Abstract
Background
Statins are the standard treatment for coronary atherosclerosis; however, some patients require additional therapies for optimal plaque regression. Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors (PCSK9i), including monoclonal antibodies and small interfering RNA-based therapies, have shown promise as adjuncts to statins, although their efficacy for coronary plaque regression, as assessed by intravascular imaging, remains uncertain.
Methods
We conducted a meta-analysis to compare the treatment efficacy of statins combined with PCSK9i (PCSK9i group) versus statins alone or statins combined with placebo (control group) in adults with coronary atherosclerosis (INPLASY registration number: INPLASY202550027). Plaque lesions were assessed using intravascular ultrasound, optical coherence tomography, coronary computed tomography angiography, and near-infrared spectroscopy. Lipid profile parameters, including low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and lipoprotein(a), were measured and analyzed.
Results
In total, 11 trials involving 2490 patients (follow-up duration: 12–78 weeks) were included. Meta-regression showed that combination therapy significantly reversed coronary artery plaque (p < 0.001). No significant difference was observed in the atheroma volume between the PCSK9i and control groups; however, fibrous cap thickness increased significantly in the PCSK9i group. Additionally, low-density lipoprotein cholesterol and lipoprotein(a) levels decreased, while high-density lipoprotein cholesterol levels increased after PCSK9i treatment.
Conclusion
PCSK9i combined with statins effectively promote coronary plaque regression, particularly in patients with acute myocardial infarction, offering a promising approach for managing coronary atherosclerosis.
Keywords
Background
Coronary artery disease (CAD) is the leading cause of morbidity and mortality worldwide, accounting for approximately one-third of all deaths. 1 The primary pathophysiological process underlying CAD is coronary atherosclerosis, which is characterized by the accumulation of lipid-rich plaques within the arterial walls. 2 Over time, these plaques can progress and obstruct coronary blood flow, leading to myocardial ischemia, infarction, and potentially fatal complications. Statins, which inhibit 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase and thus reduce low-density lipoprotein cholesterol (LDL-C) levels, have been the cornerstone of treatment for coronary atherosclerosis for decades. 3 Numerous randomized controlled trials (RCTs) and meta-analyses have demonstrated that statins significantly reduce the risk of cardiovascular events and mortality in both primary and secondary prevention settings. 4 Moreover, statins promote coronary plaque regression and stabilization, as assessed by various imaging modalities, including intravascular ultrasound (IVUS), optical coherence tomography (OCT), and near-infrared spectroscopy (NIRS).5,6 However, despite the proven efficacy of statins, some patients fail to achieve optimal LDL-C reduction and remain at high risk of cardiovascular events. 3 This residual risk has prompted the search for additional therapies that can enhance the lipid-lowering and plaque-stabilizing effects of statins. In recent years, proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors (PCSK9i) have emerged as a promising class of drugs for the management of hypercholesterolemia and coronary atherosclerosis. 7
PCSK9 is a serine protease that binds to LDL receptors on the surface of hepatocytes, promoting their degradation and thus reducing LDL-C clearance from the circulation. 7 Inhibition of PCSK9, either through monoclonal antibodies (mAbs) or small interfering RNA (siRNA), significantly reduces LDL-C levels and improves cardiovascular outcomes in patients with hypercholesterolemia. 8 Moreover, preclinical studies and small clinical trials have suggested that PCSK9 inhibition exerts plaque-stabilizing effects beyond its impact on LDL-C, possibly through anti-inflammatory and anti-apoptotic mechanisms. 9 The combination of statins and PCSK9i has the potential to enhance lipid-lowering and plaque-stabilizing effects, thereby further reducing the risk of cardiovascular events in patients with coronary atherosclerosis. However, the efficacy and safety of this combination therapy, particularly in terms of its impact on coronary plaque regression and composition, as assessed by intravascular imaging, remain unclear. To date, several clinical studies have investigated the effects of statin plus PCSK9i therapy on coronary plaque volume and composition using IVUS, OCT, coronary computed tomography angiography (CTA), and NIRS; however, the results have been inconsistent.10–15 Given the heterogeneity of the available evidence and the potential implications in clinical practice, a comprehensive meta-analysis of RCTs evaluating the efficacy and safety of statin plus PCSK9i therapy on coronary plaque regression, as assessed by intravascular imaging, is warranted. This meta-analysis aims to collate the existing data on the impact of this combination therapy on plaque volume, composition, and adverse events as well as identify potential effect modifiers and sources of heterogeneity. The findings of this study will provide valuable insights into the role of statin plus PCSK9i therapy in the management of coronary atherosclerosis and guide future research in this field.
Methods
Protocol registration
This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines
16
and was registered post-study at INPLASY with registration number
Eligibility criteria
We included clinical studies that met the following criteria:
Participants: adults (aged ≥18 years) with coronary atherosclerosis confirmed via coronary angiography or other imaging modalities. Interventions: statin therapy combined with PCSK9i (mAbs or siRNA-based therapy). Comparators: statin therapy alone or statin therapy plus placebo. Outcomes: available follow-up outcomes of intravascular imaging modalities, including IVUS, OCT, CTA, and NIRS, as well as changes in lipid profiles. Study design: parallel-group or crossover trial with a minimum follow-up duration of 3 months.
