Abstract
The L-arginine precursor, L-citrulline, re-couples endothelial nitric oxide synthase, increases nitric oxide production, and ameliorates chronic hypoxia-induced pulmonary hypertension in newborn pigs. L-arginine can induce arginase, which, in turn, may diminish nitric oxide production. Our major purpose was to determine if L-citrulline increases arginase activity in hypoxic piglet pulmonary arterial endothelial cells, and if so, concomitantly impacts the ability to increase endothelial nitric oxide synthase re-coupling and nitric oxide production. Piglet pulmonary arterial endothelial cells were cultured in hypoxic conditions with L-citrulline (0–3 mM) and/or the arginase inhibitor S-(2-boronoethyl)-L-cysteine. We measured arginase activity and nitric oxide production. We assessed endothelial nitric oxide synthase coupling by measuring endothelial nitric oxide synthase dimers and monomers. L-citrulline concentrations ≥0.5 mM increased arginase activity in hypoxic pulmonary arterial endothelial cells. L-citrulline concentrations ≥0.1 mM increased nitric oxide production and concentrations ≥0.5 mM elevated endothelial nitric oxide synthase dimer-to-monomer ratios. Co-treatment with L-citrulline and S-(2-boronoethyl)-L-cysteine elevated endothelial nitric oxide synthase dimer-to-monomer ratios more than sole treatment. Despite inducing arginase, L-citrulline increased nitric oxide production and endothelial nitric oxide synthase coupling in hypoxic piglet pulmonary arterial endothelial cells. However, these dose-dependent findings raise the possibility that there could be L-citrulline concentrations that elevate arginase to levels that negate improvements in endothelial nitric oxide synthase dysfunction. Moreover, our findings suggest that combining an arginase inhibitor with L-citrulline merits evaluation as a treatment for chronic hypoxia-induced pulmonary hypertension.
Neonates suffering from cardiopulmonary conditions associated with persistent or intermittent hypoxia are at increased risk of developing chronic and progressive pulmonary hypertension (PH), a potentially fatal disease with few therapeutic options.1–4 A contributing factor to the pathogenesis of PH is endothelial cell dysfunction characterized by decreased nitric oxide (NO) production. The amino acid L-arginine has therapeutic potential for treating PH because it may increase NO production by endothelial nitric oxide synthase (eNOS). However, studies of chronic L-arginine supplementation in adults with cardiovascular diseases associated with endothelial dysfunction have demonstrated inconsistent results and even adverse outcomes in some cases.5–8 Supplementation with L-citrulline, an L-arginine precursor, may represent an alternative approach for improving L-arginine bioavailability in order to increase NO production.
We have previously demonstrated that L-citrulline supplementation increases NO production and ameliorates PH in chronically hypoxic newborn piglets and increases NO production in pulmonary artery endothelial cells (PAECs) cultured under hypoxic conditions.9–12 As demonstrated by increasing eNOS dimers and reducing eNOS monomers, we also found that supplementation with L-citrulline re-couples eNOS in pulmonary arteries from chronically hypoxic piglets and in hypoxic PAECs. When eNOS is uncoupled, superoxide (O2–) is produced instead of NO. Indeed, consistent with eNOS re-coupling, we have shown that L-citrulline supplementation reduces O2– generation in pulmonary arteries from piglets with chronic hypoxia-induced PH and in hypoxic PAECs.
It is likely that L-citrulline enhances NO production, increases the ratio of eNOS dimers-to-monomers, and reduces O2– generation in hypoxic PAECs by increasing bioavailability of the eNOS substrate, L-arginine. Notably, L-arginine has been shown to induce the enzyme arginase which also utilizes L-arginine as a substrate. Moreover, arginase is thought to regulate NO production by competing with eNOS for their common substrate.8,13,14 Considering that supplemental L-citrulline may increase bioavailable L-arginine in hypoxic PAECs, it is possible that L-citrulline could induce arginase. Yet, the effect of supplemental L-citrulline on arginase expression and activity in hypoxic piglet PAECs is not currently known. The major purpose of this study was to test the hypothesis that supplemental L-citrulline will induce arginase expression and activity in hypoxic piglet PAECs, and if so, concomitantly impact the ability to increase NO production, elevate eNOS dimer-to-monomer ratios, and reduce O2– generation. We also wanted to determine whether L-citrulline supplementation alters the protein expression of some of the enzymes downstream from arginase involved in arginine metabolism, ornithine aminotransferase (OAT) and ornithine decarboxylase (ODC).
Methods
Ethics statement
The use of animals in conducting this research was approved by the Institutional Animal Care and Use Committee of our University which is accredited by the Association for Assessment and Accreditation of Laboratory Animal Use and conformed to US National Institutes of Health guidelines for the use of experimental animals.
PAEC isolation and culture
Mixed breed piglets of both sexes were obtained from the vendor on day of life 5. Piglets were pre-anesthetized with intramuscular ketamine (10–30 mg/kg) and acepromazine (1–3 mg/kg), anesthetized with intravenous pentobarbital sodium (50–100 mg/kg), given intravenous heparin (1000 IU/kg), then exsanguinated. A midline sternotomy was performed and the lungs excised. The main pulmonary artery was isolated and flushed with fresh phosphate-buffered saline (PBS) 4–5 times. The pulmonary artery was clamped on one end and filled with 200 μl of 0.05% trypsin-ethylenediaminetetraacetic acid for 10 min at room temperature. The effluence was transferred to culture dishes with endothelial cell growth medium (EGM; Lonza, Basel, Switzerland). The PAECs were cultured in EGM in a humidified normoxic incubator (21% O2 and 5% CO2) at 37°C for three days. The EGM was changed every 72 h and the cells continued to grow until approximately 70% confluence. Cells were sub-cultured until ready for study between passages 4–10. PAECs were identified by their cobblestone appearance and by immunoblot staining for eNOS.
PAEC Experimental Protocols
Confluent PAECs were cultured under hypoxic conditions, 4% oxygen and 5% carbon dioxide, in a humidified environment at 37°C for 48 h. To evaluate the effect of different concentrations of L-citrulline, some PAECs were treated with 0–3.0 mM L-citrulline (Sigma-Aldrich, St. Louis, MO, USA). To evaluate the effect of an arginase inhibitor, both in the presence and absence of L-citrulline, other PAECs were treated with 100 μM of S-(2-boronoethyl)-L-cysteine (BEC; Sigma-Aldrich), L-citrulline, 3 mM, or a combination of L-citrulline, 3 mM, and BEC, 100 μM.
Immunoblot analysis of arginase I and II, OAT, ODC, eNOS, and eNOS dimers and monomers
After culturing under hypoxic conditions for 48 h as described above, some PAECs were washed with PBS, covered with cell lysis buffer (Thermo Scientific, Rockford, IL, USA), protease inhibitor cocktail (Sigma-Aldrich), and phenylmethylsulfonyl fluoride (Sigma-Aldrich). The cells were scraped from the culture dish and centrifuged to separate the supernatant. Aliquots of the supernatant were stored at –80°C for later use in immunoblot analyses. Protein concentration for all homogenates was determined by Bradford assay.
