Abstract
Chronic obstructive pulmonary disease (COPD) is characterized by chronic expiratory airflow obstruction that is not fully reversible. COPD patients develop varying degrees of emphysema, small and large airway disease, and various co-morbidities. It has not been clear whether these co-morbidities share common underlying pathogenic processes with the pulmonary lesions. Early research into the pathogenesis of COPD focused on the contributions of injury to the extracellular matrix and pulmonary epithelial cells. More recently, cigarette smoke-induced endothelial dysfunction/injury have been linked to the pulmonary lesions in COPD (especially emphysema) and systemic co-morbidities including atherosclerosis, pulmonary hypertension, and chronic renal injury. Herein, we review the evidence linking endothelial injury to COPD, and the pathways underlying endothelial injury and the “vascular COPD phenotype” including: (1) direct toxic effects of cigarette smoke on endothelial cells; (2) generation of auto-antibodies directed against endothelial cells; (3) vascular inflammation; (4) increased oxidative stress levels in vessels inducing increases in lipid peroxidation and increased activation of the receptor for advanced glycation end-products (RAGE); (5) reduced activation of the anti-oxidant pathways in endothelial cells; (6) increased endothelial cell release of mediators with vasoconstrictor, pro-inflammatory, and remodeling activities (endothelin-1) and reduced endothelial cell expression of mediators that promote vasodilation and homeostasis of endothelial cells (nitric oxide synthase and prostacyclin); and (7) increased endoplasmic reticular stress and the unfolded protein response in endothelial cells. We also review the literature on studies of drugs that inhibit RAGE signaling in other diseases (angiotensin-converting enzyme inhibitors and angiotensin receptor blockers), or vasodilators developed for idiopathic pulmonary arterial hypertension that have been tested on cell culture systems, animal models of COPD, and/or smokers and COPD patients.
Introduction
Chronic obstructive pulmonary disease (COPD) is a chronic inflammatory lung disease which is characterized by airflow obstruction that is only partially reversible. 1 The main environmental risk factor for COPD is inhalation of cigarette smoke (CS). However, exposure to air pollution, occupational dusts, and smoke from burning biomass fuels can also cause COPD when the exposures are sufficiently intense or prolonged. 2 COPD affects > 5% of the world’s population, and is projected to become the third leading cause of death worldwide by 2030.3,4 COPD is associated with a significant socioeconomic burden, which is predicted to increase over the coming decades. 5 Current treatment options for COPD are limited, only partially reduce symptoms, and have not been conclusively shown to alter COPD disease progression. Thus, there is a huge unmet need to develop disease-modifying therapies for COPD.
Pulmonary pathologies in COPD patients: The chronic airflow limitation that characterizes COPD is caused by two distinct pulmonary pathologies: emphysema and small airway disease. Emphysema is characterized by loss of the alveolar walls and contributes to airflow obstruction via loss of elastic recoil and collapse of the distal airways during expiration. Emphysema leads to loss of surface area for gas exchange and can thereby lead to hypoxemia. Small airway disease contributes significantly to airflow obstruction by narrowing the lumen of small airways as a consequence of sub-epithelial fibrosis, airway inflammation, goblet cell hyperplasia, and luminal obstruction caused by inflammatory exudates and mucus. 6
Other COPD patients develop chronic bronchitis which is characterized by chronic cough and mucus hyper-secretion (for at least three months per year), and is caused by hypertrophy and hyperplasia of submucosal glands along with mucus cell metaplasia in the airways. Chronic bronchitis is associated with an accelerated rate of decline in lung function, an increased risk of developing airflow obstruction, a predisposition to lower respiratory tract infections, a higher acute exacerbation frequency, and higher overall mortality rates. 7 The relative contribution of airspace disease, large airway disease, and small airway disease to the overall clinical phenotype varies substantially between COPD patients. 8
Vascular and non-vascular co-morbidities in COPD: COPD is associated with a plethora of extra-pulmonary co-morbidities, which influence the prognosis of COPD patients. A number of COPD co-morbidities affect the vasculature and include systemic arterial hypertension which occurs in ∼ 70% of COPD patients, atherosclerosis, systemic inflammation, pulmonary arterial hypertension (PAH), cor pulmonale, and venous thromboembolism. 9 Other non-vascular co-morbidities include an increased risk for lung cancer, cachexia, osteoporosis, muscle wasting, obesity, metabolic syndrome, diabetes mellitus, anxiety, depression, sleep disturbance, and anemia.9,10 Some of these co-morbidities are linked to acute or chronic CS exposure including systemic arterial hypertension, atherosclerosis, and lung cancer. 10 PAH and cor pulmonale develop as a consequence of pulmonary vascular remodeling and loss of lung tissue during emphysema development. Other COPD co-morbidities have been ascribed to chronic systemic inflammation (metabolic syndrome, diabetes mellitus, osteoporosis, anxiety, and depression). 10
The pathogenesis of COPD and the role of extracellular matrix injury: Extracellular matrix injury was first linked to the pathogenesis of emphysema in the 1960s. In 1963, Laurell and Eriksson linked pulmonary emphysema to hereditary deficiency of alpha1-antitrypsin (AAT), the major inhibitor of neutrophil elastase in the lower respiratory tract. 11 In 1965, Gross et al. showed that delivering the elastolytic enzyme, papain, into the lungs of rats led to emphysema development. 12 Other elastin- and collagen-degrading enzymes were subsequently shown to induce emphysema development in animals.13–15 These observations led to the formulation of the proteinase-anti-proteinase hypothesis for the pathogenesis of COPD. This hypothesis, focused on proteinase-mediated injury to the extracellular matrix components of the alveolar walls, dominated the COPD research field for several decades. 16 As AAT is the major inhibitor of neutrophil elastase (NE) in the lower respiratory tract, and NE is a potent elastin-degrading proteinase, early research efforts focused on the contributions of polymorphonuclear neutrophils (PMNs). Later studies focused on macrophages which also produce elastin- and collagen-degrading enzymes.13,14,17,18 More recently, adaptive immune cells (such as CD8+ and CD4+ T cells,19–23 and B cells24,25) were identified as culprits in COPD.
Contributions of alveolar septal cell injury to COPD: The proteinase–antiproteinase hypothesis does not completely explain the loss of lung tissue occurring in CS-induced emphysema. Thus, it was subsequently hypothesized that the disappearance of the alveolar walls during emphysema development also involves the progressive loss of the cellular components of the alveolar walls (capillary endothelial and alveolar epithelial cells) in addition to loss of the extracellular matrix within the alveolar walls. In 2000, injury to the cellular components of the alveolar walls alone was shown to be sufficient for emphysema to develop in experimental animal models. Kasahara et al. reported that delivering an antagonist of the receptor for vascular endothelial growth factor (VEGF; a growth factor which has crucial activities in maintaining the homeostasis of alveolar epithelial and endothelial cells [ECs]) to rats led rapidly to airspace enlargement in the absence of inflammation. 26 Thereafter, research focused on lung epithelial cells, as they are located at the interface between the external environment and the internal pulmonary milieu, and are a major target for inhaled insults. Reactive oxygen species (ROS) contained in CS were found to induce alveolar epithelial cell injury and apoptosis. 27 Moreover, lung epithelial cells were identified as effector cells that initiate and control immune and inflammatory responses to inhaled toxins, and produce ROS and reactive nitrogen species (RNS), thereby contributing to the oxidant/antioxidant imbalance that amplifies chronic pulmonary inflammation and injury. 28
The vasculature and endothelial cells in COPD
Although ECs are also a key component of the alveolar gas exchange unit, until recently, these cells have received much less attention than alveolar epithelial cells in the pathogenesis of COPD. In the remainder of this review, we will focus on the contribution of endothelial dysfunction and injury to the pathogenesis of COPD, and provide evidence that systemic endothelial injury underlies both emphysema development and systemic COPD co-morbidities including chronic renal injury, which has only recently been identified as a co-morbidity of COPD. 29 We will review the potential mechanisms underlying the EC injury detected in COPD patients. We will also review the potential of therapeutics that target endothelial injury as novel disease-modifying therapies for COPD.
