Abstract
While parenteral prostacyclin (pPCY) therapy, delivered either subcutaneously or intravenously, is recommended for pulmonary arterial hypertension patients with severe or rapidly developing disease, some patients refuse this treatment. This study aimed to understand, directly from patients with pulmonary arterial hypertension, why pPCY was refused and, in some cases, later accepted. Interviews were conducted with 25 pulmonary arterial hypertension patients who previously refused pPCY therapy (Group A: Refused/Never initiated (n = 9) and Group B: Refused/Initiated (n = 16)). Patients in both groups believed that pPCY could improve their symptoms, slow disease progression, and provide them a greater ability to perform activities. Reasons for refusal included concern over side effects and the perceived limitations of pPCY on daily activities. Group A perceived their decision as a balance between quality of life and prolonging life and most acknowledged they would reconsider pPCY if other treatment options were exhausted. Group B cited they initiated therapy due to a worsening of symptoms, disease progression, to improve quality of life, to be there for their family, or a desire to live. Following initiation, Group B indicated their experience met expectations with reduced symptoms, slowed disease progression, and perception of improved survival; concerns related to pPCY were described as manageable. Given the efficacy of pPCY therapy, clinicians should apply knowledge of these findings in clinical practice. Patients noted improvements to parenteral pump technologies to include smaller size, water resistance, and implantability may increase their acceptance of this modality. Development efforts should focus on technologies that increase the acceptance of pPCY when indicated.
Keywords
Pulmonary arterial hypertension (PAH) is a severe chronic and progressive disease characterized by remodeling of the pulmonary vasculature with vasoconstriction and vascular smooth muscle proliferation that leads to increased pulmonary vascular resistance, right ventricular failure, and, if left untreated, ultimately death.1,2 In the United States and Europe, parenteral prostacyclins (pPCYs) are widely used to treat high-risk patients with severe or rapidly developing PAH and are often added in patients who do not respond to mono- or combination therapy.3,4 While pPCYs, delivered either subcutaneously or intravenously (IV), are associated with improved symptoms and outcomes,5–7 and guidelines support their use, 2 many patients do not receive pPCY therapy prior to their death. Unfortunately, the reasons behind this underutilization are not well understood.
A number of studies indicate that less than half of PAH patients receive pPCY therapy before death or clinical deterioration.8–11 In the Registry to Evaluate Early and Long-term PAH Disease Management (REVEAL Registry), only 43% of patients who died during the study period were receiving IV prostacyclin as monotherapy or combination therapy prior to death due to any cause. The authors also found that only 51% of patients were receiving pPCY therapy when they deteriorated to World Health Organization Functional Class IV. 8
Relatively little is known as to why pPCY therapy is not more commonly administered in patients with advanced PAH. A chart review of PAH patients who died after being treated at a large pulmonary hypertension center (n = 101) found that patients who died without pPCY therapy (N = 40) were not considered candidates (60%), had no documented evaluation for pPCY therapy (25%), or refused pPCY therapy (15%). 10 While data from one center’s experience provides some evidence, the underlying reasons for patient refusal of pPCY therapy are not well documented. Therefore, it is important to engage with patients directly to understand why they decided against starting pPCY therapy.
This patient-centered approach to examining reasons for refusal is in line with the European Society of Cardiology/European Respiratory Society guidelines for PAH, which stress that final treatment decisions should be made in consultation with the patient. 3 In addition, CHEST guidelines on pharmacologic therapy for PAH state, “all treatment decisions should be informed by patient preferences, goals, and assessments of health-related quality of life.” 4 The “patient voice” has also been emphasized by recent regulatory and policy directives provided by the Food and Drug Administration and others,3,8,12–14 particularly in PAH patients. 15 Consequently, the purpose of this study was to understand, directly from patients with PAH, why pPCY therapy was refused and, in some cases, later initiated.
Methods
Study design and participants
This qualitative study was conducted with PAH patients who had refused pPCY therapy. The study protocol and interview materials were approved by a central institutional review board. Patients were unaware of the study sponsor and were recruited from a third party’s internal database of patients who had opted to participate in qualitative research studies. Potential participants were contacted via email by the third party and asked to complete an online screening questionnaire. Eligible patients were ≥18 years of age, were diagnosed with PAH, and had been recommended pPCY therapy in the previous two years. Patients who met the eligibility criteria then completed a 45-min telephone interview conducted by a trained proctor.
