammatory cytokines play a role in the development of IPAH. Consistent with previous studies,19,20 we observed significantly elevated levels of TNF-α, IL-6, IL-1β, and MCP-1. Interestingly, in our further analysis, we found no difference in cytokines levels between WHO-FC I/II and WHO-FCIII/IV patients and a lack of correlation between cytokine levels and hemodynamic and echocardiographic parameters, which was consistent with the results shown by other studies.19,24,25 Soon et al. 25 examined serum cytokines (TNF-α, IL-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12, IL-13, and interferon-γ) in 60 idiopathic and heritable PAH patients and did not find any correlation between cytokine levels and hemodynamic parameters (mPAP, CI, and PVR), implying that inflammatory cytokines, although involved in the pathogenesis of IPAH, do not reflect the entire mechanism.
Proprotein convertase subtilisin/kexin type-9 (PCSK9), the nine-member of proprotein convertases family, exerts a critical role in LDL-C metabolism, and several studies also found that PCSK9 was involved in inflammation,6,7,11 which was further confirmed by vivo and vitro studies.26–28 IPAH as an inflammatory disease, and it is not clear whether PCSK9 is increased in IPAH. Excitingly, our study showed the serum PCSK9 was significantly higher in IPAH subjects than in control subjects, and further analysis revealed that serum PCSK9 level in WHO-FC III/IV patients was higher than that in WHO-FC I/II group. Our results demonstrated that increased serum PCSK9 could discriminate IPAH from healthy controls with high sensitivity and strongly correlated with pulmonary hemodynamics, indicating PCSK9 might be a severity biomarker for PAH. It is well known that PCSK9 is mainly expressed in the liver but is also highly expressed in many other tissues and cells, such as colon, kidney, endothelial cells, smooth muscle cells, and macrophages. 29 However, the main source of elevated serum PCSK9 in IPAH patients in our study was uncertain and further studies are needed to clarify this.
Theoretically, PCSK9 elevation could induce LDL-R degradation, resulting in a decrease in the cholesterol clearance from the circulation and consequently an increase in LDL-C concentrations. However, in our study, serum LDL-C was lower in IPAH group than in control group, which was similar to several previous studies.30,31 Further, we found no relationship between PCSK9 and LDL-C existed, which might be potentially explained as follows. First, lifestyle interventions such as diet modification could affect LDL-C levels, 32 and the discomfort caused by illness condition made sufferers subconsciously change the diet. Indeed, the phenomenon of reduced LDL-C levels has already been described in several other chronic diseases, such as chronic heart failure and rheumatoid arthritis.33,34 However, whether our participants changed their diet as well as the actual impact on LDL-C is unknown. Second, it was reported that infection or inflammation could reduce serum cholesterol as indicated by reduction of both LDL-C and HDL-C in humans,35,36 and the elevation of LDL-C caused by PCSK9 might be counteracted by some other factors, such as inflammation. Consistent with these findings, we found the serum IL-6 and MCP-1 in IPAH were inversely correlated with serum LDL-C. Third, the pro-inflammatory effect of PCSK9 in IPAH might be more prominent than LDL-C regulation. As displayed in Table 3 and Fig. 3, serum PCSK9 was positively correlated with TNF-α, IL-6, and MCP-1, as well as hemodynamic and echocardiographic parameters (PASP, PADP, mPAP, PVR, and RVD), and our findings were consistent with the notion that PCSK9 also exerted various effects on the cardiovascular system via certain mechanisms independent of LDL‐C regulation. 29
To assess the clinical significance of PCSK9 in IPAH, we examined the relationship between serum PCSK9 levels and various clinical parameters. Pearson analysis certified that serum PCSK9 levels were positively correlated with cardiac function (NT-proBNP), echocardiographic parameters (RVD), and pulmonary hemodynamics (PASP, PADP, mPAP, and PVR). To exclude the influence of other variables including TC, NT-proBNP, and IL-6, multiple regression analyses were further performed and revealed that mPAP remained strongly associated with PCSK9. ROC curve analyses showed that the PCSK9, as well as IL-6, IL-1β, and MCP-1, was a strong predictor of PAH with high sensitivity and specificity. The predictive value of NT-proBNP, a pivotal biomarker and strongly associated with exercise capacity, hemodynamics, right ventricular function, and survival in PAH,37–39 was also confirmed in our study.
This study has several limitations. First, it was a preliminary cross-sectional study to explore the relationship between PCSK9 and IPAH from the perspectives of inflammation, and whether the association between PCSK9 and PAH is causative remains unclear and needs to be further elucidated. Second, the sample size is relatively small, we cannot exclude the potential influence of all confounders, and further large-scale studies are necessary to substantiate our findings. Third, our study does not address the mechanistic and pathological role of PCSK9 in IPAH. Indeed, our animal and cell experiments are in progress. Last but not least, 6-minute walk distance (6MWD) test was not performed on each of our patients, and therefore, a complete risk stratification assessment according to the newly proposed risk stratification strategy based on four criteria and using six variables on the 6th World Symposium on Pulmonary Hypertension was impracticable. 40 We sought to compare the PCSK9 levels between patients with or without low risk factors, and found that patients without low risk factor had higher PCSK9 levels than those with three low risk factors (Supplementary Fig. 2).
In conclusion, serum PCSK9 levels were elevated in IPAH. Increased PCSK9 levels were positively correlated with inflammatory cytokines, cardiac function, and hemodynamic and echocardiographic parameters. PCSK9 was a strong predictor of IPAH and might be a novel potential prognostic and therapeutic biomarker.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Ethical approvals
All procedures were performed according to the principles of the Declaration of Helsinki and approved by the local Research and Ethics Committee of the Zhongshan Hospital. Informed written consent was obtained from all participants.
Consent to participate
All the participants gave the informed consent for the collection of data and samples.
Acknowledgements
We would like to acknowledge the contribution of the staff at the 38th ward, cardiac catheter laboratory and echocardiography units at Zhongshan Hospital.
Author contributions
Conception and design: Dongxiang Zhong, Yuan Zhang and Daxin Zhou; Administrative support: Lihua Guan, Daxin Zhou and Junbo Ge; Supply of materials and samples: Xiaochun Zhang and Dandan Chen; Data collection and collation: Dongxiang Zhong, Feng Zhang; Data analysis and interpretation: Dongxiang Zhong, Qi Jin; All authors approve and revise the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Shanghai Science and Technology Development Funds (No. 19DZ1930202).
Guarantor
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