Studies of the vasculature in human COPD patients: The human pulmonary vasculature is critical for gas exchange in the lung, and the total pulmonary vascular surface area is 90 m2. The vascular system is lined by ECs which form a continuous monolayer. 30 Early studies identified injury to pulmonary vessels in lung tissue from COPD patients. In 1959, Liebow performed a histological examination of human emphysematous lungs and observed that the alveolar septa in centrilobular emphysema are remarkably thin and almost avascular. 31 He considered that a reduction in the blood supply via the small pre-capillary blood vessels induces the disappearance of alveolar septa. However, these early findings were largely ignored. In 1998, morphological abnormalities of the endothelium of the pulmonary artery were reported to be present even in patients with mild COPD. 32 In patients with mild COPD, and also in smokers with normal lung function, the small pulmonary arteries had thickened intimas, and tobacco consumption was suggested to play a critical role in the pathogenesis of pulmonary vascular abnormalities in COPD. 32 Computed tomography (CT) scans of the lungs of COPD patients with emphysema demonstrated pruning of the peripheral vasculature (indicative of loss of small vessels) in addition to vascular remodeling in COPD lungs. 33 More recent studies have examined the role of the vascular endothelium in COPD.
Endothelial dysfunction in COPD: In 1991, Dinh-Xuan et al. reported that endothelial dysfunction occurs in the pulmonary arteries of COPD patients with end-stage disease as assessed by in vitro experiments measuring relaxation of pulmonary artery rings in response to increasing concentrations of exogenous acetylcholine and adenosine diphosphate (ADP). 34 They suggested that endothelial dysfunction in late-stage COPD might contribute to the development of PAH in COPD. However, a subsequent study reported that endothelial dysfunction is also an early feature of COPD, as normoxic patients with mild COPD, and even smokers without COPD, have evidence of endothelial dysfunction. 32 Peinado et al. assessed whether endothelial dysfunction is present in early-stage COPD and related to structural abnormalities in the pulmonary vessels. They measured endothelium-dependent relaxation mediated by nitric oxide (NO) in pulmonary artery rings from COPD patients with varying disease severity, smokers without COPD, and non-smokers that were exposed to cumulative concentrations of acetylcholine and ADP. 32 Endothelium-dependent relaxation was reduced in COPD samples, even in individuals with mild disease, and was related to airflow obstruction. Peinado et al. also detected increased intimal thickening in the small pulmonary arteries in both smokers and COPD patients when compared with those in non-smokers, indicating that endothelial dysfunction is induced by CS and can be detected before COPD develops. 32
Measuring endothelial dysfunction in COPD patients: Ultrasound-based flow-mediated vasodilation (FMD) has been widely used to measure endothelial dysfunction in COPD patients and to correlate endothelial dysfunction with clinical outcomes. Patients with impaired FMD have a reduced 6-min walk test and a worse overall prognosis.35,36 Endothelial dysfunction, measured as impaired FMD, is directly related to COPD severity as assessed by forced expiratory volume in 1 s (FEV1).37–39
Linking endothelial dysfunction and injury to lesions in the lungs and kidneys of COPD patients
COPD is associated with lesions in organs systems outside the lung, and the vascular endothelium is common to the lung and other organs affected by COPD. However, until recently it was not clear whether the pulmonary lesions and the chronic lesions in other organs in COPD patients share underlying mechanisms. Recent studies have provided evidence that endothelial dysfunction and injury contribute to emphysema development, PAH, and chronic renal injury that can develop in COPD patients.
Endothelial injury in emphysema development in COPD: In 2000, the emphysema phenotype was linked to endothelial injury. Delivering a VEGF receptor (VEGFR) antagonist to rats led rapidly to air space enlargement and pruning of the pulmonary arterial tree. 26 VEGF is a trophic factor that is crucial for the survival of ECs. Subsequent studies of emphysematous lungs confirmed that COPD patients have decreased lung levels of VEGF, reduced expression of VEGFR in pulmonary ECs, 40 apoptotic alveolar septal cells, and reduced expression of hypoxia inducible factor-1 α (HIF-1α), a transcription factor (TF) that drives the expression of genes involved in endothelial function including VEGFRs. 41 Endothelial dysfunction and injury are also induced by acute CS exposure long before emphysema develops in animal models. Brief exposure of mice to CS exacerbates lipopolysaccharide- and Pseudomonas aeruginosa-induced acute lung injury in vivo, and CS extract (CSE) increases the permeability of endothelial monolayers in vitro. 42 Moreover, a recent study identified CS-induced apoptosis of ECs in the lungs (and kidneys) of mice exposed chronically to CS, and COPD patients 29 (outlined below). Thus, data from both animal models of COPD and COPD patients and controls support the hypothesis that endothelial dysfunction and injury are key processes in the pathogenesis of emphysema.
Endothelial injury links renal dysfunction and emphysema occurring in COPD patients: Microalbuminuria (MAB) is a marker of generalized endothelial dysfunction and injury as well as endothelial dysfunction in the kidney. MAB is associated with worse cardiovascular outcomes in patients with diabetes mellitus, hypertension, and the general population.43–46 A study of a Spanish cohort of COPD patients reported that 24% of these COPD patients (versus 4% of control participants) had persistent MAB. 47 Other studies have shown an association between MAB severity and systemic inflammation in COPD patients 48 and an increased mortality risk. 49 Additionally, albuminuria correlates with the degree of hypoxemia in clinically-stable COPD patients and increases during acute exacerbations.47,50 Even though MAB, indicative of glomerular capillary injury, has been reported in COPD patients,47,50 until recently, little was known about chronic renal injury in COPD.
Renal dysfunction was linked to CS-induced lung injury in humans in a large cohort of smokers screened for lung cancer in which an association between emphysema severity and the estimated glomerular filtration rate (eGFR) was reported. 51 In a study of > 900,000 individuals followed for ten years, smokers had a twofold higher risk of dying from renal failure than non-smokers after adjusting for potential confounders. 52 Another study reported that COPD patients have a higher prevalence of both concealed and overt renal failure than age-matched controls. 53
In 2017, COPD patients were shown to have pathologic evidence of chronic renal injury to explain their MAB.
29
COPD patients were shown to have more glomerulosclerosis (shrinkage and scarring of the glomeruli) and greater renal arterial and arteriolar sclerosis than age-matched smoker and non-smoker controls.
29
COPD patients also had evidence of interstitial fibrosis and tubular atrophy, which can develop secondary to chronic glomerular injury in other chronic renal diseases affecting the glomeruli. COPD also had lower eGFR rates than control participants, and there was a direct correlation between renal function (assessed as eGFR) and pulmonary function (assessed as FEV1).
29
These renal changes were linked to endothelial injury as COPD patients had greater EC apoptosis in small vessels in both the lungs and kidneys than smoker and non-smoker controls.