Telephone interview
Patients answered a series of questions designed to assess their decision-making process when asked to consider pPCY therapy. The interview was divided into six sections: (1) decision-making process upon initial consideration of pPCY therapy; (2) perceptions of pPCY therapy; (3) primary reason for treatment decision; (4) comparison of perception to experience (for patients who eventually initiated pPCY therapy); (5) consequences of declining pPCY therapy; and (6) perceptions of new pPCY pump features. Note that questions used in the patient interviews were designed for this particular study and had not been previously validated. The complete interview discussion guide is included in the supplementary material.
Cohort creation and analysis
Respondents were evaluated overall and divided into two groups for analysis: those who refused pPCY therapy and never initiated it (Group A: Refused/Never initiated) and those who initially refused pPCY therapy but subsequently initiated it (Group B: Refused/Initiated).
Analysis
Basic descriptive statistical analysis techniques were used to summarize responses to quantitative questions and qualitative activities. Differences between continuous variables were tested using independent samples T-tests and for categorical variables chi-square tests were performed.
Results
Patient characteristics
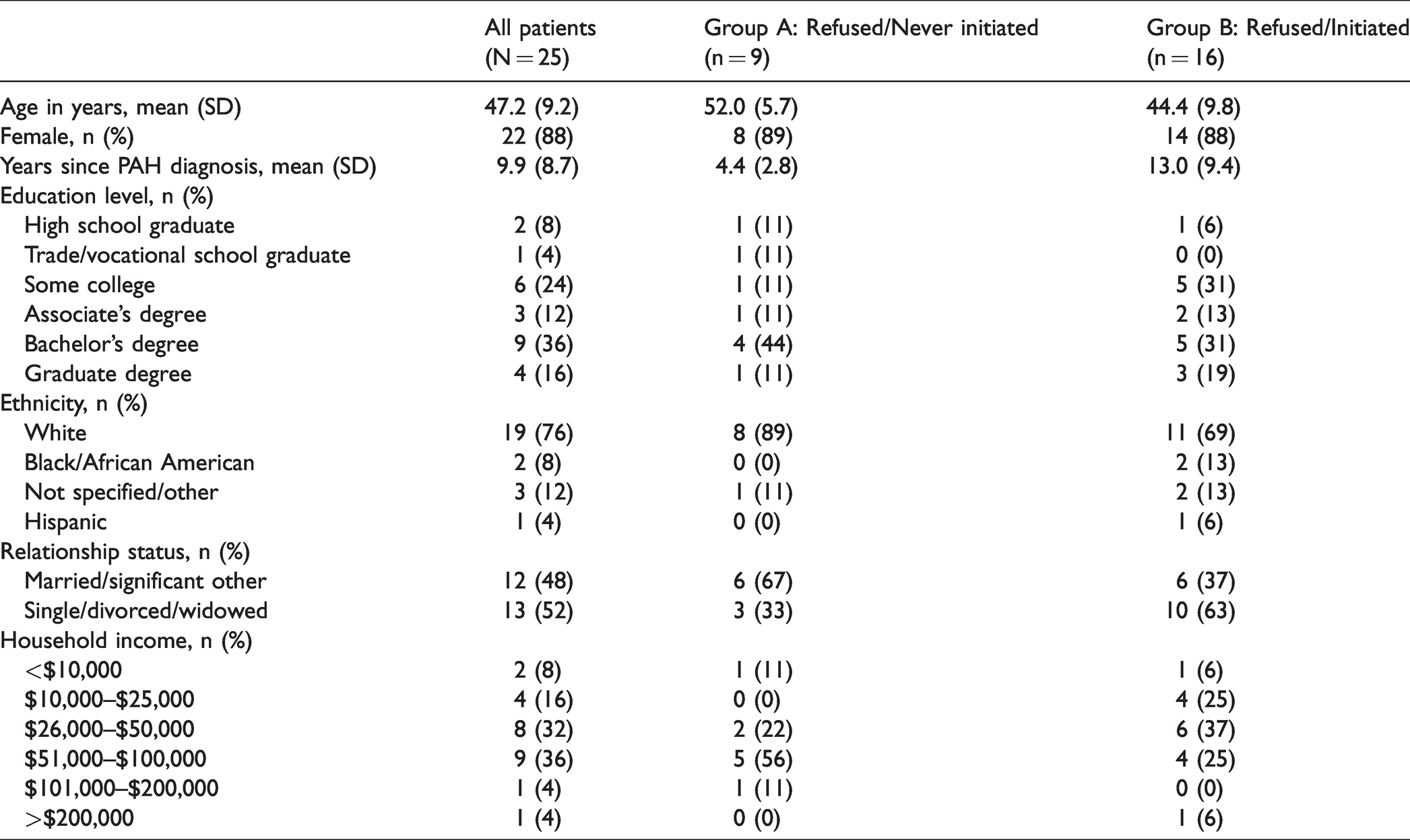
The study sample consisted of 25 PAH patients who indicated that they had refused pPCY therapy within the past two years. Table 1 provides a summary of the patients’ demographics. Patients in the overall sample had a mean (SD) age of 47.2 (9.2) years, were primarily female (88%), white (76%), and had been diagnosed with PAH for 9.9 (8.7) years. Of the 25 patients, 36% (n = 9) were in Group A: Refused/Never initiated and 64% (n = 16) were in Group B: Refused/Initiated. Patients in Group A: Refused/Never initiated were significantly older (p < .05) on average (mean = 52.0 years (5.7)) than patients in Group B: Refused/Initiated (mean = 44.4 years (9.8)). There no was difference in the proportion of females in the cohorts (p > .05). However, there was a significant difference (p < .05) between the two cohorts with respect to years since PAH diagnosis with Group B: Refused/Initiated having been diagnosed on average 13 years ago, and Group A: Refused/Never initiated diagnosed on average only four years ago. Most patients (78% in Group A: Refused/Never initiated and 69% in Group B: Refused/Initiated) reported being assessed as having severe PAH by their healthcare provider at initial diagnosis.
Patient characteristics.
Decision-making process upon initial consideration of parenteral prostacyclin therapy