We excluded observational studies, animal studies, and studies not published in English.
Information sources and search strategy
We searched the following electronic databases from inception to 31 August 2024: PubMed, Embase, Cochrane Central Register of Controlled Trials (CENTRAL), and ClinicalTrials.gov. The search strategy was developed in collaboration with a medical librarian and included a combination of keywords and controlled vocabulary terms (e.g. Medical Subject Headings terms) related to statins, PCSK9i, coronary atherosclerosis, and intravascular imaging. In addition, we manually searched the reference lists of relevant reviews and included studies to identify additional eligible trials.
Study selection
Two reviewers independently screened the titles and abstracts of all the records identified through the search strategy using the pre-specified eligibility criteria. The full texts of potentially eligible studies were retrieved and independently assessed by the same two reviewers. Any disagreements were resolved through discussion or by consulting a third reviewer if necessary. The study selection process was documented using a PRISMA flow diagram.
Data extraction
Two reviewers independently extracted data from the included studies using a standardized data extraction form. The following information was collected:
Study characteristics: first author, publication year, country, sample size, follow-up duration, and study design. Participant characteristics: age, sex, baseline LDL-C levels, baseline plaque volume, and clinical setting. Intervention and comparator details: statin type, statin dose, PCSK9i type, PCSK9i dose, and placebo. Outcomes: intravascular imaging via IVUS, OCT, CTA, or NIRS and changes in lipid profile parameters (LDL-C, high-density lipoprotein cholesterol (HDL-C), and lipoprotein(a) (Lp(a))).
Any discrepancies in the extracted data were resolved through discussion or by consulting a third reviewer if necessary.
Risk of bias assessment
Two reviewers independently assessed the risk of bias in the included studies using a tool with the following different domains of bias: (a) randomization process; (b) deviations from intended interventions; (c) missing outcome data; (d) measurement of the outcome; and (e) selection of the reported results. Each domain was judged as having a low risk, some concerns, or a high risk of bias. Any disagreements were resolved through discussion or by consulting a third reviewer if necessary (Table 1).
Quality assessment of the included articles.

(1) Representativeness of the exposed cohort; (2) Selection of the nonexposed cohort; (3) Ascertainment of exposure; (4) Demonstration that the outcome of interest was not present at the start of the study; (5) Comparability of cohorts on the basis of the design or analysis controlled for confounders; (6) Assessment of outcome; (7) Was follow-up long enough for outcomes to occur?; (8) Adequacy of follow-up of cohorts. The “?” symbol denotes missing data in original studies (e.g. fibrous cap thickness not reported).
Data synthesis and analysis
We performed a qualitative synthesis of the included studies by summarizing their characteristics, participant data, interventions, comparators, and outcomes in tables and text. If the included studies were sufficiently homogeneous in terms of design, population, interventions, and outcomes, we conducted a quantitative synthesis using random-effects meta-analyses. For the parameter plaque burden, we calculated the mean difference and its 95% confidence interval (CI) between the PCSK9i and control groups. If the plaque volume was reported using different scales or units across studies, we calculated the weighted mean difference (WMD). We assessed statistical heterogeneity using the I2 statistic. An I2 value >50% or a p-value <0.10 in the Q test indicated substantial heterogeneity. 21 If substantial heterogeneity was observed, we explored potential sources by performing subgroup analyses based on participant characteristics (e.g. age, sex, and baseline LDL-C levels), intervention characteristics (e.g. PCSK9i types), and study design (e.g. follow-up duration and imaging modality). Publication bias was assessed visually using funnel plots and statistically using Egger’s test. All statistical analyses were performed using STATA software (StataCorp LLC, College Station, TX, USA). A two-tailed p-value <0.05 was considered to indicate statistical significance.
Results
Study selection
The initial search strategy yielded 1245 records from electronic databases and 23 additional records from manual searches of reference lists. After removing duplicates, 987 unique records were screened by title and abstract, of which 138 were deemed potentially eligible and underwent full-text review. After assessing the full-text articles against the pre-specified eligibility criteria, 11 clinical studies10–15,17–21 were included in the qualitative synthesis (meta-analysis). The reasons for excluding studies during the full-text review stage were as follows: (a) nonrandomized design (n = 41); (b) ineligible population (n = 32); (c) ineligible intervention or comparator (n = 27); (d) ineligible outcomes (n = 19); and (e) insufficient follow-up duration (n = 7). The study selection process is summarized in the PRISMA flow diagram (Figure 1).

Flow diagram of study selection.
