Abstract
There are limited data investigating the epidemiology and resource utilization associated with parenteral prostacyclin use in children. We sought to examine national trends in treatment practices and resource utilization during prostacyclin initiation for pulmonary arterial hypertension (PAH) at children’s hospitals in the United States. Patients with PAH initiated on parenteral epoprostenol and treprostinil (2004–2014) were identified using a nationwide administrative database. Demographics, clinical characteristics, and resource utilization were compared between epoprostenol and treprostinil groups. Costs were indexed in 2014 US dollars. Among 1448 children admitted with a primary or secondary diagnosis of PAH, 280 (19%) were initiated on parenteral prostacyclins (epoprostenol n = 195 and treprostinil n = 85). Epoprostenol predominated early (97% of initiations in 2005); however, treprostinil predominated recently (52–67% of initiations/year). Children initiated on treprostinil had shorter ICU stays (1 [IQR = 0–4] vs. 4 [0–10] days, P < 0.001), shorter total lengths of stay (4 [2–9] vs. 8 [4–18] days, P = 0.001), and lower in-hospital mortality (1 vs. 12%, P = 0.001) with no difference in 30-day (13 vs. 19%, P = 0.19) or one-year readmission rates (56 vs. 61%, P = 0.41). Inpatient costs were lower for treprostinil initiation ($23,779 [11,830–39,535] vs. $32,976 [11,904–94,082], P = 0.03), with a greater difference in the recent era (2009–2013). Though significant variation exists regarding prostacyclin use for PAH across US centers, prostacyclins are common among children with PAH. Treprostinil initiation has been increasing and is associated with less resource utilization and lower cost compared to epoprostenol initiation. Post-discharge outcome data are needed to fully inform decision-making about the relative benefits of parental prostacyclin drug choice.
Pulmonary hypertension (PH) is a complex and devastating disease. Pulmonary arterial hypertension (PAH) is an important class of PH comprising largely patients with idiopathic PAH and familial PAH (historically classified as “primary pulmonary hypertension”).1–4 Estimates of five-year mortality for PAH are in the range of 20–30% in the pediatric population. 1
The most recent pediatric guidelines recommend continuous parenteral prostacyclin therapy with either epoprostenol or treprostinil for patients with PAH at high risk of disease progression and those that have failed oral combination therapy.5,6 Prostacyclins have been shown to improve hemodynamics, functional class, exercise capacity, and survival, but there are few data comparing the two drugs or evaluating the economic impact of these therapies.7,8
Recently, there has been increasing emphasis on enhancing healthcare value by reducing costs and improving outcomes. 9 While much PAH research has focused on outcomes, there are scant data relating to the cost associated with PAH care which is resource-intensive. A number of small cost comparison studies have estimated the relative costs of epoprostenol and treprostinil in mostly adult populations with mixed findings.10–13 None of these studies have evaluated prostacyclin use among patients with PAH or resource utilization and costs associated with prostacyclin therapy in the pediatric population.
We sought to examine national trends in the use of prostacyclin therapy and describe resource utilization associated with initiation of either epoprostenol or treprostinil in children with PAH. We hypothesized that prostacyclin therapy is frequently used in the pediatric population and that treprostinil is associated with less total resource utilization and cost than epoprostenol during initiation of therapy despite being a more expensive drug.
Methods
Data source
Data for this study were obtained from the Pediatric Health Information System (PHIS), an administrative database that contains inpatient, emergency department, ambulatory surgery, and observation encounter-level data from over 45 not-for-profit, tertiary care pediatric hospitals in the United States. These hospitals are affiliated with the Children’s Hospital Association (Overland Park, KS, USA). Data quality and reliability are assured through a joint effort between the Children’s Hospital Association and participating hospitals. Portions of the data submission and data quality processes for the PHIS database are managed by Truven Health Analytics (Ann Arbor, MI, USA). Data are de-identified at the time of data submission and data are subjected to a number of reliability and validity checks before being included in the database. This study was approved by the Institutional Review Board of the University of Pittsburgh.
Study population
All inpatient and observation encounters at PHIS member hospitals for patients aged 2–22 years at the time of admission between 1 January 2004 and 31 March 2014 were included. Patient encounters in PHIS are classified using the ubiquitous International Classification of Disease, Ninth Revision (ICD-9) coding scheme. We included patient encounters with an ICD-9 diagnosis code of “primary pulmonary hypertension” (416.0) but did not include “chronic pulmonary embolism” (416.2), “other chronic pulmonary heart diseases” (416.8), or “chronic pulmonary heart disease unspecified” (416.9), in an effort to include only those patients with idiopathic or familial PAH. The lower age limit of two years was chosen to exclude cases of acute PH related to prematurity which we also considered outside the focus of our study.
