Abstract
Prostacyclin pathway agents are a critical treatment for patients with pulmonary arterial hypertension. Seven prostacyclin pathway agents are available, including agents administered by parenteral infusion, by inhalation, and orally. Pulmonary arterial hypertension patients are now transitioned from one prostacyclin pathway agent to another with increasing frequency. Such transitions require careful downtitration and uptitration to avoid decompensation from rapid withdrawal and to achieve a patient's optimal dose based on efficacy and tolerability. Clinical guidance is especially lacking for transitions involving the newer, oral prostacyclin pathway agents; specifically, selexipag and oral treprostinil. We present three case reports of patients with pulmonary arterial hypertension who underwent one or more transition between parenteral and oral prostacyclin pathway agents, including some transitions that were successful and some that were not. These cases illustrate key considerations, such as titration protocols, patient selection, side effect management, and pharmacokinetics.
Prostacyclin pathway agents (PPAs) represent a mainstay in the treatment of pulmonary arterial hypertension (PAH). 1 Seven PPAs are currently available in the United States, including Flolan (epoprostenol), which is given by continuous intravenous (IV) infusion using a refrigerated pump; Veletri, a thermostable formulation of IV epoprostenol; Ventavis, an inhaled formulation of iloprost; three formulations of treprostinil, including Remodulin, Tyvaso, and Orenitram, which are administered via parenteral infusion, via inhalation, and orally, respectively; and Uptravi (selexipag), a prostacyclin receptor agonist that is given orally. 2
While infused PPAs provide critical therapy for patients with PAH, these agents have well-known drawbacks. 3 Patients treated with IV infusion of PPAs can experience central line infections, sepsis, bleeding, and thromboembolism. 3 Patients who receive subcutaneous (SQ) infusion can experience site pain, site reactions, and infections. 3
Patients now transition between infused and oral PPAs for a variety of reasons, including lifestyle, side effects, or complications of infused therapy, clinical worsening, and hospitalizations during which medications cannot be administered orally. 4 Transitioning between PPAs is inherently complex due to the fragility of patients and the potential for clinical worsening. 5 PPA doses must be carefully uptitrated and downtitrated, symptoms and hemodynamics must be monitored, and clinicians must manage side effects that could prevent effective dosing.5–7
A few studies have examined transitions from one infused PPA to another or between infused and inhaled PPAs. 4 However, there exist only limited data on transitioning patients between infused and oral PPAs.5,7–9 Two prospective studies have been published to date. Chakinala et al. 7 reported results for 33 patients with World Health Organization (WHO) functional class (FC) I or II PAH, classified as low risk, according to the European Society of Cardiology (ESC)/European Respiratory Society (ERS) criteria, who switched from infused to oral treprostinil using a five-day inpatient transition protocol. At 24 weeks post-transition, 31 of 33 patients remained on oral treprostinil. Maestas et al. 5 subsequently reported outcomes of 24 patients with low-risk WHO FC I or II PAH who were transitioned from infused to oral treprostinil using the same inpatient protocol. These patients were followed for two years, at which time 46% had experienced clinical worsening after an average of 557 days on oral therapy; most of these patients transitioned back to parenteral therapy. In this paper, we present three patients who were transitioned one or more times between parenterally infused and oral PPAs. These cases go beyond what was previously reported by Chakinala et al. 7 and Maestas et al. 5 in several important respects. They include not only oral treprostinil but also the newest oral PPA, selexipag. They include patients transitioned from infused to oral PPAs and from oral to infused PPAs. The cases were chosen to illustrate key aspects of such transitions, including the management of side effects, titration, patient selection, background therapy, and pharmacokinetics.
Case 1
Patient 1 is an African American male who was diagnosed with WHO Group 1 PAH (HIV-related) in August 2014, at age 42 years. Right heart catheterization (RHC) revealed mean pulmonary arterial pressure (mPAP) of 58 mmHg, pulmonary vascular resistance (PVR) of 10 Wood units (Wu), and cardiac index (CI) of 1.2 L/min/m2; echocardiography suggested severe right ventricular enlargement; and six-minute walk distance (6MWD) was 124 m. The patient was classified as New York Heart Association FC IV and high risk, with a Registry to Evaluate Early and Long-Term PAH Disease Management (REVEAL) risk score of 12. 10 He was started on IV thermostable epoprostenol, ambrisentan (10 mg QD), and tadalafil (40 mg QD). Epoprostenol was titrated to 58 ng/kg/min, then reduced to 46 ng/kg/min due to severe PPA-related diarrhea and nausea.