29
Unlike control participants, COPD patients also had double contouring of their glomerular capillary walls (Fig. 1), which is a response to repetitive injury to the endothelial wall, and is characterized by capillary wall remodeling with formation of new basement membranes and entrapment of cellular elements.
54
There was an indirect correlation between the severity of renal capillary injury (assessed as double contouring) and both renal function (as assessed by eGFR) and lung function (as assessed by FEV1) indicating that endothelial injury in the lungs and kidneys underlies the chronic injury detected in the lungs and kidneys of COPD patients.
29
Double contouring of renal capillaries indicative of repetitive endothelial injury in COPD. (a) Severe double contouring in a glomerular capillary in a renal section from a patient with COPD (red arrows). (b) A glomerular capillary in a renal section from a participant without COPD which has no double contouring. (c) A cartoon depicting deposition of additional basement membranes (double contouring) around a glomerular capillary in a patient with COPD (left) versus a normal glomerular capillary in a healthy individual that does not have double contouring (right).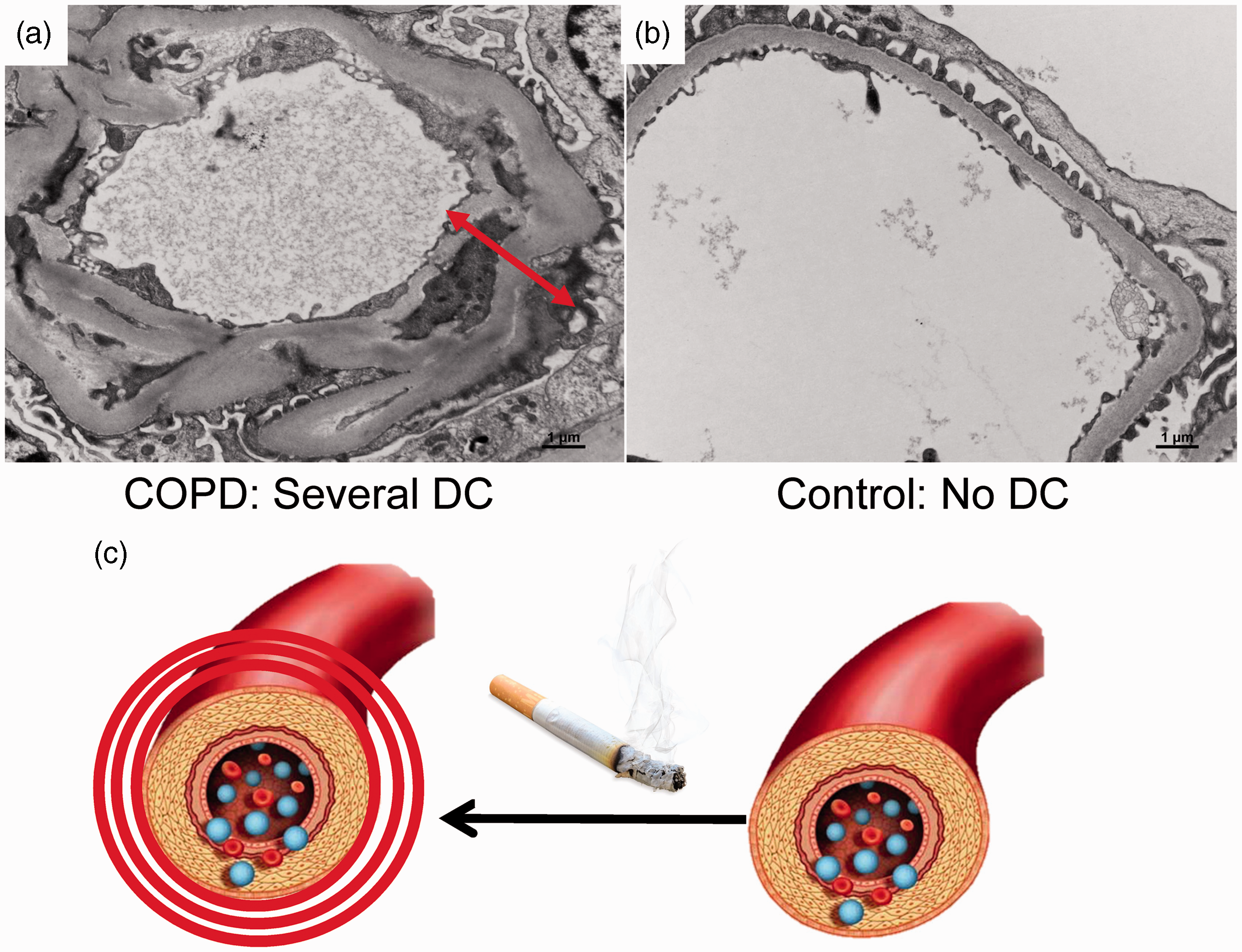
The same study reported that wild-type (WT) mice exposed to CS developed albuminuria which was detected as early as one week after initiating the CS exposures. After six months of CS exposure, the mice developed glomerulosclerosis and had increased EC apoptosis in their small vessels in both the lungs and kidneys. 29 Mice exposed chronically to CS also developed widening and flattening of the renal podocyte foot processes, 29 which is an early but non-specific sign of glomerular injury, and leads to increased glomerular permeability and proteinuria. 55 Together, the human and animal results support the notion that CS-induced endothelial injury links the chronic pulmonary and renal lesions detected in human COPD patients and CS-exposed mice.
Endothelial injury in PAH in COPD: PAH is a common co-morbidity of COPD. The incidence of mild to moderate PAH is ∼ 50% in advanced COPD and its development is associated with higher mortality rates, 56 poorer exercise capacity, 57 and more frequent exacerbations. 58 COPD patients with PAH generally have mild-to-moderate elevations in mean pulmonary arterial pressures (mPAP) and pulmonary vascular resistance (PVR), and a preserved cardiac output. 59 About 50% of COPD patients develop PAH during exercise which may be due to hypoxic pulmonary arterial vasoconstriction induced by exertion.58,60
As in idiopathic PAH (iPAH), the pulmonary arteries in patients with COPD exhibit intimal proliferation of poorly differentiated smooth muscle cells and the deposition of extracellular matrix proteins. Endothelial dysfunction occurs in the early stages of both iPAH and PAH secondary to COPD, and likely contributes to the progression of both diseases. 61 EC injury is followed by vasoconstriction and remodeling of the pulmonary arteries. Many of the factors that induce endothelial dysfunction and injury in the systemic circulation (outlined in more detail below) contribute to EC injury in the pulmonary circulation of patients with COPD and secondary PAH. These factors include increased EC expression of endothelin-162 and the receptor-II for transforming growth factor-beta, 63 and reduced EC expression of endothelial nitric oxide synthase (eNOS) 64 and prostacyclin synthase. 65 These perturbations together promote vasoconstriction and cell proliferation.
Potential mechanisms underlying endothelial dysfunction and injury in COPD
Studies of samples from COPD patients, animal models of COPD, and cell culture systems have identified a number of mechanisms by which endothelial injury/dysfunction develops in COPD. Endothelial dysfunction in the systemic and/or pulmonary circulations of COPD patients has been linked to: (1) direct toxic effects of CS on ECs;
66
(2) generation of auto-antibodies directed against ECs; (3) vascular inflammation; (4) increased oxidative stress levels in vessels inducing increases in lipid peroxidation and increased AGEs-RAGE activation; (5) reduced activation of the anti-oxidant pathways in ECs; (6) an increase in EC release of mediators with vasoconstrictor, pro-inflammatory, and remodeling activities (endothelin-1) and reduced EC expression of mediators that promote vasodilation and homeostasis of ECs (NOS and prostacyclin);
56
and (7) increased endoplasmic reticular stress and the unfolded protein response in ECs (Fig. 2).