Demographics, clinical characteristics, in-hospital outcomes, readmission rates, charges, and costs were queried. Included in this were PHIS-defined elements of surgical and medical complications as well as clinical, pharmacy, imaging, supply, and “other” charge subgroups. We defined prostacyclin initiation as the first encounter with either an epoprostenol or treprostinil charge. This method excluded the potential to “double-count” patients who were initiated on epoprostenol and transitioned during a later admission to treprostinil (or vice versa). Also, to account for potential lack of knowledge about prostacyclin use before 2004 (i.e. left-censored prostacyclin use), we excluded patients whose first prostacyclin use occurred in 2004 from our initiation analysis. To examine the frequency at which prostacyclins were used relative to the volume of patients with PAH (i.e. rate of prostacyclin initiation), we divided the number of prostacyclin initiations by the number of unique patients with PAH by year or by institution.
Charges and cost
Each PHIS hospital reports charges after adjusting for local cost-of-living difference according to the Centers for Medicare & Medicaid Services. Most hospitals report institutional cost-to-charge ratios which were used to estimate hospital costs. For initiation encounters without a reported cost-to-charge ratio (n = 32, 11%), the cost-to-charge ratio was estimated by using the median cost-to-charge ratio of initiation encounters among reporting hospitals (0.34). We then indexed all dollar amounts to 2014 US dollars using the annual consumer price index published by the US Bureau of Labor Statistics. 14
Statistics
Categorical data are presented as n (%) and continuous data as mean ± standard deviation or median [interquartile range]. Comparisons between groups were made using Wilcoxon rank-sum test for continuous variables and the chi-square or Fisher’s exact test for categorical data. Epoprostenol was available during the entire study period while treprostinil was adopted later and was not widely available at the beginning of our study period. To control for the potential confounding effect of treatment era, we performed additional, era-stratified analyses (early 2005–2008 vs. late 2009–2014) or included treatment era as a covariate in select linear and logistic regression models. Statistical tests were two-sided and P ≤ 0.05 was considered significant. Statistical analyses were performed using JMP Pro 9 (SAS Institute, Inc., Cary, NC, USA).
Results
Study population
