Abstract
The incidence of pulmonary embolism (PE) has been increasing since the introduction of the D-dimer test and CT angiography in 1998. 1 The current indication for inferior vena cava (IVC) filter is limited to patients with active bleeding complications and contraindication for anticoagulation.2–6 However, the venous thromboembolism (VTE) guidelines from the American Heart Association and the Society of Interventional Radiology recommended the IVC filter to be considered in hemodynamically unstable patients and those with limited cardiopulmonary reserve as an adjunct to anticoagulation.4,6
Whether IVC filter placement improves the survival of PE patients is unclear. A randomized, clinical trial of 129 hip fracture patients comparing IVC filter placement with conservative management showed that IVC filter placement was associated with reduced mortality (10% vs. 24%), although none of the groups received VTE prophylaxis. 7 Prevention du Risque d’Embolie Pulmonaire par Interruption Cave (PREPIC 1) trial of 400 patients with proximal deep vein thrombosis (DVT) compared IVC filter placement in addition to anticoagulation vs. anticoagulation alone. In this study, the IVC filter reduced the incidence of PE but did not affect mortality. 8
Systemic arterial hypotension, tachypnea, troponin leak, and right ventricular hypokinesis are significant prognostic factors in mortality associated with PE and represent the population with complicated PE. 9 In these patients with complicated PE, the IVC filter appears to have a mortality benefit. Wadhwa et al. 10 used the National Inpatient Sample (NIS) database to demonstrate that IVC filter placement improves survival in PE patients with congestive heart failure (adjusted hazard ratio (0.53, 95% CI 0.51–0.55, p < 0.001). In another study, Stein and Matta 11 utilized the same NIS database (1999–2008) to analyze 21,905 unstable patients (combined shock and mechanical ventilation) and demonstrated decreased mortality in all age groups. Another study using the Premier database (2010–2014) analyzed data for 4284 unstable patients and reported similar results. 12 However, neither the Stein nor the Premier study addressed anticoagulation status or performed risk-adjustment for confounders. Whether IVC filter placement improves mortality in PE complicated by acute respiratory failure, acute myocardial infarction, or in patients requiring thrombolytics has not been explored.
In this study, we sought to address whether IVC filter placement is associated with a reduction in mortality in complicated PE patients which included four subgroups; (A) PE complicated by acute myocardial infarction—representing troponin leak, (B) PE complicated by acute respiratory failure—representing limited lung reserve, (C) PE complicated by shock—representing limited cardiac reserve, and (D) PE treated with thrombolytics—representing massive and submassive PE population.
Methods
Data source
Discharge data were obtained from the National Inpatient Sample (NIS 2002–2014). NIS is the largest all payer-inpatient care database in the United States. NIS is a stratified systemic random sample, which approximates to 20% of all the discharges. The data are de-identified and publicly available; hence, the ethical board review approval was not required.
Study population and outcomes
ICD-9-CM coding was used to retrieve the study population, define all complications, and define Elixhauser comorbidities.
13
All ICD-9-CM codes used in the study are listed in Supplemental Table 1. From 2002 to 2014, a total of 254,465 patients were identified as complicated PE study population which included acute PE patients complicated with acute myocardial infarction, acute respiratory failure, shock, and patients requiring thrombolytics, after excluding missing dead or alive cases (N = 1404). We identified up to 15 diagnoses for all the conditions including acute PE in our study, since there is a concern for low capture of event rates when the diagnostic code is limited to the primary diagnosis,
14
and the complications such as acute respiratory failure and shock are more likely to be listed as primary diagnosis for coding purposes.
15
The acute PE coding by definition excludes cases with a personal history of PE or chronic PE. In patients with a personal history of PE and chronic PE, the possibility of the complication not being related to the PE is higher; thus, we chose to exclude these diagnosis codes in our analysis. We also excluded the cases of septic, iatrogenic (can include septic PE), and saddle PE. Central emboli have an increased risk of mortality compared to distal emboli.
16
Since the mortality outcomes of central PE cannot be generalized to distal PE, we chose to exclude saddle PE in our study. The complicated PE patients were further subgrouped into four target subpopulations (Fig. 1); (A) PE complicated with acute myocardial infarction (N = 81,683), (B) PE complicated with acute respiratory failure (N = 64,310), (C) PE complicated with shock (N = 82,975), and (D) PE patients requiring thrombolytics (N = 25,495). The IVC filter was placed during the same admission in which acute complicated PE was identified. Separate subgroups included (B1) acute respiratory failure with mechanical ventilation, (B2) acute respiratory failure with anticoagulation contraindications, and (C1) shock with anticoagulation contraindications.
