Abstract
Alveolar capillary dysplasia with misalignment of the pulmonary veins (ACDMPV) is a rare congenital pulmonary disease that affects newborns. Most patients with ACDMPV are born at full term and are healthy. The main clinical manifestations are refractory pulmonary hypertension and pulmonary failure with gastrointestinal, urinary, or cardiac malformations. ACDMPV often progresses rapidly, but no conventional biological or imaging tests other than genetic testing are available for its diagnosis. Lung biopsy is currently the gold standard for diagnosis. We herein report two cases of ACDMPV confirmed by pathological examination and discuss their ultrasonographic findings.
Keywords
Introduction
Alveolar capillary dysplasia with misalignment of the pulmonary veins (ACDMPV) is a congenital lung disease that can result in persistent pulmonary hypertension of the newborn (PPHN). Affected patients die of respiratory failure. The main pathological changes in ACDMPV are decreased density and abnormal positioning of the pulmonary capillaries, thickening and hypertrophy of the small pulmonary arterioles, immature lobular development, lymphangiectasia, muscularization of the distal arterioles, and misalignment of the pulmonary veins.1–4 Since the description of the first autopsy case of a term infant with alveolar capillary dysplasia in 1981, 5 approximately 200 cases have been reported worldwide, and these cases showed a relatively even geographical distribution.6–29 Because the prenatal histories in these cases were generally normal and no characteristic manifestations were noted in the imaging examinations,30–33 clinical evidence for diagnosis is still lacking. With the development of ultrasound, lung ultrasound (LUS) has become an extremely important bedside imaging tool in the neonatal intensive care unit (NICU). It is often used for the differential diagnosis and follow-up of pulmonary diseases.34–36 LUS has no risk of radiation and is reproducible but operator-dependent. A-lines, B-lines, and the lung sliding sign can be used to identify ACDMPV. When the ultrasound probe is perpendicular to the pleura, a linear high-echo reflection parallel to the pleura line is generated because of the multiple reflections formed by the reverberation effect located below the pleura line. In the two-dimensional grayscale mode, A-lines present as a series of smooth, clear, and regular linear strong echo reflections parallel to the pleura line. 30 In the presence of an allergic reaction, alveolar capillary expansion, higher permeability, fluid and fiber infiltration, pulmonary edema, and interlobular septal thickening occur; B-lines in such cases present as multiple ultrasound reflections into the space between the adjacent lung lobules and the space between the pleura and adjacent lung lobule. 30 The lung sliding sign occurs in the presence of horizontal relative sliding of the visceral pleura and parietal pleura with the movement of pulmonary respiration and can be seen at the pleura line under real-time ultrasound. In this study, we report two cases of ACDMPV diagnosed by pathological examination and present and analyze their LUS patterns.
Case reports
Case 1
A female neonate born at 37 weeks’ gestation was admitted to our institution with a 9-hour history of cyanosis. Her Apgar scores were within the normal range. The cyanosis had developed within 3 hours after her uneventful delivery, and her oxygen saturation (O2Sat) was 68%. Her symptoms did not improve considerably after administration of oxygen via a high-frequency ventilator. She was transferred to the NICU. Physical examination showed low O2Sat, cyanosis, and scattered sputum sounds in both lungs; there were no obvious abnormalities of the heart and abdomen. Routine blood test results were normal. A bedside echocardiogram showed patent ductus arteriosus and pulmonary hypertension. LUS (GE Vivid-E9 Dimension Color Doppler ultrasound machine with GE LOGIQ E9 line-array probe 9L-D [frequency of 3–10 MHz] and convex array probe C2-9 [frequency of 4.5–9 MHz0]; GE Healthcare, Chicago, IL, USA) showed dense B-lines on the lung surface and a lack of pulmonary consolidation (Figure 1). Because the patient was 12 hours old at the time of examination, the B-lines were interpreted as the manifestation of neonatal lung transition. Chest X-ray (CXR) examination showed diffuse haziness in both lungs. Treatment for hypoxemia was immediately provided with a mechanical ventilator, and inhaled nitric oxide (iNO) was administered to reduce the pulmonary arterial pressure. Obvious improvements in her condition were noted.

