Abstract
Parenteral prostanoids are effective for improving outcomes in patients with pulmonary arterial hypertension. However, subcutaneous or intravenous delivery via an external pump places a significant burden on patients. Consequently, the Implantable System for Remodulin© (treprostinil) was developed and is associated with a low rate of complications (United Therapeutics (Research Triangle Park, NC) in collaboration with Medtronic, Inc. (Mounds View, MN)). The current real-world experience study evaluated pulmonary arterial hypertension patients' perceptions of their quality of life, ability to perform activities of daily living, perceptions on the benefits and risks of the implantable system, and their social interactions before and after receiving the implantable system. Pulmonary arterial hypertension patients who had been transitioned from an external infusion pump to the implantable system completed a mix of quantitative and qualitative questions administered online over the course of a six-day period. A total of 20 patients completed the study. All patients reported that their quality of life, confidence out in public, and ability to travel long distances had improved. Over 90% of patients reported that their overall level of independence was better since receiving the implantable system, and most patients indicated that their ability to independently perform specific activities of daily living had improved. Responses to the qualitative questions suggested that the implantable system saved time, improved interpersonal relationships, and increased freedom. Results from this real-world patient experience study suggest this novel delivery system provides improvements in factors that are of substantial importance to patients.
Introduction
Parenteral treprostinil, delivered either subcutaneously (SC) or intravenously (IV), is widely used in the United States and Europe for pulmonary arterial hypertension (PAH) patients with more severe or rapidly-developing disease1,2 and is effective for improving symptoms and outcomes.3,4 However, SC administration is associated with infusion site pain and reaction, which can limit patient tolerability,3,5,6 and IV administration has the inherent risk of catheter-related bloodstream infections (BSIs)7,8 and concomitant increase in morbidity and mortality. 9 To utilize parenteral therapy, patients or caregivers must manage pump settings, maintain sterile technique, dilute the medication (IV), and clean and care catheter sites. Given the inconvenience and adverse events (AEs) associated with SC/IV external pump administration, PAH patients can experience significant treatment-related burden, particularly with respect to health-related quality of life (HRQoL).10–14 Moreover, patients point to these burdens as central reasons for poor treatment acceptance and subsequent changes in their treatment. 10 Given the risks and complexities associated with SC/IV administration, patients and physicians are often reluctant to use parenteral prostanoids, which may be critical medications in the management of pulmonary hypertension for many patients. 6
A number of studies have identified specific components of HRQoL that are negatively impacted by PAH.11,12 PAH patients in general experience significantly less “free-living activity,” as assessed by daily step count, 15 as well as a significant reduction in physical activities.16–19 This lack of mobility has a significant impact on activities of daily living, which some PAH patients have a particularly hard time performing independently.10,11,19 Patients also report that PAH has a significant and negative impact on their social functioning and relationships,10–12,19 often experiencing social isolation.12,13 A recent systematic review found that the prevalence of depression, anxiety, and panic in PAH patients ranged from 7.5% to 53%. 20 A series of studies by Matura and colleagues also reported that over 50% of PAH patients report sleep difficulties.21–23
To overcome the significant burden associated with external pump delivery systems, the Implantable System for Remodulin
24
was developed. Approved by the United States Food and Drug Administration in 2017,
25
the fully implantable, programmable delivery system consists of the SynchroMed II implantable pump, the associated programmer, and an implantable catheter designed to prevent occlusion (see Fig. 1). The implantable system is indicated for use in adult patients with New York Heart Association functional class I, II, and III PAH.
25
Results from the DelIVery study, a multicenter, prospective, single-arm trial in 60 patients with PAH, detail that over a six-month follow-up period, the implantable system was associated with a statistically significant reduction in the rate of catheter-related complications, with no reported catheter-related BSIs or catheter occlusions.
26
The efficacy of treprostinil was maintained via the implantable system from baseline to six months post-implant. In addition, plasma treprostinil concentrations one week post-implant were highly correlated with baseline concentrations. All patients rated the treatment as good to excellent, and 79% of patients believed the effectiveness of the implantable system was better than expected. Average delivery system management time decreased to 75%, from 2.5 h per week to 0.6 h per week, which included travel time to the clinic for pump refills. Long-term results of the DelIVery study were recently published, further demonstrating the safety profile of the implantable system over approximately 282 patient-years.
27
Investigators recommend continued patient treatment at specialized PAH centers to preserve the risk-benefit ratio of the implantable system, until training is disseminated to other sites.