Mechanisms by which endothelial dysfunction and injury is induced in COPD and leads to end-organ injury affecting the lungs and kidneys in COPD. CS components have direct toxic effects on ECs. CS also leads to increased oxidative stress levels in the lungs and kidneys. Oxidative stress increases the generation of AGEs in these organs, and AGEs bind to and activate RAGE. RAGE signaling in ECs induces endothelial dysfunction and injury (in part, by activating NF-κB), and this process further increases tissue oxidative stress levels and inflammation. RAGE activation also increases RAGE expression which amplifies endothelial injury and end-organ injury. Increased oxidative stress levels in tissues and components of CS (including oxidants and acrolein) also induce endoplasmic reticulum (ER) stress and the unfolded protein response in ECs leading to endothelial dysfunction/injury. CS and oxidative stress both induce senescence of ECs which contributes to endothelial dysfunction. CS changes the pattern of vaso-active mediators produced by ECs. These changes include increased generation of vasoconstrictors (such as endothelin-1) and reduced production of vasodilators such as prostacyclin and NO (due, in part, to reduced expression of prostacyclin synthase and NOS by ECs). Endothelial injury induced by the processes mentioned above leads to the generation of neo-epitopes against which auto-antibodies are generated. The binding of anti-EC antibodies to ECs further increases endothelial injury and amplifies tissue inflammation. The endothelial dysfunction and injury that are induced by all of these pathways lead to apoptosis of ECs. EC apoptosis in the lungs leads to loss of the alveolar walls and emphysema development. EC apoptosis in other organs leads to chronic end-organ injury. CS-induced injury in the kidney (detected as MAB) is characterized by glomerulosclerosis and secondary tubular atrophy and interstitial fibrosis.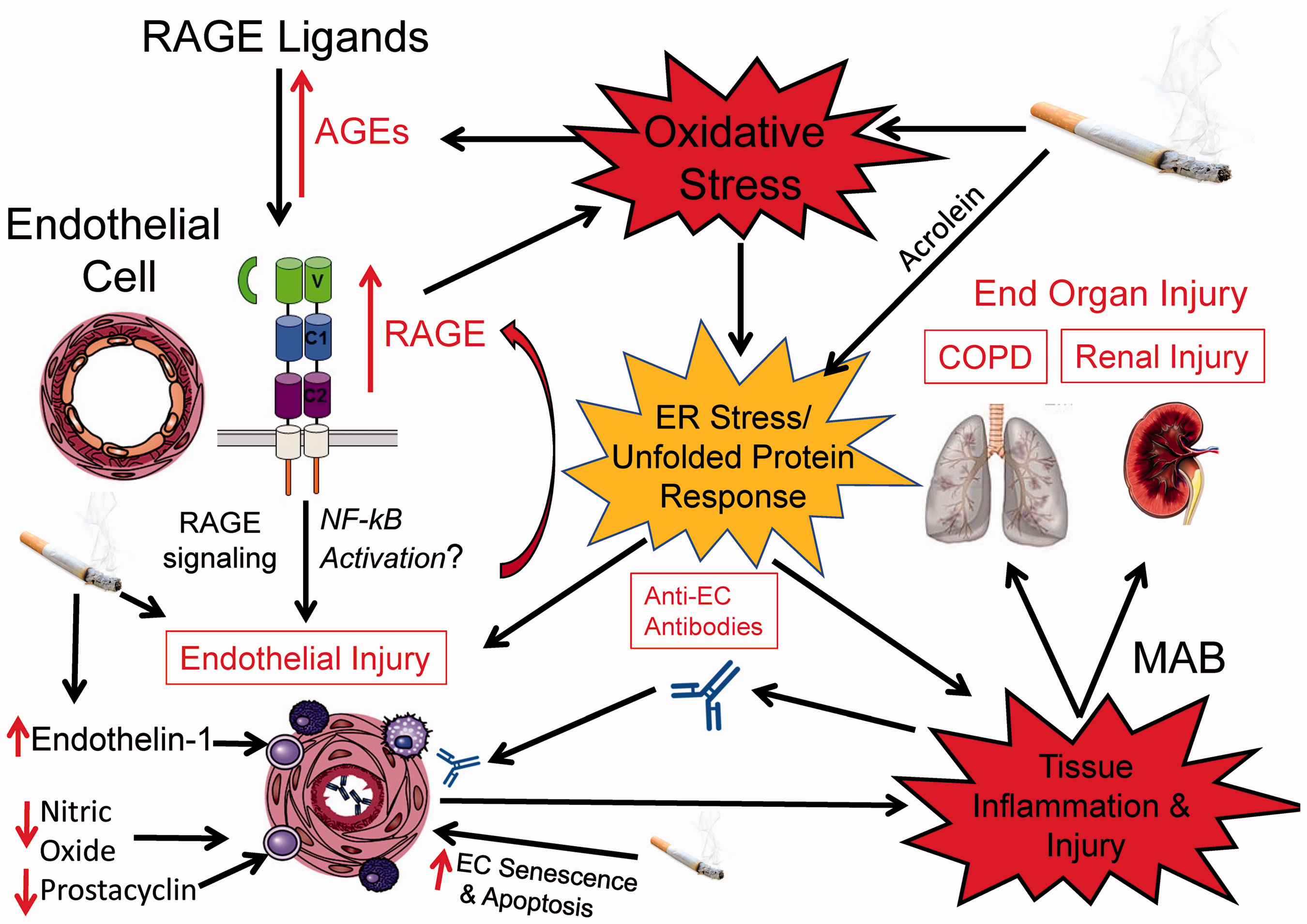
Direct toxic effects of CS: CSE directly induces apoptosis of ECs in vitro, and components of CS (including nicotine and its metabolites, acrolein, superoxide anion, and hydroxyl radicals) have been detected in the circulation where they can interact directly with ECs in different organs.67–70 As outlined above, inhaling CS increases the permeability of the alveolar-capillary barrier, 42 and this likely enables CS components to enter the circulation and directly injure ECs in the pulmonary and systemic circulations.
As the kidneys (unlike the lungs) are not directly exposed to CS, circulating components of CS are likely to be toxic to renal as well as pulmonary ECs. While it is clear that CSE injures ECs in vitro (see above), these findings have been largely ignored or characterized as clinically irrelevant, yet they provide mechanistic insights and support the notion that emphysema is, at least in part, a vascular problem. Thus, the “vascular COPD phenotype” is a clinically entity that has been under-recognized, and endothelial dysfunction and injury may be a common pathogenic mechanism linking emphysema development in COPD to several common COPD co-morbidities in addition to chronic renal injury including atherosclerosis, skeletal muscle wasting, and osteoporosis. Additional studies are needed to test this hypothesis.