The patient improved on triple combination therapy, with RHC in July 2016 showing an mPAP of 26 mmHg, PVR of 1.22 Wu, and CI of 4.01 L/min/m2. However, he experienced repeated line complications, including three hospitalizations for line sepsis and four visits to the emergency room. As a result, the decision was made in November 2016 to transition him to oral selexipag. Prior to transitioning, his 6MWD was 440 m, and he was classified as low-risk (REVEAL risk score of 4).
He was transitioned in a community cardiology practice by the pulmonary hypertension nurse practitioner using the 20-week outpatient protocol shown in Fig. 1. Oral selexipag (starting dose of 200 µg BID) was initiated after his epoprostenol had been titrated down to 8 ng/kg/min. It was discontinued when he reached a dose of 800 µg BID of selexipag. His selexipag dose reached 1600 µg BID in April 2017. Twelve weeks later, RHC showed a mildly increased mPAP (37 mmHg); however, the patient's 6MWD was nearly unchanged at 420 m, and he reported feeling well, so he was followed without further changes. Three months later, his 6MWD had improved to 540 m. The patient has continued triple therapy, with RHC in October 2018 showing an mPAP of 28 mmHg, PVR of 2.20 Wu, CI of 3.0 L/min/m2, and an evaluation in February 2019 showing New York Heart Association FC I to II symptoms with a 6MWD of 530 m.
Transition timeline—Patient 1. Titration of PPA doses during outpatient transition from IV epoprostenol to PO selexipag.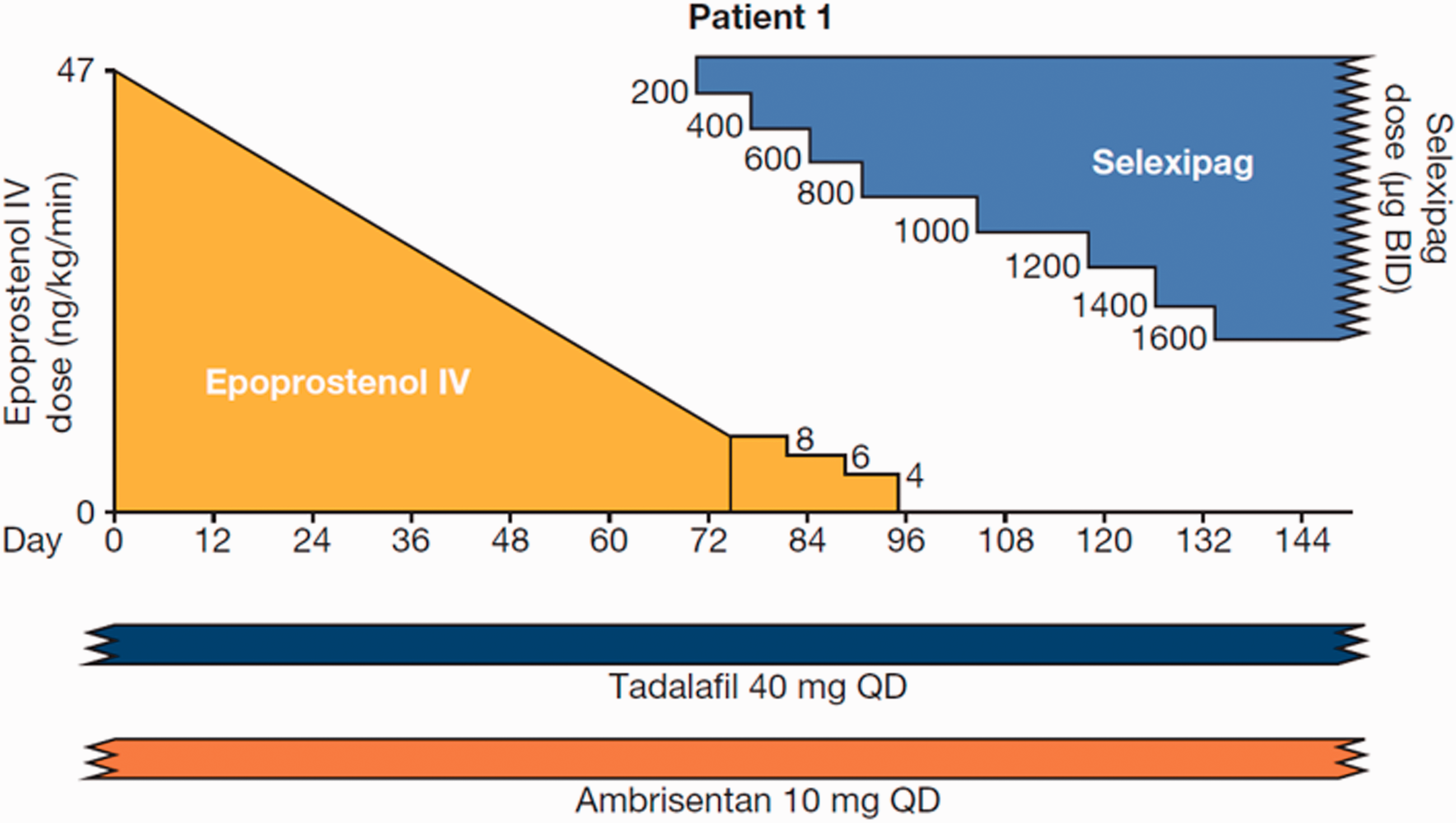
Careful consideration was given prior to transitioning this patient from an IV to an oral PPA. On his prior triple regimen, he had improved hemodynamically with near normalization of his PVR and CI; he also had a substantially improved 6MWD. While this patient's transition was completed prior to wide use of formal risk assessment tools, when the REVEAL tool was applied, his risk category was low just prior to transition. We closely monitored his FC and 6MWD during the transition period.