Overview of the Implantable System for Remodulin.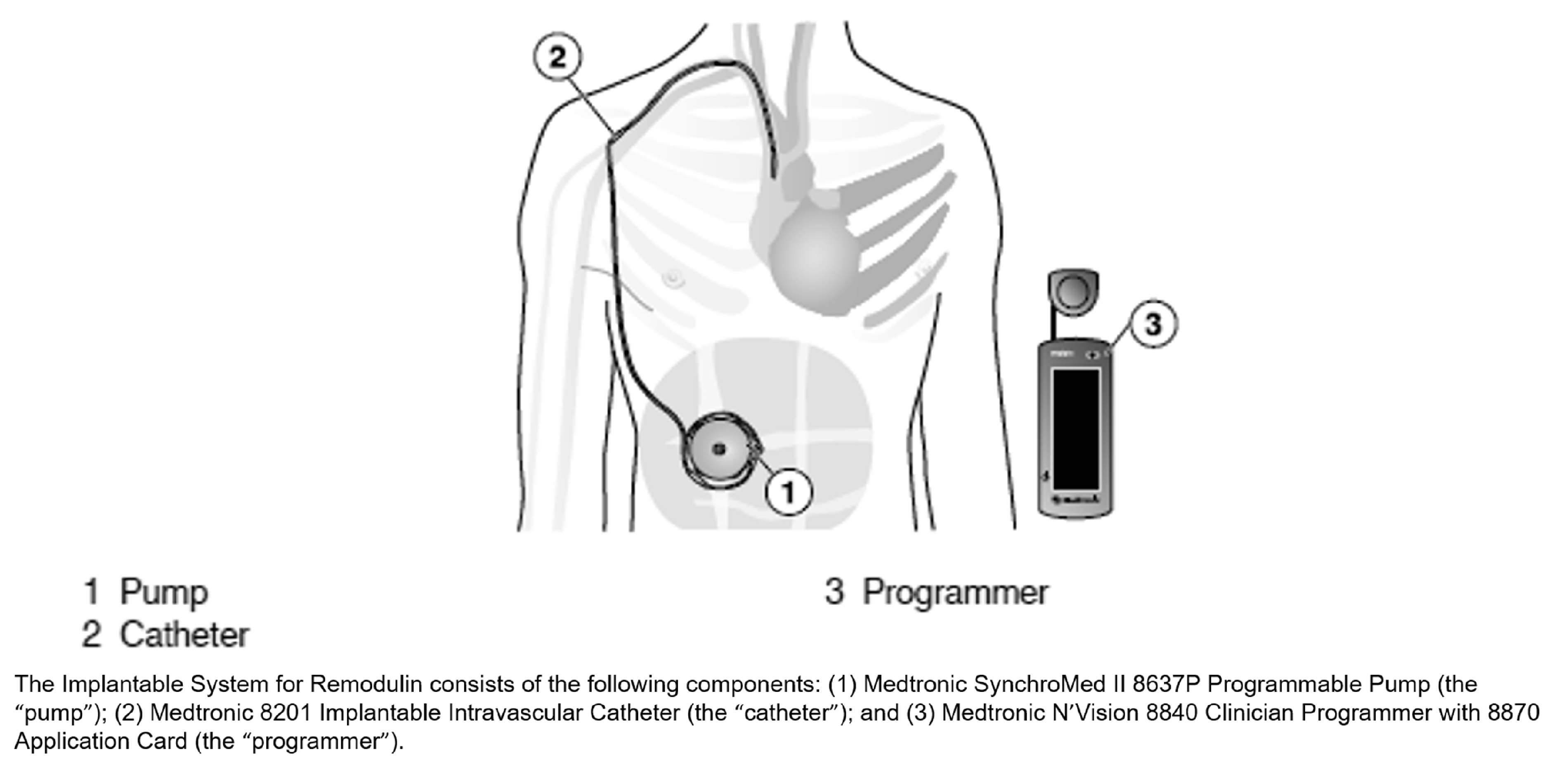
While traditional outcomes in PAH, such as functional classification, exercise capacity, and cardiopulmonary hemodynamics, are meaningful assessment parameters, quality of life (QoL) outcomes and patient experience are also important considerations in choosing therapy. 28 CHEST guidelines on pharmacologic therapy for PAH state that “all treatment decisions should be informed by patient preferences, goals, and assessments of health-related quality of life”. 2 The “patient voice” has also been emphasized by recent regulatory and policy directives provided by the Food and Drug Administration and others,1,29–32 particularly in PAH patients. 33 The current study was designed to evaluate patient perceptions of QoL, ability to perform activities of daily living, and social interactions, before and after receiving the implanted system in the DelIVery study. 26
Methods
Study design
The current study collected both quantitative and qualitative data over six consecutive days. Each day, patients logged into a Health Insurance Portability and Accountability Act compliant online platform and spent 15–30 min completing a quantitative portion (days 1 and 2) followed by a series of qualitative activities (days 3–6). The names of patients and their responses were blinded to everyone except the research moderator. The protocol was approved by a central institutional review board, and all participants completed an online informed consent form prior to initiating any activities. Data collection was retrospective (particularly as it relates to experience on SC/IV medication delivery with external pump) from patient recollection.