Study characteristics
The 11 included clinical studies were published between 2016 and 2024 and involved a total of 2490 participants with coronary atherosclerosis. The sample sizes of individual studies ranged from 37 to 484 participants. The mean age of the participants ranged from 58.3 to 67.8 years, and the proportion of male participants ranged from 62.5% to 88.2%. The baseline mean LDL-C levels ranged from 68.4 to 127.8 mg/dL. The interventions included statin therapy combined with PCSK9i, such as evolocumab, alirocumab, and inclisiran. The follow-up duration ranged from 12 to 78 weeks. The key characteristics of the included studies are summarized in Table 2.
Characteristics of the included studies.
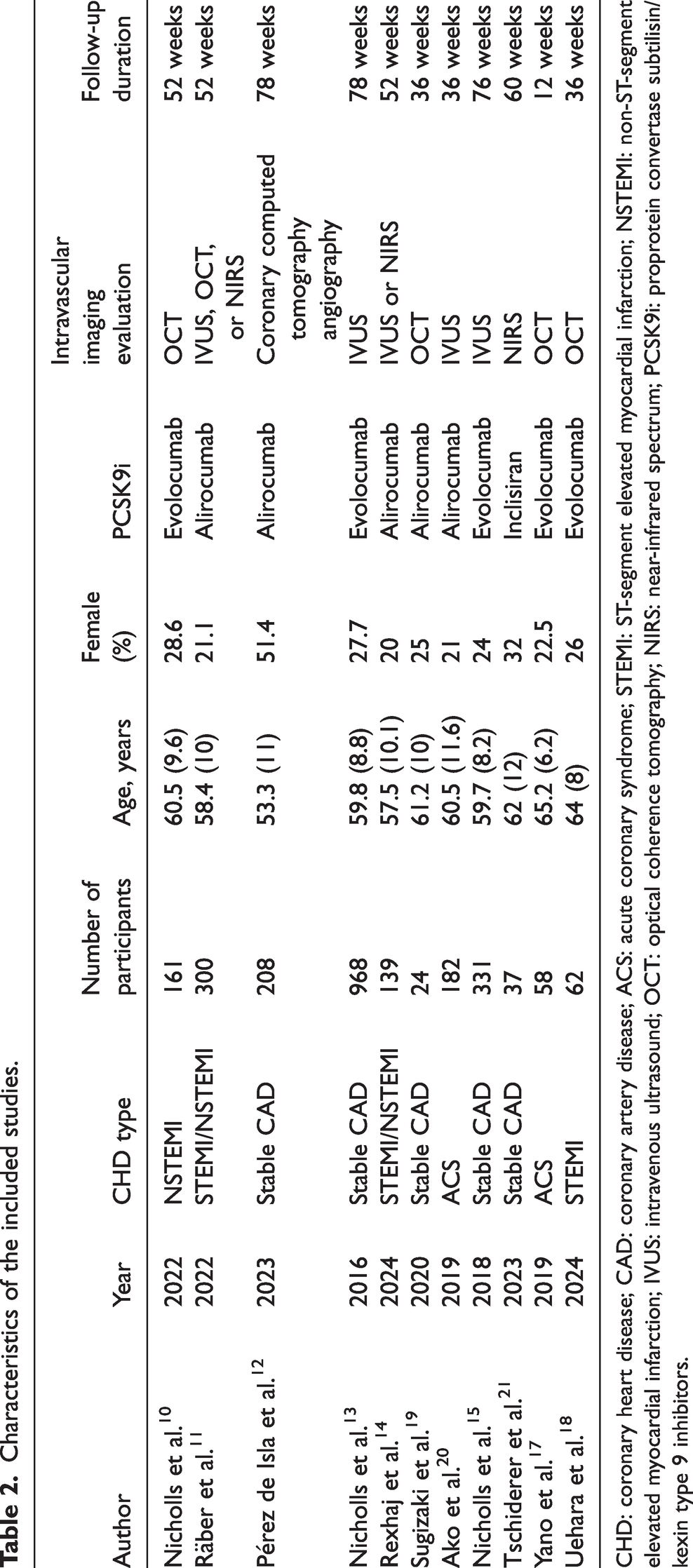
CHD: coronary heart disease; CAD: coronary artery disease; ACS: acute coronary syndrome; STEMI: ST-segment elevated myocardial infarction; NSTEMI: non-ST-segment elevated myocardial infarction; IVUS: intravenous ultrasound; OCT: optical coherence tomography; NIRS: near-infrared spectrum; PCSK9i: proprotein convertase subtilisin/kexin type 9 inhibitors.