Study population schematic. The flowchart describes the scheme of sub-classification of acute pulmonary embolism (PE) for propensity score (PS) match analysis with their weighted data counts. IVC filter†: inferior vena cava filter. Propensity score-matched outcomes in pulmonary embolism complicated with acute myocardial infarction and acute respiratory failure. OR: odds ratio; SEM: standard error of mean.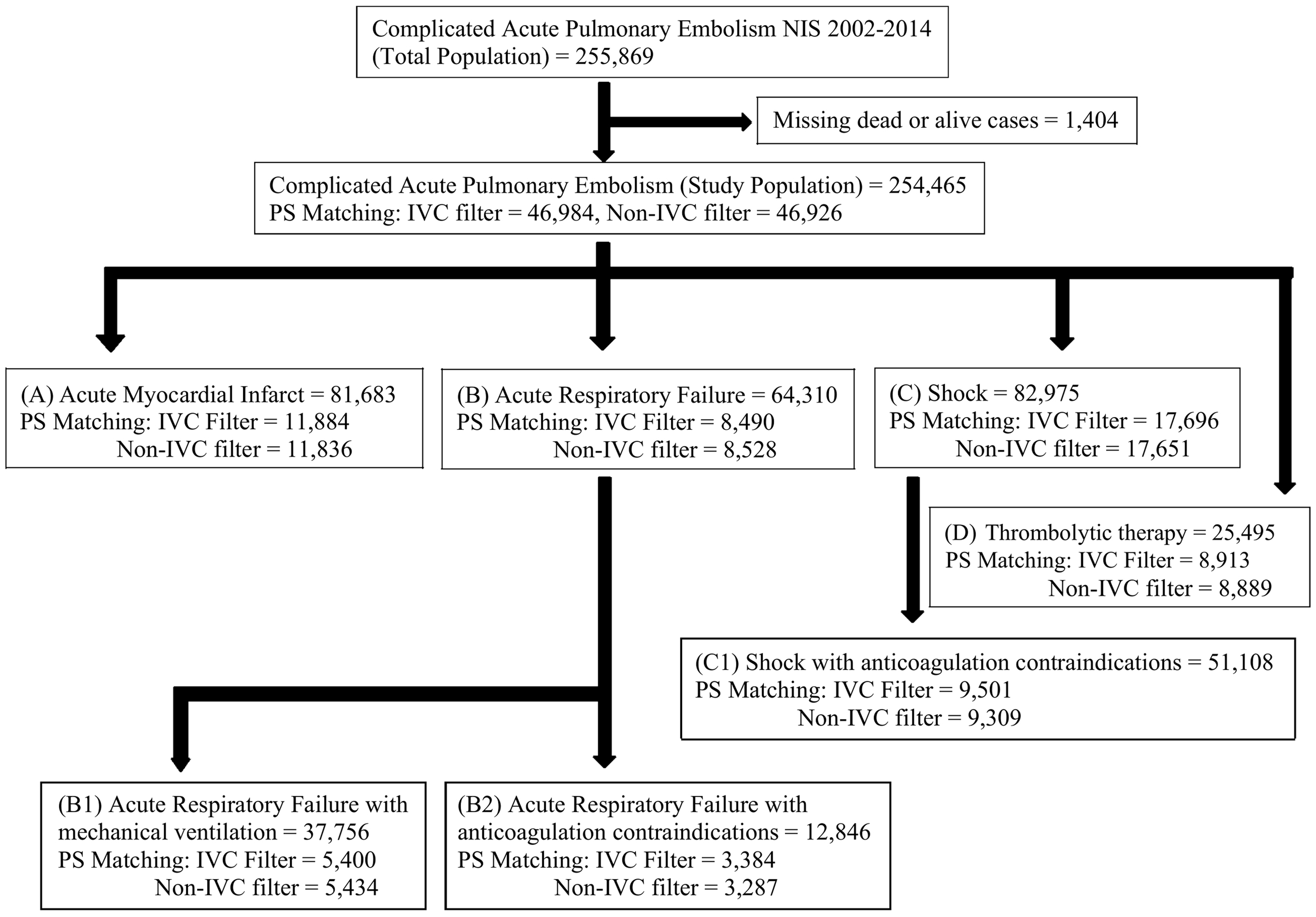
Propensity score-matched outcomes in pulmonary embolism complicated with shock and thrombolytic treatment.
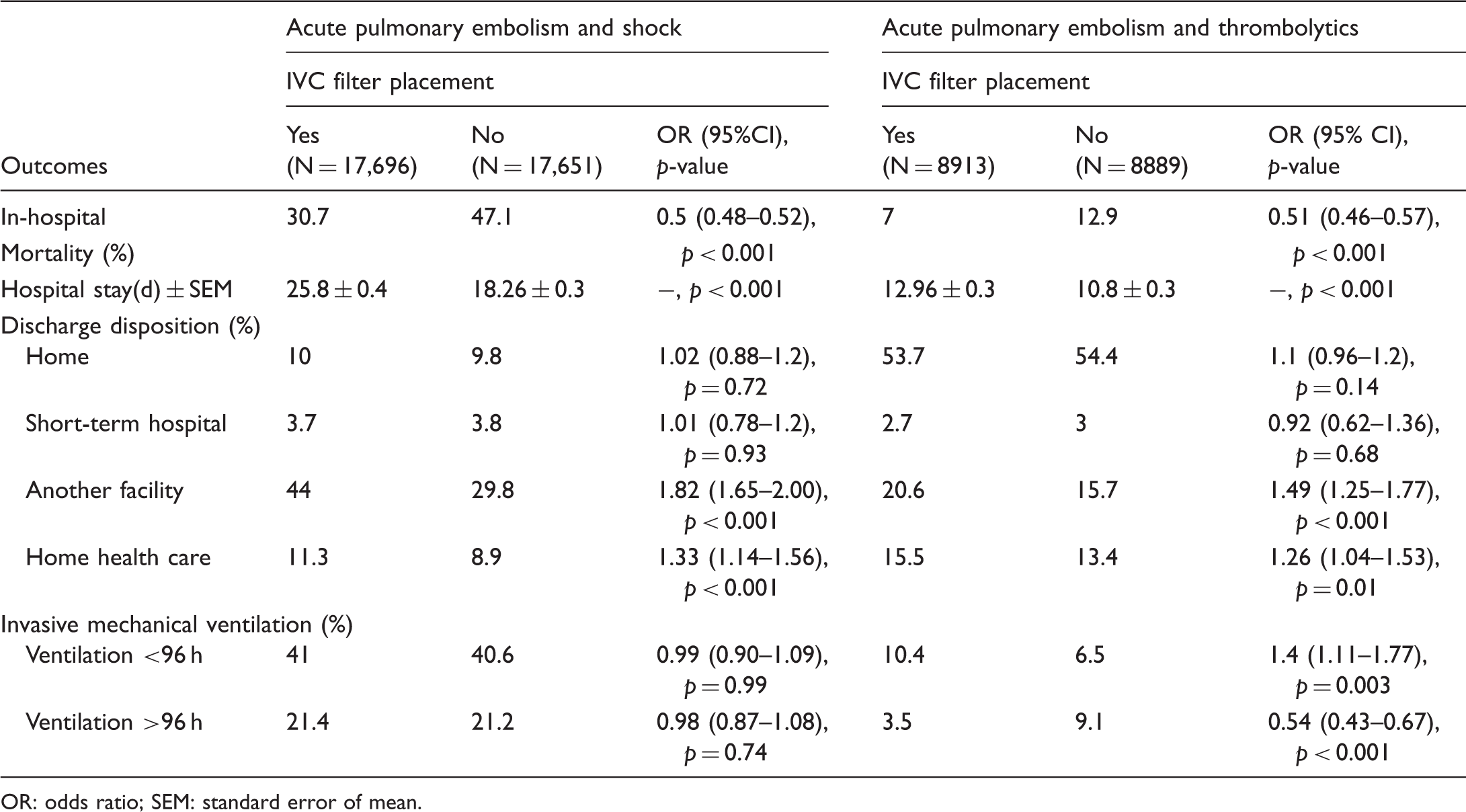
OR: odds ratio; SEM: standard error of mean.
