Abstract
Aim:
We evaluated the lipid-lowering (LL) effect of proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9i) in patients with heterozygous familial hypercholesterolemia (HeFH) treated with LL-drugs and lipoprotein apheresis (LA).
Patients and Methods:
The PCSK9i treatment (evolocumab 420 mg/4 weeks, alirocumab 150 mg/2 weeks, or alirocumab 75 mg/2 weeks: 9, 6, and 2 patients, respectively) was initiated in patients with HeFH (n = 17; aged 35-69 years, 10 men, previously treated with statins + ezetimibe ± colesevelam and LA sessions for 2-12 years). A lipid profile was obtained before and immediately after the LA session and before, 1 and 2 months after switching to PCSK9i treatment. The duration of PCSK9i therapy ranged from 3 to 18 months.
Results:
Median total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and triglycerides (TG) levels before LA were 268, 198, 46, and 126 mg/dL, respectively, and decreased (at the end of the LA session) to 117, 50, 40, and 51 mg/dL, respectively (P < .001 for TC and P = .001 for all other comparisons). The median time-averaged LDL-C levels following LA were 155 (121, 176; median [25th, 75th percentile]) mg/dL. Median TC, LDL-C, and TG levels before PCSK9i therapy were 269, 190, and 127 mg/dL and decreased to 152, 100, and 95 mg/dL, respectively (P = .002, P < .002, and P < .03, respectively). Steady LDL-C levels with PCSK9i treatment were significantly lower compared with time-averaged LDL-C levels following LA (median value: 100 vs 155 mg/dL; P = .008). With PCSK9i, from 13 patients with CHD, 6 (46.1%) patients achieved LDL-C <70 mg/dL, and 2 patients (15.4%) achieved LDL-C <100 mg/dL. Lipoprotein apheresis was discontinued in all patients except for 2 who continued once monthly.
Conclusions:
PCSK9i can reduce LDL-C more consistently over time compared with a transient decrease following LA in HeFH patients. PCSK9i therapy may reduce the frequency of LA. Larger trials are required to establish the clinical implications of PCSK9i in patients previously on LA.
Keywords
Introduction
Familial hypercholesterolemia (FH) is an autosomal, in most cases, dominant disorder with a gene dosage effect, resulting in elevated plasma levels of low-density lipoprotein cholesterol (LDL-C) and an increased risk of premature atherosclerotic cardiovascular disease (ASCVD). 1 Familial hypercholesterolemia is defined as homozygous (HoFH) or heterozygous (HeFH). The plasma LDL-C levels are frequently more than twice higher in HoFH compared with HeFH, although there can be some overlap in these values. 2
The prevalence of HeFH is the highest among genetic disorders (1 in 200 to 250 births). 3,4 Familial hypercholesterolemia is still underdiagnosed and undertreated in the general population. 4 Nordestgaard et al 4 evaluated approximately 180 countries/territories and found that in most countries <1% of FH patients were diagnosed with only few exceptions (71% diagnosed in the Netherlands, 43% in Norway, 19% in Iceland, 13% in Switzerland, 12% in the UK, and 6% in Spain). 4 If untreated, 50% of men and 30% of women with HeFH may develop ASCVD before the age of 50 and 60 years, respectively. 5 Thus, lipid-lowering (LL) treatment is crucial for these patients. Furthermore, patients with HeFH are likely to require >1 medication to achieve target LDL-C levels; in more severe cases, lipoprotein apheresis (LA) is required. 6 Lipoprotein apheresis is usually performed every 1 to 2 weeks with each session lasting about 2 to 3 hours to remove about 60% to 70% of the LDL-C. 6 However, LDL-C reduction with LA is transient and a rebound elevation occurs after 10 to 14 days. 6 Nevertheless, LA treatment has increased life expectancy in HoFH patients. 7
Proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9i) represent a LL drug class that can decrease LDL-C levels by up to 70%. 8 The PCSK9 is involved in LDL receptor degradation. It follows that decreasing the availability of PCSK9 will increase LDL receptors and decrease the concentration of LDL in the circulation. 8 Since PCSK9i can produce mean reductions in LDL-C similar to LA, we wanted to evaluate the efficacy and safety of PCSK9i in HeFH patients treated previously with classical LL drugs and LA.