For arginase I and II, OAT, ODC, and eNOS analysis, equal volumes of sample buffer composed of Tris-Glycine SDS 2× (Novex, Carlsbad, CA, USA) with 5% 2-mercaptoethanol (Sigma-Aldrich) were added to aliquots of the supernatant and heated to 80°C for 15 min. Using previously described methods, equal amounts of protein were loaded onto tris-glycine pre-cast 4–20% polyacrylamide gels (Invitrogen, Carlsbad, CA, USA) followed by electrophoresis at 120 volts for 2 h and then the protein was transferred to polyvinylidene difluoride membrane. 15 To block non-specific protein binding, the membrane was then incubated at room temperature in PBS and 0.1% Tween-20 (PBS-T, Sigma-Aldrich) containing 5% non-fat dried milk and then washed in PBS-T containing 1% non-fat dried milk. To detect the protein of interest, the membrane was incubated overnight with arginase I (Abcam, Cambridge, MA, USA) 1:500, arginase II (Santa Cruz Biotechnology, Santa Cruz, CA, USA) 1:500, OAT (ThermoFisher, Waltham, MA, USA) 1:1000, ODC (Abcam) 1:500, or eNOS (BD Transduction Laboratories, San Jose, CA, USA) 1:1000, all diluted in PBS containing 0.1% Tween-20 and 1% non-fat dried milk as carrier buffer. After washing with PBS-T and 1% non-fat dried milk, the membrane was then incubated with anti-rabbit (for arginase I and II, OAT, and ODC; Cell Signaling Technology, Danvers, MA, USA) or anti-mouse (for eNOS; Cell Signaling Technology) IgG horseradish peroxidase (HRP)-linked secondary antibody diluted in the carrier buffer (1:2500) for 60 min. For eNOS dimers and monomers, we used nonsonicated and nonboiled lysates and low-temperature SDS-PAGE as previously described. 16 Membranes were developed using enhanced chemiluminescence reagents (PerkinElmer, Waltham, MA, USA). The chemiluminescent signal was captured using an iBright FL1500 imaging system (ThermoFischer) and the bands for each protein were quantified using the iBright Analysis Software. After washing and stripping the membrane, we followed a similar procedure to re-probe the membranes for beta-actin (1:100,000) and anti-mouse IgG HRP-linked secondary antibody (1:2500, Cell Signaling Technology).
Arginase activity
After culturing under hypoxic conditions for 48 h as described above under PAEC Experimental Protocols, arginase activity was measured in some PAECs by colorimetric determination of urea produced by arginase using an arginase assay kit (Abnova, Taipei City, Taiwan). Arginase activity was normalized to protein content which was determined by Bradford assay.
O2– measurement
Using a modification of our previously described methods, a lucigenin (N,N′-Dimethyl-9,9′-biacridinium dinitrate; Sigma-Aldrich)-enhanced chemiluminescence assay was used to measure O2–. 17 After culturing under hypoxic conditions for 48 h as described above under PAEC Experimental Protocols, some PAECs were washed with PBS. The PAECs were then incubated under hypoxic conditions for an additional 60 min in Krebs Hepes buffer with A-23187 (10 μM; Biomol, Hamburg, Germany), L-arginine (10 μM; Sigma-Aldrich), and the same concentrations of L-citrulline (0–3 mM) and/or BEC (100 μM) as used for the initial 48 h incubation. Next, the PAECs were placed into the luminometer (Titertek Instruments, Huntsville, AL, USA) and background relative light units (RLU) were measured. Then, lucigenin (5 μM) was added and lucigenin-treated RLU measurements were obtained. The background RLU measurements were subtracted from lucigenin-treated RLU measurements. RLU was then normalized to protein content which was determined by Bradford assay.
NO measurement
Using a modification of previously described methods, NO concentrations were measured by chemiluminescence analysis. 11 PAECs were cultured under hypoxic conditions for 48 h as described above under PAEC Experimental Protocols. The PAECs were washed with PBS, then incubated under hypoxic conditions for an additional 60 min in Krebs-Hepes buffer with A-23187 (10 μM), L-arginine (10 μM), and the same concentrations of L-citrulline (0–3 mM) and/or BEC (100 μM) as used for the initial 48 h incubation. Aliquots of the Krebs-Hepes buffer were injected into the reaction chamber of a chemiluminescent NO analyzer (Sievers, Weddington, NC, USA). The reaction chamber contained vanadium (III) chloride in 1 M HCl at 90°C to reduce nitrite and nitrate to NO gas. N2 gas was used to transfer the NO gas to a gas bubble trap where HCl vapor was removed by 1 M NaOH. A standard curve was generated by adding 1, 2.5, 5.0, 10, 15, and 20 μM of NaNO3 to distilled water and assaying. NO concentration was determined by normalizing NO produced to protein content which was determined by Bradford assay.
Statistical analysis
Data are presented as mean ± SEM. Data were compared by one-way ANOVA with Fisher’s protected least significant difference post hoc comparison test. p-Values <0.05 were considered significant.
Results
We first evaluated the effect of L-citrulline treatment on arginase I and II protein amounts and arginase activity. Arginase I protein amounts did not differ between L-citrulline-treated and untreated hypoxic PAECs regardless of L-citrulline concentration (Fig. 1a). In contrast, although not significantly altered by treatment with 0.5 mM L-citrulline, when treated with higher L-citrulline concentrations (1 and 3 mM), arginase II protein amounts were greater for L-citrulline-treated than untreated hypoxic PAECs (Fig. 1b). Arginase activity was similar for L-citrulline-treated and untreated hypoxic PAECs when treated with the lowest L-citrulline concentration, 0.1 mM. However, when treated with two higher L-citrulline concentrations, 0.5 and 3 mM L-citrulline, arginase activity was greater for L-citrulline-treated than untreated hypoxic PAECs (Fig. 1c). These findings indicate that dependent on the concentration, L-citrulline treatment increased arginase II protein levels and arginase activity in hypoxic PAEC. Notably, treatment with 3 mM L-citrulline increased both arginase II protein levels and arginase activity in hypoxic PAECs.