Demographics and clinical characteristics during prostacyclin initiation.
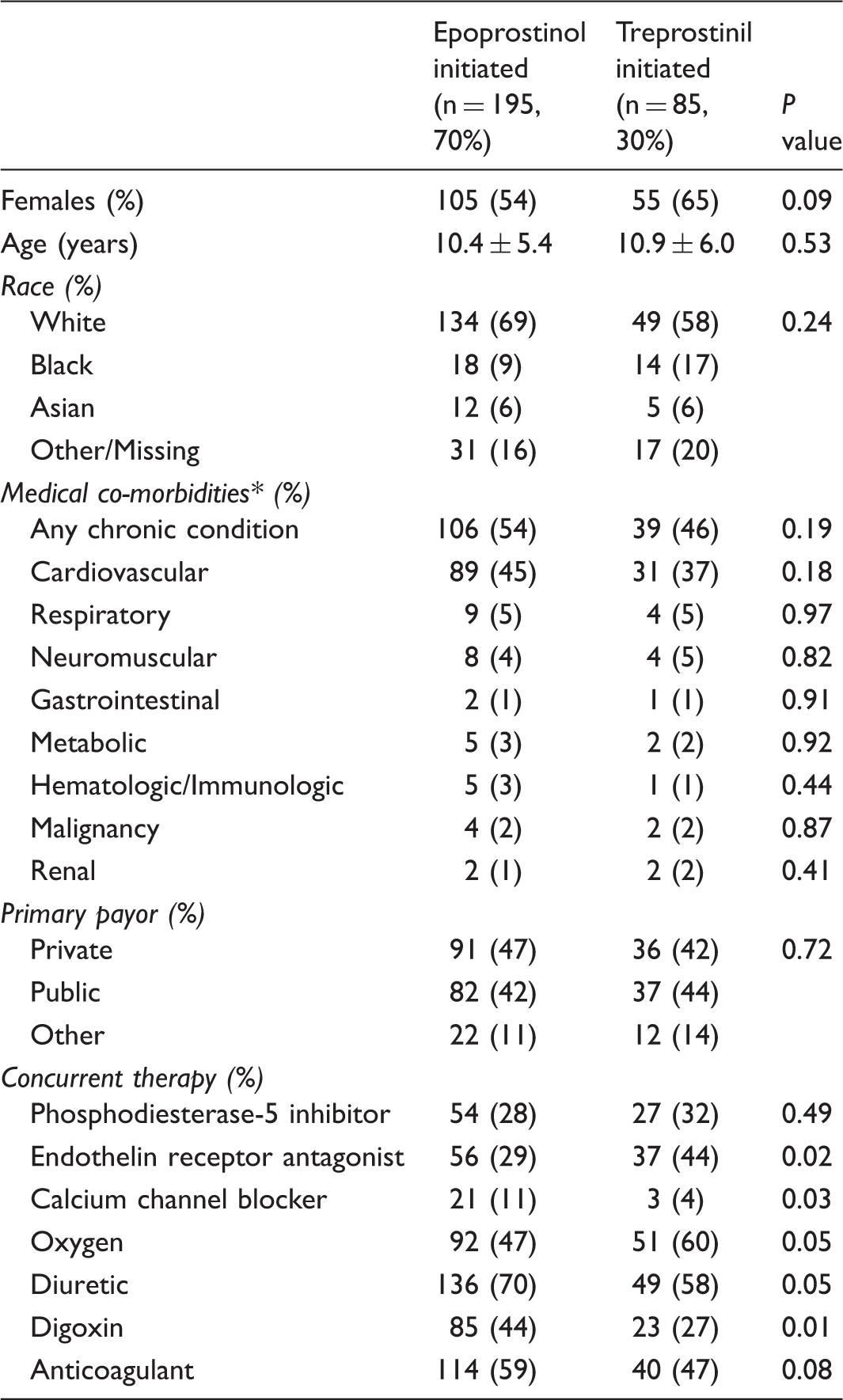
Data presented as n (%) or mean ± SD.
As defined by PHIS.
Prevalence of prostacyclin initiation
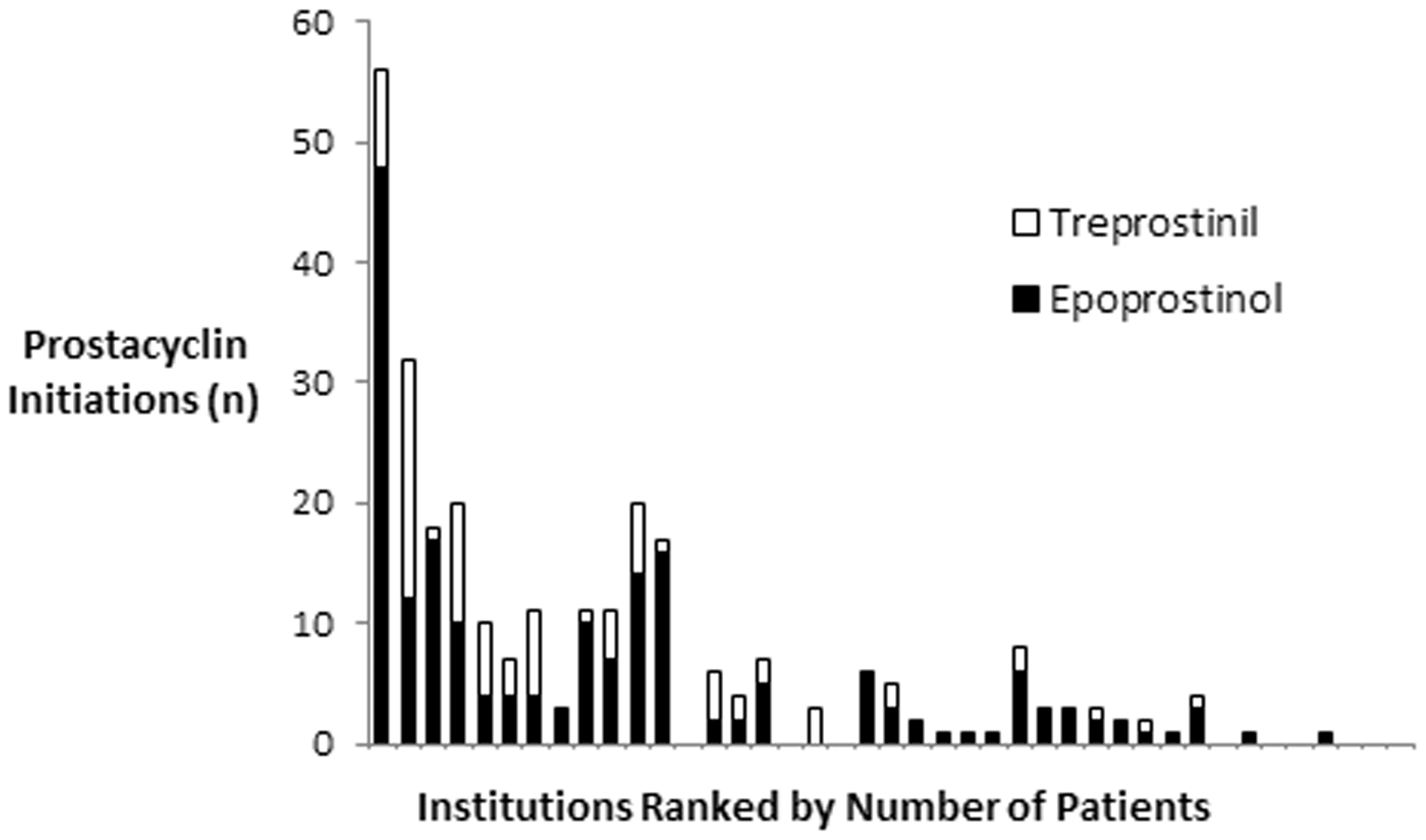
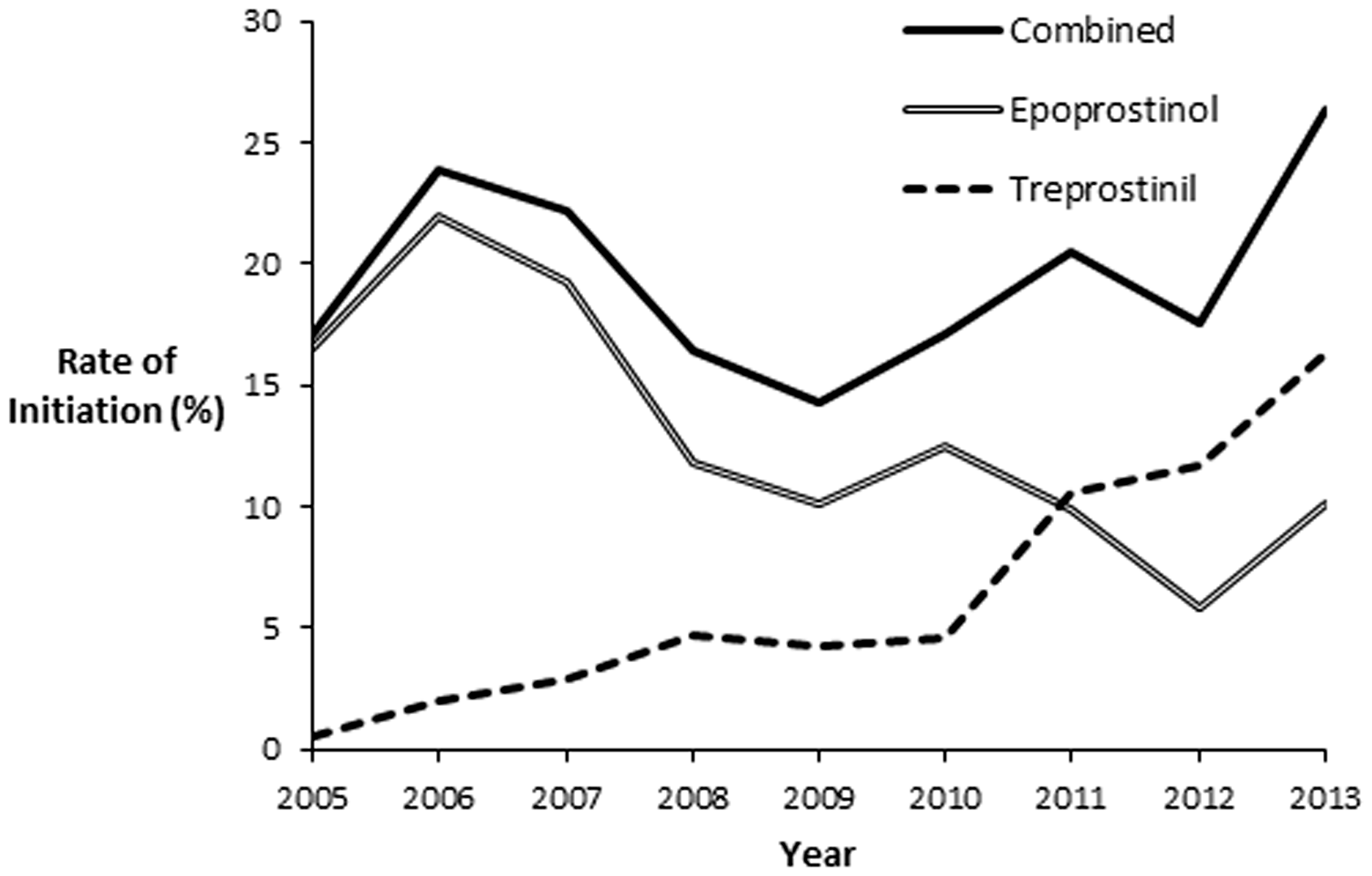
Figure 1 shows the prevalence of prostacyclin initiation as a function of institutional PAH volume as measured by patients hospitalized for PAH. The median proportion of PAH patients initiated on prostacyclins per hospital was 16% with a range of 0–53%. Among centers with >50 patients admitted with a diagnosis of PAH (n = 8), the prevalence of parental prostacyclin initiation was in the range of 6–29%. Figure 2 displays the relative proportions of epoprostenol and treprostinil initiations at each hospital. Eight of the 41 (20%) centers with PAH admissions had no patients initiated on parental prostacyclins. Except for a single institution (#18) with all four prostacyclin initiations as treprostinil, epoprostenol initiation showed no association with institutional PAH volume. As shown in Fig. 3, epoprostenol predominated over treprostinil early, accounting for 97% of total initiations in 2005. However, since 2011, treprostinil initiation has been predominant (52–67% of initiations/year).