Patients and Methods
Patients included in the present study had a HeFH phenotype and were currently treated in Onassis Cardiac Surgery Center. They were not controlled by maximum tolerated LL agents and required LA. The diagnosis of HeFH was based on the Simon Broome criteria, 9 which incorporate LDL-C levels, clinical signs, and family history of premature ASCVD in a first-degree relative, to generate a score that leads to a classification of either “definite” or “probable” FH.
Patients were treated with a low-fat diet and maximum tolerated doses of a statin: atorvastatin 80 mg/daily (2 patients) or rosuvastatin 40 mg/daily (15 patients) plus ezetimibe (17 patients) and/or colesevelam 1875 mg/daily (3 patients). Lipoprotein apheresis was performed biweekly for 2 to 12 years, due to their inability to achieve LDL-C targets. 1
Patients were then allocated to a PCSK9i without LA but maintaining other treatment and lifestyle advice. The study was approved by the establishment ethics committee and informed consent was obtained from all participants.
Diagnosis of HeFH Phenotype
All patients were diagnosed with HeFH phenotype based on the following clinical criteria. 9
Definite
Plasma LDL-C in adults >4.9 mmol/L (190 mg/dL) and in children >4 mmol/L (155 mg/dL) plus DNA-based evidence or tendon xanthomas in the patient or in a first- or second-degree relative.
Possible
Plasma LDL-C in adults >4.9 mmol/L (190 mg/dL) and in children >4 mmol/L (155 mg/dL) and family history of myocardial infarction at an age of <50 year in a second-degree relative or at an age of <60 year in a first-degree relative or family history of LDL-C in adult first- or second-degree relative >4.9 mmol/L (190 mg/dL) or >4 mmol/L (155 mg/dL) in a child or sibling aged <16 years.
Lipoprotein Apheresis Procedure
Patients required LA treatment due to inability to reach target LDL-C levels despite treatment with diet and LL drugs, according to guidelines (see discussion for a description of various LDL-C targets). The polyacrylate whole blood adsorption (direct adsorption of lipoproteins) was used. Direct adsorption of lipoproteins 500, 750 (500 + 500), and 1250 (500 + 750) absorbers were incorporated in the extracorporeal circuit. Direct adsorption of lipoproteins primer solution, acid citrate dextrose formula A (ACD-A) solution, blood lines, and hemadsorption monitor 4008 ADS (Fresenius HemoCare Adsorber Technology GmbH) were used. Prior to the session, the absorbers were rinsed with 3 × 2000 mL of primer solution at a flow rate of 400 mL/min. The first 2 L contained 20 000 IU of heparin. The absorbers were saturated with citrate during priming. Prior to the session, the patients received bolus intravenous heparin, followed by an ACD-A infusion during the session. Acid citrate dextrose formula A was first mixed with the patient’s blood at a ratio of 1:20 and reduced to 1:40 after 1500 mL of blood were treated. The treatment volume per session was 2500 to 4500 L. Two bilateral vascular accesses were established by venepuncture, generally in the median cubital veins. At the initiation of the session, the patient was only connected to the afferent line of the extracorporeal circuit. All sessions were carried out under blood pressure and electrocardiogram monitoring.
Laboratory Measurements
At the beginning of LA, before any interventions and particularly before heparin administration, and at the end of each session (just before removing the needle from the afferent arm), a blood sample was drawn to measure fasting lipids. Blood was collected in tubes containing ethylenediaminetetraacetic acid. Plasma TC, triglyceride (TG), and high-density lipoprotein cholesterol (HDL-C) levels were measured using enzymatic colorimetric methods on a Roche Integra Biochemical analyzer with commercially available kits (Roche). Serum LDL-C levels were calculated using the Friedewald formula 10 (of note that Friedewald formula underestimates LDL-C values) for TG levels <4.5 mmol/L (400 mg/dL): calculated LDL-C (mg/dL) = TC – HDL-C − TG/5. Fasting blood samples were also obtained before initiation of PCSK9i treatment as well as 1 and 2 months after (to assess whether changes in lipid parameters were stable). Apart from lipids, plasma glucose, serum creatinine, urea, uric acid, alanine aminotransferase (ALT), aspartate aminotransferase (AST), and thyroid tests (thyrotropin, TSH) were measured in patients on PCSK9i using a Roche Integra Biochemical analyzer and commercially available kits (Roche). All samples were analyzed within 24 hours.