Statistical analysis
Survey specific analysis tools were used to account for the stratification, clustering, and data sampling to report weighted data estimates.
20
Propensity score matching (PSM) was used as the preferred method of analysis for our observational data because the degree of overlap of baseline covariates between the two arms can be explicitly examined, and it allows separation of the design from the analysis of the study, in contrast to regression-based analysis.
21
PSM using a greedy-nearest neighbor 1:1 matching without replacement and caliper 0.2 was used to generate IVC filter and control groups for complicated PE and each of the seven subgroups of interest (Fig. 1). The propensity score-matched IVC filter and the control groups were created so that mortality rates can be easily compared. The propensity of IVC filter scores was estimated via unweighted logistic regression model,
22
with age, sex, race, lower extremity DVT, PE comorbidities, and Elixhauser comorbidities as covariates. Propensity score matching was performed in R PS MatchIt extension package version 3.0.4 in SPSS
23
(Supplemental Data 3). The 45 co-variates in complicated PE were compared via standardized mean differences generated by the software (Fig. 2) to check for balance between the IVC and control group (Supplemental Data 4).
24
Standardized mean differences of covariates in full complicated PE cohort (age, gender, race, Elixhauser, and PE comorbidities) before and after matching. The Elixhauser co-morbidities are well-aligned—within 10% standardized difference—even before matching. The PE comorbidities distribution shows a slight variation on the standardized difference scale before matching. Post matching, all the comorbidities are perfectly balanced in the IVC filter and non-IVC filter group.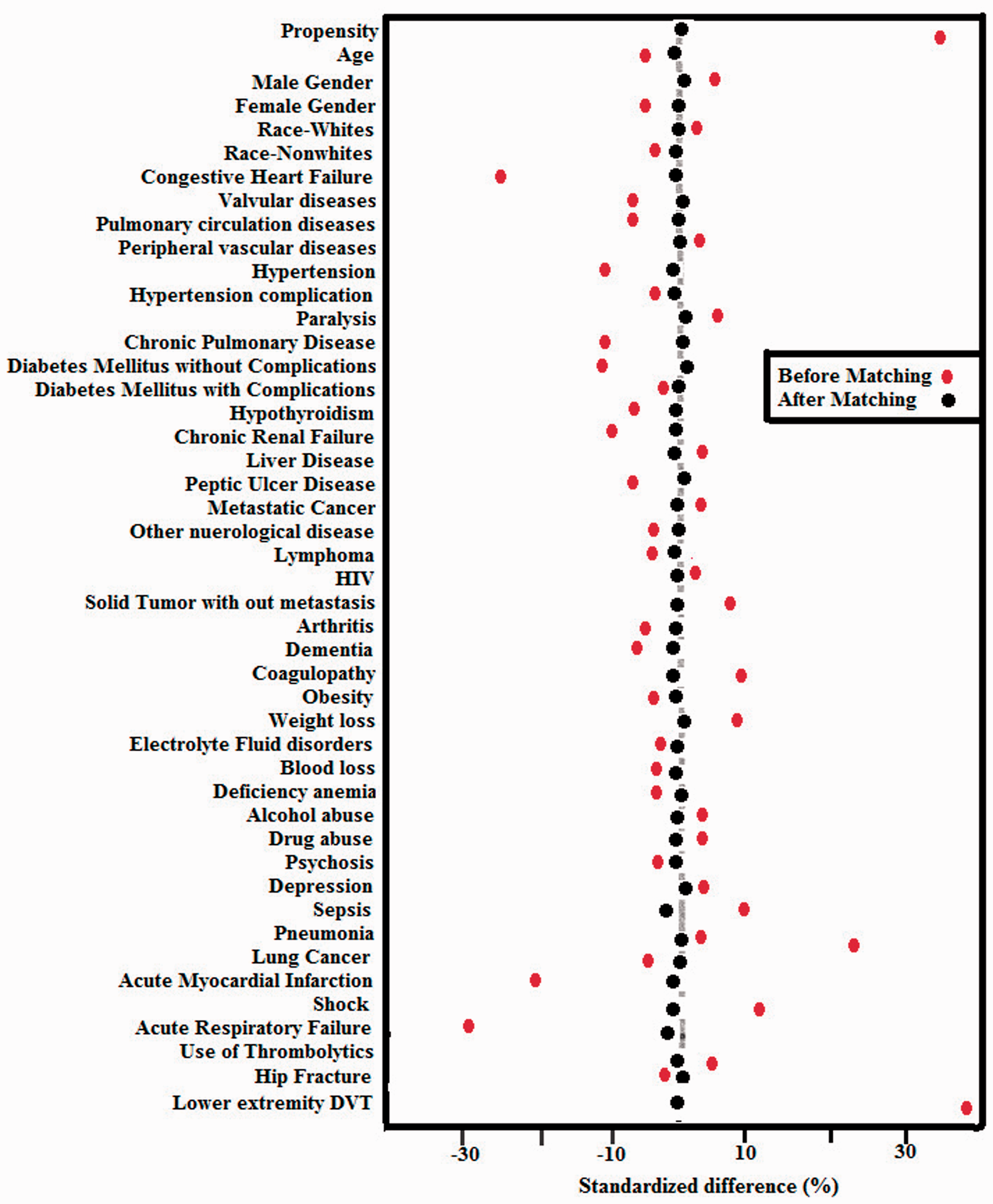
After generating the IVC and control groups using PSM, the effect of IVC filter on mortality was estimated using odds ratio (OR), and the number needed to treat (NNT) on the weighted data. Confidence intervals for the NNT were calculated using the Altman method. 25 We reported E-values for sensitivity analysis in our observational study to assess the minimum strength of association any unmeasured confounding factor would need to have with the IVC filter placement and the mortality outcome to explain away the observed treatment-outcome association. 26 For the residual confounder to explain the observed association, the unmeasured confounder should have a risk ratio associated with the treatment-outcome greater than the E value.