Ultrasonographic images of the lungs 12 hours after birth. (a) B-line was observed on the long-axis image of the inferior vena cava and on most of the lung surface (white arrow). Only a few A-lines were present in the right anterior lung. (b) The pulmonary sliding sign and B-line were observed in the dynamic images. (c) The pleural lines (white triangles) were thickened, and their echo responses were uneven.
At 22 hours after birth, the neonate’s O2Sat and blood pressure began to progressively decrease. Three hours later (25 hours after birth), symptoms of brain damage and heart failure appeared. Her pulmonary arterial pressure did not decrease despite the continued administration of iNO. Right heart failure and neonatal shock then occurred. The doctors suggested a genetic test, but the parents refused. The neonate finally died 30 hours after birth.
Because of the presence of persistent pulmonary hypertension and sensitivity to iNO only at the initial stage, ACDMPV was highly suspected. Thus, an ultrasound-guided lung puncture was performed 1 hour after death. LUS showed diffuse dense B-lines on the lung surface and thickened pleural lines on the local lung, and A-lines were still present (Figure 2). We obtained a lung tissue sample (area of 1.5 × 0.2 cm2), and the pathological examination results (Figure 3) were consistent with congenital ACDMPV.

Ultrasound-guided lung biopsy in Case 1. (a) Lung ultrasound image of the right anterior and upper chest wall showed thickened pleural lines (white triangles) on the local lung surface and sporadic B-lines (white arrows) in all hypoechoic images. (b) The surface of the lung was filled with dense B-lines (white arrows) with lengths of >3 mm and (c) Using lung ultrasound guidance, the operator obtained lung tissue in the right anteroinferior direction. The arrowhead indicates the puncture needle.

Pathologic examination findings of puncture biopsy of right lung tissue in Case 1. (a) Field of view centered on an arteriole and its surrounding venules. The venules grew in nests to form capillary loops (hematoxylin and eosin staining, 10 × 10) and (b) Extensive widening of the alveolar septum. Small venules grew in nests to form capillary loops (hematoxylin and eosin staining, ×40). The lesion was consistent with congenital alveolar capillary dysplasia with misaligned pulmonary veins.
Case 2
A 1-hour-old male neonate born at 39 weeks’ gestation was admitted to our institution with congenital anal atresia. His Apgar scores were within the normal range, and he had no respiratory symptoms. The screening results suggested a high risk of Down syndrome, and prenatal ultrasound showed that he had no gallbladder. Physical examination revealed a single transverse palmar crease on his right palm. After admission, routine blood tests showed no obvious abnormalities. CXR examination exhibited multiple consolidations in both lungs. An echocardiogram revealed patent ductus arteriosus and pulmonary hypertension. LUS using the same system as in Case 1 showed dense B-lines and a small number of A-lines in some lung fields with the lung sliding sign (Figure 4). The child was diagnosed with congenital anal atresia, congenital omphalocele, and neonatal wet lung.

Partial echocardiography and lung ultrasound images in Case 2. (a) Short-axis image of the ventricle showed dense B-lines (white arrows) on the adjacent lung surface at the level of the papillary muscle of the left ventricle. (b) Long-axis image of the inferior vena cava also showed dense B-lines (white arrows) on the surface of the lung. (c) Inflow tract of the right ventricle around the sternum showed dense B-lines (white arrows) on the surface of the right lung and (d–f) Lung ultrasound showed diffuse B-lines (white arrows) on most of the lung surface, and a small number of A-lines (white triangles) were observed on the left side of the lung.