Rate of prostacyclin initiation as a function of the volume of PAH patients by hospital. Each data point represents a unique hospital. Median prevalence of prostacyclin initiation was 16% with a range of 0–53%. Relative proportion of epoprostenol vs. treprostinil use during prostacyclin initiation by hospital. Hospitals are ranked from most PAH patients (left) to the least PAH patients (right). Epoprostenol use (vs. treprostinil use) was in the range of 0–100% of prostacyclin initiations per hospital. Annual rate of prostacyclin initiations during study period. Rate of initiation defined as the number of prostacyclin initiations divided by the number of new PAH cases reported at each hospital.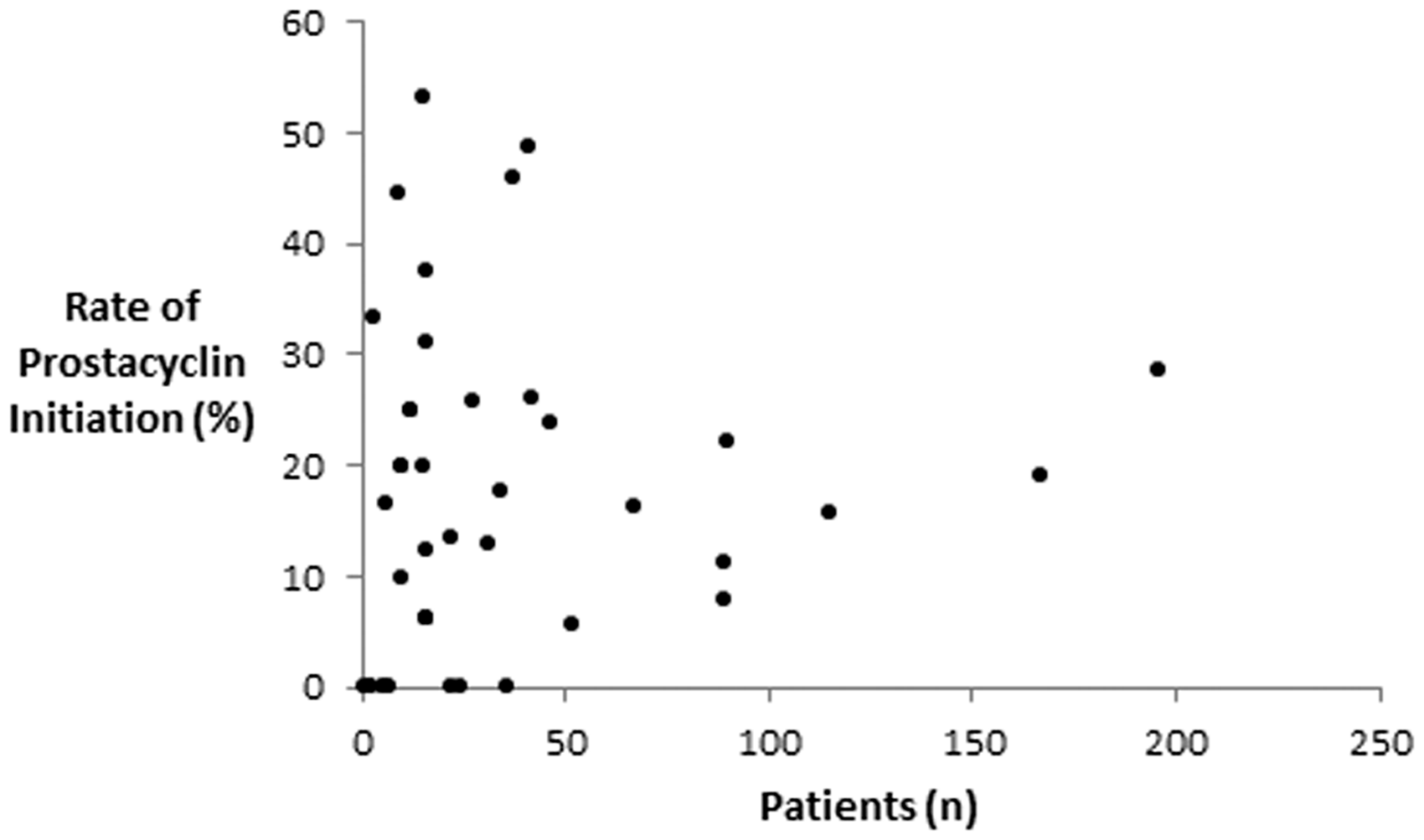
Resource utilization and inpatient outcomes
Resource utilization, in-hospital outcomes, and hospital readmission rates at the time of prostacyclin initiation.