Case 2
Patient 2 is a Hispanic female who was diagnosed with WHO Group 1 pulmonary arterial hypertension (scleroderma-associated) in March 2014, at age 55 years. She exhibited moderate-to-severe hemodynamic impairment (mPAP: 40 mmHg, PVR: 6.39 Wu, CI: 2.64 L/min/m2), preserved right heart function (right ventricular ejection fraction of 41%), and 6MWD of 329 m. She initiated treatment with SQ treprostinil in April 2014, titrating to 47 ng/kg/min. Her 6MWD improved to 381 m after two months of treatment, and after three months of treatment, RHC revealed a slight improvement in mPAP (36 mmHg) and PVR (5.91 Wu). At this point, she exhibited WHO FC II symptoms and was classified as intermediate risk, according to the ESC/ERS criteria. 1 However, she suffered severe recurrent infusion site pain and asked to switch to oral treprostinil.
Tadalafil (40 mg QD) was added in July 2014 to provide background therapy for the transition. In October 2014, she underwent a four-day transition, following the inpatient protocol published by Chakinala et al.
7
The transition occurred at a leading pulmonary hypertension academic center and involved the collaborative efforts of the pulmonary hypertension nurse practitioner, the pulmonary hypertension physician, the inpatient pharmacist, and bedside nurses on a dedicated pulmonary hypertension inpatient unit. As shown in Fig. 2a, oral treprostinil (TID) was started simultaneously with the first dose reduction of SQ treprostinil; thereafter, the oral dose was increased, and the SQ dose decreased every eight hours. The dose of oral treprostinil was 9 mg TID at discharge and was later increased to 9.5 mg TID.