Participants
Participants were drawn from the DelIVery study. 26 Patients meeting the following requirements at DelIVery study enrollment were included: (1) stable PAH (World Health Organization Group 1), (2) receiving a continuous infusion of treprostinil via an external pump, and (3) had no PAH treatment added for at least two months prior to enrollment. 26 Patients in the DelIVery study were transitioned from either IV or SC treprostinil to the implantable system, although patients receiving SC treprostinil were required to be switched to IV infusion at least 30 days, and be stable for those 30 days prior to getting the implantable system. 26 Patients were contacted by the study coordinator or nurse at their current study site and informed of the opportunity to participate in the current study. If patients expressed interest, they contacted the research moderator by email or by phone and were deemed eligible for inclusion in the real-world study if they: (1) continued to receive treprostinil via the implantable system; (2) could read, write, and speak English; and (3) agreed to keep study information confidential.
Quantitative assessments
On the first day of the study, patients completed questions assessing demographic characteristics and their previous experiences with SC/IV treprostinil with an external pump.
On the second day, patients were asked about complications with their SC/IV external pump. Patients responded to 10 questions regarding whether the implantable system improved their overall QoL, confidence out in public, independence, and activities of daily living relative to their previous mode of administration. For example, patients were asked: “Overall, has getting the implantable pump made your QoL better or worse compared to (SC or IV) infusion ?”, and they responded by selecting one of the following response options: “a lot better,” “a little better,” “no change,” “a little worse,” or “a lot worse.” Patients were also asked what they have been able to do with the implantable system that they were not able to do, or found difficult to do, with their previous SC and/or IV external pump.
Qualitative assessments
On the third day of the study, patients were asked to respond to two open-ended questions on how previous SC/IV infusion via an external pump and the implantable system impacted relationships with family, loved ones, and caregivers.
On day four, patients searched online and selected images that most closely represented what their life was like on their previous SC/IV external pump and what their life has been like since receiving the implantable system. Patients also responded to a series of open-ended questions regarding their experiences with previous SC/IV infusion and the implantable system.
On day five, patients participated in a “Community Sharing Wall,” by posting about what they liked the most and the least about SC/IV infusion and the implantable system, and by reacting to other patients' posts.
On day six, patients were asked to write a letter to someone considering the implantable pump, describe their experience with the implantable pump, explain how it has changed their lives, and offer advice.
Data analysis
Basic descriptive statistical analysis techniques were used to summarize responses to quantitative questions and qualitative activities. No formal statistical analysis was performed.
Results
Patient characteristics
At the time of recruitment, 46 of the 60 patients from the DelIVery clinical trial remained on treatment. Patients were drawn from all but one DelIVery clinical trial site, which did not participate due to contracting considerations. Twenty-one of 46 eligible patients elected to participate in this real-world patient experience study. Responses from one patient were excluded after incorrectly identifying themselves as previously receiving SC therapy. Mean age of the real-world study sample was 52.56 years (SD = 12.82), which was similar to the overall age of patients in the DelIVery study (mean = 50.10; SD = 13.5). The study included five men. Patients who participated in the study had, on average, been using the implantable system for five years (SD = 0.70). Patients had been receiving therapy via an external pump for five years (SD = 0.70) on average prior to receiving the implantable system.
Quantitative results (days 1 and 2)
Of the 20 patients in the study, eight patients completed only the IV-related survey questions and six patients answered only the SC survey questions, while another six patients answered both the IV- and SC-related questions. Consequently, 14 patients responded to the IV-related questions (previous IV infusion group) and 12 patients responded to the SC-related questions (previous SC infusion group).