Immortality bias arises when patients who received filter were alive at the time of intervention and patients who did not receive the filter may have died before a filter could be placed. To address immortality time bias, we performed a secondary analysis; patients with filters, being alive at the time of filter placement on the day of admission were matched with the non-filter group, being alive at a similar time after diagnosis. If the IVC filter placement was associated with significant mortality difference after matching the two groups when they were alive on the day of admission, immortality bias was deemed unable to explain the results. 27 Statistical analysis of the data was performed using SPSS Statistics for Windows, Version 23.0. (Armonk, NY).
Results
Filter in complicated PE
Of 254,465 complicated PE patients, 49.8% were males. The mean (SEM) age was 66.4 (0.071) years, and the mean (SEM) length of stay was 14.1 (0.07) days. The details of the baseline characteristics of the complicated PE population are shown in Supplemental Table 2.
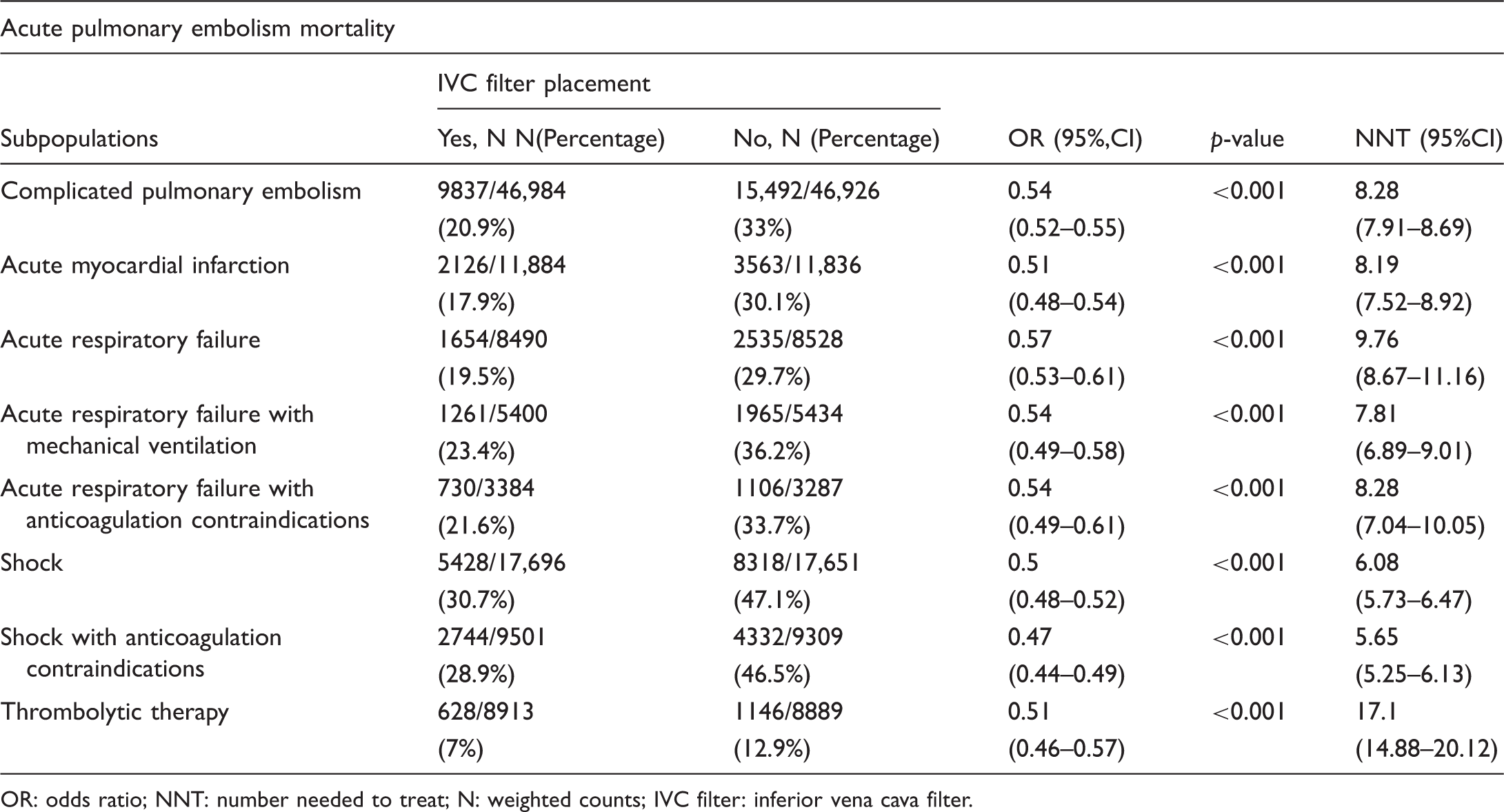
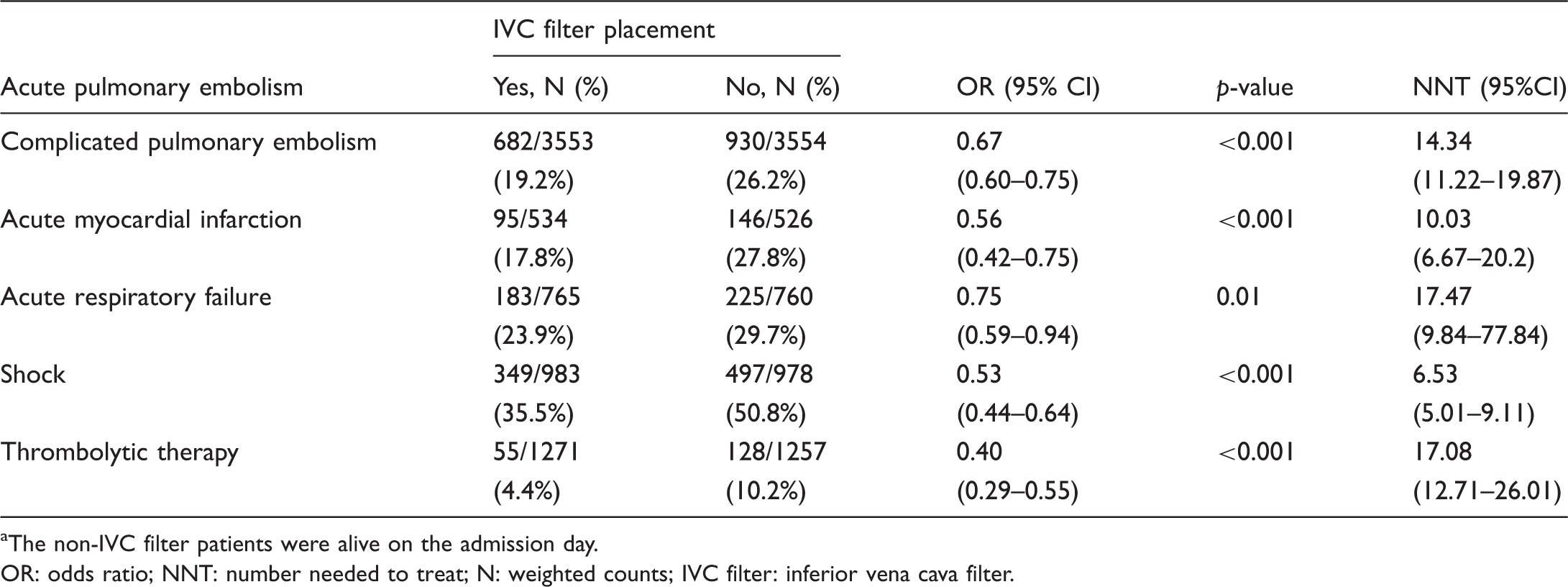
The standardized mean differences of all the covariates were less than 10% of absolute values after propensity score matching (Fig. 2). The 45 co-variates were well-balanced between the filter and non-filter groups. The IVC filter placement in complicated PE was associated with lower mortality rates vs. the propensity-matched controls (20.9% vs. 33%; NNT = 8.28, 95% confidence interval (CI) 7.91–8.69, E-value = 2.53), (Table 3, Fig. 3). The IVC filter, when placed on the day of admission, was associated with decreased mortality (19.2% vs. 26.2%, p < 0.001; NNT = 14.34, 95% CI 11.22–19.87) (Table 4).
Comparison of propensity score matched in-hospital mortality with and without IVC filter placement in complicated PE and subgroups. Inferior vena cava (IVC) filter placement was associated with decreased mortality in all the studied subgroups of pulmonary embolism (PE) in the propensity score (PS) matching. †ARF: acute respiratory failure; ‡ NNT: number needed to treat. Propensity score-matched in-hospital mortality in complicated pulmonary embolism and subgroups. OR: odds ratio; NNT: number needed to treat; N: weighted counts; IVC filter: inferior vena cava filter. Propensity score-matched in-hospital mortality in complicated pulmonary embolism and subgroups with filter placement on admission day.