At the time their provider recommended pPCY therapy, 68% of patients perceived their health as poor/very poor, 24% as fair, and 8% as good. Similarly, 68% of patients reported that their provider also told them their condition was severe/very severe and 12% reported moderate, while 20% did not provide this information.
Patients most commonly reported being told by their provider that starting pPCY therapy was necessary due to the following reasons: (1) their pressures/echo findings or the severity of their disease, and that it was the “gold standard”; (2) that it is the most direct/consistent and aggressive therapy; and (3) that it would extend their lives. When asked if their provider shared information about the positive and negative effects of therapy, 44% (22% of Group A: Refused/Never initiated; 56% of Group B: Refused/Initiated) of patients indicated that both were mentioned and 44% (66% of Group A: Refused/Never initiated; 31% of Group B: Refused/Initiated) said only positive effects were communicated to them (12% did not respond). The potential benefits communicated to patients included improving symptoms, functionality, and disease progression, while the negative effects pertained to the side effects of the therapy.
When describing their initial reaction to being asked to consider pPCY therapy, 100% of patients described having a negative reaction to their provider’s recommendation. These reactions were marked frequently with terms such as “scared,” “fear,” “permanent,” “interfere,” and “being attached.” Moreover, most patients were worried about side effects associated with parenteral administration and the impact pPCY therapy would have on their daily lives, specifically by reducing their freedom or ability to engage in daily and leisure activities.
Patients were asked what online research or other resources they used when deciding on pPCY therapy. The most common sources included other patients with PAH, family and friends, medical providers, support groups (e.g. Pulmonary Hypertension Association), and social media. In fact, 100% of patients in Group B: Refused/Initiated indicated that they sought out additional information regarding pPCY therapy, while only 56% of patients in Group A: Refused/Never initiated did any additional research.
Specific topics patients researched included medication side effects, therapy and administration options, financial resources to pay for therapies, general disease information, and infection risk/central line information. Interestingly, 48% of patients (22% Group A: Refused/Never initiated; 69% Group B: Refused/Initiated) stated they reached out to others on pPCY therapy, either online or through support groups, as part of their research.