Among questions regarding the implantable system experience, 100% of the patients reported that they could not “feel the implantable system delivering medication,” and all patients agreed with the statement “the benefits of the implantable pump were worth the risks.” Twelve of the 14 (86%) previous IV infusion patients stated that external IV infusion took more time than the implantable system and the remaining two stated that they both took the same amount of time. Ten of the 12 (83%) previous SC infusion patients reported that external SC infusion took more time than the implantable system. One SC patient stated that the two delivery methods take the same amount of time, while another stated that the implantable system took more time, but that it was worth the extra travel time required to refill the pump. Note that patients only had their implanted system at the study site, which may have been an extended distance from their home. This may have resulted in the real perception that it took more time than the use of their delivery system prior to implantation.
Eleven of 14 patients (79%) in the previous IV infusion group and 4 of 12 patients (33%) in the previous SC infusion group recalled more PAH-related emergency room visits (over an average one-year time frame) during the time they were on an external pump. One patient from the previous IV infusion group and two patients from the previous SC infusion group recalled more PAH-related emergency room visits on the implantable pump. The remaining patients in both groups either stated that the number of emergency room visits were similar or could not remember.
The impact of the implantable system on patient QoL, satisfaction, and activities of daily living are provided in Figs. 2 and 3 for previous IV and SC patients, respectively. All patients reported that they experienced improved overall QoL, improved confidence out in public, and improved ability to travel long distances with the implantable system. Except for one patient from the previous IV infusion group, all patients reported that sleep had improved since receiving the implantable pump. Most patients in both groups indicated that the implantable system had made working (e.g. paid employment or volunteering) at least a little better.
Impact of the implantable system on quality of life and activities of daily living—previous IV patients (N = 14). Impact of the implantable system on quality of life and activities of daily living—previous SC patients (N = 12).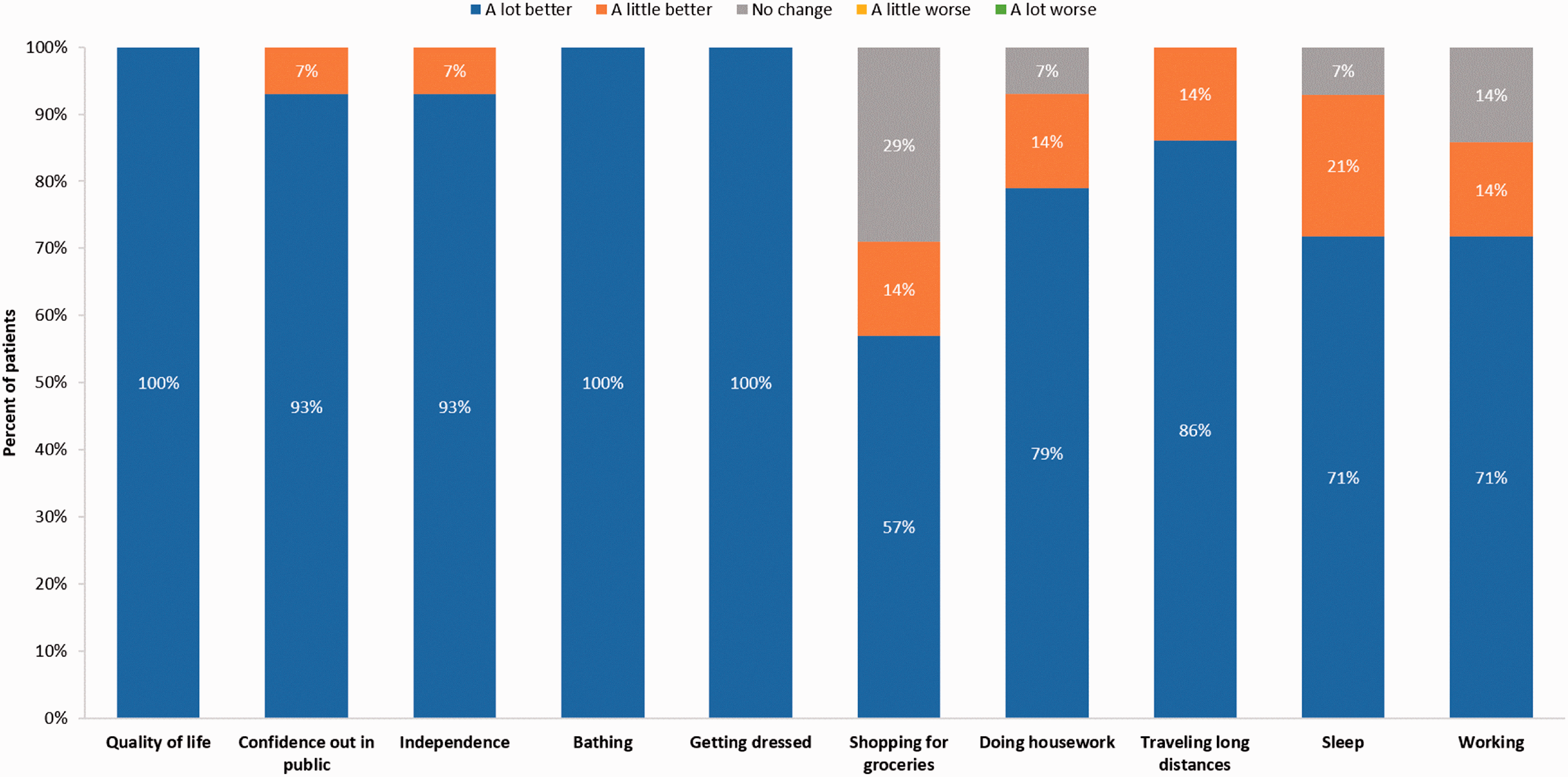
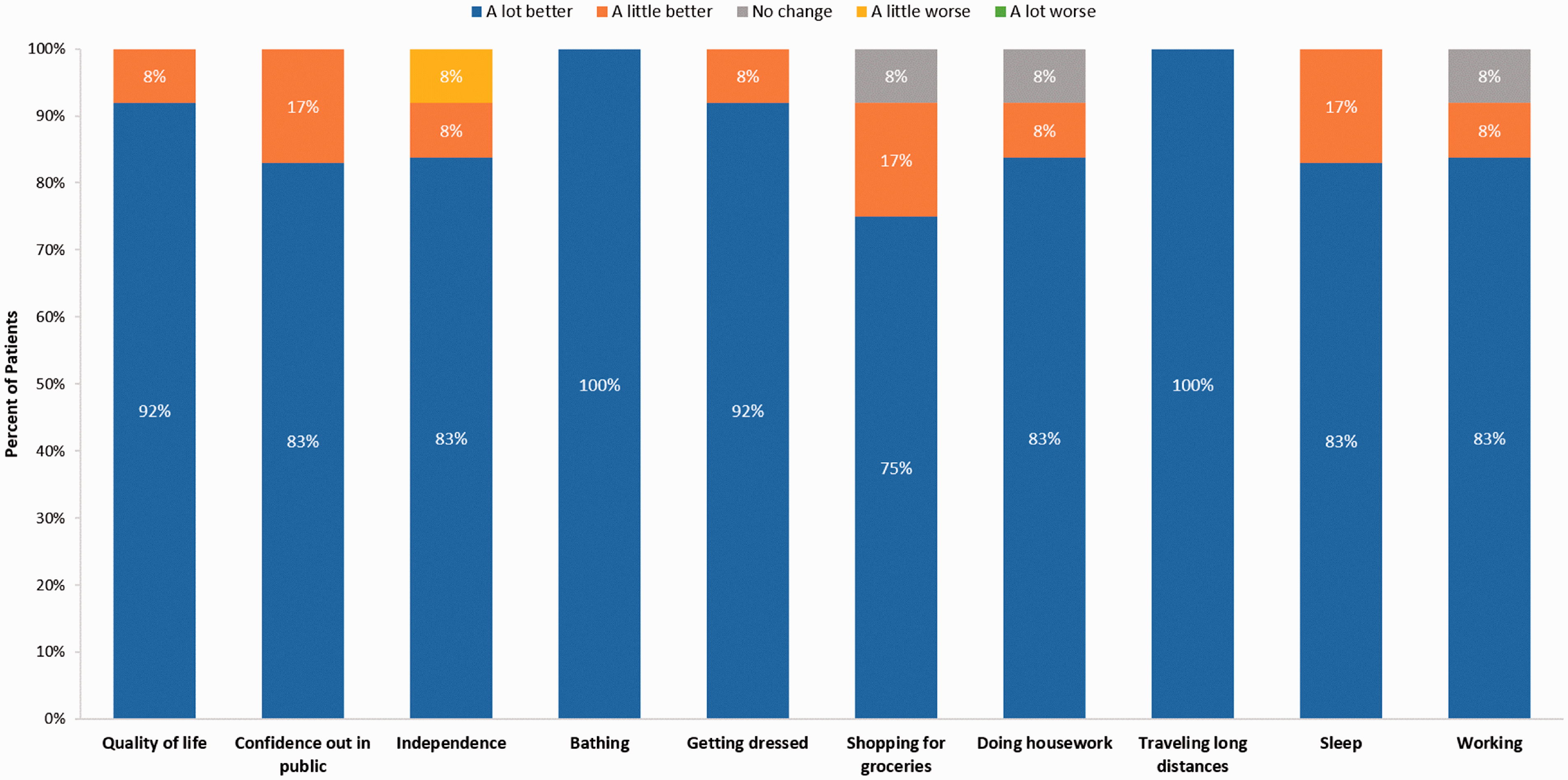
Over 90% of patients in both groups reported their overall level of independence was improved since receiving the implantable system. One patient in both groups indicated that their level of independence was “a little better,” and one patient in the SC group indicated that it was “a little worse.” All patients reported that the implantable system made bathing “a lot better.” Similarly, all patients but one in the SC group, indicated that getting dressed had become “a lot better” with the implantable system. Over 90% of patients in both groups reported improved ability to do housework and over 70% of SC and 90% of IV patients reported improved ability to shop for groceries.