Effects of PCSK9i treatment on LDL-C, HDL-C, and Lp(a) levels
After control or PCSK9i treatment, the LDL-C level10–15,17–19 in the control group was higher than that in the PCSK9i group (pooled WMD = 52.265, 95% CI: 45.093–59.437, mg/dL), and variation in WMD attributable to heterogeneity was detected (84.8%) (Figure 2(a), upper). The absolute change in LDL-C level10,11,13,15,20 was also higher in the control group than in the PCSK9i group (pooled WMD = 46.742, 95% CI: 9.723–83.761, mg/dL), and variation in WMD attributable to heterogeneity was detected (p < 0.01) (Figure 2(a), lower). The HDL-C level10–15,17,18 in the control group was lower than that in the PCSK9i group (pooled WMD = −2.785, 95% CI: −4.365 to −1.204, mg/dL), and variation in WMD attributable to heterogeneity was not detected (Figure 2(b), upper). The absolute change in HDL-C level10,11,13,15,20 tended to be lower in the control group than in the PCSK9i group (pooled WMD = −1.438, 95% CI: −2.957 to 0.081, mg/dL), and variation in WMD attributable to heterogeneity was detected (98.8%) (Figure 2(b), lower). The Lp(a) level10–15,18 in the control group tended to be higher than that in the PCSK9i group (pooled WMD = 3.495, 95% CI: −0.463 to 7.454, nmol/L), and variation in WMD attributable to heterogeneity was detected (65.1%) (Figure 2(c), upper). The absolute change in Lp(a) level10,11,13,15,20 was significantly higher in the control group than in the PCSK9i group (pooled WMD = 19.248, 95% CI: 2.831–35.665, nmol/L), and variation in WMD attributable to heterogeneity was detected (99.9%) (Figure 2(c), lower). Some of the included studies were sub-analyses of the PACMAN-acute myocardial infarction (AMI) and GLAGOV studies. Upon re-examination, these studies included distinct patient cohorts that did not overlap with their parent trials. However, to ensure rigor, we excluded these sub-analyses and confirmed that the results remained unchanged (LDL-C: pooled WMD = 52.123, 95% CI: 43.897–60.350, mg/dL; Lp(a): pooled WMD = 3.428, 95% CI: −0.696 to 7.553) (Supplemental Figures 1 and 2).

Effects of PCSK9i treatment on LDL-C, HDL-C, and Lp(a) levels. (a) LDL-C level in the control group was higher than that in the PCSK9i group (upper); the absolute change in LDL-C level was also higher in the control group than in the PCSK9i group (lower). (b) HDL-C level in the control group was lower than that in the PCSK9i group (upper); the absolute change in HDL-C level tended to be lower in the control group than in the PCSK9i group (lower) and (c) Lp(a) level in the control group tended to be higher than that in the PCSK9i group (upper); the absolute change in Lp(a) level was significantly higher in the control group than in the PCSK9i group (lower) and PCSK9i: proprotein convertase subtilisin/kexin type 9 inhibitors; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; Lp(a): lipoprotein(a).
Effects of PCSK9i on plaque regression
After control or PCSK9i treatment and at the end of the follow-up period, the minimum fibrous cap thickness10,11,17–19 in the control group was lower than that in the PCSK9i group (pooled WMD = −18.647, 95% CI: −27.381 to −9.912, μm), and variation in WMD attributable to heterogeneity was not detected (Figure 3(a)). In addition, the maximum lipid arc10,11,17–19 was significantly higher in the control group than in the PCSK9i group (pooled WMD = 11.760, 95% CI: 7.439–16.080, °), and variation in WMD attributable to heterogeneity was not detected (31.5%) (Figure 3(b)). However, the atheroma volume11–13,15,20 showed no significant difference between the control and PCSK9i groups (pooled WMD = 3.777, 95% CI: −8.282 to 15.837, mm3), and variation in WMD attributable to heterogeneity was not detected (10.3%) (Figure 3(c)). There was a substantial difference in the assessment results of plaque burden between modalities, especially invasive and noninvasive assessments (e.g. resolution, variability, and analysis segments); therefore, the CTA study was excluded. However, even after excluding the CTA study, the assessment results showed no significant difference between the control and PCSK9i groups (pooled WMD = 3.143, 95% CI: −8.935 to 15.222, mm3) (Supplemental Figure 3). In contrast, the maximum lipid-core burden index within 4 mm (maxLCBI4 mm)12,17,21 was significantly higher in the control group than in the PCSK9i group (pooled WMD = 49.453, 95% CI: 25.859–73.046), and variation in WMD attributable to heterogeneity was not detected (Figure 3(d)). In summary, intravascular imaging indicated that PCSK9i, including RNA interference of PCSK9, had positive effects on plaque regression.