Statistical Analysis
All continuous variables were expressed as median (25th and 75th percentile), unless otherwise stated. Given the sample size, nonparametric tests were preferred. Related samples were compared using the Wilcoxon signed-rank test. All tests were 2-tailed and P < .05 was considered significant. The efficacy of LA-LDL-C (time-averaged levels [CAVG] of LDL-C) was evaluated by using a modified version of the Kroon et al formula 11 : CAVG = CMIN + K (CMAX-CMIN); CMIN = LDL-C level immediately after LA and CMAX = LDL-C level at the start of the subsequent procedure. Kroon et al 11 proposed using a K = 0.73. Statistical analysis was performed using the Statistical Package for Social Sciences software (release 17.0, SPSS Inc.).
Results
The baseline characteristics of the patients are summarized in Table 1. All patients had “definite” HeFH, according to the Simon Broome criteria. In 6 patients, DNA analysis was performed and confirmed a single mutated allele of the LDLR (LDL receptor) gene. Of the 17 patients, 10 were males. The mean age of the total population was 54.6 ± 14.0 years. Thirteen patients had a history of coronary heart disease (CHD), 3 were hypertensive, and 1 had diabetes mellitus type 1. None of the patients were smokers or obese. From 13 patients with CHD, 12 patients underwent revascularization procedures (percutaneous coronary intervention and/or coronary artery bypass grafting), 2 patients underwent aortic valve replacement, and 7 patients had carotid arteries stenosis (3 were operated). Coronary heart disease was excluded in 4 patients by negative thallium heart scan test or coronary computed tomography angiography. An annual carotid ultrasound was performed in all patients, whereas ultrasonography for other peripheral arteries was not evaluated routinely.
Baseline Characteristics of the Patient Population (n = 17).

Abbreviation: BMI, body mass index.
Nine patients received evolocumab (subcutaneously, 420 mg/4 weeks) and 8 patients received alirocumab (subcutaneously, 75 mg/2 weeks [2 patients] and 150 mg/2 weeks [6 patients]). The median (25th and 75th percentile) changes in TC, LDL-C, HDL-C, and TG levels immediately after LA were −160 (118, 183), −149 (114, 159), −5 (5, 8), and −85 (44, 101) mg/dL, respectively (P < .001 for TC and P = .001 for all other comparisons; Table 2). Figure 1A shows the median acute changes of LDL-C and Figure 1B in each of 5 consecutive LA sessions. The median time-averaged LDL-C levels during LA was 155 (121, 176) mg/dL (Table 2). Lipoprotein apheresis in the majority of the patients was performed biweekly for 2 to 12 years.
Lipid Changes Before and After the 2 Treatments.a

Abbreviations: HDL-C, high-density lipoprotein cholesterol; LA, lipoprotein apheresis; LDL-C, low-density lipoprotein cholesterol; PCSK9i, proprotein convertase subtilisin/kexin type 9 inhibitor; TG, triglyceride; TC, total cholesterol.
a Lipids values are median (25th, 75th percentile) and in mg/dL. To convert TG from mg/dL to mmol/L divide by 88.6. To convert TC, LDL-C, and HDL-C from mg/dL to mmol/L divide by 38.7.