a
The non-IVC filter patients were alive on the admission day. OR: odds ratio; NNT: number needed to treat; N: weighted counts; IVC filter: inferior vena cava filter.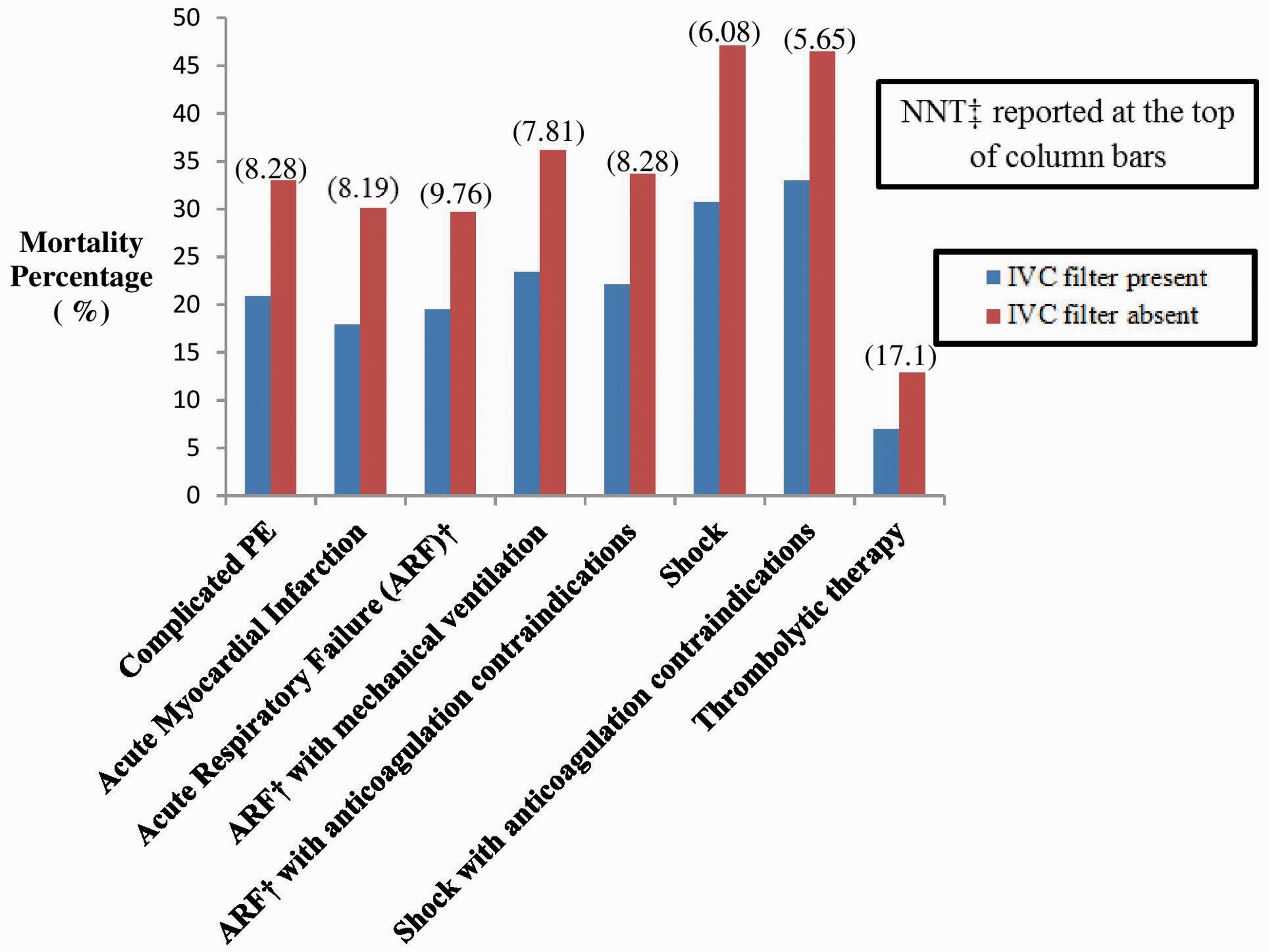
Filter in PE complicated by acute myocardial infarction
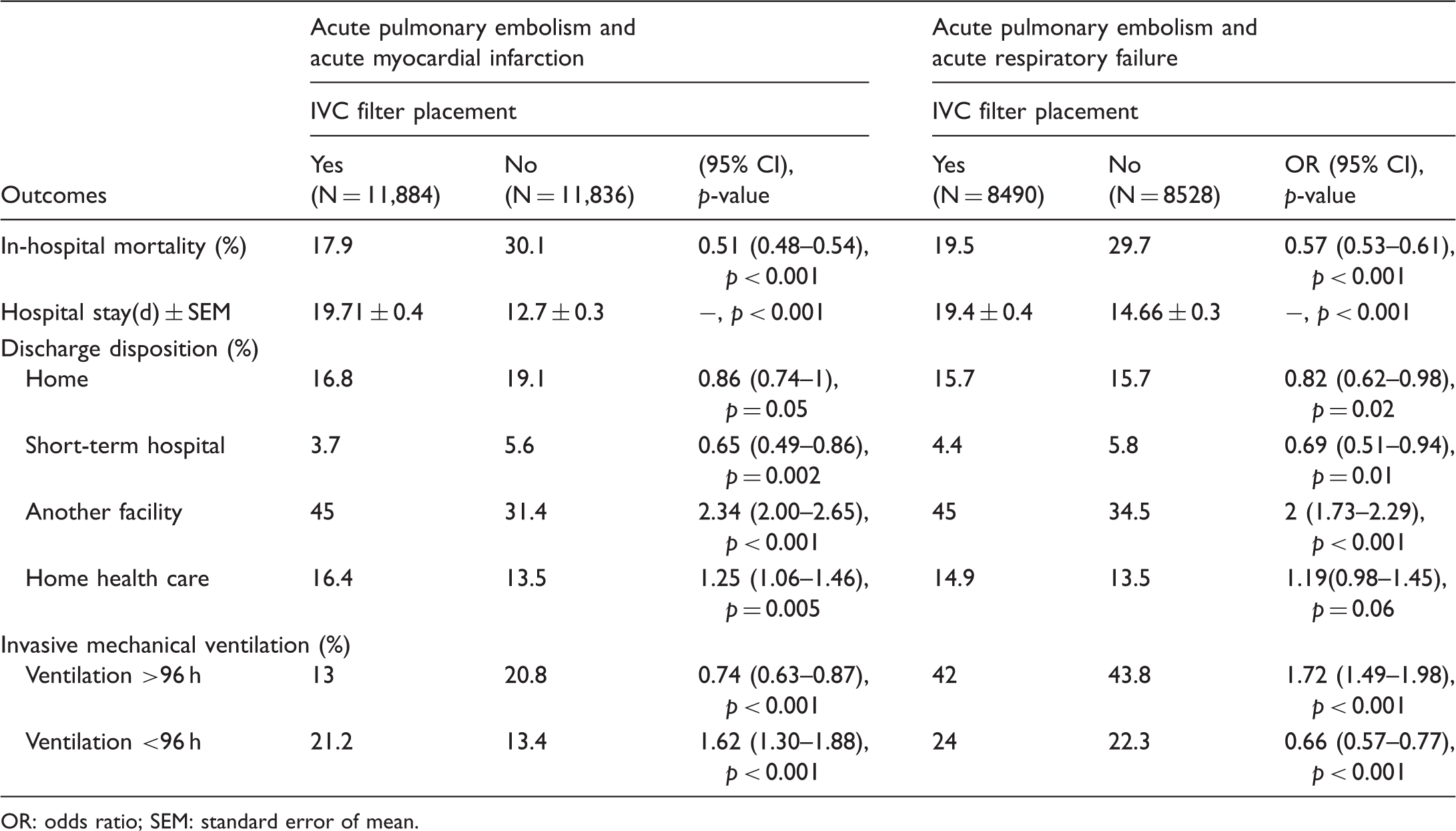
Of 81,683 patients with PE complicated by acute myocardial infarction, 14.5% underwent IVC filters placement. IVC filter placement was associated with mortality benefit (17.9% vs. 30.1%; NNT = 8.19, 95% CI 7.52–8.92, E-value = 2.76) (Table 2) even on the day of admission (17.8% vs. 27.8%, p = 0.008; NNT = 10.03, 95% CI 6.67–20.2) (p < 0.001 for both) (Table 4). Increased length of stay was observed with IVC filter placement (19.71 vs. 12.79 days), p < 0.001.
Filter in PE complicated by acute respiratory failure
Out of 64,310 PE with acute respiratory failure patients, 13.2% of the population had an IVC filter placed. IVC filter placement was associated with decreased acute respiratory failure mortality (19.5% vs. 29.7%; NNT = 9.76, 95% CI 8.67–11.16, E-value = 2.38).
In the subgroup analysis of patients with acute respiratory failure and anticoagulation contraindications, IVC filter placement was associated with decreased mortality (21.6% vs. 33.7%; NNT = 8.28, 95% CI 7.04–10.05). The mortality benefit was observed in patients who required mechanical ventilation (23.4% vs. 36.2%; NNT = 7.81, 95% CI 6.89-9.01) (p < 0.001 for all the subgroups). The IVC filter was associated with decreased mortality in patients with acute respiratory failure on the day of admission (23.9% vs. 29.7%, p = 0.01; NNT = 17.47, 95% CI 9.84-77.84). The acute respiratory failure subgroup was linked to an increased mean length of stay (19.4 vs. 14.6 days, p < 0.001).
Filter in PE complicated by shock
IVC filter was placed in 6.1% of the population with PE complicated with shock. The IVC filter group with shock was associated with decreased mortality (30.7% vs. 47.1%; NNT = 6.08, 95% CI 5.73–6.47, E-value = 2.43).
In the subgroup analysis, IVC filter was associated with a mortality benefit in PE complicated shock and anticoagulation contraindications (33% vs. 46.5%; NNT = 5.65, 95% CI 5.25–6.13). We observed similar association in PE patients complicated with shock when IVC filter was placed on the day of admission (35.5% vs. 50.8%, p < 0.001; NNT = 6.53, 95% CI 5.01–9.11).
