Abstract
Implantable pumps for intravenous treprostinil provide a promising option to overcome drawbacks of parenteral prostanoid administration with external pumps in pulmonary hypertension. We retrospectively analyzed 85 patients undergoing implantation in a single center since 2010. In our cohort, serious complications were rare, and flow rate increase over time warrants careful monitoring.
Despite the availability of a range of oral treatment options, parenteral prostacyclins remain a cornerstone in the treatment of pulmonary arterial hypertension (PAH). According to current guidelines, parenteral prostacyclin is recommended as part of combination therapy regimen in case of inadequate response to initial treatment, while in high risk, the guidelines call for initial combination including parenteral prostacyclin. 1 In clinical routine, however, administration of prostacyclin using external pumps is challenging and associated with infusion-site reactions for subcutaneous administration in the majority of patients or rare but possibly life-threatening catheter-related infections for the intravenous route.2,3 Therefore the availability of a fully implantable pump for intravenous treprostinil in 2009 (Lenus pro®, Tricumed Medizintechnik GmbH, Kiel, Germany) provided a promising option to administer treprostinil avoiding local side effects of subcutaneous treatment and in parallel limit the risk for catheter-related infections. Nevertheless, this approach requires surgical intervention which is associated with significant risk for patients with pulmonary hypertension. 4 Therefore, an interdisciplinary expert panel defined criteria and procedures for pre-, peri-, and postoperative management of patients at our center. Key responsibility of the PAH physician is achieving adequate clinical response by optimizing PAH-specific therapy. All patients are started on subcutaneous treprostinil and uptitrated in an outpatient setting under supervision of specialized nursing staff. Implantation of a pump for intravenous treprostinil can only be proposed if the patient is stable, devoid of signs of right heart failure, and additionally suffers from significant side effects such as severe site pain or repeated infections at the infusion site. Patients unable to handle the external infusion pumps as well as patients suffering from the stigma of a visible external pump are also considered eligible after uptitration. After clearance from the PAH team, the patient is independently assessed by the anesthesiologist and the surgeon. Both anesthesia and surgery are performed by dedicated physicians only. All implantations are performed in general anesthesia including extended hemodynamic monitoring. Postoperatively blood pressure, oxygen saturation, and heart rate are continuously monitored for 24 h. Pump refills are done by dedicated staff only at our outpatient clinic.
We performed a retrospective database analysis of all pump implantations performed at our center between September 2010 and November 2019 with a focus on procedure- and system-related events.
During the study period, 85 patients (47 female, 38 male) underwent pump implantation; 58 patients were diagnosed with PAH, 17 patients with inoperable chronic thromboembolic pulmonary hypertension, 9 patients with combined pre- and postcapillary pulmonary hypertension, and 1 patient with group V pulmonary hypertension. Six patients were referred for pump implantation from other hospitals and followed up outside our center. Mean age at the time of implantation was 66 years (range: 16–85), mean time on subcutaneous treprostinil was 9 months (range: 1–78), and mean treprostinil dose was 26.1 ng/kg/min (range: 7.4–120.8) as shown in Fig. 1.
Month on s.c. Therapy and Treprostinildose at Implantation.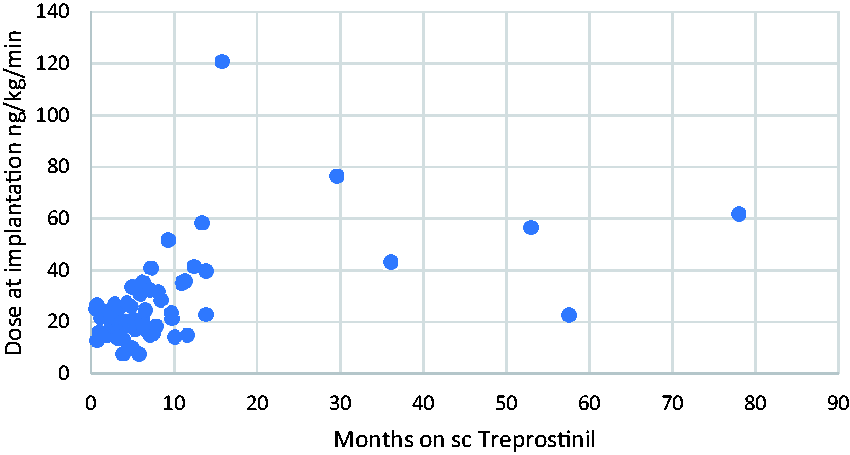
The reason for pump implantation was significant site pain in 77 patients (90.6%), inability to handle the infusion pump in 1 patient, and psychological reasons in 7 patients. In 70 patients, Lenus pro® pumps (reservoir volume 40 ml, flow rate of 1.3 ml/day in 69 patients, 60 ml and 2 ml/d in one case) were implanted. Fifteen patients were implanted within a clinical trial using the Siromedes® pump (Tricumed Medizintechnik GmbH, Kiel, Germany).
Intraoperatively, we observed one case of ventricular tachycardia caused by the central catheter, the patient recovered without sequelae, two cases of pneumothorax as a known complication of puncture of the vena subclavia, and one case of hematothorax in a patient with concomitant hematological malignancy were managed successfully during postoperative stay. A hypotensive episode in one patient required catecholamine support during the postoperative night. Mild seroma was observed in seven patients.