Because the newborn’s condition had become stable by 31 hours after birth, he underwent surgical treatment of the anal atresia and omphalocele. However, PPHN manifested after the operation. Continuous iNO was administered, and sildenafil and bosentan were provided to immediately reduce the pulmonary arterial pressure; however, no obvious clinical effect was seen. Multiple CXR examinations showed persistent pulmonary consolidation, and echocardiography revealed pulmonary hypertension. According to the clinical profile and imaging results, the child was finally diagnosed with primary PPHN. Multiple LUS scans (Table 1) at 2, 9, 12, 13, 15, 18, and 21 days after birth showed that the B-line distribution had gradually increased. To determine the cause of the patient’s multiple malformations, we performed genetic tests 15 days after birth. The results revealed heterozygous variation of the FOXF1 gene (Figure 5), which suggested that the underlying cause of PPHN was likely to be ACDMPV. Therefore, pulmonary surfactant (PS) was given 15 days after birth, and the O2Sat improved. Twenty-four days after birth, we performed an ultrasound-guided lung puncture. LUS before the operation showed dense B-lines that were fused into the waterfall sign in both lungs. Lung tissue (area of 1.5 × 0.2 cm2) was obtained.
Series of follow-up lung ultrasound examinations in Case 2.
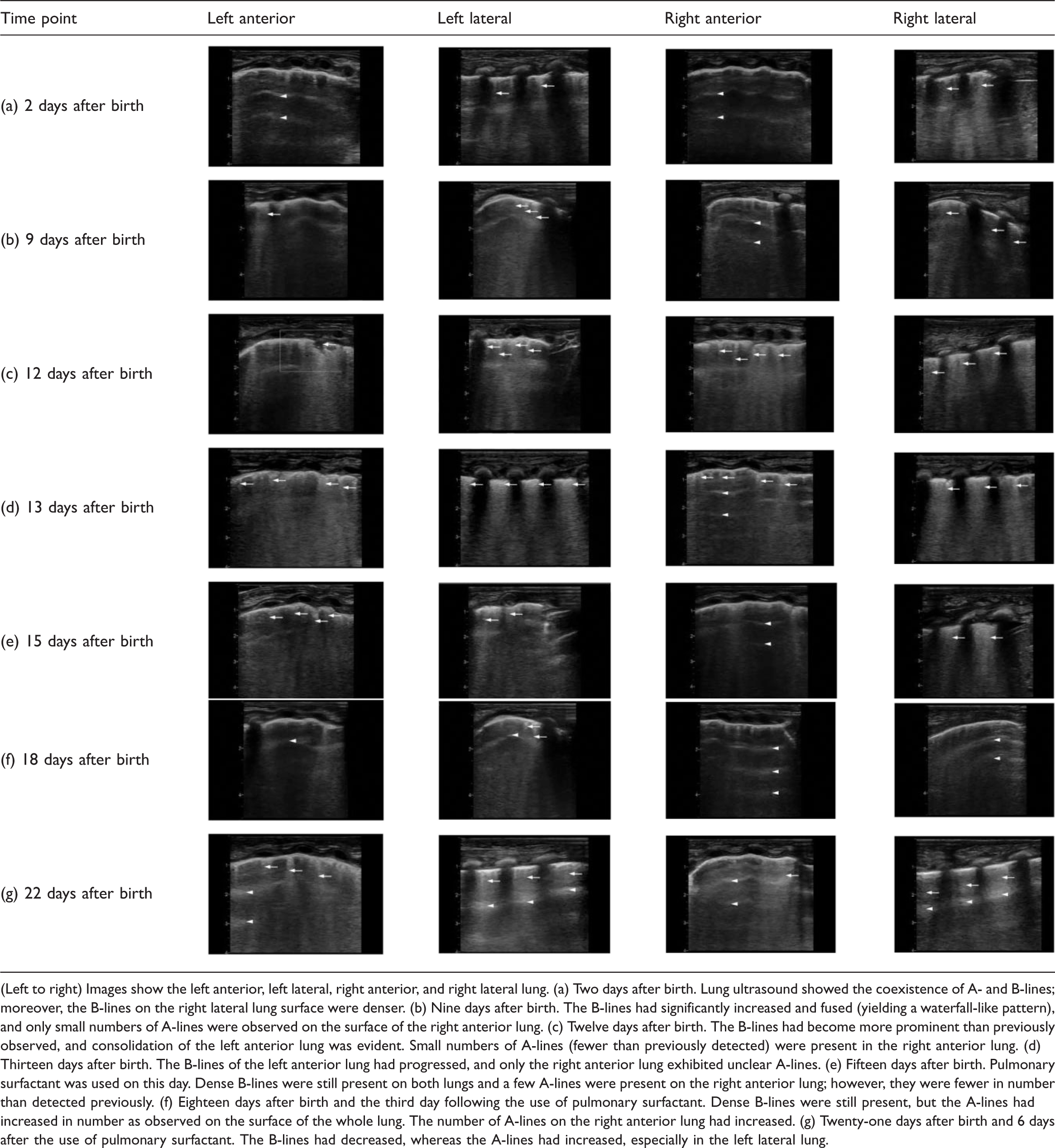
(Left to right) Images show the left anterior, left lateral, right anterior, and right lateral lung. (a) Two days after birth. Lung ultrasound showed the coexistence of A- and B-lines; moreover, the B-lines on the right lateral lung surface were denser. (b) Nine days after birth. The B-lines had significantly increased and fused (yielding a waterfall-like pattern), and only small numbers of A-lines were observed on the surface of the right anterior lung. (c) Twelve days after birth. The B-lines had become more prominent than previously observed, and consolidation of the left anterior lung was evident. Small numbers of A-lines (fewer than previously detected) were present in the right anterior lung. (d) Thirteen days after birth. The B-lines of the left anterior lung had progressed, and only the right