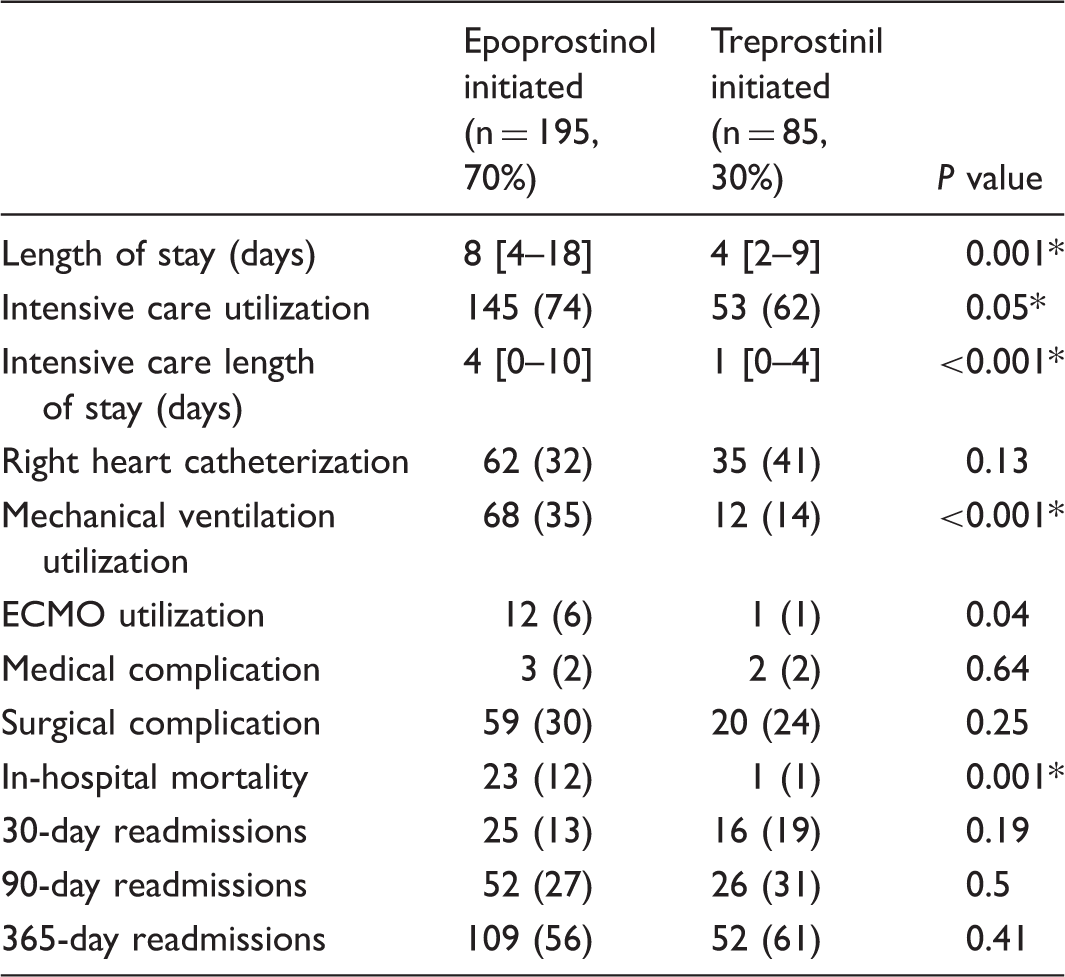
Data presented as number (%) or median [IQR].
Remained significant in multivariate analysis.
ECMO, extracorporeal membrane oxygenation.
Readmission following prostacyclin initiation
All cause readmissions at 30, 90, and 365 days following the prostacyclin initiation encounter were queried. We found no differences in the frequency of readmissions between those initiated on epoprostenol vs. treprostinil (Table 2).
Charges and cost
Hospital charges and costs at the time of prostacyclin initiation.