(a) Transition timeline—Patient 2 (infused to oral). Titration of PPA doses during inpatient transition from SQ treprostinil to PO treprostinil. (b) Transition timeline—Patient 2 (oral to infused). Titration of PPA doses during inpatient transition from PO treprostinil to SQ treprostinil.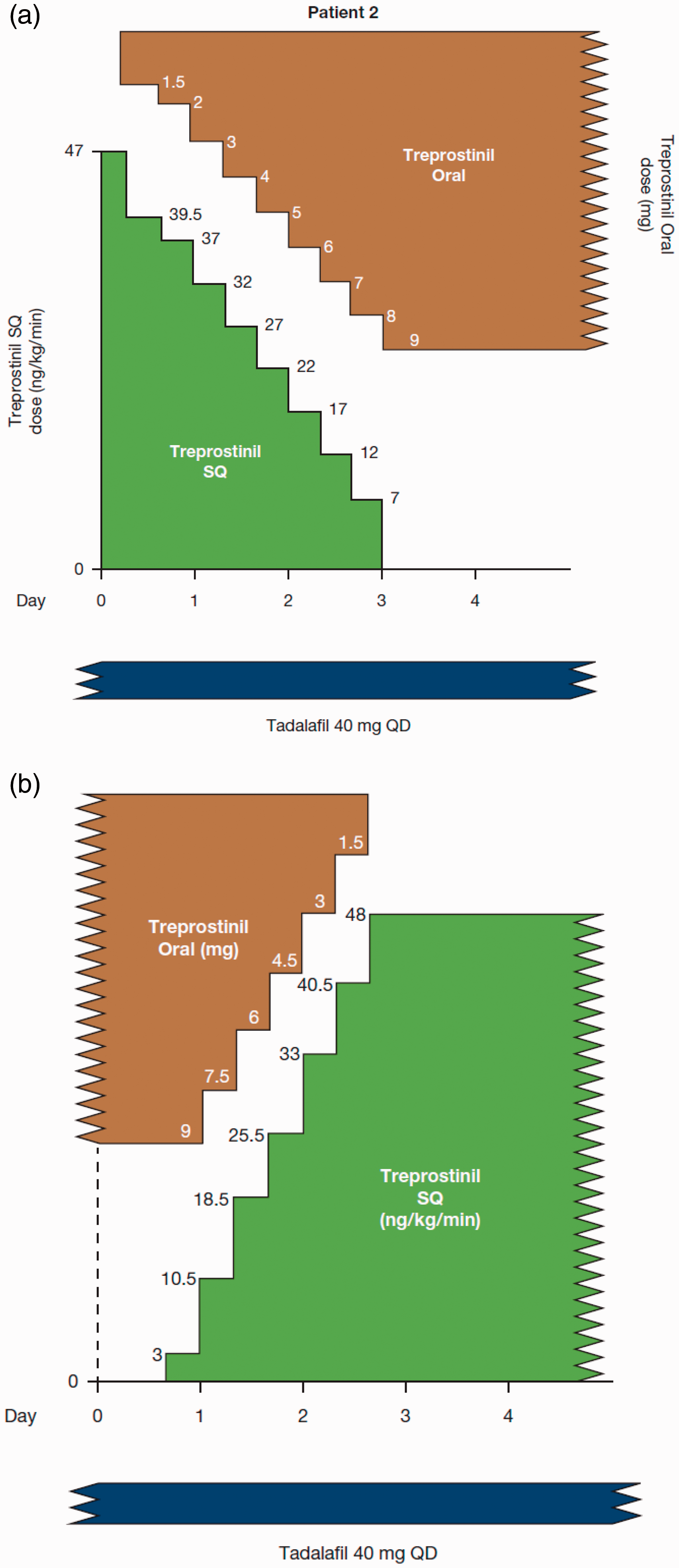
The patient's PAH symptoms were well controlled for several months following transition, and her 6MWD improved to 459 m. However, due to progressively worsening side effects, her dose was reduced to 8.75 mg, and she began skipping doses. She deteriorated to WHO FC IV, with a 6MWD of 203 m and a right ventricular ejection fraction of 30%. In May 2015, RHC showed an mPAP of 58 mmHg, PVR of 18.2 Wu, and CI of 1.79 L/min/m2, placing her in the ESC/ERS high-risk category.
She was hospitalized and transitioned back onto SQ treprostinil over three days, as shown in Fig. 2b. She was discharged at an SQ dose of 48 ng/kg/min, and over several months, she titrated up to 75 ng/kg/min, with improving symptoms. RHC in January 2017 showed improved hemodynamics (mPAP: 28 mmHg; PVR: 5.1 Wu; CI: 2.97 L/min/m2). Her 6MWD has improved to 330 m, and she continues on SQ treprostinil and oral tadalafil.