At the time of evaluation, mean follow-up was 25 months (range: 1–104), and mean treprostinil dose was 54.19 ng/kg/min (range 20.3–132.7). Ten unplanned surgical interventions were observed during total 2095 patient-months. Refixation of the pump after rupture of fixation of the pump was necessary in four patients, and six surgical interventions were necessary for catheter dislocations. These were mainly due to mechanical reasons quite atypical for patients with PAH like extensive arm movements during gardening or excessive skiing and mountainbiking leading to two interventions in one single patient. The observed incidence of serious complications related to the pump and catheter per 1000 treatment-days was 0.063 and 0.095, and total incidence of serious events including the peri-procedural events and one refill complication was 0.254 per 1000 treatment-days and 0.366 for all observed events. Importantly, no case of catheter-related infection nor surgical-site infection was observed. Our experience compares favorably to a recent publication from German centers 7 who observed 0.60 serious adverse events per 1000 treatment-days. However, comparison of results is difficult, as procedures at multiple centers do not follow the same uniform standard as in our hospital.
An important finding in the long-term management of our patients was the increase in the flow rate of the pump. Possibly, chemical substances within the treprostinil solution cause changes at the glass capillary regulating the continuous flow. As already published by other groups,5–7 this increase occurs gradually over time. Richter and coworkers recently showed that flow rate increase above the upper limit of technical deviation (7.5%) is evident after three years. 8 These results are consistent with the findings in our cohort. However, we have seen increasing flow rate as early as 12–15 months after implantation in single pumps. This early increase was within the limit of technical deviation in all cases, however, followed by further gradual increase of the flow rate. Flow rate can be calculated by measuring reflow volume at refill and dividing administered volume by days since last refill. To increase accuracy of this calculation, the pump producer recommends using a “reference flowrate” which is the arithmetic mean of the last three calculated flow rates. To be able to also foresee trends, we implemented a dedicated documentation system to not only document reflow volumes but also the development of flow rate changes over time. This allows us to adapt treprostinil dose and in the further course change the refill interval from four to three weeks. So far this was necessary in nine of our patients; in one case, we had to shorten the refill interval to two weeks for a flow rate increase from originally 1.3 ml/day to 2.3 ml/day. This led to a planned pump replacement after 78 months of therapy with the first pump. During the observation period, pump change was performed in another nine patients; in six cases, pumps were changed before increase in flow rate would have required a two-week refill interval which was considered unacceptable by the patients, in three patients, the original pump was replaced by a pump with a 60 ml reservoir to facilitate further dose increase. All replacements were performed without complication by the dedicated surgical team. We did not observe any serious events related to flow rate increase. With the availability of generic treprostinil (Trisuva®, Amomed Pharma GmbH, Vienna, Austria) in 2018, we gradually switched all our patients from the originator to this more cost-effective product, as stability data for both implantable pumps used at our center were available. Evaluation of flow rates demonstrated no impact of this intervention on pump function, neither in newly implanted nor in pumps implanted before the product switch.
During more than 2000 refill procedures, we have observed a single complication, when the patient reported dizziness after refill. For significant hypotension, the patient was admitted to our ICU, and according to our standards, the pump was emptied immediately and filled with saline. Reflow indicated a paravasate of 4 ml. Using catecholamines, blood pressure was stabilized in the ICU, the patient was weaned off catecholamines and could be discharged from hospital three days later. Of note, this complication occurred in late 2019 after nearly 10 years of refill experience.
Clearly, the retrospective nature of the evaluation is a limitation of our study. However, we are convinced that our real-life experience supports growing knowledge on the management of this additional therapeutic option.
In summary, implantation of an infusion pump for intravenous treprostinil was safe and feasible in our cohort. Given a strict interdisciplinary approach, surgery can safely be performed in this group of seriously ill patients even at advanced age. Unplanned surgical interventions associated with the pump system were extremely rare (0.158 per 1000 treatment days) and could safely be performed by dedicated staff. The risk of catheter-related infections seems to be neglectable. Careful control and documentation of flow rate at every refill is of utmost importance as flow rate increase within the limit of technical deviation was predicting future development of flow rate. Refill complications are rare but possibly serious; management of this treatment should therefore be done at expert centers only.
Implanted pumps could help to overcome the drawbacks of external pumps for parenteral prostacyclin in PAH. Investment in the development of improved devices and prospective clinical trials are warranted.
Footnotes
Ethical approval
At our center, data on pulmonary hypertension patients are collected in the Elisabethinen Linz Pulmonary Hypertension Registry (ELPHREG). ELPHREG is approved by the concerned ethics committee, and written informed consent was obtained from all patients.
Author contributions
Regina Steringer-Mascherbauer, Elisabeth Sigmund, Charlotte Huber, and Josef Aichinger were responsible for treatment of pulmonary hypertension, optimizing specific treatment preoperatively, and long-term care including pump refills. Maria Lummersdorfer and Dagmar Engleder provided anesthesia. Reinhold Függer and Uwe Fröschl were the dedicated surgeons. Regina Steringer-Mascherbauer performed the data analysis and prepared the manuscript draft. All authors contributed equally to the final manuscript.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Regina Steringer-Mascherbauer has received research funding, fees for lectures, and travel/accommodation/meeting expenses from Actelion, AOP Orphan, GSK, MSD, and OrphaCare outside the submitted work. Maria Lummersdorfer and Reinhold Függer report fees for lectures and travel/accommodation/meeting expenses from AOP Orphan. Elisabeth Sigmund and Charlotte Huber have received travel/accommodation/meeting expenses from Actelion, AOP Orphan, and MSD. The other authors report no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Elisabethinen Linz Pulmonary Hypertension Registry (ELPHREG) is supported by AOP Orphan Pharmaceuticals AG and GSK.