Impact of the implantable system on patient activities—example responses.

Notes: Patient responses are provided verbatim. The above represents responses from four distinct patients.
IV: intravenous; SC: subcutaneous.
Qualitative results
Eleven patients completed the qualitative questions for IV external infusion and nine patients completed the questions for SC external infusion. Patients were assigned based on whether they had been on IV or SC for at least a year. Due to time commitment, patients that previously received both IV and SC for at least a year were asked to only answer questions for one route of administration. An attempt was made to obtain a balanced number of responses for IV and SC groups.
Day 3
IV/SC infusion and implantable system impact on relationships—example responses.
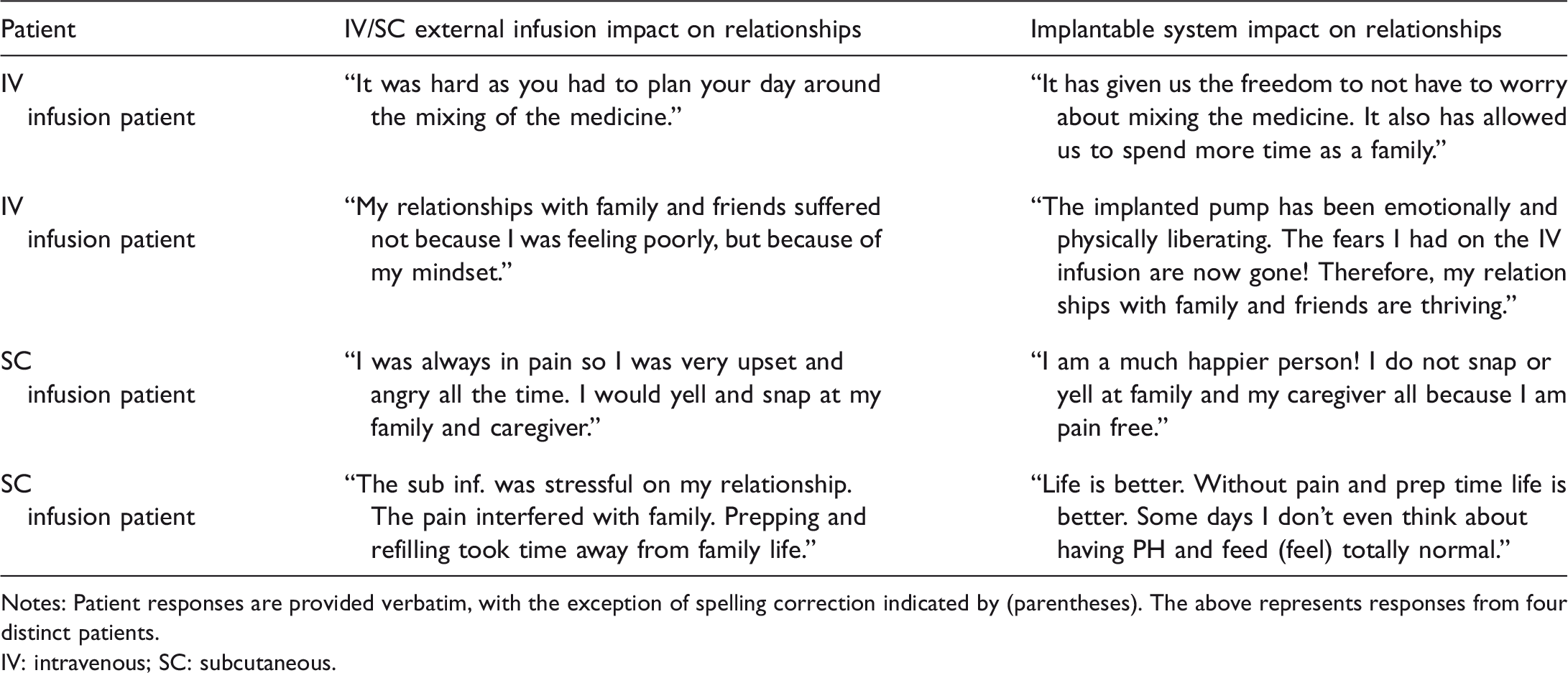
Notes: Patient responses are provided verbatim, with the exception of spelling correction indicated by (parentheses). The above represents responses from four distinct patients.