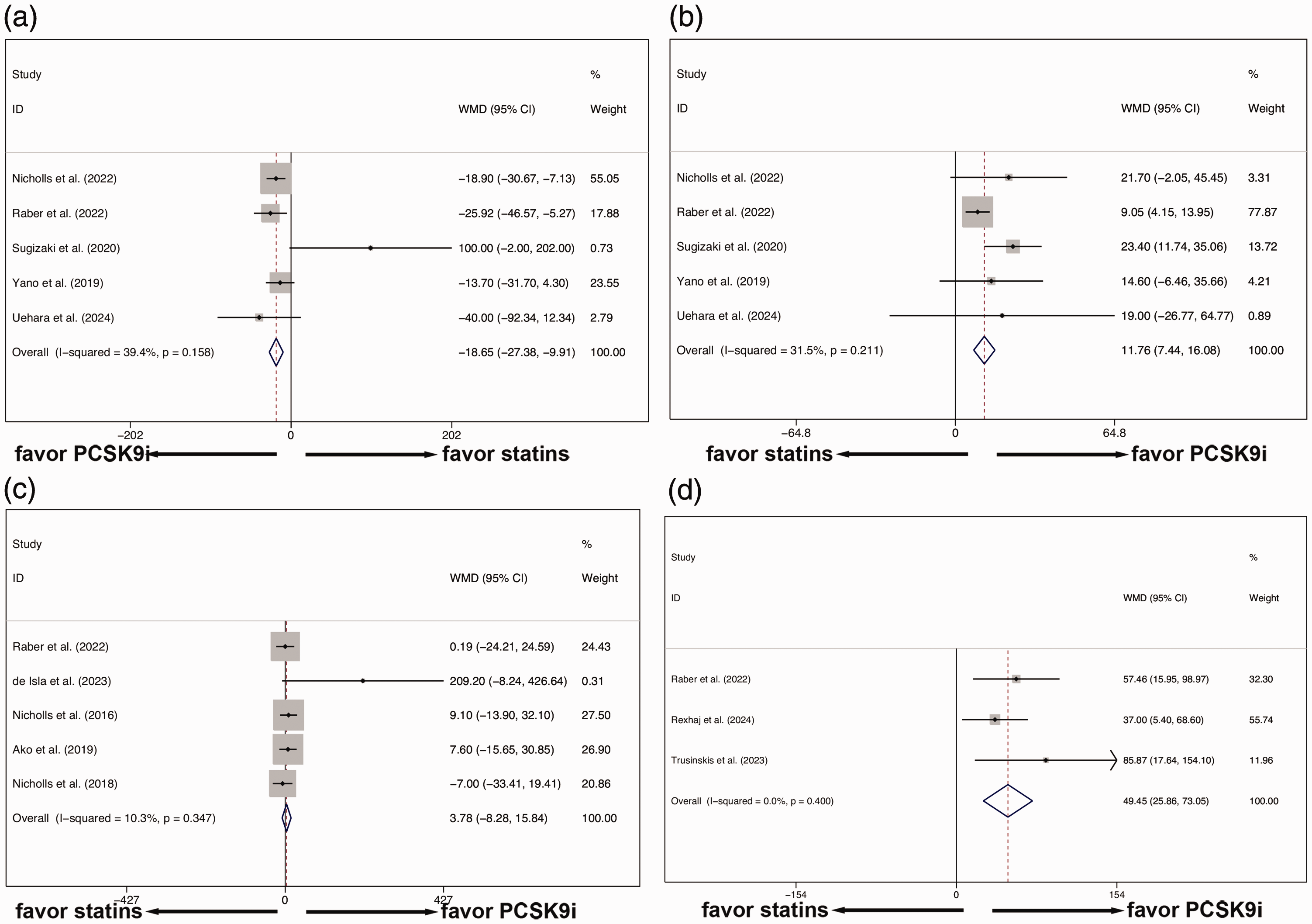
Effects of PCSK9i treatment on plaque burden and plaque regression. (a) The minimum fibrous cap thickness in the control group was lower than that in the PCSK9i group. (b) The maximum lipid arc was significantly higher in the control group than in the PCSK9i group. (c) The atheroma volume showed no significant difference between the control and PCSK9i groups and (d) the maxLCBI4 mm was significantly higher in the control group than in the PCSK9i group. PCSK9i: proprotein convertase subtilisin/kexin type 9 inhibitors; maxLCBI4 mm: maximum lipid-core burden index within 4 mm.
Effects of PCSK9i treatment on plaque regression in the AMI and stable CAD subgroups
Considering that the etiology of the included studies was characterized as two main types—AMI and stable CAD—we further explored the effects of PCSK9i treatment on plaque regression in the AMI and CAD subgroups. In the AMI subgroup, the minimum fibrous cap thickness in the control group was lower than that in the PCSK9i group (pooled WMD = −19.523, 95% CI: −28.290 to −10.756, μm); however, in the stable CAD subgroup, the minimum fibrous cap thickness in the control group was higher than that in the PCSK9i group (pooled WMD = 100, 95% CI: −2 to 202, μm) (Figure 4(a)). In addition, in both AMI and stable CAD subgroups, the maximum lipid arc was significantly higher in the control group than in the PCSK9i group (pooled WMD = 9.909, 95% CI: 5.258–14.560, and pooled WMD = 23.400, 95% CI: 11.736–35.064, respectively) (Figure 4(b)). In the AMI subgroup, the atheroma volume in the control group showed no significant difference compared with that in the PCSK9i group (pooled WMD = 4.074, 95% CI: −12.759 to 20.906, mm3). Similarly, in the stable CAD subgroup, the atheroma volume in the control group showed no significant difference compared with that in the PCSK9i group (pooled WMD = 3.465, 95% CI: −13.822 to 20.751, mm3) (Figure 4(c)). Furthermore, after excluding the CTA study, the atheroma volume showed no significant difference between the control and PCSK9i groups (pooled WMD = 2.156, 95% CI: −15.185 to 19.497, mm3) (Supplemental Figure 4). In summary, subgroup analysis suggested that AMI patients can potentially benefit from PCSK9i treatment in terms of plaque regression.