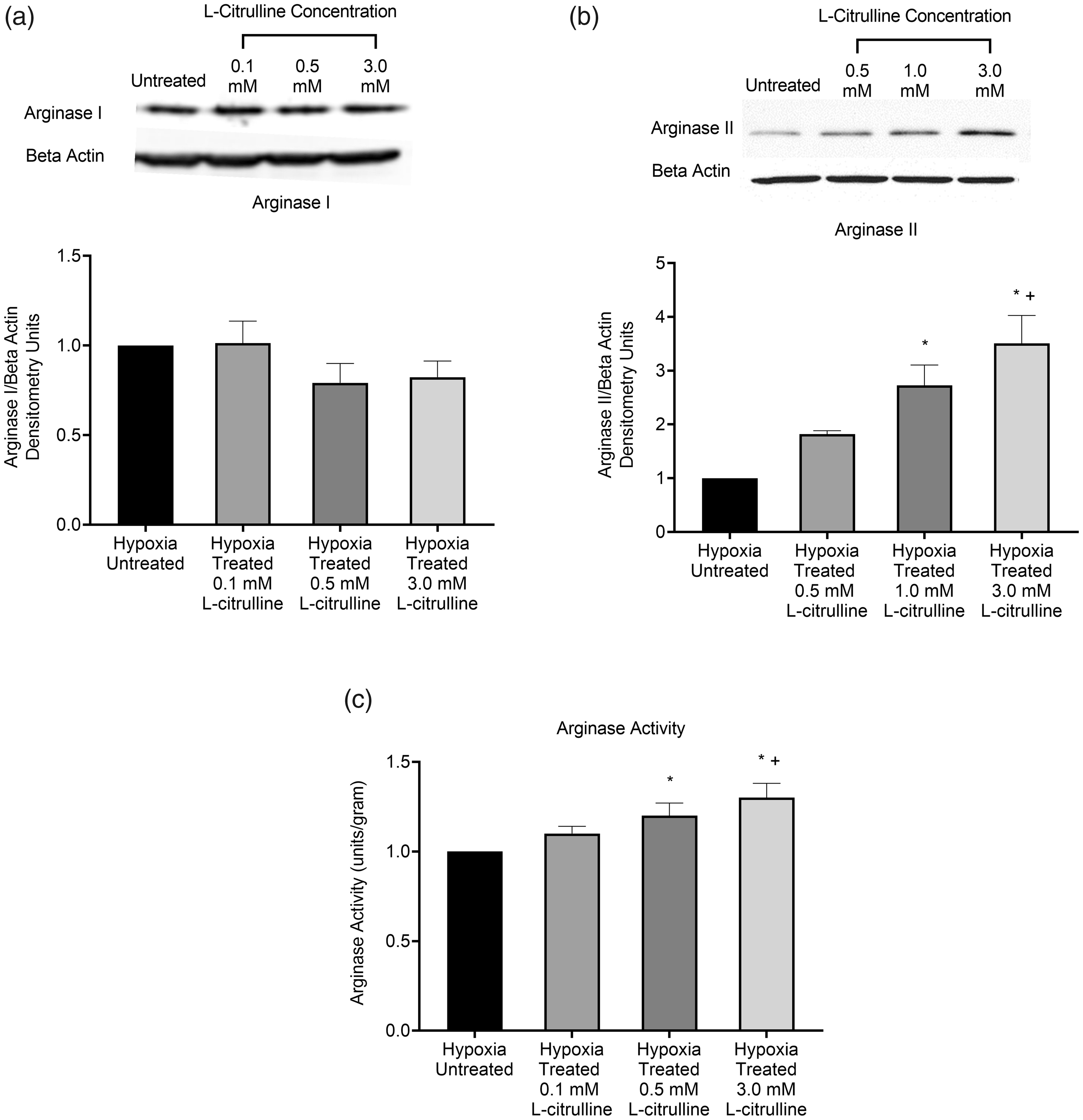
Arginase I and II protein levels and arginase activity in hypoxic PAECs treated with and without L-citrulline. (a) Arginase I protein levels were similar between untreated and L-citrulline-treated hypoxic pulmonary artery endothelial cells (PAECs) regardless of concentration of L-citrulline (n = 7 for each group). (b) Arginase II protein levels were greater for hypoxic PAECs treated with either 1.0 mM or 3.0 mM L-citrulline than for untreated hypoxic PAECs. Moreover, arginase II protein levels were grater for hypoxic PAECS treated with 3.0 mM L-citrulline compared to 0.5 mM L-citrulline (n = 6 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from hypoxic treated with 0.5 mM L-citrulline). (c) Arginase activity was greater for hypoxic PAECs treated with either 0.5 mM or 3.0 mM L-citrulline than for untreated hypoxic PAECs. Moreover, arginase activity was greater for hypoxic PAECs treated with 3.0 mM L-citrulline compared to 0.1 mM L-citrulline (n = 11 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from 0.1 mM L-citrulline).
We next evaluated the effect of L-citrulline treatment on OAT and ODC protein levels in hypoxic PAECs. This is because we wondered whether L-citrulline concentrations that increase arginase activity would also impact the enzymes, OAT and ODC, both of which utilize L-ornithine, a downstream product of L-arginine metabolism by arginase. We found that OAT and ODC protein levels in hypoxic PAECs treated with 3.0 mM L-citrulline were slightly decreased compared to untreated hypoxic cells (Fig. 2a and b); whereas OAT and ODC protein levels were similar between untreated hypoxic PAECs and those treated with lower (0.1 and 0.5 mM) L-citrulline concentrations.