Generation of auto-antibodies directed at EC components: Some COPD patients have circulating anti-antibodies directed against ECs. 71 CS-induced direct injury to ECs may lead to the generation of neo-epitopes that trigger an immune response in susceptible individuals. This immune response may include the generation of anti-endothelial antibodies that form immune complexes with their cognate antigens on ECs to thereby amplify EC dysfunction and injury, and increase tissue inflammatory responses (Fig. 2). 71
Vascular inflammation: Systemic inflammation occurring in COPD patients may contribute to the development of both pulmonary 32 and systemic 39 endothelial dysfunction thereby contributing to the development of emphysema, PAH, and chronic renal injury in COPD patients. ECs are activated in the pulmonary vessels of COPD patients, as assessed by increased expression of inducible adhesion molecules (E- and P-selectin, and intercellular adhesion molecule-1) and this is likely mediated by the exposure of these cells to inflammatory mediators generated in the inflamed lungs of COPD patients. 72 Activated ECs release cytokines, including TNF-α and IL-1β,73,74 which contribute to both emphysema development and the deposition of extracellular matrix proteins around the small airways of CS-exposed mice.75,76 MacNee et al. showed that when humans inhale CS, the transit of polymorphonuclear neutrophils in the pulmonary circulation is delayed, and their capability to interact with the pulmonary endothelium is increased which induces pro-inflammatory changes in the ECs via the release of mediators of inflammation. 77 Proteinase inhibitors that are upregulated during inflammatory responses have also been linked to endothelial dysfunction and injury in COPD. SERPINF1 expression is upregulated in human emphysematous lungs, and this proteinase inhibitor inhibits angiogenesis by inducing EC apoptosis. 78 AAT is an acute phase protein and its circulating levels increase during inflammatory responses. AAT is taken up by pulmonary ECs and inhibits intracellular active caspase-3 and therefore EC apoptosis in vitro and in animal models of COPD. 79
Pathways induced by increased oxidative stress: Oxidative stress plays a key role in the pathogenesis of the pulmonary component of COPD. 80 CS contains large numbers of ROS and RNS. ROS and RNS are generated by circulating inflammatory cells activated by CS. Activation of ECs by oxidative stress promotes intravascular micro-thrombosis, reduces blood flow, and further activates inflammatory cells to release ROS and RNS. 81 In addition to being activated by ROS and RNS, ECs are also an important source of ROS via activation of xanthine oxidase, NADH/NADPH oxidase, and uncoupled eNOS. EC-derived ROS and RNS contribute to vascular dysfunction and tissue injury in other diseases. 82 COPD patients have higher oxidative stress levels in plasma samples and ECs than control participants.83–85 Increased oxidative stress levels in vessels promote endothelial dysfunction and injury by inducing peroxidation of the lipid components of ECs, increasing activation of the receptor for advanced glycation end-products (RAGE), and inducing cellular senescence.
Lipid peroxidation: Endothelial dysfunction occurring in other diseases has been linked to lipid peroxidation of components of ECs.32,86–88 Increased systemic oxidative stress levels in COPD patients increase the generation of lipid peroxidation products including malondialdehyde (MDA) 85 and 4-hydroxy-2-nonenal (4-HNE). 84 Plasma MDA levels increase as COPD severity increases (as assessed by FEV1 percent predicted).85,89 Pulmonary EC levels of 4-HNE-modified proteins are higher in formers smokers with COPD compared with former smokers without COPD, and there is an inverse correlation between 4-HNE adduct levels in pulmonary ECs and the FEV1 percent predicted value. 84 Thus, the increased oxidative stress levels in the vessels of COPD patients may contribute to endothelial dysfunction and injury and loss of the alveolar walls, in part, by increasing peroxidation of the lipid components of ECs.
The advanced glycation end-products (AGEs) and receptors for AGEs (RAGE) pathway: Excessive RAGE signaling is linked to MAB occurring in animal models of diabetes mellitus.90,91 As outlined below, RAGE has been strongly linked to COPD previously but not to endothelial injury or MAB occurring in COPD patients. RAGE is a transmembrane receptor that is ubiquitously expressed and is activated when ligands, including AGEs, high mobility group box 1 (HMGB1), and calgranulins bind to this receptor (Fig. 2). 92 Signaling via RAGE increases the release of pro-inflammatory mediators by activating TFs including nuclear factor of kappa light polypeptide gene enhancer in B-cells (NF-κB), Egr-1, and JAK/STAT, leading to tissue inflammation and injury. 92
Genome-wide association studies have linked single-nucleotide polymorphisms in the RAGE (AGER) locus to COPD development and most strongly to the emphysema phenotype.93,94 RAGE promotes the development of emphysema in experimental animals as CS-exposed RAGE-deficient (Ager−/−) mice are protected from emphysema development and lung inflammation when exposed to CS. 95 Conditional over-expression of RAGE in the airways of mice in an inducible fashion leads to increased lung inflammation, progressive airspace enlargement, and loss of microvascular ECs in the lung.96–98 Interestingly, soluble RAGE (generated by proteolytic shedding of RAGE from cell surfaces) functions as a decoy receptor which blocks the binding of RAGE ligands to the transmembrane form of RAGE expressed on cell surfaces.92,99 Reduced plasma soluble RAGE levels are linked to the presence of emphysema in humans. 100
Recently, oxidative stress-induced increased RAGE signaling was linked to endothelial injury in the lungs and kidneys of human COPD patients and CS-exposed mice. 29 This study reported higher RAGE staining in ECs in both the lungs and kidneys of human COPD patients compared with ECs in smokers and non-smoker controls. Higher RAGE staining in ECs in both the lungs and kidneys was also detected in WT mice exposed to CS versus air. AGEs are ligands for RAGE, and their generation is induced by CS and oxidative stress which is increased in COPD tissues.80,101 Increased RAGE activation by ligands including AGEs also leads to increased RAGE expression. 102 Thus, the hypothesis that CS increases tissue oxidative stress levels to promote the generation of AGEs, thereby causing RAGE activation, which, in turn, increases the expression of RAGE was then tested by these authors. Oxidative stress, AGEs, and RAGE levels were increased in pulmonary and renal tissue homogenates in CS-exposed mice. Immunostaining studies showed that the increases in AGEs and RAGE staining occurred mostly in glomerular and pulmonary ECs in COPD patients and CS-exposed mice. 29
AGE–RAGE interactions activate several TFs including NF-κB. RAGE-mediated activation of ECs via NF-κB activation increases EC production of pro-inflammatory, pro-coagulant, and vasoactive genes. 103 However, RAGE also signals via other TFs including Egr-1 and JAK/STAT. 92 It is unclear which intracellular pathways downstream of RAGE lead to endothelial dysfunction and pro-inflammatory signaling in the lungs and kidneys of COPD patients. CS may induce endothelial dysfunction by activating RAGE via pathways other than the oxidative stress-AGEs pathway. For example, RAGE is activated by ligands other than AGEs, and the levels of these other RAGE ligands (including HMGB1 and calgranulins) are elevated in lung and plasma samples from COPD patients.104,105
Senescence of ECs: Lung tissue from COPD patients has an increased percentage of ECs that are senescent, and pulmonary ECs from COPD patients cultured ex vivo develop replicative senescence earlier than cells from control participants. 106 Pulmonary ECs from COPD patients have reduced telomerase activity, shorter telomeres, and higher p21 and p16 expression levels than cells from control participants. 106 Oxidative stress and CS both contribute to the induction of senescence in other cells,107,108 and thus are likely to be inducers of senescence in ECs in both the systemic and pulmonary circulations of COPD patients. Senescent pulmonary epithelial cells and ECs release inflammatory mediators that may increase the local and systemic inflammation detected in COPD patients.106,109,110
Reduced activation of anti-oxidant pathways
Nuclear factor-erythroid 2-related factor 2 (NRF2): One of the most important anti-oxidant pathways in the lung is the NRF2 pathway. NRF2 is a TF which responds to oxidative stress by binding to anti-oxidant response elements in the nucleus to promote the transcription of many antioxidants and cyto-protective genes. Nrf2-deficient mice develop exaggerated CS-induced pulmonary emphysema, lung inflammation, lung oxidative stress levels, and alveolar septal cell apoptosis. 111 Although NRF2 is most highly expressed by epithelial cells and inflammatory cells in the lung, it is also expressed by lung ECs. 112 NRF2 expression and activation is reduced in macrophages and epithelial cells in COPD lungs,113,114 and NRF2 expression increases in peripheral blood mononuclear cells from patients with COPD after they stop smoking. 85 NRF2 protects the endothelium from oxidant-induced injury that occurs during aging, and in patients with diabetes mellitus and hypertension. 115 Exposing human ECs to serum from smokers reduced NRF2 expression by the ECs in vitro, 83 but whether reduced expression or activation of NRF2 occurs in ECs in COPD lungs has not been evaluated. Future studies should determine whether CS reduces NRF2 expression in ECs in the systemic and/or pulmonary circulation to induce endothelial dysfunction and injury and thereby to contribute to emphysema development, PAH, chronic renal injury, and other co-morbidities in COPD patients.