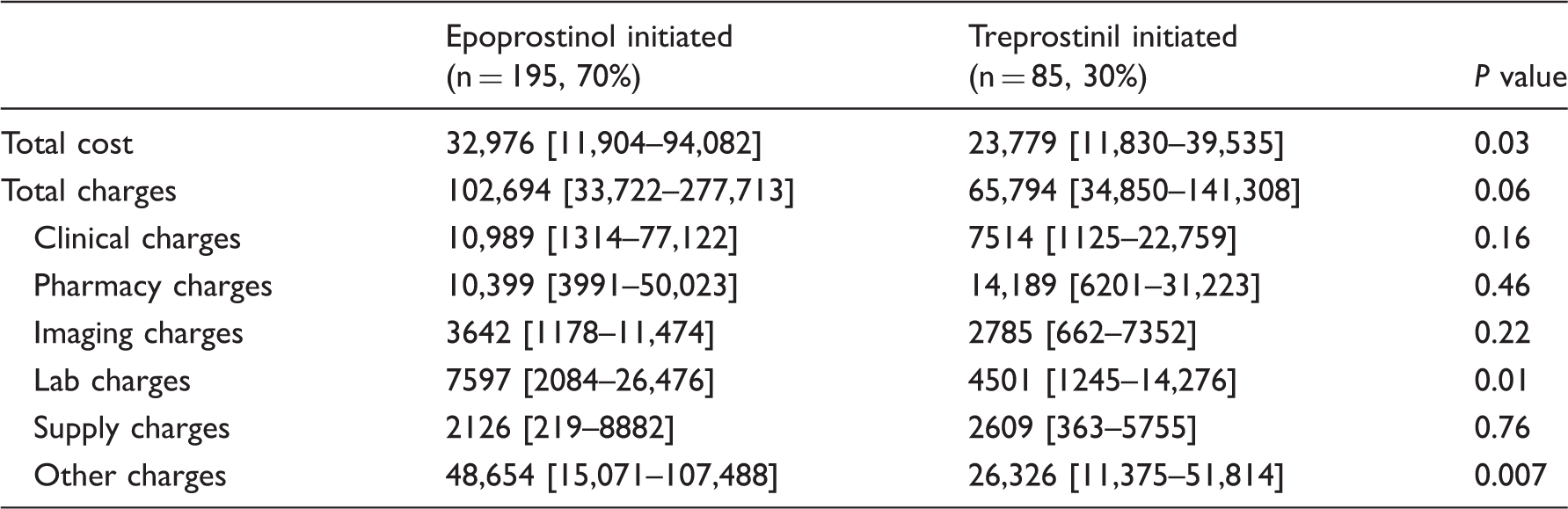
Data presented as median [IQR].
All data in 2014 USD.
Charges and costs at the time of prostacyclin initiation during late era (2009–2013).
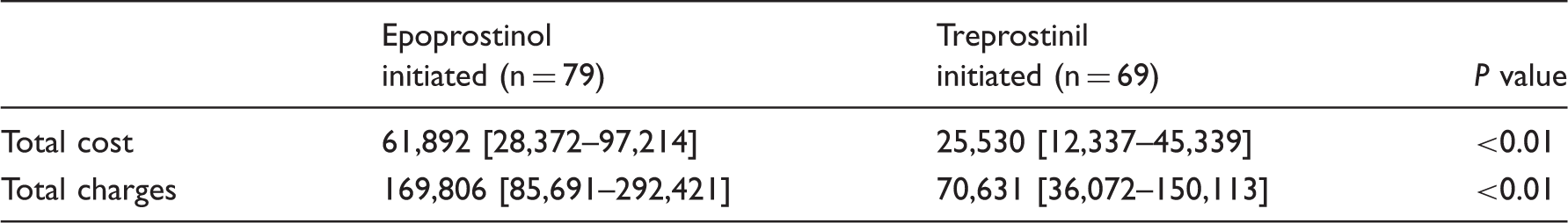
Data presented as median [IQR].
All data in 2014 USD.
Discussion
This study demonstrates that initiation of parenteral prostacyclin therapy is common among children who are admitted with PAH to US children’s hospitals, and there has been an increase in prevalence of children initiated on parental prostacyclins over the past five years. We have also shown that epoprostenol, once the only option for parenteral prostacyclin treatment, has been overtaken by treprostinil as the most commonly initiated parental prostacyclin in the pediatric PAH group in the US. Median hospital costs associated with treprostinil initiation were 39% less than those associated with epoprostenol initiation, with shorter lengths of stay and lower resource utilization. While epoprostenol was widely used throughout the study period, intravenous and subcutaneous treprostinil became more widely utilized during the late years of the study period (albeit at a poorly defined time), potentially contributing to the significant era effect we observed. Nonetheless, the differences we observed in cost and resource utilization remained after accounting for drug era, suggesting that the associations of treprostinil with lower cost and charges are not fully due to differences in practice norms or experience managing pediatric PAH during the earlier years of the analysis when epoprostenol initiation was much more common.