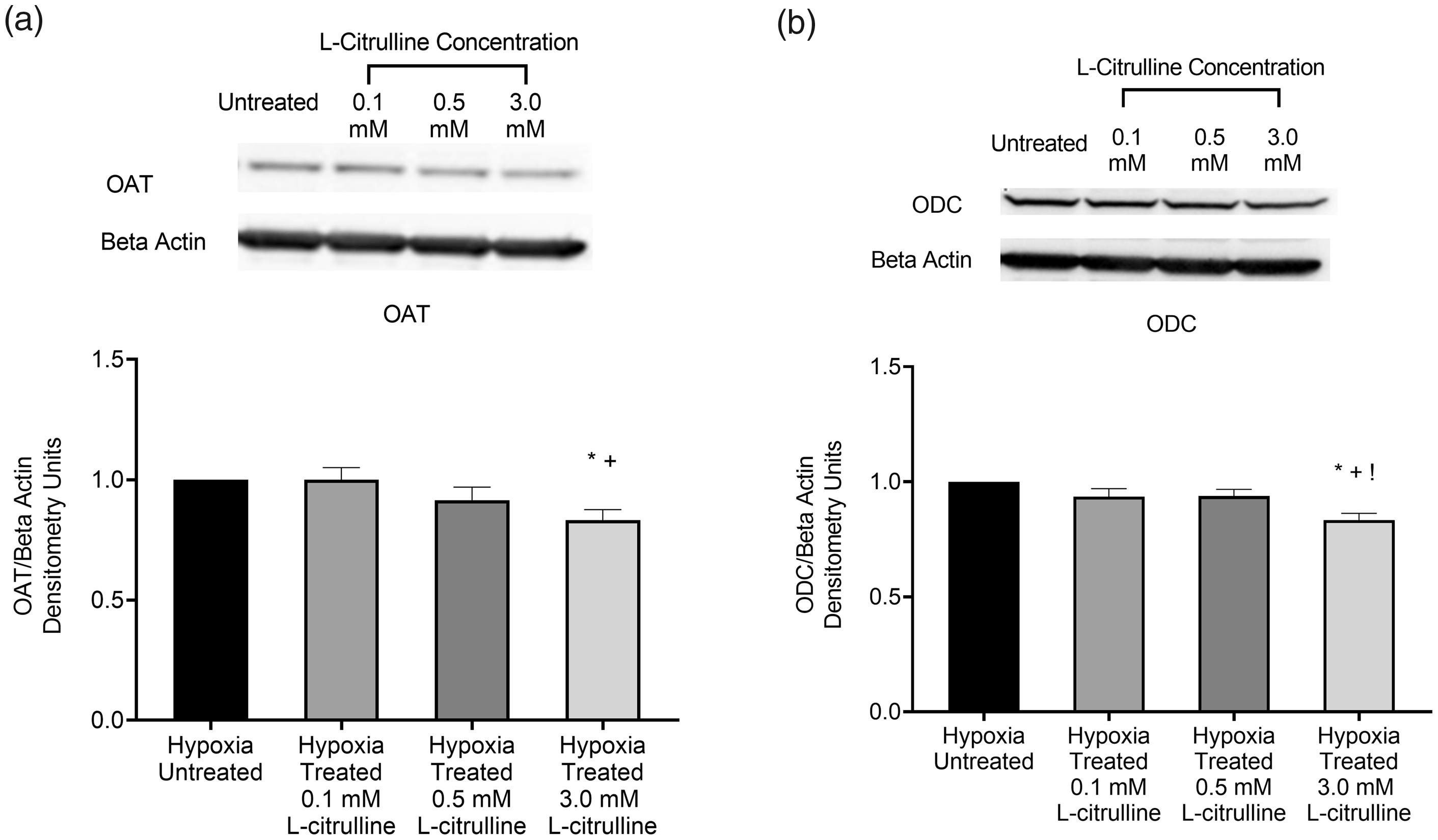
OAT and ODC protein levels in hypoxic PAECs treated with and without L-citrulline. (a) Ornithine aminotransferase (OAT) protein levels were slightly lower in hypoxic pulmonary artery endothelial cells (PAECs) treated with 3.0 mM L-citrulline compared to levels measured in untreated and 0.1 mM L-citrulline treated groups of hypoxic PAECs (n = 7 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from 0.1 mM L-citrulline). (b) Ornithine decarboxylase (ODC) protein levels were slightly lower in hypoxic PAECs treated with 3.0 mM L-citrulline compared to ODC protein levels measured in all other groups of hypoxic PAECs (n = 6 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from 0.1 mM L-citrulline, !p < 0.05 from 0.5 mM L-citrulline).
We also evaluated the effect of treatment with L-citrulline on NO production in hypoxic PAECs. Since L-citrulline is a precursor for L-arginine, the substrate for both eNOS and arginase, we were particularly interested in the impact on NO production from a concentration of L-citrulline that increased arginase activity (Fig. 1c). We found that, for all concentrations of L-citrulline, hypoxic PAECs treated with L-citrulline demonstrated greater NO production than untreated hypoxic PAECs (Fig. 3a). Furthermore, hypoxic PAECs treated with highest L-citrulline concentration, 3.0 mM, demonstrated greater NO production compared to the lowest L-citrulline concentration, 0.1 mM (Fig. 3a). Thus, even when hypoxic PAEC were treated with L-citrulline concentrations that elevated arginase activity (Fig. 1c), L-citrulline increased NO production.
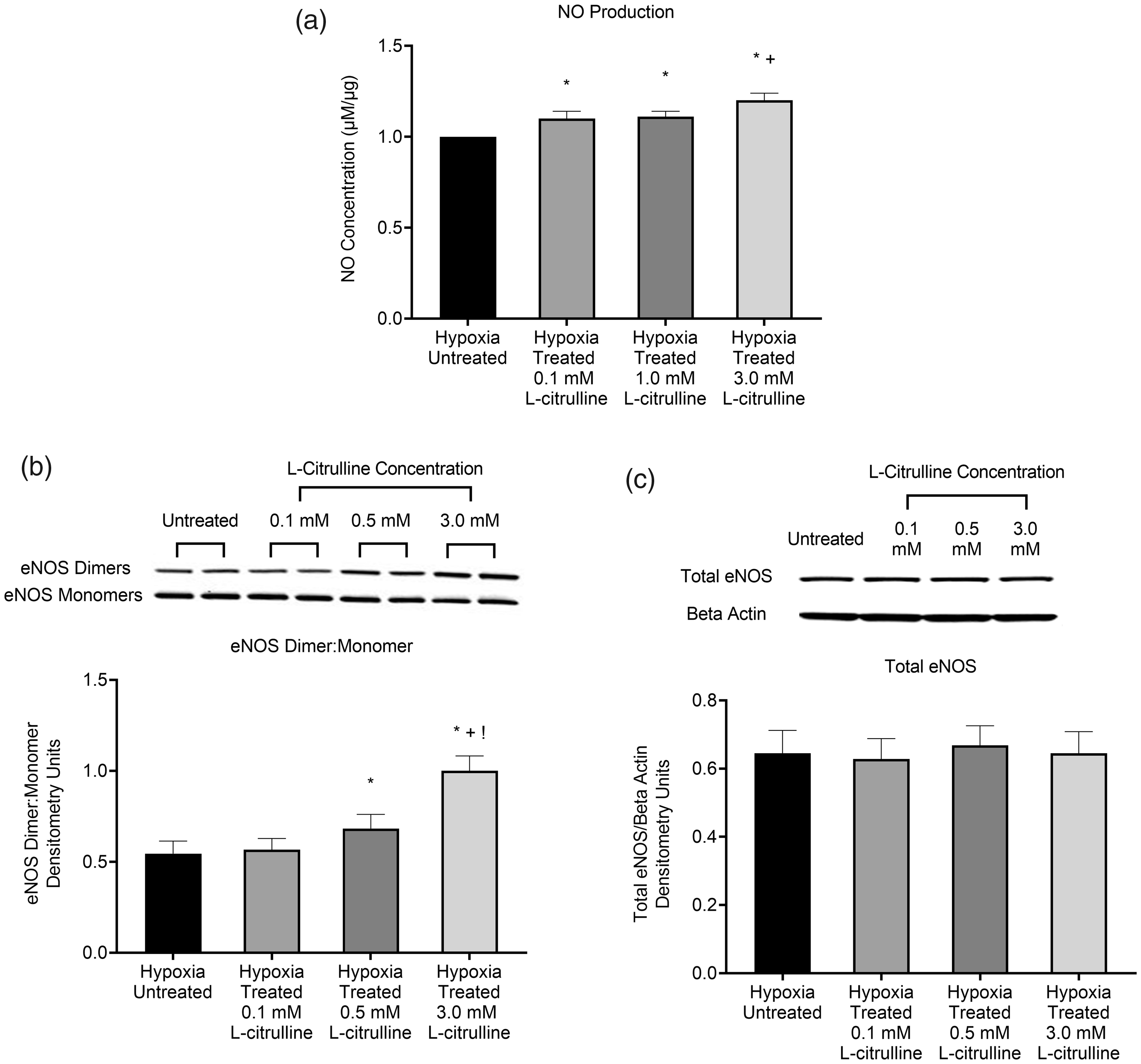
NO production, eNOS dimer-to-monomer ratios, and total eNOS protein levels in hypoxic PAECs treated with and without L-citrulline. (a) Nitric oxide (NO) production was greater for all groups of L-citrulline-treated hypoxic pulmonary artery endothelial cells (PAECs) than for untreated hypoxic PAECs. In addition, NO production was greater for hypoxic PAECs treated with 3.0 mM L-citrulline compared to 0.1 mM L-citrulline (n = 14 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from 0.1 mM L-citrulline). (b) Endothelial nitric oxide synthase (eNOS) dimer-to-monomer ratios were greater for hypoxic PAECs treated with either 0.5 or 3.0 mM L-citrulline than for untreated hypoxic PAECs. In addition, eNOS dimer-to-monomer ratios were greater for hypoxic PAECs treated with 3.0 mM L-citrulline than for all other groups (n = 12 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from hypoxic 0.1 mM L-citrulline, !p < 0.05 from hypoxic 0.5 mM L-citrulline). (c) There was no difference in total eNOS protein levels amongst the groups of hypoxic PAECs (n = 6 for each group).