IV: intravenous; SC: subcutaneous.
Day 4
Symbolic images of life on IV/SC and implantable system—example responses.

Note: The above represents responses from four distinct patients.
IV: intravenous; SC: subcutaneous.
All patients uploaded positive images to symbolize their lives since receiving the implantable system, and commented that the images represented happiness, release, and, most frequently, freedom. When asked what they liked most about the implantable system, almost all patients remarked that the system gave them freedom. In addition, the diminished occurrence of AEs from the previous external infusion was frequently mentioned. The initial surgery and travelling to refill the pump were aspects patients did not like about the implantable system.
Day 5
The central theme running through patients posts on the “Community Sharing Wall” (see online Appendix B, Table 1) when asked what they liked most about their previous SC/IV external infusion was that it delivered the medicine they needed to stay alive. SC patients overwhelmingly stated they disliked the site pain the most. IV patients said it was the fear of infections they disliked the most. All patient responses are provided in online Appendix A, Table 4.
When asked to write down what they liked most about the implantable system, patients stated that they had less fear of complications, less self-consciousness and isolation, and less hassle. Both SC and IV patients overwhelmingly stated that the implantable system provided them with a level of freedom that they had not enjoyed on their previous external infusion. The most commonly mentioned dislike about the implantable system was the travel and time associated with having to get it refilled. All patient responses are provided in online Appendix B, Table 1.
Day 6
In the letters written to those considering the implantable system, most patients mentioned that on their external infusion pump, they experienced significant AEs (e.g. infection and site pain) and that they always felt “tethered” to the external pump. One example letter is provided in Fig. 4; all patient letters are provided in online Appendix A, Table 5. All patients noted that their lives after receiving the pump had markedly improved, citing that the implantable system was “life-changing” and had given them “freedom” to do the things they wanted to do. In addition, patients pointed to their increased happiness and independence afforded by the implantable system. They also mentioned the positive impact the implantable system had on their relationships. The most frequently cited negative aspect of the implantable pump was the time and travel inconvenience involved with refilling the pump.
Example of letter to someone considering the implantable system.
Discussion
Despite the clinical efficacy of treprostinil and other prostacyclin analogs, PAH patients experience a significant number of side effects with delivery via an external pump. These side effects, coupled with the logistic and pragmatic considerations of an external infusion pump, have a significant and negative impact on patients' overall QoL.10–12 Using a mixed-method design, the current study provided a unique window into PAH patients' perceptions of their QoL while on SC/IV treatment via an external infusion pump and subsequently on the implantable system. 33 Patients report an improved overall experience with the implantable system compared to their previous IV/SC external infusion pumps, specifically in areas of QoL, satisfaction, and activities of daily living.
The original DelIVery trial reported that all patients reported their satisfaction with the therapy as at least “good” and that patients remained satisfied with the implantable system. 26 In the current real-world patient experience study, all patients indicated that the benefits associated with the implantable system greatly outweighed the risks. Many patients recalled fewer PAH-related emergency room visits since receiving the implantable system, relative to an average year on their previous external infusion pump. Most patients also indicated that the time associated with the implantable system was less than their previous mode of administration.