A, LDL-C levels before and after LA. B, LDL-C changes before and after 5 LA sessions. C, LDL-C levels before and 1 month after PCSK9i treatment. D, LDL-C levels before and after the first and second dose of PCSK9i treatment. In the box-plots, the horizontal dark lines within boxes represent the median value of each variable while the bottom and top of the boxes represent the 25th and 75th percentile, respectively. Outliers are marked with a dot and extreme outliers with an asterisk (the case number in the current series for each is also indicated). LA indicates lipoprotein apheresis; LDL-C, low-density lipoprotein cholesterol; PCSK9i, proprotein convertase subtilisin/kexin type 9 inhibitors.
The major adverse events concerning the LA sessions were allergic reactions manifested as shortness of breath, facial flushing, nausea, vomiting, and severe hypotension (due to the use of polyacrylate and dextran sulfate columns that convert kininogen to bradykinin, thus leading to marked increases in bradykinin levels, which do not apply to other LA systems). These side effects were most likely seen during the first LA sessions.
In our patients during the LA sessions, 1 patient presented an episode of bradykinin syndrome with Quincke’s edema (transitory, localized, noninflammatory edema of subcutaneous tissue or mucous of mouth, lips, tongue or larynx, and others and may result in fatal asphyxiation in the absence of specific treatment) and lumbar pain. Minor side effects such as puncture difficulties, technical problems, headache, dizziness, or hypotensive periods occurred occasionally (Table 3). All the patients were rechallenged, uneventfully. From our previous report, 12 we found an average annual ASCVD event rate of 5.5%.
Possible Side Effects of LA and PCSK9i in the Study Population.

Abbreviations: LA, lipoprotein apheresis; PCSK9i, proprotein convertase subtilisin/kexin type 9 inhibitors.
a In 1 patient.
b In 2 patients.
The median (25th and 75th percentile) changes in TC, LDL-C, and TG concentrations following PCSK9i therapy were: −97 (−39, −160), −62 (−26, −131), and −63 (−14, −76) mg/dL (P = .002, P < .002, and P < .03, respectively). Of note, the TG levels after LA may be lower due to heparin administration and LA can chronically lower baseline LDL-C (see discussion). High-density lipoprotein cholesterol increased nonsignificantly from a median (25th and 75th percentile) value of 43 (39, 56) to 45 (33, 52) mg/dL (P = .213; Table 2
LA and PCSK9i therapy produced a median absolute LDL-C decrease of 149 mg/dL (69.8%) and 62 mg/dL (37.4%), respectively (P = .013); the corresponding changes for TG levels were 85 mg/dL (55.4%) and 63 mg/dL (46.5%; P = .2). Steady LDL-C levels with PCSK9i treatment were significantly lower compared with time-averaged LDL-C levels following LA (median value: 100 vs 155 mg/dL; P = .008). No gender-related differences were observed.
LDL-C Target Attainment
Recommended LDL-C targets at the time the study was conducted 4,13 -15 for FH patients (adults <2.5 mmol/L [<100 mg/dL] and adults with CHD or diabetes mellitus <1.8 mmol/L [<70 mg/dL]) in our study cohort were as follows.
Immediately after LA and without PCSK9i use, 9 patients (6 patients with CHD) achieved LDL-C <70 mg/dL and 3 patients <100 mg/dL (2 patients with CHD), which was transient, and a rebound elevation occurred; the corresponding number of patients following PCSK9i treatment was 6 and 2, respectively, Table 4. Specifically, from 13 patients with CHD, 6 (46.1%) patients achieved LDL-C <70 mg/dL, 2 (15.4%) patients achieved LDL-C <100 mg/dL, and 5 patients achieved LDL-C >100 mg/dL (38.5%). All patients with LDL-C >100 mg/dL were advised to continue LA sessions. Two patients continued LA as before, 2 patients decreased the rate to once monthly (although the advice was for biweekly) and 1 patient decided to stop LA. We had some difficulty to convince patients with LDL <100 mg/dL to continue LA. For the 4 patients without CHD, 1 patient achieved LDL-C <70 mg/dL, 2 patients <100 mg/dL, and 1 patient achieved LDL-C >100 mg/dL after PCSK9i. All 4 patients without CHD stopped LA.
LDL-C Target Attainment.