Mediators with vasoactive properties
NOS and NO: NO is a gaseous signaling molecule which is produced by ECs and other cells. NO has important beneficial effects on vessels including reducing vascular smooth muscle tone, inhibiting smooth muscle proliferation and migration, promoting EC homeostasis, inhibiting platelet aggregation, and suppressing of the release of inflammatory mediators from ECs. 116 NO is generated by three isoforms of NOS: neuronal NOS (nNOS, NOS1); inducible NOS (iNOS, NOS2); and endothelial NOS (eNOS, NOS3). Although all three isoforms are expressed by ECs, eNOS is mainly responsible for production of NO by ECs.117–119 Systemic endothelial dysfunction has been linked to reduced release of bioavailable NO by ECs. CS reduces EC generation of bioavailable NO by: (1) inducing epigenetic silencing of eNOS; 120 (2) increasing protein kinase C-mediated phosphorylation of eNOS to reduce eNOS activity; 121 and (3) generating oxidants (including superoxide anion) that interact with NO to convert NO to peroxynitrite (ONO2-) which has pro-inflammatory activities and induces cellular injury. 122
Reduced eNOS expression in vessels contributes to the pulmonary vasoconstriction and the excessive growth of the tunica media observed in patients with iPAH. 123 Reduced endothelial-mediated generation of NO also occurs in the pulmonary vessels of smokers and COPD patients. Heavy smokers have reduced eNOS expression in their pulmonary arterial ECs. 64 COPD patients with PAH have reduced expression of eNOS in their pulmonary arteries and eNOS expression is inversely related to COPD severity. 124 Thus, downregulation of NO generation may also contribute to endothelial dysfunction during the development of both emphysema (Fig. 2) and PAH in COPD patients.
Prostacyclin: Prostacyclin is synthesized by prostacyclin synthase and is released, along with NO, by ECs. 125 Prostacyclin induces vasodilation, and inhibits platelet aggregation and mitogenesis of various cells. 126 Reduced prostacyclin expression by ECs has been linked to endothelial dysfunction in COPD. Pulmonary ECs in COPD patients have lower prostacyclin synthase levels than cells in lungs from control participants.65,127,128 Increased EC apoptosis was associated with reduced expression of prostaglandin synthase in ECs in the lungs of COPD patients. 129 In addition, CSE reduces the expression of prostacyclin synthase in human ECs in vitro. 65 Furthermore, mice with lung-specific over-expression of prostacyclin synthase were protected from CS-induced apoptosis of pulmonary ECs, 65 but neither emphysema development nor PH were measured in this study. Additional studies are needed to determine whether the reduced expression of prostacyclin by ECs that is induced by CS exposure contributes to endothelial dysfunction in both the systemic and pulmonary circulations of COPD patients to promote emphysema development, PAH, and possibly other COPD co-morbidities.
Endothelin-1: The endothelium also releases mediators having deleterious activities on vessels including endothelin-1. 126 Endothelins are peptides with vasoconstrictor activities consisting of 21 amino acids. There are three endothelin isoforms (endothelin-1, -2, and -3) that bind to four endothelin receptors, ETA, ETB1, ETB2, and ETC. 130 Endothelin-1 is the predominant isoform expressed in the vasculature and is the most potent vasoconstrictor that is known to date. Endothelin-1 induces vascular dysfunction in other diseases via its potent vasoconstrictor, pro-inflammatory and mitogenic activities, and its capacity to induce the release of free radicals from ECs and platelet activation. 131 Endothelin-1-induced endothelial dysfunction has been implicated in other diseases including iPAH, 132 and the vasculopathy that occurs in patients with diabetes mellitus 133 and systemic sclerosis. 134 Plasma endothelin-1 levels are increased in patients with stable COPD and inversely related to COPD severity as assessed by baseline FEV1. 135 Plasma endothelin-1 levels increase further during acute exacerbations of COPD and correlate inversely with the degree of oxy-hemoglobin desaturation. 135 Arterial endothelin-1 levels are elevated in COPD patients that have nocturnal oxy-hemoglobin desaturation. 136
These data suggest that hypoxia drives endothelin-1-mediated endothelial dysfunction in COPD. However, studies evaluating the efficacy of endothelin receptor antagonists in animal models of COPD have not yet been conducted to test this hypothesis. It is noteworthy that mice lacking endothelin-1 only in podocytes or vascular ECs are protected from chronic renal injury and MAB in models of diabetes mellitus and renal ischemia-reperfusion injury.137,138 Also, transgenic mice over-expressing human endothelin-1 develop age-related glomerulosclerosis and interstitial fibrosis associated with increased renal EC apoptosis.139,140 However, studies of the pulmonary and renal phenotypes of CS-exposed endothelin-1 gene-targeted mice have not yet been published.
Renin-angiotensin-aldosterone system: Angiotensin-II is another vaso-active mediator that has potential to contribute to endothelial dysfunction in COPD patients as it potently stimulates vasoconstriction in the systemic and pulmonary circulations, and has pro-inflammatory and pro-fibrotic activities (Fig. 3).
141
Angiotensin II is generated by angiotensin-converting enzyme-1 (ACE-I) which is highly expressed by pulmonary ECs and its expression increases during chronic hypoxia. ACE-I is a metalloproteinase which converts inactive angiotensin-I to active angiotensin-II (Fig. 3). There are no published studies of ACE-I or angiotensin II levels in COPD patients with the emphysema-predominant phenotype. However, several studies have linked ACE-I genetic polymorphisms and angiotensin-II to PAH in COPD patients.