anterior lung exhibited unclear A-lines. (e) Fifteen days after birth. Pulmonary surfactant was used on this day. Dense B-lines were still present on both lungs and a few A-lines were present on the right anterior lung; however, they were fewer in number than detected previously. (f) Eighteen days after birth and the third day following the use of pulmonary surfactant. Dense B-lines were still present, but the A-lines had increased in number as observed on the surface of the whole lung. The number of A-lines on the right anterior lung had increased. (g) Twenty-one days after birth and 6 days after the use of pulmonary surfactant. The B-lines had decreased, whereas the A-lines had increased, especially in the left lateral lung.

Results of first-generation sequencing verification of the FOXF1 gene variation in families with ACDMPV showed heterozygous variation of the FOXF1 gene in children with ACDMPV, and the mutation site was c376_377insT; p(.Pro126fs). No mutation was found at this site in either parent.
At 26 days after birth, the patient’s O2Sat and blood pressure decreased again, finally leading to his death. The pathological examination results indicated ACDMPV (Figure 6).

Histologic examination findings of ultrasound-guided biopsy of the right lung in Case 2. (a, b) The lung interstitium was widened and the pulmonary capillary density was decreased (hematoxylin and eosin staining: a, ×100; b ×40) and (c) Misaligned pulmonary veins were located within the pulmonary arteriole sheath (Masson’s trichrome stain, ×100).
Review of the literature
A literature search of imaging findings (CRX, computed tomography [CT], or LUS) in several Chinese databases (including the China National Knowledge Infrastructure, China Biology Medicine, and Wanfang Data databases) and PubMed up to December 2021 was performed. Using the term “alveolar capillary dysplasia,” 31 articles (48 cases) that included definite imaging findings and diagnoses were found. Of these 48 cases, 44 had descriptions of CXR findings, 13 had descriptions of chest CT findings, and only 4 had descriptions of LUS findings. The detailed information on these patients and their imaging findings are provided in Supplementary Tables S1 and S2.