Patient perceptions of parenteral prostacyclin therapies
When asked about the perceived benefits of pPCY therapy, all but three patients (Group A: Refused/Never initiated, n = 2; Group B: Refused/Initiated, n = 1) were able to name at least one perceived benefit. The most mentioned benefits included an expectation that pPCY therapy would improve their symptoms, slow disease progression, increase their ability to perform activities, and improve their quality of life (QoL).
Despite the acknowledgment of these benefits, all 25 patients expressed fear and concern about the impacts of pPCY therapy. All patients mentioned at least two concerns, of which the most common included the impact of wearing an external pump for an interminable amount of time on their daily life, an inability to swim/challenges showering, administering the medication, the potential for line infections for intravenous use and site pain for subcutaneous use, and their ability to work or care for children while on pPCY therapy.
Primary reason for treatment decision
Patients were asked how they ultimately decided to initiate pPCY therapy or not, and a summary of their responses is provided in Table 2. Patients in Group A: Refused/Never initiated perceived their decision as a balance between QoL and prolonging life. Most would reconsider pPCY therapy if their disease started to progress rapidly or if they felt there were no other options. Patients in Group B: Refused/Initiated most frequently stated they did so due to a worsening of symptoms, disease progression, to improve QoL, to be there for their family, or due to a desire to live.
Patient reasoning for parenteral prostacyclin therapy decision.

Comparison of perception to experience with parenteral prostacyclin therapy
Patients in Group B: Refused/Initiated (n = 16) were asked about their perceptions regarding pPCY before starting therapy as well as their experience after starting therapy. Six patients reported they had initiated IV therapy and four patients reported they had initiated subcutaneous (SC) therapy. Six patients reported they had initiated an IV or SC therapy and subsequently switched to the alternative. By and large patients’ actual experiences with pPCY treatment matched their perceptions with regard to potential benefits of prostacyclin therapy including reduced symptoms, slowed disease progression, and increased survival. All patients that responded indicated that pPCY therapy had met their expectation with respect to its impact on survival, while 31% of patients indicated that their actual length of survival had exceeded their expectations. In addition, patients anticipated the occurrence of common side effects associated with pPCY therapy, including subcutaneous site-pain and line-related issues, and stated their experience was as expected. Sixty-three percent reported at least one unplanned hospitalization or emergency room (ER) visit due to their pPCY therapy. All patients were on an IV prostacyclin and cited central line-related complications such as line repairs, line clots, or infections. With respect to QoL, 56% of patients anticipated that pPCY therapy would have a positive impact on their QoL, 31% believed it would have a negative impact, and 3% of patients believed it would have positive and negative effects. When asked about their actual experience, 81% of patients suggested that their QoL had improved since starting pPCY therapy. Of the seven patients who perceived managing/physically wearing the pump would negatively impact their QoL, five of the patients characterized their actual experience as positive stating it was “less of a big deal,” or just “a new routine,” and something you “learn to live with.” With respect to the expected experience with administration of pPCY treatment, most patients expressed significant reservations and even fear. After starting therapy, all but one patient that previously expressed concern indicated that with training and experience the self-administration was not difficult and was not a major issue.
Consequences of declining parenteral prostacyclin therapy
Patients were asked whether they experienced unplanned hospitalizations or ER visits related to their PAH in the time since declining pPCY therapy. Patients had a mixed experience across both groups, with 32% (45% of Group A: Refused/Never initiated; 25% of Group B: Refused/Initiated) reporting at least one unplanned hospitalization or ER visit since declining pPCY therapy.
Perceptions of new pump/device features
Patients were asked to rate (1 = not at all impactful; 5 = extremely impactful) the extent to which several new pump/device features would impact their interest in utilizing a pPCY therapy (Fig. 1). The new pump/device features patients reported to be the most impactful included water resistance, a pre-filled pump, implantability, and smaller size. The ability to have a pre-filled pump was rated the most impactful by Group A: Refused/Never initiated, while water resistance was rated the most impactful by Group B: Refused/Initiated. When asked which pump feature(s) would change patients’ minds about declining a pPCY therapy, the most frequently mentioned features were implantability (60%) followed by water resistance (36%).