The Renin-Angiotensin-Aldosterone System (RAAS). ACEi and ARBs were originally designed to inhibit the RAAS. Renin is an enzyme that converts angiotensinogen (produced by the liver) to inactive angiotensin I (Ang I). ACE-I is a metalloproteinase which is highly expressed by pulmonary ECs. ACE-I converts inactive Ang I to active angiotensin II (Ang II). Ang II has potent vasoconstrictor activities along with pro-inflammatory and pro-fibrotic activities. ACEi inhibit the enzymatic activity of ACE-I. ARBs compete with Ang II for Ang type I receptors and thereby block Ang II signaling.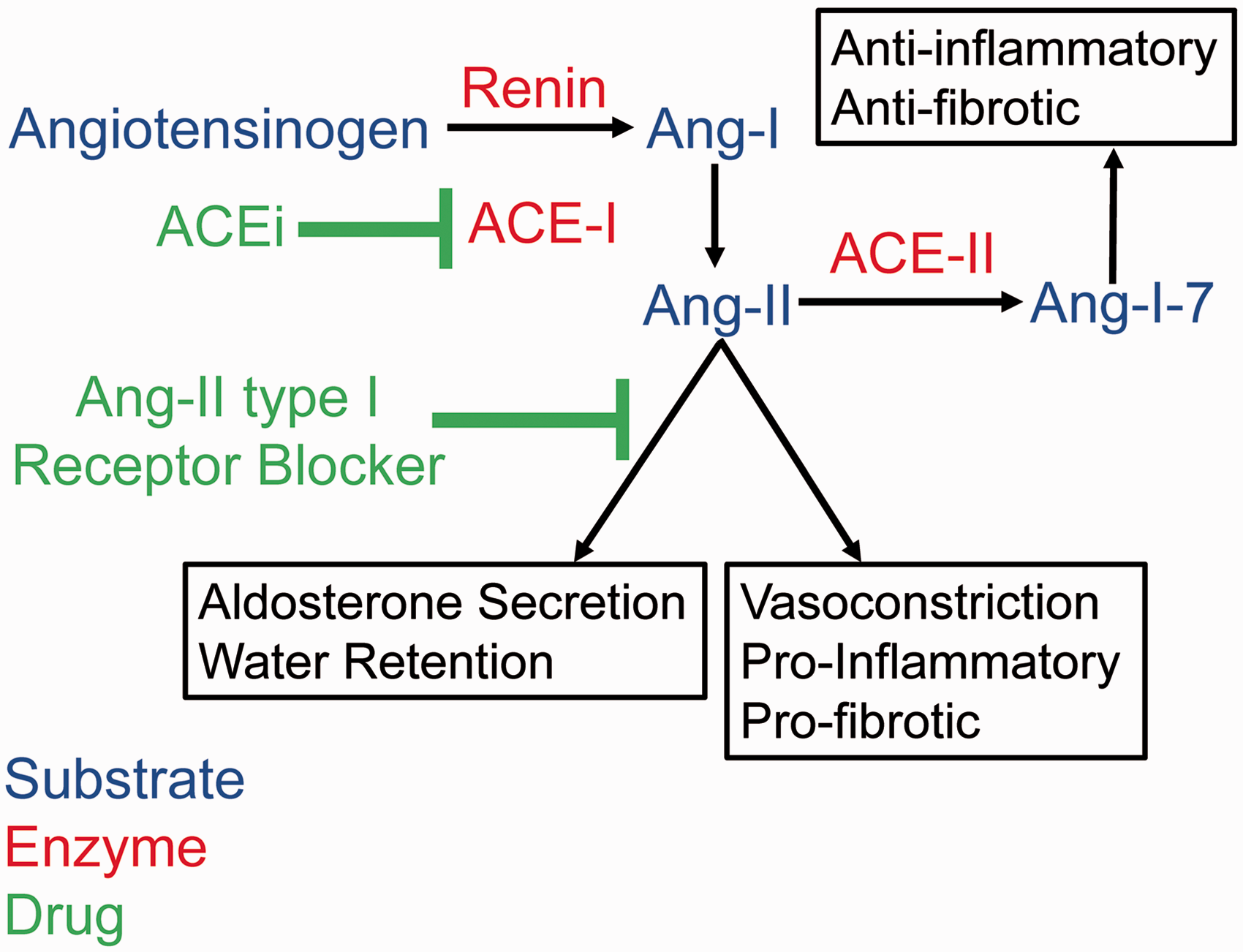
The ACE-I gene contains a polymorphism based on the presence (insertion [I]) or absence (deletion [D]) within an intron of a 287 base-pair nonsense DNA domain, resulting in three genotypes (DD homozygote, II homozygote, and DI heterozygote). 142 The ACE DD genotype is associated with increased circulating and cellular concentrations of ACE-I, and an increased risk of cardiovascular disease. 142 The DD genotype was associated with PAH that develops during exercise and was associated with impaired tissue oxygenation in two studies of small cohorts of COPD patients.143,144 In another cohort of 63 male COPD patients, the DD ACE-I genotype was strongly linked to endothelial dysfunction suggesting that the D variant is linked to abnormal vascular responses. 145 However, additional studies of larger cohorts of COPD patients including women are needed to confirm this observation. A study of a small cohort of hypoxemic COPD patients with PAH and cor pulmonale showed that the angiotensin-II receptor blocker, losartan, reduced the mPAP and systemic vascular resistance, and improved cardiac output. These results suggest that increased angiotensin II-mediated signaling contributes to the PAH co-morbidity in human COPD patients, but additional studies of larger cohorts of COPD patients with PAH are needed to confirm these results.
There are no reports in the literature linking high angiotensin-II levels to endothelial dysfunction in COPD patients without PAH. However, treating mice chronically exposed to CS with an angiotensin receptor blocker (losartan) protected the animals from CS-induced increases in lung oxidative stress levels, alveolar septal cell apoptosis, and emphysema development. 146 The authors attributed the effects of losartan to inhibition of transforming growth factor-β (TGF-β) signaling in the lung, but they did not measure plasma angiotensin-II levels or albuminuria in the CS-exposed and losartan-treated mice. Additional studies are needed to determine whether angiotensin-II signaling contributes to generalized endothelial dysfunction and injury, alveolar septal cell apoptosis, emphysema development, PAH, and chronic renal injury in humans with COPD.
Endoplasmic reticulum (ER) stress and the unfolded protein response: ER stress is another mechanism that may contribute to endothelial dysfunction in COPD. ER stress triggers the unfolded protein response (UPR), which is a group of pathways activated by the accumulation of improperly folded proteins in cells. ER stress and the UPR are triggered by various stressors that disrupt successful maturation of proteins in the ER by interfering with proper folding, assembly, and post-translational modification. Proteins enter the ER as unfolded polypeptide chains and the capacity of the ER to fold proteins is regulated by a signal transduction pathway that uses sensors facing the ER lumen. ER stress reduces the protein load entering the ER and increases the capacity of the ER to handle the unfolded proteins. If homeostasis cannot be achieved by these processes, the UPR induces other signaling pathways that induce inflammation and cellular apoptosis.
Prolonged activation of the unfolded protein response promotes EC death during the development of other diseases including atherosclerosis. 147 Studies of whole lung samples from CS-exposed mice revealed that: (1) CS-induced oxidative stress impaired oxidative protein folding in the ER and triggered an ER stress response; and (2) CS impaired oxidative protein folding by reducing the formation of disulfide bonds through excessive post-translational oxidation of the enzyme, protein disulfide isomerase, an ER luminal protein which contributes to disulfide bond formation and isomerization. 148 Although oxidative stress may trigger ER stress and the UPR, other components of CS may also contribute. Systemic delivery of a component of CS (acrolein) to rats increased lung levels of acrolein-protein adducts and this change was associated with increased ER stress and an UPR in the lungs, along with increased alveolar septal cell death and emphysema development. 149 Inhibiting CS-induced ER stress in mice by treating them with 4-phenylbutyric acid reduced pulmonary inflammation, alveolar septal cell apoptosis, and emphysema development. 150 ER stress, activation of the UPR, and increased levels of acrolein-protein adducts occur in ECs in the small vessels in the lungs of COPD patients.149,151 Thus, ER stress induced by the effects of circulating components of CS or CS-induced increases in oxidative stress levels in small vessels leads to an increased unfolded protein response in ECs that likely contributes to endothelial dysfunction and injury, alveolar septal cell apoptosis, and emphysema development and possibly co-morbidities in human COPD patients (Fig. 2).