To explore the effect of L-citrulline on the state of eNOS coupling, we then evaluated the effect of L-citrulline on eNOS dimer-to-monomer ratios in hypoxic PAECs. Uncoupled eNOS can be demonstrated as a loss of eNOS dimer formation and an increase of eNOS monomers. We found that eNOS dimer-to-monomer ratios did not differ between untreated hypoxic PAECs and those treated with the lowest L-citrulline concentration, 0.1 mM (Fig. 3b). However, when compared to untreated hypoxic PAECs, eNOS dimer-to-monomer ratios were greater for hypoxic PAECs cultured in the presence of the two higher L-citrulline concentrations, 0.5 and 3 mM (Fig. 3b). Moreover, eNOS dimer-to-monomer ratios were much greater for hypoxic PAECs cultured in the presence of the highest L-citrulline concentration, 3.0 mM, when compared to all other groups (Fig. 3b). Total eNOS was similar for PAECs from all groups regardless of L-citrulline concentration (Fig. 3c). Thus, L-citrulline increased eNOS dimer-to-monomer ratios even when hypoxic PAEC were treated with a concentration of L-citrulline (3 mM) that elevated both arginase II protein levels and arginase activity (Fig. 1c).
We also wanted to determine the effect of different L-citrulline concentrations on O2– production. For all concentrations of L-citrulline, hypoxic PAECs treated with L-citrulline generated less O2– than untreated hypoxic PAECs (Fig. 4). Furthermore, hypoxic PAECs treated with the highest L-citrulline concentration, 3.0 mM, generated less O2– compared to the lowest L-citrulline concentration, 0.1 mM (Fig. 4). These findings show that L-citrulline treatment reduced the amount of O2– generated by hypoxic PAEC even using concentrations that were found to increase arginase activity (Fig. 1c).
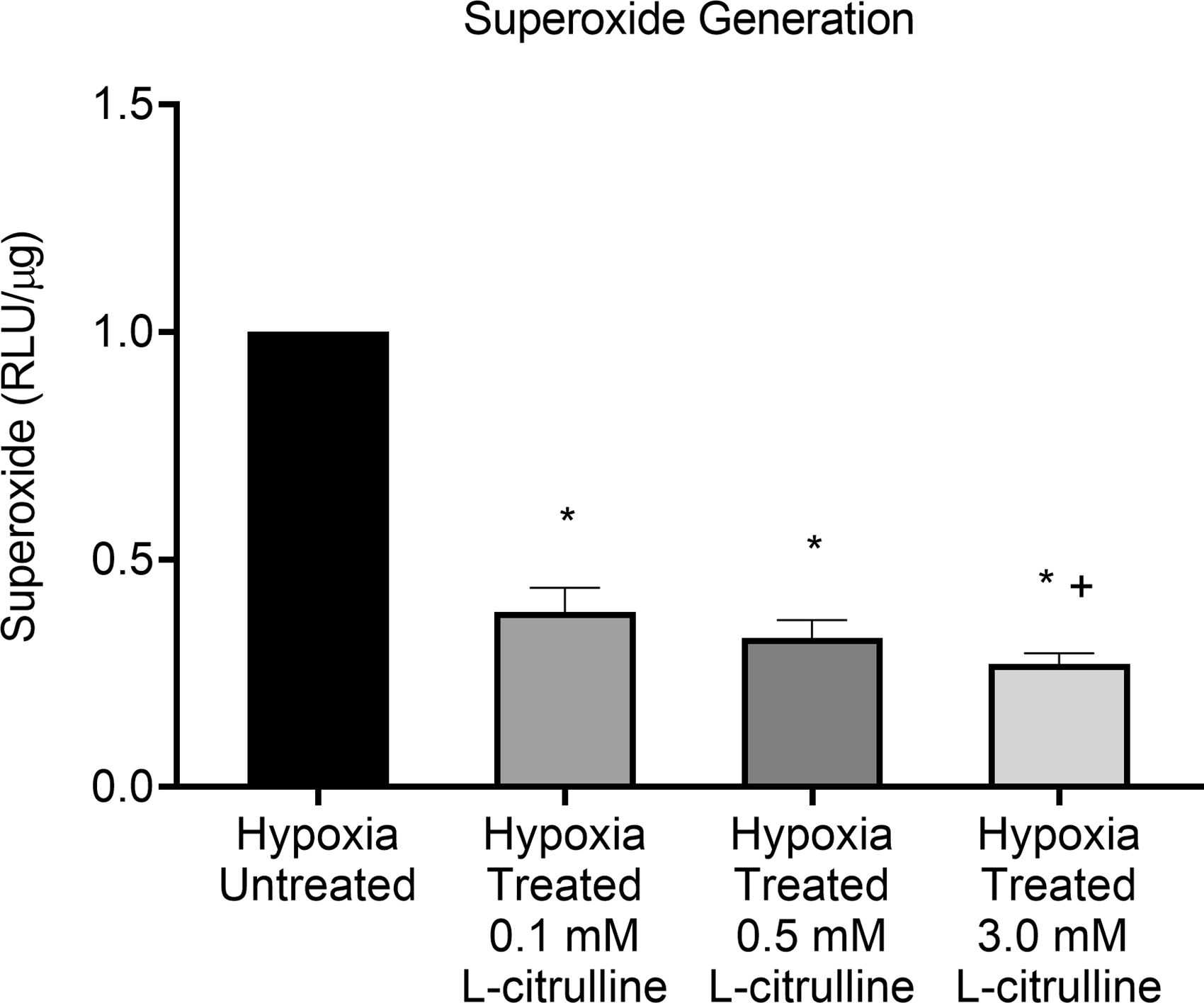
O2– generation in hypoxic PAECs treated with and without L-citrulline. Superoxide (O2–) generation was less for all groups of L-citrulline-treated hypoxic pulmonary artery endothelial cells (PAECs) than for untreated hypoxic PAECs. In addition, O2– generation was less for hypoxic PAECs treated with 3.0 mM L-citrulline compared to 0.1 mM L-citrulline (n = 12 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from 0.1 mM L-citrulline).