Case 3
Patient 3 is a Caucasian-Asian female who was diagnosed with WHO Group 1 pulmonary arterial hypertension (methamphetamine-associated) in October 2006, at age 29 years. She exhibited WHO FC III symptoms, with RHC showing an mPAP of 56 mmHg, PVR of 13 Wu, and CI of 1.47 L/min/m2. During 2007 and 2008, she initiated oral sildenafil (20 mg TID), ambrisentan (10 mg), and IV epoprostenol (42.5 ng/kg/min). She continued this regimen until 2017, when she expressed interest in switching to oral selexipag. RHC at that point showed an mPAP of 42 mmHg and a PVR of 5.08 Wu. She was transitioned at an accredited PAH comprehensive care center under the direction of her pulmonary hypertension physician in coordination with his pulmonary hypertension nurse navigator. At that time she was being treated with maximal therapy, i.e. a regimen incorporating drugs targeting the three known pathophysiological pathways in PAH: a PDE5i, an endothelin receptor antagonist, and a PPA.
As shown in Fig. 3a, she underwent a nine-week outpatient transition starting in December 2017. Selexipag (200 µg BID) was started prior to her first IV epoprostenol dose reduction. She then increased selexipag in increments of 200 µg BID at one to two week intervals based on her prostacyclin side effects and pulmonary hypertension symptoms. After she reached her maintenance dose (1600 µg BID) in February 2018, IV epoprostenol was continued for another three weeks at a residual dose of 1.5 ng/kg/min to allow additional time to assess for signs of clinical worsening before the infusion line was removed—a precaution that was undertaken because she had transitioned onto selexipag more quickly than her physician recommended. In July 2018, RHC revealed improved hemodynamics (mPAP: 34 mmHg, PVR: 3.5 Wu, and CI: 2.86 L/min/m2).
(a) Transition timeline—Patient 3 (infused to oral). Titration of PPA doses during outpatient transition from IV epoprostenol to PO selexipag. (b) Transition timeline—Patient 3 (oral to infused to oral). Titration of PPA doses during temporary transition from PO selexipag to IV and inhaled epoprostenol, while patient was intubated in intensive care unit.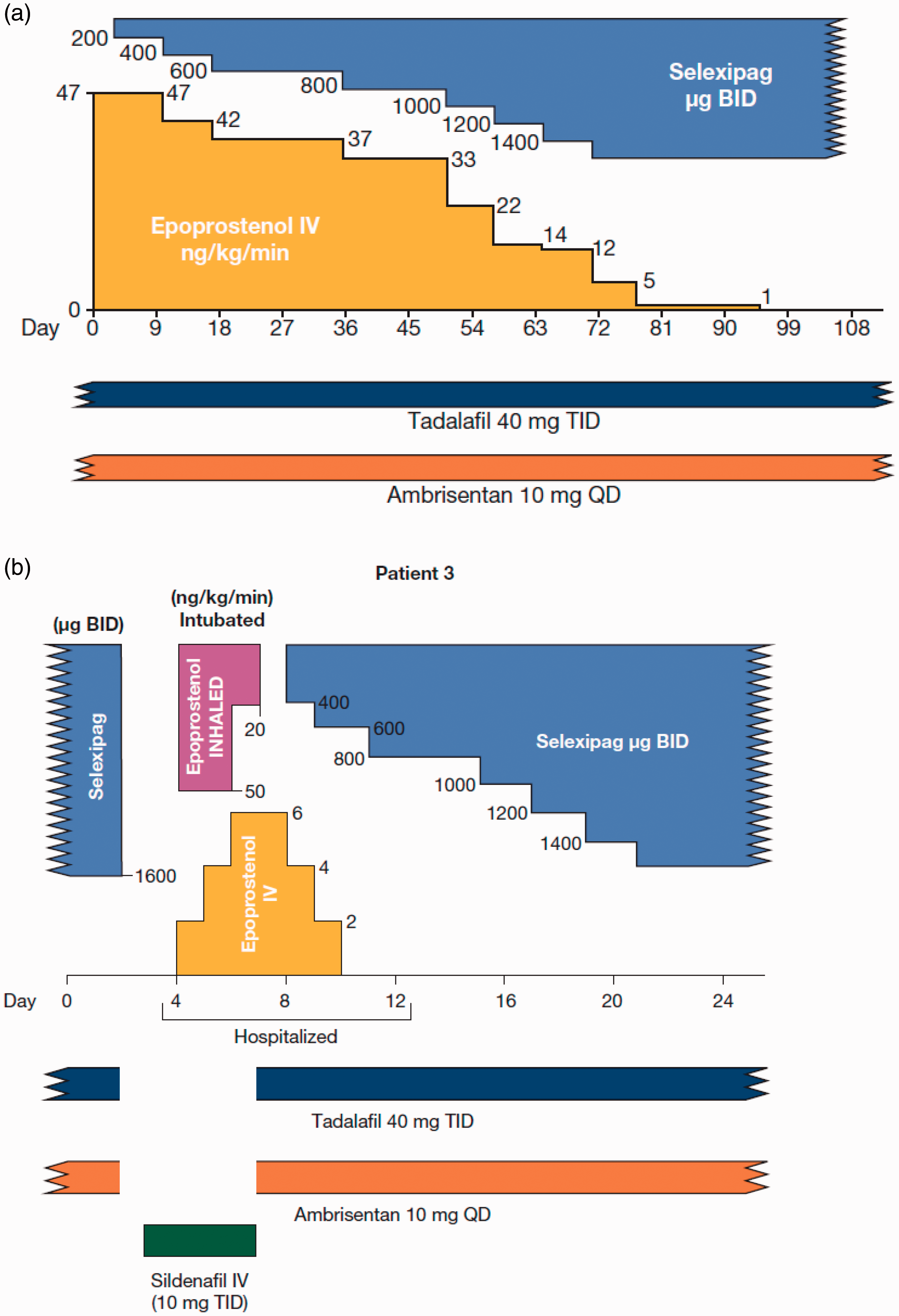
In November 2018, the patient presented at the emergency room with altered mental status and somnolence, and was diagnosed with severe sepsis related to Escherichia coli urinary tract infection. She exhibited acute respiratory failure secondary to sepsis and was admitted to the intensive care unit and intubated. She was treated with IV antibiotics, volume expansion, and vasopressors. Because she had missed her PAH medications for 24 h before entering the hospital and because intubation prevents administration of oral PAH medications, she was started on IV sildenafil (10 mg every eight hours) and IV epoprostenol; in addition, she was started on inhaled epoprostenol to