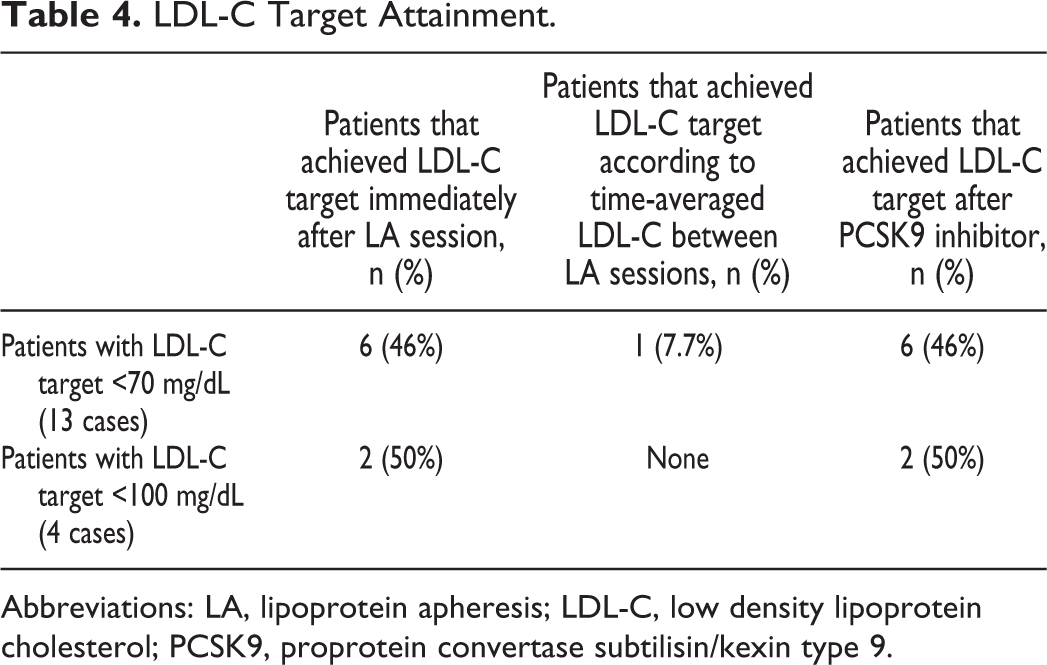
Abbreviations: LA, lipoprotein apheresis; LDL-C, low density lipoprotein cholesterol; PCSK9, proprotein convertase subtilisin/kexin type 9.
Plasma glucose, serum creatinine, urea, uric acid, ALT, AST, and thyroid tests remained within normal values during treatment with PCSK9i. No serious side effect was reported. One patient complained of skin irritation at the PCSK9i injection site.
Discussion
In the present study, HeFH patients treated with LA and maximum tolerated doses of non-PCSK9i LL drugs were switched to PCSK9i (+ continuing the other LL therapies) to assess whether the recommended LDL-C goals were achieved.
Pfohl et al 16 reported that mean pre-LA LDL-C levels reduced by 30% after 6 months and 36% after 18 months of LA.
Only few studies evaluated the effect of LA and PCSK9i 16,17 in patients with HeFH. Kawashiri et al 17 switched 11 HeFH patients from biweekly LA to a biweekly subcutaneous injection of evolocumab 140 mg and reported a 62.5% reduction in LDL-C levels with evolocumab.
In our study, the median reduction of LDL-C with PCSK9i was 37.4%, while it was 69.8% with LA (acute decrease). Furthermore, PCSK9i did not decrease HDL-C concentrations in contrast to what is observed after LA. Lappegård et al 16 observed that LA led to significant reductions in large and intermediate size LDL particles and a nonsignificant reduction in small LDL particles in 3 HoFH patients, whereas evolocumab nonsignificantly reduced the latter LDL subclasses which are more atherogenic. Hohenstein et al 18 reported that in a German Lipoprotein Apheresis Unit, the authors concluded that only 10% to 15% of their patients might stop LA following PCSK9i therapy. 19 However, no information about how many patients had HoFH was provided.