We wanted to further evaluate the impact of arginase activity and L-citrulline treatment on NO production, the state of eNOS coupling, and O2– generation in hypoxic PAECs. To do this, we performed studies with the arginase inhibitor, BEC, and a concentration of L-citrulline that increased arginase activity, 3 mM (Fig. 1c). Hypoxic PAECs treated with L-citrulline, BEC, or the combination of BEC and L-citrulline demonstrated increased NO production compared to untreated hypoxic PAECs but there was no significant difference in NO production between the three treatment groups (Fig. 5a). All treatments, both separately and combined, increased eNOS dimer-to-monomer ratios above those found in untreated hypoxic PAECs (Fig. 5b). A similar increase in eNOS dimer-to-monomer ratios was found in hypoxic PAECs treated separately with BEC or L-citrulline; whereas a greater increase in eNOS dimer-to-monomer ratios was found in hypoxic PAECs that received combined treatment compared to those that were treated with either alone (Fig. 5b). There was no difference in total eNOS between untreated hypoxic PAECs and those treated with any of the three treatment strategies (Fig. 5c). All three treatment strategies reduced O2– generation in hypoxic PAECs to levels below those measured in untreated hypoxic PAEC (Fig. 6). Similar to the impact on eNOS dimer-to-monomer ratios, the combination of BEC and L-citrulline decreased O2– generation more than treatment with either alone (Fig. 6) and the change in O2– generation did not differ between those receiving therapy with either alone (Fig. 6). Thus, inhibiting arginase with BEC increased NO production, eNOS dimer-to-monomer ratios, and reduced O2– generation in a fashion similar to treatment with 3 mM L-citrulline. The finding of a greater impact of combined vs single therapy on O2– production and eNOS dimer-to-monomer ratios, suggests that adding a treatment that inhibits arginase could augment some effects of treatment with 3 mM L-citrulline, i.e. a concentration of L-citrulline that increases arginase II protein and arginase activity (Fig. 1b and c).
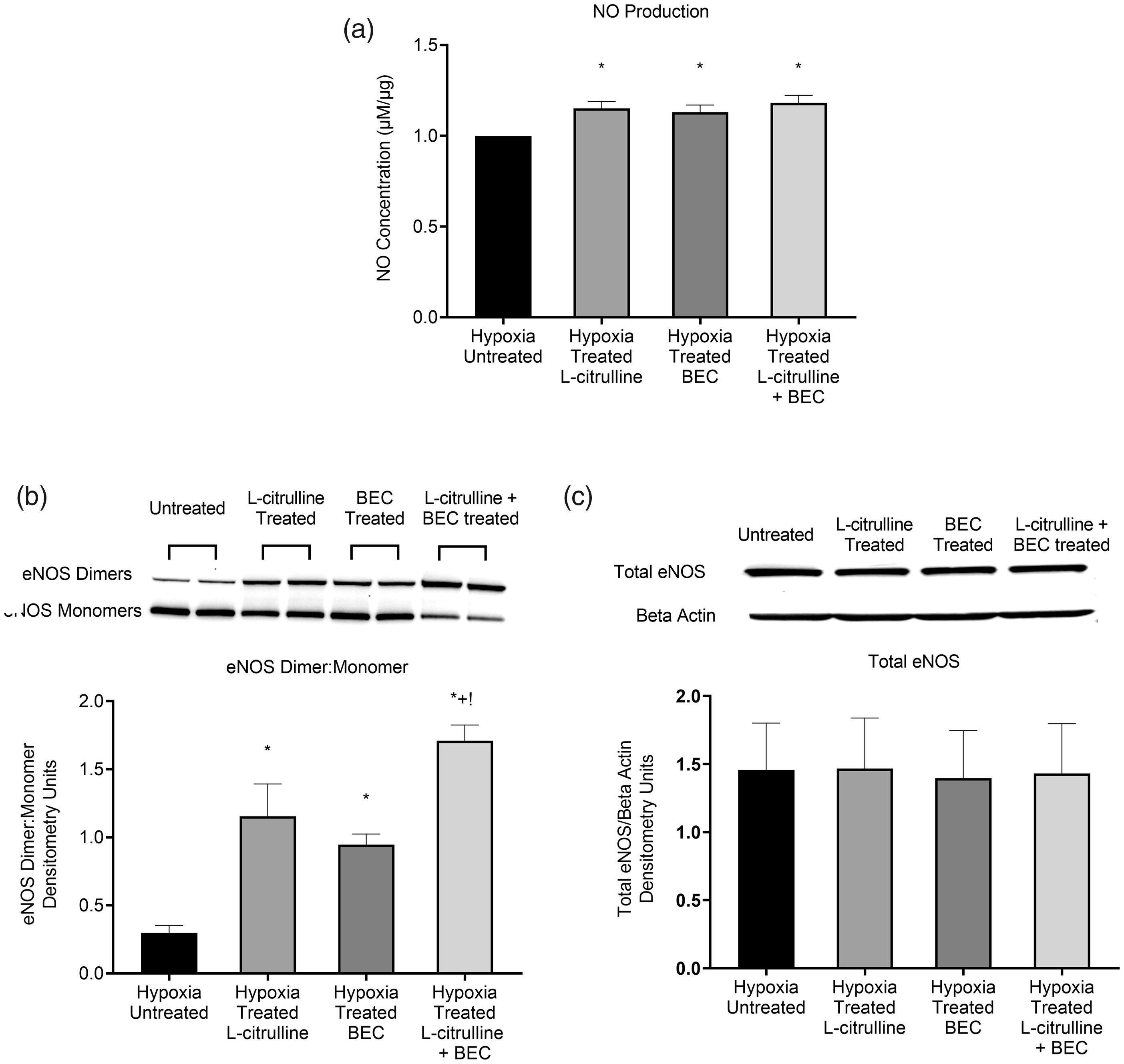
NO production, eNOS dimer-to-monomer ratios, and total eNOS protein levels in hypoxic PAECs treated with L-citrulline and/or the arginase inhibitor, BEC. (a) Regardless of treatment strategy, nitric oxide (NO) production was greater for all three groups of treated hypoxic pulmonary artery endothelial cells (PAECs) compared to untreated hypoxic PAECs. There was no significant difference in NO production between the three treatment groups (n = 14, *p < 0.05 from hypoxic untreated). (b) Regardless of treatment strategy, endothelial nitric oxide synthase (eNOS) dimer-to-monomer ratios were greater for all three groups of treated hypoxic PAECs compared to untreated hypoxic PAECs. eNOS dimer-to-monomer ratios did not differ between PAECs treated solely with either L-citrulline or the arginase inhibitor S-(2-boronoethyl)-L-cysteine (BEC). However, eNOS dimer-to-monomer ratios were greater for hypoxic PAECs co-treated with L-citrulline and BEC than for hypoxic PAECs treated with either L-citrulline or BEC alone (n = 10 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from hypoxic 3.0 mM L-citrulline, !p < 0.05 from hypoxic 100 μM BEC). (c) There was no difference in total eNOS protein levels amongst the groups of hypoxic PAECs (n = 4 for each group).