manage her acute respiratory failure. As shown in Fig. 3b, she was extubated and awakened on hospital day 4. She immediately resumed oral sildenafil, ambrisentan, and selexipag. She discontinued IV epoprostenol two days after extubation and reached a selexipag dose of 1600 µg BID 14 days after extubation. In March 2019, four months after discharge, echocardiography showed reduced right ventricular systolic function but normal right ventricular and right atrial volumes. The patient continues on oral therapy.
Discussion
Optimal strategies for transitions between infused and oral PPAs.

PAH: pulmonary arterial hypertension; PDE5i: phosphodiesterase type 5 inhibitor; PHA: Pulmonary Hypertension Association; PPA: prostacyclin pathway agent.
For Patient 2, the gradual onset of side effects played a pivotal role in her eventual decline on oral treprostinil by limiting the maximum tolerated dose and by prompting skipped doses—similar to the observations reported by Chakinala et al. 7 and Maestas et al. 5 Chakinala et al. 7 suggested that the onset of side effects with oral treprostinil was likely related to fluctuations in circulating drug levels; patients transitioning from infused to oral treprostinil tolerated higher total daily doses with TID compared to BID administration, 7 which is likely due to the lower peak to trough ratio of 2.5 with TID dosing compared to 7 with BID dosing. 11 It is worth noting that the tendency for drug level fluctuations may differ from one oral PPA to another. The active metabolite of selexipag has a plasma half-life of 9–14 h, significantly longer than the 4.5 h half-life reported for treprostinil, which may limit the potential for fluctuations in drug levels.12,13
The clinical deterioration of Patient 2 did not emerge until seven months post-transition, which is similar to the delayed worsening reported by Maestas et al. 5 That study found mPAP and PVR prior to transition to be significant, independent predictors of clinical worsening up to two years post-transition, with a PVR of 4.16 Wu identified as an optimal cutoff between patients who did and did not experience clinical worsening. Viewed in the context of that study, Patient 2's pretransition hemodynamics (mPAP: 36 mmHg, PVR: 5.91 Wu) may have been suboptimal for transition. 5 Although she was placed on tadalafil background therapy prior to the transition, it is possible that more aggressive treatment with a third agent (an endothelin receptor antagonist) or a higher dose of SQ treprostinil would have improved her pre-transition hemodynamics and increased her chances of succeeding long term on oral treprostinil. Optimized hemodynamics supported by appropriate background therapy may set the stage for a successful transition by giving patients sufficient cardiopulmonary reserve to withstand an “aim-low” protocol, with a low initial oral PPA target dose.