The IVC filter group had increased mean length of stay (25.8 vs. 18.26 days; p < 0.001) and a higher percentage of these patients were discharged either to another facility (44% vs. 29.8%; OR = 1.84, 95% CI 1.76–1.92) or home with home health care (11.3% vs. 8.9%; OR = 1.30, 95% CI 1.21–1.39) p < 0.001 for both (Table 3).
Filter in PE receiving thrombolytics
An IVC filter was placed in 1.9% of the 24,595 PE patients that received thrombolytics. In the PSM analysis of this population, patients with IVC filters were observed to have lower in-hospital mortality (7% vs. 12.9 %; NNT 17.1, 95% CI 14.88–20.12, E-value = 3.01). IVC filter placement on the day of admission was associated with lower mortality (4.4% vs. 10.2%; NNT = 17.08, 95% CI 12.71–26.01) (p < 0.001 for all). As with the other subgroups, IVC filter patients had a slightly prolonged length of stay (12.96 vs. 10.8 days) and were more likely to be discharged to another facility (20.6% vs. 15.7%; OR = 1.39, 95% CI 1.29–1.50) (p < 0.001 for all) (Table 3).
Discussion
To our knowledge, this is the first risk-adjusted analysis of patients with complicated PE to show that IVC filter placement is associated with significantly decreased mortality. In the secondary analysis, after matching the patients on propensity score in the IVC filter arm that received IVC filter on the day of admission and non-IVC filter arm, after excluding the patients that were deceased on the day of admission, the IVC filter placement was associated with significant mortality benefit in complicated PE and all the subgroups (Table 4). The possibility of the immortality bias was ruled out with the propensity score matching on the alive subjects in both arms on the day of admission. 28 In addition, immortality bias is unlikely on the day of admission since mere hours have elapsed between the time of admission and the IVC filter placement, during which immortality bias can occur. Therefore, we concluded that the immortality bias could not explain the association of the IVC filter placement with decreased mortality in complicated PE and its subgroups.