Discussion
PPHN is a clinical manifestation of ACDMPV. For neonates with hypoxemia, a confirmed diagnosis is needed before an etiological treatment can be implemented. On the premise that the underlying cause of pulmonary hypertension cannot be diagnosed, the neonates in these two cases only responded briefly to the PPHN treatment, developed persistent pulmonary hypertension, and then died. Thus, the diagnosis of ACDMPV still requires additional research. Only about 10% of cases reported to date have been familial. 37 Thus, it is difficult for the clinician to advise the patient’s family based only on the results of a genetic test for the diagnosis of ACDMPV, as the patient’s family history usually suggests.
The gold standard for diagnosing ACDMPV is histological examination of the lung. 33 According to the pathological findings of ACDMPV reported to date, the histopathological features of this disease are mainly decreased density and abnormal positioning of the pulmonary capillaries as well as thickening of the walls of small peripheral pulmonary arteries and the pulmonary interstitium. In addition, immature lobular development, lymphangiectasia, muscularization of distal arterioles, and misalignment of the pulmonary veins result in PPHN.1–4,30,38 All the pathological changes in our two patients were consistent with the features of ACDMPV. Nevertheless, the clinical applicability of histological examination findings is limited: lung tissue sampling requires that the patient has a highly stable clinical condition, and it should be conducted before extracorporeal membrane oxygenation. 39 Most cases of ACDMPV progress rapidly, as in Case 1 of the present study; the referred child died of heart failure and shock soon after her condition began to progress. Thus, it is difficult or impossible to complete standard histological examinations while the patient is still alive. According to the case reports published to date, approximately 90% of ACDMPV cases are diagnosed postmortem.4,40 Therefore, LUS, as a noninvasive imaging modality that does not involve radiation, could be an examination technique of choice for neonates with idiopathic hypoxemia who do not respond to PPHN treatment. It is rapid and inexpensive and can be performed at the bedside.
LUS in these two cases showed dense B-lines on the lung surface. This finding was consistent with the thickening of the interlobular septa as shown by pathologic examination. The thickening of the alveolar interstitium as shown by light microscopy was more obvious in Case 2 than in Case 1. Correspondingly, the density of the B-lines pm LUS was higher in Case 2 than in Case 1. The B-lines worsened gradually before the death of the patient in Case 2: the B-lines gradually became denser, and a small portion of them fused into the waterfall sign. This suggests that the density of the B-lines on LUS in patients with ACDMPV might be related to the severity of the interlobular lesions. Del Rey Hurtado de Mendoza et al. 41 analyzed four children with ACDMPV and found that A-lines were present in all patients, but no B-lines were observed. In the present study, we used high-frequency ultrasound combined with echocardiography to completely observe the whole lung, and B-lines were observed. Thus, according to the pathological examination, B-lines might be an important manifestation of LUS in patients with ACDMPV, but this requires confirmation in more systematic studies. Therefore, a diagnosis of ACDMPV should be seriously considered in neonates with idiopathic hypoxemia who do not respond to PPHN treatment and who display dense B-lines on the lung surface.
When neonates show low O2Sat, clinicians often use CXR examination to predict the underlying cause. However, according to the literature review shown in Supplementary Tables S1 and S2, most of the CXR images of patients with ACDMPV showed diffuse haziness or pneumothorax, most likely caused by mechanical ventilation. Therefore, CXR imaging does not show features specific for ACDMPV. Most CT scans demonstrated a ground-glass opacity (least specific), and only two cases showed thickened peribronchial soft tissue. By contrast, chest CT revealed more severe lung lesions than CXR; nevertheless, it is still difficult to determine the presence of ACDMPV based on these findings. 42 There are no absolute defined limits for patient radiation exposure. However, according to the ALARA principle, it is reasonable to keep the doses as low as possible or achievable, especially for children. 42 Therefore, LUS is an attractive option. Notably, neither CXR nor CT is recommended for monitoring and following up children with ACDMPV; instead, the application of dynamic ultrasound has been proposed. 43
LUS is a well-developed technique worldwide.44–46 In some NICUs, LUS examinations have partly replaced the routine CXR examinations for diagnosing and differential neonatal lung diseases. 47 At the same time, LUS can accurately depict the locations of lesions to guide lung tissue puncture biopsy, which can also provide important clinical assistance for diagnosing lung diseases. LUS might be useful in the diagnosis of ACDMPV.