Patients indicated that the implantable system made their overall QoL, confidence out in public, and independence “a lot better” compared with SC/IV external infusions. These reported improvements in QoL are particularly interesting given that the DelIVery trial 26 found no significant improvement in QoL as assessed by the Cambridge Pulmonary Hypertension Outcome Review. 34 In addition, most patients reported that the implantable system resulted in significant improvements in sleeping and working as well as performing activities of daily living such as bathing, getting dressed, shopping for groceries, and doing housework. These factors are particularly important given that previous studies suggest that PAH patients experience significant difficulties in these areas and, in turn, experience significant impacts on their overall QoL.11,12
In the qualitative portion of the survey, two overarching themes emerged. The first was based on the responses of what patients' lives were like on their previous IV/SC external infusion and centered on the fear and impact of AEs, specially, infection associated with IV administration and pain caused by SC administration. Patients indicated that the worry about and/or presence of AEs had significant impact on their social relationships and was a main drawback of that therapy option. In addition, patients suggested a diminished occurrence of AEs that were once present with previous external infusion (e.g. SC site pain and IV infection) after receiving the implantable system. Furthermore, patients reported experiencing an improvement in their overall QoL, particularly their relationships with family members and caregivers. The second and perhaps more pervasive theme was the lack of freedom experienced on the external infusion pump and the gain of freedom when moving to the implantable system. Patients commented that all aspects of their lives were negatively impacted by being “tethered” to the external pump. Switching to the implantable system allowed them to engage with others and in their environment more freely. All patients commented at least once that they had significantly more freedom on the implantable system, aside from having to travel for refills. These emerging themes highlight the advantage of supplementing clinical efficacy endpoints such as the six-minute walk with a mixed-method approach to capturing the “voice” of the patient. Consequently, collecting qualitative data to understand and evaluate patients QoL should be applied to other PAH treatments.
While the findings from this study shed light on the real-world experiences of PAH patients with the implantable system, they should be considered alongside some limitations. First, patients' perceptions of their lives on the IV/SC may have been biased because they were currently on the implantable system and had not been on IV/SC for multiple years when answering the study questions from memory. Second, due to the progressive nature of PAH, patients may have had a different health status when evaluating their experience on the implantable system than when they had been on the previous external pump. Finally, sample selectivity is a real concern. Twenty-three percent of DelIVery patients were no longer on treatment at the time of recruitment for the current study, due to death or transplantation. Of those remaining on treatment, only 46% participated in this elective study. Of note is that at the time of submission of this paper, all surviving patients have undergone elective replacement of their implanted pumps due to pumps reaching “end-of-life” criteria. All patients enthusiastically elected to undergo pump replacement (alternative to pump replacement was reverting to IV/SC delivery of treprostinil via an external pump).
In summary, results from this real-world experience study highlight substantial improvements in patient satisfaction, QoL, and activities of daily living associated with switching from IV or SC external infusion to the implantable delivery system. This novel implantable delivery system appears to provide improvements in factors that are of importance to patients but may not always be captured by traditional outcome measures.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020907881 - Supplemental material for Implantable system for treprostinil: a real-world patient experience study
Supplemental material, sj-pdf-1-pul-10.1177_2045894020907881 for Implantable system for treprostinil: a real-world patient experience study by Shelley Shapiro, Robert C. Bourge, Patti Pozella, David F. Harris, Erick H. Borg and Andrew C. Nelsen in Pulmonary Circulation
Footnotes
Author contributions
P.P., D.F.H., A.C.N., S.M.S., and R.C.B. participated in the design of the study. D.F.H. was responsible for data collection. P.P., D.F.H., and E.H.B. were responsible for data analysis. All authors contributed to the interpretation of the study results and to the writing of this manuscript.
Consent
The study was conducted on a HIPPAA-compliant online platform. Before entering the platform for the first time, patients completed an online Informed Consent Form.
Ethical approval
Study was approved by Western Institutional Review Board (approval #2016921).
Conflict of interest
S.M.S., R.C.B., and D.F.H. have received consulting fees from United Therapeutics Corporation. P.P., E.H.B., and A.C.N. are employees at United Therapeutics Corporation.
Acknowledgements
The authors wish to thank the investigators and institutions from which these patients were drawn: University of Alabama at Birmingham (Robert C. Bourge), Birmingham, AL; University of Chicago Medical Center (Mardi Gomberg-Maitland), Chicago, IL; VA Greater Los Angeles Healthcare System and UCLA School of Medicine (Shelley M. Shapiro), Los Angeles, CA; Orlando Regional Medical Center (James H. Tarver III), Orlando, FL; Aurora St. Luke’s Medical Center (Diane L. Zwicke), Milwaukee, WI; Arizona Pulmonary Specialists, Ltd (Jeremy P. Feldman), Phoenix, AZ; Washington University School of Medicine (Murali M. Chakinala), St. Louis, MO; Mayo Clinic Rochester (Robert P. Frantz), Rochester, MN; and University of Texas Southwestern Medical Center (Fernando Torres), Dallas, TX. Editorial assistance was provided by Jason Allaire and Tina Hong of Generativity Solutions Group, funded by United Therapeutics Corporation.
Funding/Guarantor
This study was funded by United Therapeutics Corporation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