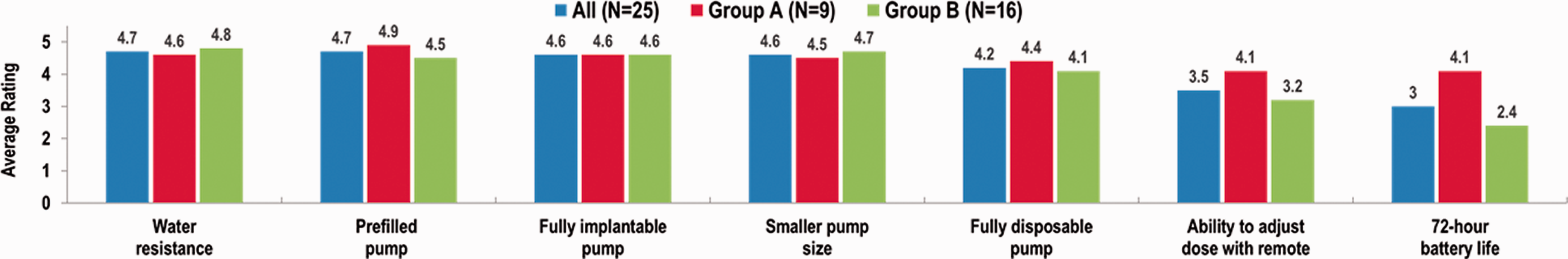
Impact of new pump/device features on patients’ interest in using a parenteral prostacyclin therapy.
Discussion
The current investigation represents one of the first attempts to understand the thought process and rationale surrounding PAH patients’ decisions to refuse or initiate pPCY therapy. The results of this patient-centered qualitative study have identified a number of important themes and considerations.
First, most patients indicated that their provider had brought up pPCY therapy because their condition warranted an aggressive approach. Most patients also acknowledged their poor health at that time, but still initially decided to decline parenteral treatment. Those patients who ultimately started pPCY therapy suggested they did so because “they had no other choice.” It appears that patients perceived their disease had progressed to the point that failure to do anything would result in their premature death. Patients who did not start pPCY therapy indicated they wanted to try an oral therapy first or wanted to wait to use pPCY therapy as a “last resort,” which is not surprising given that these patients had been living with the diagnoses, on average, for four years compared to 13 years in patients who eventually initiated pPCY therapy.
Second, while about half of patients reported being told about both the positive and negative effects of pPCY therapy by their provider, a similar proportion recalled only being told of the positive effects. This may reflect recall bias since the typical discussion around initiation of parenteral therapy typically includes extensive discussion of the complexity and side effects (authors’ viewpoint). A prior single-center study found that in 25% of PAH patients who died without receiving pPCY therapy, their medical record included no documented discussion of why pPCYs were never started. 10 The lack of documentation makes it difficult to ascertain if the rationale to not start pPCYs was due to patient choice, the patient not being an appropriate candidate, or lack of provider recommendation in a timely manner. Additionally, our findings suggest that patients who did more of their own research (e.g. online, support groups, talking to PAH patients on pPCY therapy) were more likely to initiate therapy after initially refusing. Moreover, a large proportion of those who eventually started therapy sought out information directly from their peers who had experience using a pPCY therapy.
Third, the findings of this study suggest that patients’ concerns about starting pPCY therapy primarily centered around its impact on various dimensions of QoL. Patients were worried that the potential side effects of the parenteral route of administration and the pump used to administer prostacyclin therapy could negatively affect their QoL by limiting their freedom and ability to engage in daily activities. However, at the same time, many also expressed that their QoL might improve with pPCY therapy by reducing symptoms and slowing disease progression. In fact, most of the patients in Group B: Refused/Initiated indicated that their QoL improved upon initiating therapy, which is consistent with studies that evaluated QoL following initiation of pPCY therapy.16,17 Moreover, these patients indicated that their initial concerns of the pump affecting their QoL were mostly unwarranted.
Fourth, the study identified several potential characteristics of pump/device features that patients suggested could lead to increased adoption of pPCY therapy. These characteristics included water resistance, a pre-filled pump, implantability, and smaller device size. These findings suggest that continued work is needed in the development of patient-centered technologies with features that help patients to feel unencumbered by their pump/device as they engage in daily activities. However, it is imperative that providers and patients fully weigh the risks and benefits of any new technology when making treatment decisions. This is particularly important for newly introduced medical devices for patients with PAH where enhanced features may improve the patient experience but may come with an increase in safety risks.