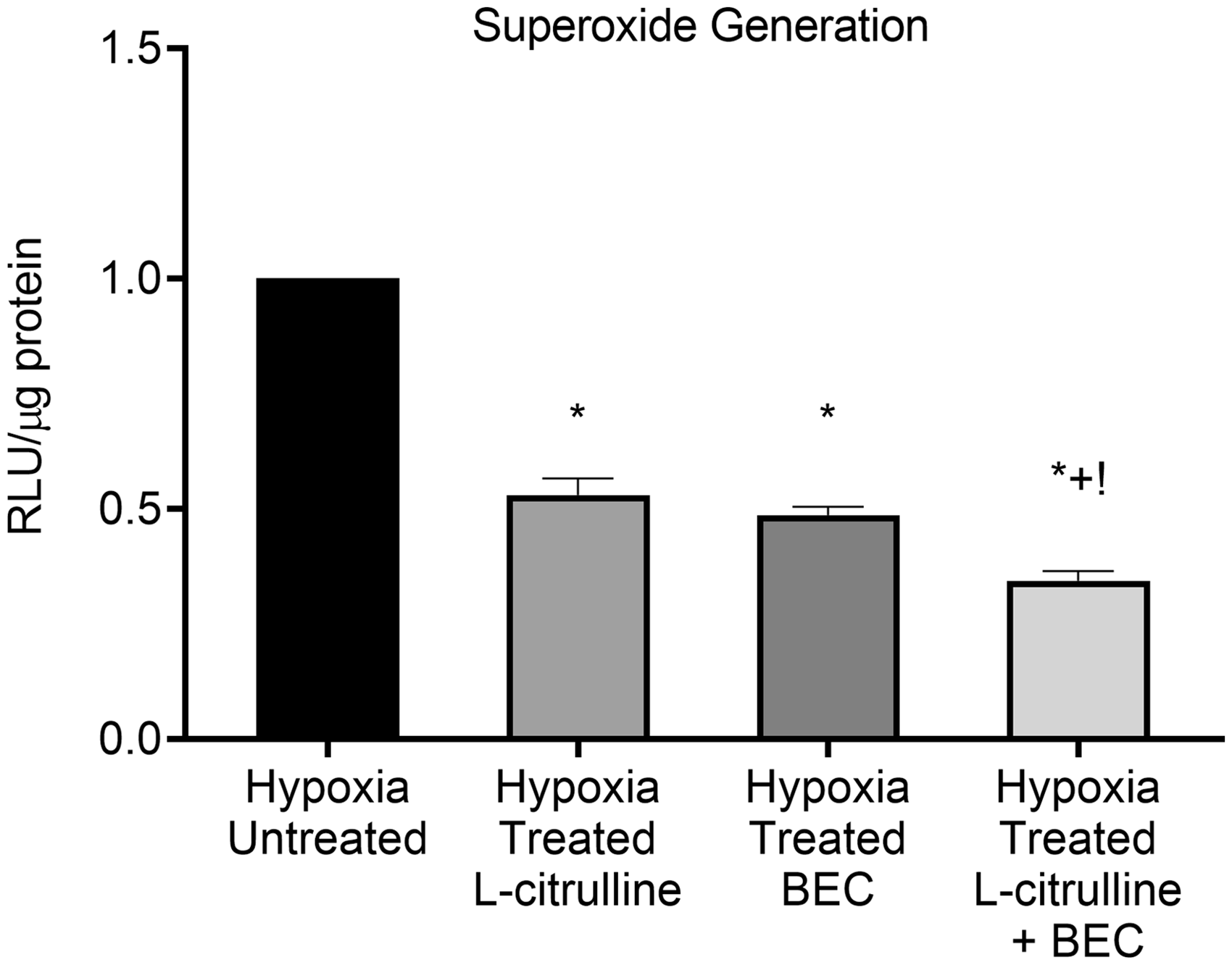
O2– generation in hypoxic PAECs treated with L-citrulline and/or the arginase inhibitor, BEC. Regardless of treatment strategy, superoxide (O2–) generation was less for all three groups of treated hypoxic pulmonary artery endothelial cells (PAECs) compared to untreated hypoxic PAECs. The amount of O2– generated did not differ between PAECs treated solely with either L-citrulline or the arginase inhibitor S-(2-boronoethyl)-L-cysteine (BEC). However, the amount of O2– generated was less for hypoxic PAECs co-treated with L-citrulline and BEC than for hypoxic PAECs treated with either L-citrulline or BEC alone (n = 10 for each group, *p < 0.05 from hypoxic untreated, +p < 0.05 from 3.0 mM L-citrulline, !p < 0.05 from 100 μM BEC).
Discussion
In this investigation, we report some novel effects of L-citrulline supplementation in PAECs from newborn piglets cultured under hypoxic conditions. In particular, we show for the first time that L-citrulline induces arginase activity and arginase II protein amounts in hypoxic piglet PAECs.
Arginase is an enzyme that metabolizes L-arginine into L-ornithine and urea for many normal biologic purposes. However, arginase has been implicated in some forms of cardiovascular disease due to its ability to decrease NO production in endothelial cells, alter NO-mediated vasodilation, and contribute to vascular remodeling in certain disease states, including PH.14,18–21 Arginase exists in two isoforms, arginase I and II, which have distinct subcellular localization in the cytosol and mitochondria respectively. Although there are differences in the tissue distribution of the isoforms, both arginase I and II are present in many cell types.14,18,19,22,23 Of particular interest, both isoforms have been demonstrated in endothelial cells from a variety of species and vascular beds, yet, arginase II may be the predominant endothelial isoform.24–27
In this study, we demonstrate that both arginase I and II are present in hypoxic piglet PAECs but only arginase II protein levels were increased with L-citrulline supplementation and only at concentrations of 1.0 mM or greater (Fig. 1a and b). The increase in arginase II protein levels but not arginase I may be due to differences in regulation because arginase I and II are encoded by separate genes and are known to be regulated by different stimuli.14,19 We also show that when a concentration of 0.5 mM L-citrulline is achieved or surpassed, arginase activity is increased in hypoxic PAECs (Fig. 1c). Prior studies in pig endothelial cells have shown that arginase expression and/or activity can be regulated by hypertension, hydrogen peroxide, uric acid, vascular wall shear stress, oxidized low density lipoprotein, and lipopolysaccharide.24,28–31 We add to the literature by demonstrating that L-citrulline can increase arginase II protein levels and arginase activity in hypoxic piglet PAECs.