The clinical manifestation of pulmonary hypertension with diffuse B-lines in two-dimensional LUS should be differentiated from bronchopulmonary dysplasia, neonatal respiratory distress syndrome, and meconium aspiration syndrome. Children with bronchopulmonary dysplasia exhibit hypovascularity and abnormal function and structure of blood vessels. This leads to a decrease in the gas exchange area and to pulmonary hypertension. 48 The sonographic features of LUS mainly include an abnormal pleural line, diffuse B-lines (pulmonary interstitial syndrome), and vesicle inflation signs, among others. 49 The lack of PS in children with neonatal respiratory distress syndrome leads to alveolar collapse, decreased inflation, alveolar and interstitial edema, and thickening of the interlobular septum, finally resulting in significant changes in the gas-to-water ratio in lung tissue. The sonographic features of LUS mainly include an abnormal pleural line, bilateral diffuse B-line fusion into the waterfall sign, and lung consolidation. 50 By contrast, LUS in neonates with meconium aspiration syndrome shows uniform changes in both lungs, serious alveolar rupture, and pneumothorax; disappearance of the lung sliding sign; and a complete A-line sign. 50 The clinical manifestations and imaging results are similar and can be differentiated by the responses to PS treatment and ventilator-assisted ventilation.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221126876 - Supplemental material for Ultrasound findings in neonates with alveolar capillary dysplasia with misalignment of the pulmonary veins: report of two cases
Supplemental material, sj-pdf-1-imr-10.1177_03000605221126876 for Ultrasound findings in neonates with alveolar capillary dysplasia with misalignment of the pulmonary veins: report of two cases by Yan-bing Lin, Bei Xia, Juan Cao and Zi-Jian Tang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605221126876 - Supplemental material for Ultrasound findings in neonates with alveolar capillary dysplasia with misalignment of the pulmonary veins: report of two cases
Supplemental material, sj-pdf-2-imr-10.1177_03000605221126876 for Ultrasound findings in neonates with alveolar capillary dysplasia with misalignment of the pulmonary veins: report of two cases by Yan-bing Lin, Bei Xia, Juan Cao and Zi-Jian Tang in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605221126876 - Supplemental material for Ultrasound findings in neonates with alveolar capillary dysplasia with misalignment of the pulmonary veins: report of two cases
Supplemental material, sj-pdf-3-imr-10.1177_03000605221126876 for Ultrasound findings in neonates with alveolar capillary dysplasia with misalignment of the pulmonary veins: report of two cases by Yan-bing Lin, Bei Xia, Juan Cao and Zi-Jian Tang in Journal of International Medical Research
Supplemental Material
sj-pdf-4-imr-10.1177_03000605221126876 - Supplemental material for Ultrasound findings in neonates with alveolar capillary dysplasia with misalignment of the pulmonary veins: report of two cases
Supplemental material, sj-pdf-4-imr-10.1177_03000605221126876 for Ultrasound findings in neonates with alveolar capillary dysplasia with misalignment of the pulmonary veins: report of two cases by Yan-bing Lin, Bei Xia, Juan Cao and Zi-Jian Tang in Journal of International Medical Research
Footnotes
Acknowledgements
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
The parents of the patients described in this report provided their written consent to publish images and other clinical information pertaining to their children. The parents of the patients understand that their children’s names and initials have not been published and that due efforts were made to conceal their identities. All experiments were performed in accordance with relevant guidelines and regulations. This study was approved by the ethical committee for medical research at Shenzhen Children’s Hospital (Reference Number: 2022039).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