While the results from the qualitative study are compelling, they should be considered alongside several caveats. First, the generalizability of the study findings may be limited due to relatively small and select sample of PAH patients and an even smaller number of patients that refused and never initiated pPCY therapy. Further research including a larger and more heterogeneous sample is required. Second, the small sample and qualitative nature of the study limits the ability to test for differences between the two patient cohorts. Third, the retrospective nature of the study may introduce some recall bias, particularly when patients in Group B: Refused/Initiated were asked to recall their decision to initially refuse pPCY therapy. However, an attempt was made to mitigate this bias by including patients that had been recommended pPCY therapy within the previous two years. Fourth, while this study examined patients who were deemed appropriate candidates for pPCY therapy by their providers, there are scenarios where a PAH patient may not be considered an appropriate candidate by their healthcare provider due to comorbidities or a history of non-compliance with medications.10 Given that healthcare providers frequently determine that patients are not candidates for pPCY therapy additional research examining treatment decisions from the clinician’s perspective is also needed. Fifth, the current study focused on one mode of treatment administration at the exclusion of others. As more therapies have become available to treat PAH, patients have the option to choose other clinically indicated therapies over pPCY therapy. Finally, the questions used in the patient qualitative interviews were designed specifically for this study and had not been previously validated. Certain questions pertaining to new pump and device features may have led patients to endorse certain aspects of PAH devices in development. While some study authors are employed by companies that design, develop, and commercialize parenteral devices for PAH, and thus are keenly interested in patient feedback on device features, the authors do not endorse any particular product or product feature. As stated earlier, device selection and PAH medication choice encompasses a variety of factors and should be at the discretion of the healthcare provider and the patients they treat.
Despite these limitations, the results of this study have several important clinical implications. Given that patients who eventually initiated pPCY therapy reported being told their disease had significantly progressed and that further delaying treatment could have serious consequences, clinicians should consider stressing the severity of patients’ conditions when recommending pPCY therapy. The use of newly developed risk assessment tools could aid in the identification of high-risk patients and objectively communicate that risk to the patient. 16 In addition to the objective assessment of disease severity, clinicians should consider addressing patient perceptions of pPCY therapy’s negative impact on QoL. It is clear that some patients have a preconceived notion that their QoL, as it relates to mobility and independence, may be negatively impacted by wearing an external pump. However, clinicians should communicate that many patients who start therapy report that their earlier concerns ultimately became less significant and they in essence “get used to” the pump. At the same time, clinicians should consider highlighting that most patients report that other aspects of their QoL, specifically symptoms and impact of their disease, often improve with pPCY therapy. Taken together, the results of the current study highlight the importance of taking a patient-centered approach to understanding patients’ treatment decisions. Given the efficacy of pPCY therapy, the application of these findings to discussions around initiating therapy in clinical practice may increase the likelihood that patients will move to pPCY therapy when indicated.
Supplemental Material
sj-pdf-1-pul-10.1177_20458940211046761 - Supplemental material for Reasons for refusing parenteral therapy: a qualitative study of patients with pulmonary arterial hypertension
Supplemental material, sj-pdf-1-pul-10.1177_20458940211046761 for Reasons for refusing parenteral therapy: a qualitative study of patients with pulmonary arterial hypertension by Kellie Morland, Amresh Raina, Abigail Nails, Peter Classi, Martine Etschmaier and Robert P. Frantz in Pulmonary Circulation
Footnotes
Authors’ contributions
All authors contributed to the development of this paper.
Acknowledgements
The authors wish to thank Jason Allaire, PhD of Generativity Health Economics and Outcomes research for providing editorial assistance with this paper.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KM, AN, and PC are employees of and own stock in United Therapeutics. RPF has received fees from Janssen for steering committee and advisory board participation; fees from Liquidia and Gossamer Bio for advisory board participation; and coverage of travel expenses from United Therapeutics for advisory board participation. AR has received research support from United Therapeutics, Liquidia, Actelion, Phase-Bio, and Complexa. ME is an employee of Xcenda, LLC, which received financial support for the conduct of this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by United Therapeutics Corporation.
Guarantor
United Therapeutics Corporation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