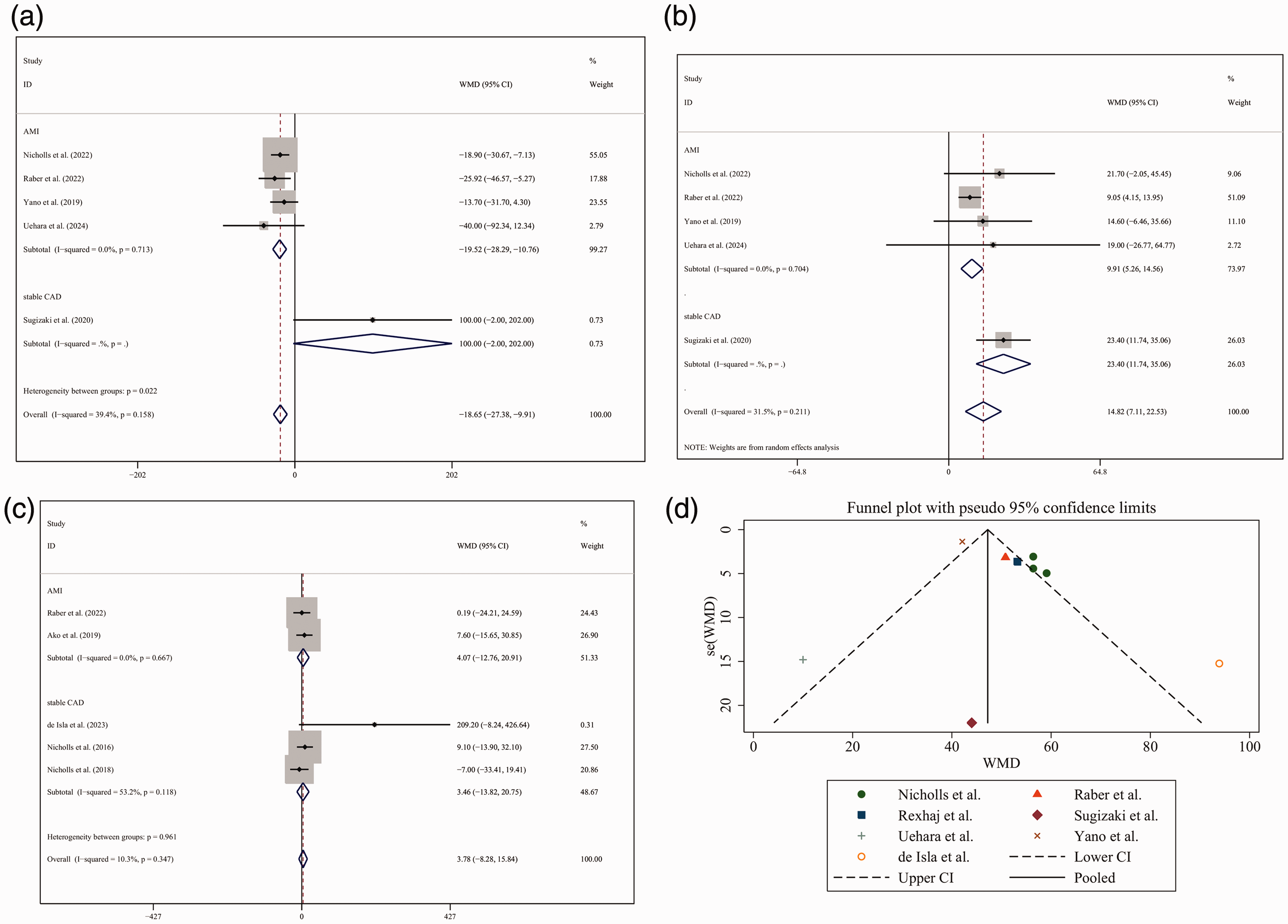
Effects of PCSK9i treatment on plaque regression in the AMI and stable CAD subgroups. (a) In the AMI subgroup, the minimum fibrous cap thickness in the control group was lower than that in the PCSK9i group; however, in the stable CAD subgroup, the minimum fibrous cap thickness was higher in the control group than in the PCSK9i group. (b) In both AMI and stable CAD subgroups, the maximum lipid arc was significantly higher in the control group than in the PCSK9i group. (c) In both AMI and stable CAD subgroups, the atheroma volume in the control group showed no significant difference compared with that in the PCSK9i group and (d) Funnel plot showing publication bias. PCSK9i: proprotein convertase subtilisin/kexin type 9 inhibitors; AMI: acute myocardial infarction; CAD: coronary artery disease.