Inconsistent with our findings, other authors have suggested that L-citrulline does not induce arginase and may be a non-competitive inhibitor of arginase in some species and cell types.8,32,33 In fact, L-citrulline reversed hyperoxia-induced elevations in arginase activity in lung tissue from a newborn rat model of bronchopulmonary dysplasia. 34 It is possible that there are differences in regulation of arginase between species, animal ages, cell types, and that these differences are further affected by the presence of stimulating or inhibiting factors dependent on disease condition. It is also possible that regulation of arginase will depend on the precise location of endothelial cells along the longitudinal axis of the pulmonary circulation. For example, it is possible that findings with microvascular endothelial cells will differ from findings with endothelial cells derived from the main pulmonary artery as used in this study.
In this study, we did not elucidate the exact mechanism by which L-citrulline induced arginase expression and activity. However, it is known that via a two-step enzymatic process, L-citrulline is the precursor for L-arginine synthesis in PAECs.8,20 In turn, L-arginine can induce arginase expression and activity in endothelial cells.8,35 Thus, it is possible that L-citrulline increased arginase activity in hypoxic PAECs by elevating levels of L-arginine in the subcellular compartments available to arginase I and/or arginase II.
Consistent with our previous studies, in this study, we found that supplementation with the L-arginine precursor, L-citrulline, increased NO production, elevated eNOS dimer-to-monomer ratios, and reduced O2– generation in hypoxic PAECs.10,16 These effects indicate that L-citrulline improves eNOS dysfunction in hypoxic PAECs by re-coupling eNOS. This positive impact of L-citrulline on NO signaling occurred even when concentrations of L-citrulline were used that induced arginase II expression and arginase activity. Yet, arginase has been shown to compete with eNOS for the common substrate, L-arginine, and thereby modulate NO production.14,20,24,35,36 Thus, it is possible that even greater improvements in NO production and eNOS re-coupling would have been found if induction of arginase had not occurred.
Other authors have demonstrated that arginase inhibition in endothelial cells increases NO production and there is increasing recognition that arginase inhibition may represent a potential therapeutic approach for treating diseases in which arginase dysregulation is a contributing factor, including PH.13,36–38 Our findings with the arginase inhibitor, BEC, are consistent with these possibilities. Specifically, we found that treatment with BEC increased NO production, enhanced eNOS dimer-to-monomer ratios, and reduced O2– generation in hypoxic PAECs (Figs 5a and b and 6). Moreover, we found that the increase in eNOS dimer-to-monomer ratios and the reduction in O2– generation were greater when hypoxic PAEC were treated with a combination of BEC and a concentration of L-citrulline that induced arginase than treatment with either alone (Figs 5a and b and 6). Hence, our results extend those of others and suggest that arginase inhibition has the potential to improve the impact of L-citrulline on eNOS dysfunction in hypoxic conditions.
To further evaluate the impact of L-citrulline-induced arginase activity, we measured the protein levels of ODC and OAT which are two downstream enzymes that utilize the L-ornithine produced during the metabolism of L-arginine by arginase. The products of ODC and OAT play an important role in endothelial and smooth muscle cell proliferation and collagen synthesis, respectively, and therefore are thought to contribute to pathologic vascular remodeling in some forms of PH.19,39 Considering our result that L-citrulline increases arginase activity, we expected an increase in ODC and OAT protein abundance due to the likely increase in the shared substrate and product of arginase activity, L-ornithine. However, we found that ODC and OAT protein levels were not increased at any concentration of L-citrulline that we studied, including concentrations that increased arginase activity in hypoxic PAECs (Fig. 2a and b). It is important to note that it remains possible that L-citrulline increased the activity of one or both of these proteins without increasing their protein amounts.
The therapeutic impact of our findings merit comment. We previously found that L-citrulline supplementation inhibited the development of chronic hypoxia-induced PH in newborn piglets. 9 We also previously found that the mean plasma L-citrulline concentrations in untreated hypoxic piglets were 0.1 mM; while, dependent on the dose used for treatment, the L-citrulline-treated hypoxic piglets achieved mean trough plasma L-citrulline concentrations of 0.2 mM and 0.5 mM. 9 These findings are of interest because L-citrulline treatment had an ameliorative effect on the development of PH even though some piglets achieved plasma concentrations of L-citrulline that we found to induce arginase activity in our current study. Moreover, we also previously found that the pulmonary arteries from the L-citrulline-treated piglets produced more NO, generated less O2–, and had higher eNOS dimer-to-monomer ratios than the untreated piglets. 9 In other words, when combined, findings from our previous and current study show that L-citrulline supplementation improves NO signaling in vivo, even in the presence of L-citrulline concentrations that induce arginase activity in vitro (Fig. 1c). Nonetheless, the dose-dependent effect of L-citrulline on arginase activity found in our current study should raise the concern that that there could be an in vivo concentration of L-citrulline that elevates arginase activity sufficiently to negate the beneficial impact of L-citrulline on NO signaling and PH in chronically hypoxic piglets.
It also merits comment that to date findings from our studies show that L-citrulline inhibits the progressive development of chronic hypoxia-induced PH. Whether or not L-citrulline can actually reverse well-established PH has yet to be clearly shown and will require further investigative efforts.
In summary, L-citrulline increases arginase II protein levels and arginase activity in hypoxic piglet PAECs in a dose-dependent fashion. None of the concentrations of L-citrulline used in this study elevated arginase activity sufficiently to negate the ability of L-citrulline to improve eNOS dysfunction in PAECs from newborn piglets cultured under hypoxic conditions. The arginase inhibitor, BEC, improved eNOS dysfunction in hypoxic PAECs in a manner similar to L-citrulline. Moreover, combining an arginase inhibitor with L-citrulline treatment had a greater impact on some parameters of eNOS dysfunction in hypoxic PAECs than treatment with either alone. Taken together, these findings suggest arginase inhibition may have potential as an adjunctive approach to L-citrulline therapy and that there may be a dose-dependent concentration that should not be exceeded when considering the use of L-citrulline to treat PH in conditions associated with chronic hypoxia.
Footnotes
Author contributions
Conception and design of study: M.S.D., C.D.F.; data acquisition: M.S.D., Y.Z., M.R.K.; analysis and interpretation of data: M.S.D., Y.Z., M.R.K, C.D.F.; drafting of manuscript: M.S.D., C.D.F.; read and approved the final version of the manuscript: M.S.D., Y.Z., M.R.K, C.D.F.
Guarantor
C.D.F.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.D.F. is listed as a co-inventor on a patent for the use of L-citrulline as a therapeutic treatment for lung conditions.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Heart, Lung, and Blood Institute (RO1-HL-097566, R56-HL-097566).