Recommended LDL-C targets 4,13 -15 for FH patients with CHD in our study cohort were achieved with PCSK9i administration in 46% of patients. PCSK9i effectively reduce Lp(a) by about 25% to 30% 20,21 ; unfortunately, we did not evaluate this effect. Data from the FOURIER (Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk) trial showed that at 48 weeks, evolocumab reduced Lp(a) by 26.9% (6.2% to 46.7%). However, this reduction was lower in patients with higher baseline Lp(a) levels, possibly due to the reduced clearance of the smaller isoforms or other as yet undefined mechanisms. 19
Our HeFH patients were treated with LA to achieve LDL-C target values, in accordance with the international consensus-integrating lipoprotein apheresis document. 22 The post-LA treatment rebound of serum TGs takes 1 to 2 days reaching 83% of pretreatment levels; TC and LDL-C take 8 to 13 days. 11 The recovery half-times (T 50%) for the posttreatment rebound of LDL-C and Lp(a) values is about 4 days. This means that most of HeFH patients have high levels of LDL-C between LA sessions or this treatment should be performed twice weekly. Of note, LA treatment is performed only in selected centers and many patients have to cover long distances to reach the nearest center. Additionally, there are adverse effects (most likely at the first sessions) such as allergic reactions manifested as shortness of breath and/or facial flushing, nausea vomiting, serious hypotension, and there are other limitations such as venepuncture difficulties and technical problems. 6 All the above could affect patient quality of life (QOL), although this has not been confirmed in trials. Rosada et al 23 evaluated QOL in 29 patients on regular LA treatment using the Medical Outcomes Study 36-item Short-Form Health Survey (SF-36), the Beck Depression Inventory (BDI), and a newly developed questionnaire for assessing QOL in patients undergoing LA. They showed that the procedure itself is generally tolerated without major complaints, suggesting that the benefits of LA exceed any negative effects on patient’s QOL. 23
Current guidelines suggest the use of PCSK9i on top of diet as well as the maximum tolerated LL in statin-intolerant patients, to achieve LDL-C goals. 24 However, PCSK9i drugs are costly; this may minimize their use in clinical practice. 24 It should be noted that PCSK9i, although adding extra costs to health care systems, 24 are cheaper than LA sessions (yearly costs for PCSK9i are less than one-fifth of the costs of LA and recently after the price reduction are even less). 18 They also offer a better QOL for patients, based on the comments above. Thus, even after overlooking its complexities and difficulties another problem to using LA is the high cost. 25 The average annual expenditure of a biweekly regimen of LA can range from US$33 187 (Germany) to US$114 478 (USA) per patient. 25 In Greece, the cost of biweekly regimen of LA is approximately 26 000 Euros per patient. This questions whether health care systems can meet the demand for LA and particularly considering the availability of PCSK9i.
Spitthöver et al 26 studied a cohort of 110 patients with established CVD; the use of PCSK9i on top of routine care increased the proportion of patients attaining the LDL-C target concentration by 41.8% overall; in 55.5% of patients who received PCSK9i and LA combination therapy, the proportion of patients attaining the LDL-C target increased to 54.1%. Furthermore, among patients on chronic LA, 18.1% terminated LA, whereas LA frequency was reduced by 25.5% in the rest of them during this cohort study. 26 Zenti at al 27 reported a case of HeFH patient who had statin intolerance; combination therapy (ie, PCSK9i and LA) decreased LDL-C by 49.4%. Thus, patients who do not achieve an LDL-C <70 mg/dL should remain on LA treatment. However, the frequency of LA is a significant factor affecting compliance. Patients must usually arrange an entire day dedicated to the procedures (involving transport), and it is no surprise that the higher the frequency of LA sessions, the lower the adherence. In the Rosada et al study 23 from 29 patients (see above), 11 patients mentioned the long distance from home to the apheresis center and the monotonous hospital meals. In the present study, the median time-averaged LDL-C levels following LA was 155 mg/dL which is >50% higher compared with what was achieved after switching to PCSK9i treatment. In principle, if a similar difference is confirmed by larger studies, this difference could prove to be a disadvantage because the evidence from several trials supports the “lower is better” concept for LDL-C levels. 23 Also, we do not know whether the variation in LDL-C levels after apheresis is equally effective as a steady lowering of LDL-C levels. The median time-averaged LDL-C levels following LA may not be an accurate representation of the real therapeutic effect.