E values were reported as an alternative to sensitivity analysis in our observational study. E value accounts for the maximum bias an unmeasured confounder can induce. It represents the minimum strength of association on the risk ratio scale that an unmeasured confounder would need to have with the IVC filter and mortality to be able to explain away the IVC filter-mortality association. It is a quantifiable measure that is related to the evidence of causality. In our observational study of mortality data, E values were consistently higher than 2 in complicated PE and its subgroups. These results make it unlikely for any unmeasured confounder to be able to explain away the IVC filter-decreased mortality association. 29
There was no substantial difference in the distribution of the covariates of 30 Elixhauser comorbidities (<10% standardized difference) even before propensity score matching. The PE comorbidities determined by us using logistic regression exhibited a slight covariate difference before PSM (Fig. 2). The use of primarily balanced data even before PSM in most of the co-variates and the large E values in complicated PE and its subgroups makes it unlikely for an unmeasured confounder or covariate variable to explain away the IVC filter-decreased mortality association. 29 Post matching, all the covariates were well-balanced (Fig. 2).
Severe PE can result in acute right ventricular strain and dysfunction. A severe mismatch between oxygen demand and supply, especially in patients with pre-existing coronary artery disease can lead to troponin leak and acute myocardial infarction.30,31 Elevated troponin in the setting of PE has been associated with higher mortality. 12 Decreased mortality was observed with IVC filter placement in PE complicated with acute myocardial infarction in our study. In the PREPIC 2 trial, 65% of the patients in both arms had evidence of right ventricular dysfunction either by echocardiogram or elevated troponin. Only 6 out of 200 (3%) patients with IVC filter placement and 3 out of 199 (1.5%) of deaths were attributed to PE. 32 The trial was underpowered to detect any mortality or PE recurrence difference. The higher mortality noticed in our study in PE complicated by acute myocardial infarction—17% with IVC filter vs. 30.1% without IVC filter—discerned the effect of IVC filter on mortality.
Acute hypoxemic respiratory failure is one of the significant indicators of PE severity and is part of the Pulmonary Embolism Severity index.33,34 However, the impact of IVC filter in PE patients complicated with acute respiratory failure independent of other medical conditions has not been studied well to date. PSM analysis showed that IVC filter placement in PE complicated with acute respiratory failure was associated with lower in-hospital mortality. The mortality benefit was observed in patients with anticoagulation contraindications and patients requiring mechanical ventilation in further subgroup analysis.
In the PE complicated by shock, the IVC filter was associated with mortality improvement. Importantly, in the subgroup analysis of these patients with anticoagulation contraindications, IVC filter placement was again associated with a mortality benefit. These results are consistent with those reported by Stein et al.11,12 who studied unstable patients (shock with mechanical ventilation) on retrospective databases and with the guidelines provided by most professional societies.4,6,35 Similarly, our findings are also consistent with the mortality benefit described in the ICOPER registry, where patients with massive PE who received an IVC filter demonstrated decreased mortality (10.1% vs. 43.3%; HR 0.12, p = 0.02). 36
We also demonstrated that IVC filter placement was associated with lower mortality in PE patients who received thrombolytics (Table 3). This is in agreement with the mortality benefit reported using the Premier administrative database 2010–2014, although this study did not account for the confounders. 37
We hypothesize that in patients with complicated PE who have limited lung and cardiac reserve and are at high risk of another PE, IVC filter placement decreases the recurrence of PE, 38 which would otherwise prove fatal. The high E-values in our study reinforce the robustness of the results from our observational study. The results from this national observational study call for the need for prospective studies and clinical trials to further evaluate the outcomes of IVC filters in complicated PE.
Limitations
Our study has certain limitations. The sequence of the events that led to death, including the definitive cause of death could not be established. The use of ICD-9-CM codes has limited sensitivity in claims data and does not necessarily capture all the clinical events, 14 including post-discharge outcomes. Our study did not evaluate the role of IVC filter in saddle PE, and the conclusions do not apply to saddle PE. We recognize that the accuracy of the interpretation of the data results is contingent upon the correct ICD-9-CM coding in claims data. The ICD-9-CM coding for contraindications to anticoagulation may not correctly identify all the patients that did not receive anticoagulation and may include cases that had complications to anticoagulation. A final limitation is the inability of PSM to address unmeasured confounding; only measured confounders (age, sex, race, PE comorbidities, and Elixhauser comorbidities) were adjusted for in estimating the effect of IVC filter on mortality. However, as explained, E-value >2 makes it unlikely for unmeasured confounder or covariate to explain away the observed mortality difference associated with IVC filter placement in complicated PE.
Conclusion
IVC filter placement is associated with decreased in-hospital mortality in patients with acute PE complicated by acute myocardial infarction, acute respiratory failure, shock, and in those who received thrombolytics. These results call for the need for prospective studies and clinical trials to further assess the benefit of IVC filter in complicated PE.
Muhammad H Gul and Zin M Htun contributed equally to this work and are joint first authors.
Supplemental Material
PUL882636 Supplemetal Material - Supplemental material for Clinical outcomes of inferior vena cava filter in complicated pulmonary embolism
Supplemental material, PUL882636 Supplemetal Material for Clinical outcomes of inferior vena cava filter in complicated pulmonary embolism by Muhammad H. Gul, Zin M. Htun, Joseph Rigdon, Belinda Rivera-Lebron and Vinicio de Jesus Perez in Pulmonary Circulation
Footnotes
Acknowledgments
We would like to thank Gustavo Davila Heresi, MD, for ideas on improving the project
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Ethical approval was not needed, as NIS is de-identified and commercially available.
Guarantor
MHG had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis, including and especially any adverse effects.
Contributorship
Muhammad H. Gul and Zin Mar Htun contributed equally to this work and are joint first authors. ZMH, JR, BRL, VJP contributed substantially to the study design, methodology and interpretation, and the writing of the manuscript. The authors have no conflict of interest to disclose. No external funding was received for this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