Publication bias
The funnel plot was symmetrical, suggesting no publication bias in the included studies (Figure 4(d)). Egger’s test did not indicate any significant small-study effects (p > 0.05).
Discussion
This systematic review and meta-analysis of 11 clinical studies involving 3846 participants with coronary atherosclerosis revealed that statin therapy combined with PCSK9i significantly reduced plaque volume compared with statin therapy alone or statin therapy plus placebo (control treatment). This finding was consistent across subgroups based on the type of PCSK9i or statin used. The combination therapy also significantly reduced the LDL-C and Lp(a) levels compared with control treatment. However, the HDL-C level was significantly elevated in the PCSK9i group. The primary finding of this meta-analysis is that statin therapy combined with PCSK9i reduces coronary plaque volume and increases minimum fibrous cap thickness, which is consistent with the finding of previous studies investigating the effects of aggressive lipid-lowering therapies on coronary atherosclerosis. The GLAGOV trial, 13 one of the largest RCTs included in this meta-analysis, demonstrated that the addition of evolocumab to statin therapy resulted in a greater decrease in the plaque volume compared with statin therapy alone. Similarly, the ODYSSEY J-IVUS trial 20 reported that alirocumab combined with statin therapy led to a significant reduction in the plaque volume compared with statin monotherapy in Japanese patients with CAD. The observed reduction in the plaque volume with the combination of statins and PCSK9i may be attributed to the potent LDL-C-lowering effects of these therapies. PCSK9i, including mAbs and siRNA-based therapies, substantially reduce LDL-C levels by increasing the expression of LDL receptors on the surface of hepatocytes. 22 This mechanism of action complements the effects of statins, which inhibit HMG-CoA reductase and reduce LDL-C synthesis. 23 In addition, the level of HDL-C, the so-called good cholesterol, was higher in the PCSK9i group. Moreover, Lp(a), which is gradually gaining importance in atherosclerosis, showed significant correlation with PCSK9i treatment. The synergistic effect of these two classes of drugs on LDL-C lowering may contribute to the reduction in plaque volume observed in this meta-analysis. In addition to their effects on LDL-C, statins and PCSK9i may also exert pleiotropic effects that contribute to plaque regression. Statins have been shown to reduce inflammation, improve endothelial function, and enhance the stability of atherosclerotic plaques. 24 Similarly, PCSK9i have been suggested to possess anti-inflammatory properties, as evidenced by the reduction in C-reactive protein (CRP) levels observed in this meta-analysis and previous studies. 25 Although the circulating CRP levels did not change, PCSK9i reduced intra-plaque inflammation (e.g. reduction in tumor necrosis factor-α and interleukin-6 levels) and enhanced plaque stability (e.g. increase in collagen content and sirtuin 3 levels). The anti-inflammatory effects of these therapies may play a role in reducing the plaque volume and improving clinical outcomes in patients with coronary atherosclerosis. This study revealed that the differential efficacy of PCSK9i between stable CAD and AMI subgroups can be attributed to distinct pathophysiological and cardiovascular risk characteristics between these conditions. The potential mechanism could be owing to differences in atherosclerotic plaque stability, baseline LDL-C levels, inflammatory response, and patients’ metabolic responses.
The findings of this meta-analysis have important implications for the management of patients with coronary atherosclerosis. Current guidelines recommend statins as the first-line therapy for LDL-C lowering and cardiovascular risk reduction. However, some patients may not achieve optimal LDL-C levels or may continue to experience cardiovascular events despite statin therapy. The addition of PCSK9i to statin therapy may provide an effective option for these patients, as demonstrated by the reduction in plaque volume and LDL-C levels in the current meta-analysis. The use of intracoronary imaging techniques, such as IVUS and OCT, may help identify patients who are most likely to benefit from the combination therapy and guide treatment decisions. Our study adds significant value by including five additional trials (total 11 vs. 6 in the prior meta-analysis) and extending the follow-up duration (up to 78 vs. 24 weeks). We also stratified the outcomes by clinical presentation (acute coronary syndrome vs. chronic coronary syndrome) and analyzed plaque composition (e.g. fibrous cap thickness), which were not addressed in the previous work.
Limitations
Several limitations of this meta-analysis should be acknowledged. First, there was substantial heterogeneity among the included studies in terms of patient characteristics, intervention protocols, and imaging methods used to assess plaque volume. Although subgroup analyses did not identify any significant sources of heterogeneity, the variability among studies may limit the generalizability of our findings. Second, most of the included studies had a relatively short follow-up duration, and the long-term effects of statin therapy combined with PCSK9i on plaque volume and clinical outcomes remain uncertain. Third, the assessment of plaque volume using IVUS or OCT may not fully capture the complex morphology and composition of atherosclerotic plaques, which may have important implications for plaque stability and clinical events. Despite these limitations, the present meta-analysis provides the most comprehensive evidence to date on the efficacy and safety of statin therapy combined with PCSK9i in the treatment of coronary atherosclerosis. The findings support the use of this combination therapy as a promising approach to reduce plaque volume and improve lipid profiles in patients with CAD. However, further research is needed to confirm these findings and address the limitations of the current evidence. We extracted absolute plaque volume changes (not percentages) from the original studies. For example, Nicholls et al. 13 (GLAGOV) reported a between-group difference of −5.8 mm³, which aligns with our pooled estimate (WMD −3.8 mm³, 95% CI −8.3 to 15.8). Variability across studies (e.g. follow-up duration and baseline LDL-C) likely explains the nonsignificant pooled result.