One final observation is notable regarding Patient 2: when she switched back to infused therapy after worsening on oral treprostinil, she required nearly twice her original dose of SQ treprostinil to re-stabilize her symptoms and hemodynamics, which is similar to patients in the Maestas et al.'s study. 5 This illustrates the seriousness of decisions to transition patients onto oral PPA therapy and the importance of careful patient selection and optimization of hemodynamics prior to the transition to maximize the likelihood of success.
As with Patient 2, Patient 3 also had hemodynamics (mPAP: 42 and PVR: 5.08) that may not have made her an obvious candidate for oral PPA therapy. In addition to that, however, she suffered from a longstanding lack of family and psychosocial support, making compliance with infused therapy all the more challenging. Her case illustrates that decisions about transitioning to oral therapy should include broader consideration of the entire patient rather than hemodynamic numbers alone. It is worth noting that following the transition to selexipag, her hemodynamics actually improved. Another recent case series 8 documents the transitioning of two patients with WHO FC IV PAH symptoms from infused PPA therapy to selexipag carried out for similar reasons. It will be important, in the future, to document the long-term outcomes of such patients on oral PPAs.
It is notable that although Patient 3 entered the intensive care unit with acute respiratory failure due to sepsis, she showed no specific signs of PAH worsening (based on clinical examination and echocardiography), despite missing her medications for 24 h. This initial absence of PAH worsening may reflect her long-term maintenance on background endothelin receptor antagonist and PDE5i therapy, as well as the long systemic persistence of selexipag, with active metabolite still present in the bloodstream for 24–36 h after the last dose. 12 The fact that Patient 3 had already been on selexipag, with only a brief interruption, facilitated her titrating rapidly back up to her individualized maintenance dose (1600 µg BID) only 14 days after extubation.
Finally, the experience of Patient 3 reflects the growing reality that as PAH patients survive longer, they may require multiple transitions between oral and infused PPAs due to evolving life situations, health status, emergencies, and elective hospitalizations. It is important to plan ahead for this eventuality.
Conclusion
Our case series illustrates best practices for undertaking transitions between infused and oral PPAs, as well as potential problems that can be anticipated. Tolerability and fluctuations in drug levels represent major barriers to long-term success in patients who transition from infused to oral PPAs. The tolerability of these transitions is influenced by many factors. Using a lower starting dose, followed by slower uptitration, during or after the transition, may aid in reaching the eventual oral target dose. This “aim low” approach is facilitated by aggressive background therapy to optimize hemodynamics ahead of the transition. In addition, it should be emphasized that an individualized approach to transitioning is important. Each patient should be titrated based on their particular side effects and PH symptoms. Multiple transitions between infused and oral PPAs are likely to become more frequent as patients continue to survive longer, with evolving needs. Future research may focus on the use of risk assessment tools to determine the optimal patient candidate to transition, as well as the clinical significance of these therapy transitions. In addition, utilization of risk assessment tools may provide valuable measures of clinical stability versus early decline.
Footnotes
Acknowledgments
This manuscript was written independently by the authors with writing support provided by Twist Medical and funded by Actelion Pharmaceuticals, Inc. Actelion Pharmaceuticals, Inc., reviewed the manuscript for scientific accuracy only. All statements and opinions expressed in the manuscript are those of the authors and do not necessarily reflect those of Actelion Pharmaceuticals, Inc., or its representatives.
Contributorship
Each author contributed a case and related discussion. A medical writer supported the authors in collating the information and ensuring consistency across the manuscript sections. All authors critically edited the manuscript and provided final approval for manuscript submission.
Conflict of interest
T.S. has received consulting and speaker fees from Actelion and has served on advisory boards for Actelion and Bayer; L.H. has served as an unpaid consultant for Actelion; and R.H. has received speaker fees from Gilead and has served on advisory boards for Actelion, Gilead, GlaxoSmithKline, and United Therapeutics.
Funding
Funding for medical writing support was provided by Actelion Pharmaceuticals, Inc. However Actelion Pharmaceuticals, Inc did not participate in the development of the manuscript, except for one courtesy review for accuracy only.
Guarantor
Therese Sargent, Lillian Hansen, and Robin Hohsfield.