In the Alirocumab in patients with heterozygous familial hypercholesterolaemia undergoing lipoprotein apheresis: ODYSSEY ESCAPE trial, treatment of HeFH patients undergoing LA with a PCSK9i (alirocumab) resulted in an additional 54% reduction in LDL-C. 28 Patients were randomly assigned to alirocumab 150 mg (n = 41) or placebo (n = 21) every 2 weeks for 18 weeks. Based on the trial criterion of reducing LDL-C by ≥30% below the baseline value on LA, 63% of patients on alirocumab were able to discontinue LA altogether and over 90% to halve its frequency. 28
The use of combination treatment (PCSK9i + LA) to achieve the recommended LDL-C treatment goals may be useful. Such a therapeutic strategy will permit the use of LA to a more extensive patient population based on the increased intervals between LA sessions and the resulting fall in cost.
The present study has limitations. Firstly, it involved a small number of patients, but this was due to the selection of a specific patient population, that is, HeFH patients not controlled by maximum tolerated LL agents and requiring LA. Furthermore, PCSK9i are available in Greece only after approval by a national insurance committee. Follow-up was short, thus not permitting any conclusions regarding the efficacy and safety of PSCK9i in the long-term in this patient population. Further research is needed in this field. It will be difficult to compare studies where apheresis is replaced by PCSK9i because the effectiveness of the PCSK9i will depend on the functionality of the LDLR, as discussed above. However, it should be noted that 6 patients with CHD who achieved recommended LDL-C target (<70 mg/dL) 14 with PCSK9i treatment had an LDL-C <55 mg/dL, meaning that they achieved the LDL-C target recommended by new just published by European Society of Cardiology and the European Atherosclerosis Society guidelines. 29 Also, PCSK9i can effectively reduce Lp(a) by about 25% to 30% 30,31 ; unfortunately, we did not evaluate this effect in all patients.
Conclusions
This study provides insight into the characteristics and treatment patterns of FH patients, who are not at LDL-C target with LL-drugs and LA. Switching from LA to PCSK9i therapy led to a smaller but sustained, LDL-C targeting effect, whereas HDL-C levels did not decrease. Larger trials are required to establish the clinical implications of PCSK9i in patients previously treated by LA. Combination treatment with the addition of PCSK9i therapy may reduce the frequency of LA and may be useful for such patients. Because most studies comparing any treatment with LA are likely to be relatively small (due to various factors including patient availability, heterogeneity, and funding), there are a need for multicenter studies, international registries, and meta-analyses.
Footnotes
Author Contributions
Conceptualization: Vana Kolovou, Genovefa D. Kolovou, Niki Katsiki and Dimitri P. Mikhailidis, data collection: Nikoletta Karampetsou and Sophie Mavrogieni, formal analysis: Stamatis Makrygiannis, writing: Vana Kolovou, Genovefa D. Kolovou, and editing: Stamatis Makrygiannis, Athanasios Manolis, Andreas Melidonis, Dimitri P. Mikhailidis, Niki Katsiki, Sophie Mavrogieni, Genovefa D. Kolovou. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
N.K. has given talks, attended conferences, and participated in trials sponsored by Amgen, Astra Zeneca, Bausch Health, Boehringer Ingelheim, Elpen, Mylan, Novo Nordisk, Sanofi, and Servier; D.P.M. has given talks and attended conferences sponsored by Amgen, Novo Nordisk, and Libytec. G.K. has given talks, attended conferences, and participated in trials sponsored by Amgen, Angelini, MSD, Lilly, Vianex, and Sanofi-Aventis and has also accepted travel support to attend conferences from Amgen, Sanofi, and MSD. A Melidonis has given talks, attended conferences, and participated in trials sponsored by Novo, Glaxo, Libytec, and Lilly.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