Future studies should investigate the long-term effects of statin therapy combined with PCSK9i on plaque volume, plaque composition, and clinical outcomes in larger and more diverse patient populations. The optimal duration and intensity of the combination therapy also need to be established, along with the most appropriate patient subgroups who are likely to benefit from this approach. Additionally, the cost-effectiveness of the combination therapy should be evaluated, considering the high cost of PCSK9i compared with statins. Post-study registration may have introduced bias to our results and is a limitation of this study.
Conclusions
This meta-analysis demonstrates that statin therapy combined with PCSK9i significantly reduces coronary plaque volume and improves lipid profile parameters compared with statin therapy alone or statin therapy plus placebo in patients with coronary atherosclerosis. These findings support the use of statin therapy combined with PCSK9i as a promising approach for the management of coronary atherosclerosis, particularly in AMI patients who do not achieve optimal LDL-C levels or continue to experience cardiovascular events despite statin therapy. Further research is needed to confirm these findings, establish the long-term efficacy and safety of the combination therapy, and identify the most appropriate patient subgroups for this approach.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251361962 - Supplemental material for Multimodal assessment of treatment with proprotein convertase subtilisin/kexin type 9 inhibitors combined with statins for regulating coronary artery plaque regression in patients with chronic/acute coronary syndrome: A meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605251361962 for Multimodal assessment of treatment with proprotein convertase subtilisin/kexin type 9 inhibitors combined with statins for regulating coronary artery plaque regression in patients with chronic/acute coronary syndrome: A meta-analysis by Dexiao Yuan, Ting Cheng, Zhihua Cao, Fang Wang and Yongpeng Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251361962 - Supplemental material for Multimodal assessment of treatment with proprotein convertase subtilisin/kexin type 9 inhibitors combined with statins for regulating coronary artery plaque regression in patients with chronic/acute coronary syndrome: A meta-analysis
Supplemental material, sj-pdf-2-imr-10.1177_03000605251361962 for Multimodal assessment of treatment with proprotein convertase subtilisin/kexin type 9 inhibitors combined with statins for regulating coronary artery plaque regression in patients with chronic/acute coronary syndrome: A meta-analysis by Dexiao Yuan, Ting Cheng, Zhihua Cao, Fang Wang and Yongpeng Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605251361962 - Supplemental material for Multimodal assessment of treatment with proprotein convertase subtilisin/kexin type 9 inhibitors combined with statins for regulating coronary artery plaque regression in patients with chronic/acute coronary syndrome: A meta-analysis
Supplemental material, sj-pdf-3-imr-10.1177_03000605251361962 for Multimodal assessment of treatment with proprotein convertase subtilisin/kexin type 9 inhibitors combined with statins for regulating coronary artery plaque regression in patients with chronic/acute coronary syndrome: A meta-analysis by Dexiao Yuan, Ting Cheng, Zhihua Cao, Fang Wang and Yongpeng Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-4-imr-10.1177_03000605251361962 - Supplemental material for Multimodal assessment of treatment with proprotein convertase subtilisin/kexin type 9 inhibitors combined with statins for regulating coronary artery plaque regression in patients with chronic/acute coronary syndrome: A meta-analysis
Supplemental material, sj-pdf-4-imr-10.1177_03000605251361962 for Multimodal assessment of treatment with proprotein convertase subtilisin/kexin type 9 inhibitors combined with statins for regulating coronary artery plaque regression in patients with chronic/acute coronary syndrome: A meta-analysis by Dexiao Yuan, Ting Cheng, Zhihua Cao, Fang Wang and Yongpeng Wang in Journal of International Medical Research
Footnotes
Acknowledgements
Not applicable.
Author contributions
Dexiao Yuan and Yongpeng Wang conducted the studies, collected the data, and drafted the manuscript. Ting Cheng and Zhihua Cao designed and performed the statistical analysis. Fang Wang helped in drafting the manuscript. All authors have read and approved the final manuscript.
Consent for publication
Not applicable.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
This meta-analysis did not require ethical approval, as it involved the synthesis of data from previously published studies. No new human participants were involved, and no primary data were collected. All included studies were assumed to have obtained prior ethical approval in accordance with applicable standards.
Funding
This study was supported by the project of Shanghai Changning District Science and Technology Commission (CNKW2024Y50).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