One possible reason for the cost, charge, and resource utilization associations we found could be a quicker initial dose titration with fewer initial side effects. Though we could find no study which directly compared side-effect profiles of the two medications given parenterally, our clinical experience and a review of the reported frequency of some of the common prostacyclin side effects (flushing, jaw pain, diarrhea, headache) suggests this is a plausible assertion.15 Another possible reason could be that less sick patients were initiated on treprostinil. Because the PHIS database is an administrative dataset which does not collect clinical findings, hemodynamics, or echocardiographic parameters, we cannot exclude this possibility. Though endothelin receptor antagonists were more likely to be prescribed in those initiated on treprostinil therapy, while calcium channel blockers, diuretics, and digoxin were less likely to be prescribed, we believe this reflects an era difference and not a difference in disease severity between the groups.
To our knowledge, this is the largest cohort of pediatric PAH reported and the first to describe use of, and resource utilization surrounding, parental prostacyclin therapy on a national scale. Current guidelines recommend continuous parenteral prostacyclin therapy for advanced disease (World Health Organization functional class III–IV) or for patients who have failed to respond to oral therapies.5,6 Recent reports, however, have suggested that treatment with prostacyclins earlier in the disease course may be beneficial.16,17 The rate of intravenous prostacyclin use in the REVEAL registry was 26% in adults and 29% in children, both of which are greater than the 16% we observed in this pediatric cohort.1,18 This may suggest underutilization of parental prostacyclins in children with PAH who are cared for at US pediatric hospitals, some of which are not PAH specialty centers, or alternatively is a result of our conservative method for identifying prostacyclin initiations. Because of the lack of baseline hemodynamic and functional class data in our cohort, caution with respect to drawing firm conclusions on the use of prostacyclins between our cohort and the pediatric REVEAL cohort is important. Of note, the pediatric REVEAL cohort appeared to be enriched for patients with more significant PAH based on reported demographics of 49% with functional class III–IV and group mean pulmonary arterial pressure 72 ± 17 mmHg with pulmonary to systemic vascular resistance ratio of 0.8 ± 0.5. 1
We found a wide degree of institutional variation in prostacyclin initiation, particularly among centers with lower volumes of PAH patient encounters. The centers following the largest number of patients with PAH (>50) had less variation in their parenteral prostacyclin use with rates in the range of 15–30%. Current recommendations suggest prostacyclin therapy be administered at experienced centers given the complexities of prostacyclin administration.19,20 High volume centers have been demonstrated to have superior outcomes for congenital heart surgery21,22 and in pediatric heart transplantation.23,24 Whether this also applies to pediatric PAH care is unclear and not answerable by our analysis. Because we were unable to assess longitudinal outcomes such as progression to lung transplantation or death, we are unable to comment on whether the variability we observed in parental prostacyclin initiation among centers is clinically meaningful.
There are a number of limitations to this study that must be considered. The PHIS database incorporates robust validation measures, but medical coding errors may exist. PAH does not have an associated ICD-9 code but we believe our surrogate ICD-9 code (“primary pulmonary hypertension”) has a high sensitivity and specificity to identify our intended study population. Nonetheless, we cannot exclude the possibility that prostacyclin use in the immediate postoperative period after congenital cardiac surgery may have been captured in some proportion of the cases analyzed. Also, in-hospital outcome measures may also be prone to case ascertainment error as has recently been described in the congenital heart disease population. 25 Furthermore, the PHIS database is an administrative database which lacks granular clinic data preventing a robust comparison between baseline illness severity between the treatment groups. As a result, we were unable to compare the groups with respect to severity of PAH (i.e. functional class, pre-therapy hemodynamics) and we cannot exclude that the resource utilization, LOS, and total hospital costs may be related to the severity of patients’ clinical condition and not to the type of prostacyclin. A portion of patients in our cohort may have received inhaled prostacyclin therapy that may not have been captured and we were not reliably able to determine subcutaneous from intravenous delivery of treprostinil which may have had impacts on outcomes and resource utilization. The PHIS database lacks the ability to capture out-of-hospital clinical outcomes hindering fully informed decision-making on drug choice.
Prostacyclin use is common in children with PAH and treprostinil has become the most frequently initiated prostacyclin. There is significant variation across institutions regarding use of prostacyclin, suggesting the need for further recommendations. Our data suggest less resource utilization and lower cost for those patients initiated on treprostinil; however, more robust data from clinical registries are needed to support these findings.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This work was supported by the Patrick Dick Memorial Fund for Pediatric Cardiology Research at the Heart Institute at Children's Hospital of Pittsburgh of UPMC.