All of the above-mentioned pathways have been linked to endothelial dysfunction and injury in human smokers, COPD patients, and/or animal models of COPD. However, it remains unclear which pathways are the most important ones in the pathogenesis of different COPD phenotypes and which should be targeted to improve endothelial integrity in COPD patients. Additional research is needed to address these questions.
Therapeutic approaches targeting endothelial dysfunction in COPD
Progress in developing novel disease-modifying therapies for COPD has lagged far behind that for other chronic lung diseases, despite the enormous healthcare burden associated with COPD. However, endothelial injury represents a potential new therapeutic area for COPD, as therapies exist to treat endothelial dysfunction and disease progression in diseases other than COPD including iPAH, diabetes mellitus, and systemic arterial hypertension.
Drugs targeting vasoactive mediators: Drugs that correct the imbalance between vasoconstrictor and vasodilator mediators have potential to improve endothelial dysfunction and injury and limit the progression of PAH, and possibly other vascular phenotypes, and emphysema in COPD patients. Endothelin receptor antagonists and prostanoids are effective treatments for iPAH,152,153 and also reduce the endothelial dysfunction that occurs in patients with diabetes mellitus that have MAB, 154 systemic sclerosis, 155 and ischemia-reperfusion injury. 156 Some of these iPAH drugs have been tested in COPD patients with PAH, animal models of COPD, and/or cell culture models of COPD.
Endothelin receptor antagonists, prostacyclin analogs, and nitric oxide: Retrospective analyses have been conducted on COPD patients with PAH receiving various iPAH therapies targeting vaso-active mediators (including prostacyclin analogs and endothelin receptor antagonists) via compassionate use programs. These studies report that iPAH therapies, when used alone or in combination, improved clinical symptoms and pulmonary hemodynamic parameters.157,158 Although these therapies led to worsening of the PaO2, this was not clinically-significant and did not lead to specific PAH therapy withdrawal in any patient.157,158
A double-blind, placebo-controlled 12-week clinical trial of oral bosentan (a dual inhibitor of ETA and ETB) was conducted in 30 patients with severe or very severe COPD who developed PAH only during exercise. 159 Bosentan therapy did not improve exercise capacity, lung function, PAP, or maximal oxygen uptake. Bosentan-treated patients had a decrease in PaO2, an increased alveolar-arterial gradient, and a deterioration in quality-of-life scores. 159 Clinical trials of selective endothelin A receptor antagonists have not yet been conducted on COPD patients. Fundamental differences between iPAH and COPD-related PAH may explain why non-selective endothelin receptor antagonists have not consistently been shown to have efficacy in reducing PAH in COPD patients. For example, iPAH is characterized by progressive pulmonary vascular remodeling, right heart failure with elevated filling pressures, and low cardiac output. 160 In contrast, pulmonary vascular remodeling in COPD-related PAH usually progresses very slowly and rarely causes low output right ventricular failure. 161
Pulsed inhaled NO and O2 therapy versus O2 therapy alone over three months were tested in a randomized clinical trial of 40 COPD patients with secondary PAH. 162 Inhaled NO therapy reduced PAPs and PVR, and increased exercise tolerance without decreasing arterial oxygenation. 162 NO donors that increase tissue NO levels are being developed as novel therapies for other diseases.163,164 A long-lasting albumin-based NO donor (S-nitrosated human albumin) was tested in murine models of chronic renal disease and was shown to have efficacy in reducing renal fibrosis, inflammation, oxidative stress, and TGF-β levels. 165 Thus, NO donor approaches could be tested as disease-modifying therapies for limiting the progression of COPD phenotypes in which endothelial dysfunction plays a significant role.
Infusion of the prostacyclin analog, Iloprost, reduced endothelial injury following ST-segment elevation myocardial infarction, 166 but Iloprost has not been tested in clinical trials of COPD patients. However, prostacyclin analogs have therapeutic efficacy in animal models of emphysema, and this is mediated, in part, by their effects on endothelial dysfunction and injury. For example, delivering Iloprost by the intranasal route reduced pulmonary and systemic inflammation, lung oxidative stress levels, and remodeling around small pulmonary vessels in a murine model of COPD. 167 Treating CS-exposed rats with the synthetic prostacyclin analog, Beraprost sodium, reduced emphysema development and this was associated with reduced alveolar septal cell apoptosis, pulmonary inflammation, and lung anti-oxidant levels. 168 Treating human EC cultures with Iloprost or Beraprost sodium inhibited CSE-induced apoptosis of ECs in vitro.65,129
Based upon these data, randomized, placebo-controlled clinical trials of iPAH therapies should be conducted on carefully selected COPD patients with and without PAH to determine whether they have therapeutic efficacy not only in ameliorating secondary PAH, but also on other COPD phenotypes linked to the vascular phenotype including emphysema and chronic renal injury.
Drugs targeting oxidative stress
Endogenous anti-oxidants: Clinical trials of supplementation with exogenous anti-oxidant (including glutathione precursors, N-acetyl cysteine, vitamins C and E, and β-carotene) in COPD patients have yielded mixed results.80,169,170 For example, N-acetyl cysteine improved symptoms of chronic bronchitis, decreased airway bacterial colonization, and reduced the rate of re-hospitalization of COPD patients by 30%. 171 In another study, treating COPD patients with N-acetyl cysteine for two years slowed the rate of decline in FEV1. 172 However, the effects of anti-oxidant therapies on endothelial dysfunction and the vascular phenotypes in COPD patients have not been examined.
Increasing endogenous anti-oxidant pathways to improve endothelial dysfunction has recently been explored in animal models of COPD. Treating WT mice that were chronically exposed to CS with a potent activator of NRF2 decreased lung oxidative stress levels, alveolar septal cell apoptosis, emphysema development, and PAH. 173 NRF2 activation in ECs improves endothelial dysfunction in murine models of diabetes mellitus and atherosclerosis.174,175 Sulforaphane, a derivative of cruciferous vegetables, stimulates NRF2 activity in vitro and in vivo to increase anti-oxidant gene expression.176,177 The results of a phase 2, randomized, placebo-controlled trial evaluating the effects of daily dosing with oral sulforaphane for four weeks on NRF2 target gene expression in patients with COPD were recently reported. This study showed that sulforaphane therapy neither increased NRF2 target gene expression nor altered levels of anti-oxidants or markers of inflammation. 178 However, endothelial dysfunction was not evaluated in this study.
Drugs targeting the renin-angiotensin system: The development of MAB in patients with diabetes and systemic arterial hypertension is a clinical indication for initiating therapy with angiotensin-converting enzyme inhibitors (ACEi) and angiotensin receptor antagonists (ARBs). 179 The use of ACEi and ARBs in diabetes mellitus and hypertension improves not only endothelial function (as assessed by MAB), but also reduces the progression of chronic renal injury in these patients. 180 ACEi were developed to inhibit activation of the renal-angiotensin-aldosterone pathway by inhibiting ACE-I mediated conversion of angiotensin-I into active angiotensin-II (Fig. 3). However, ACEi have also antioxidant properties 181 by reducing the production of reactive carbonyl precursors for AGEs, chelating transition metals, and inhibiting various oxidative steps thus reducing AGEs generation. 181 In humans with diabetes mellitus, and animal models of diabetes mellitus, ACEi therapy attenuated renal injury and MAB reducing endothelial injury by reducing oxidative stress to inhibit activation of RAGE.182–185
A recent study
29
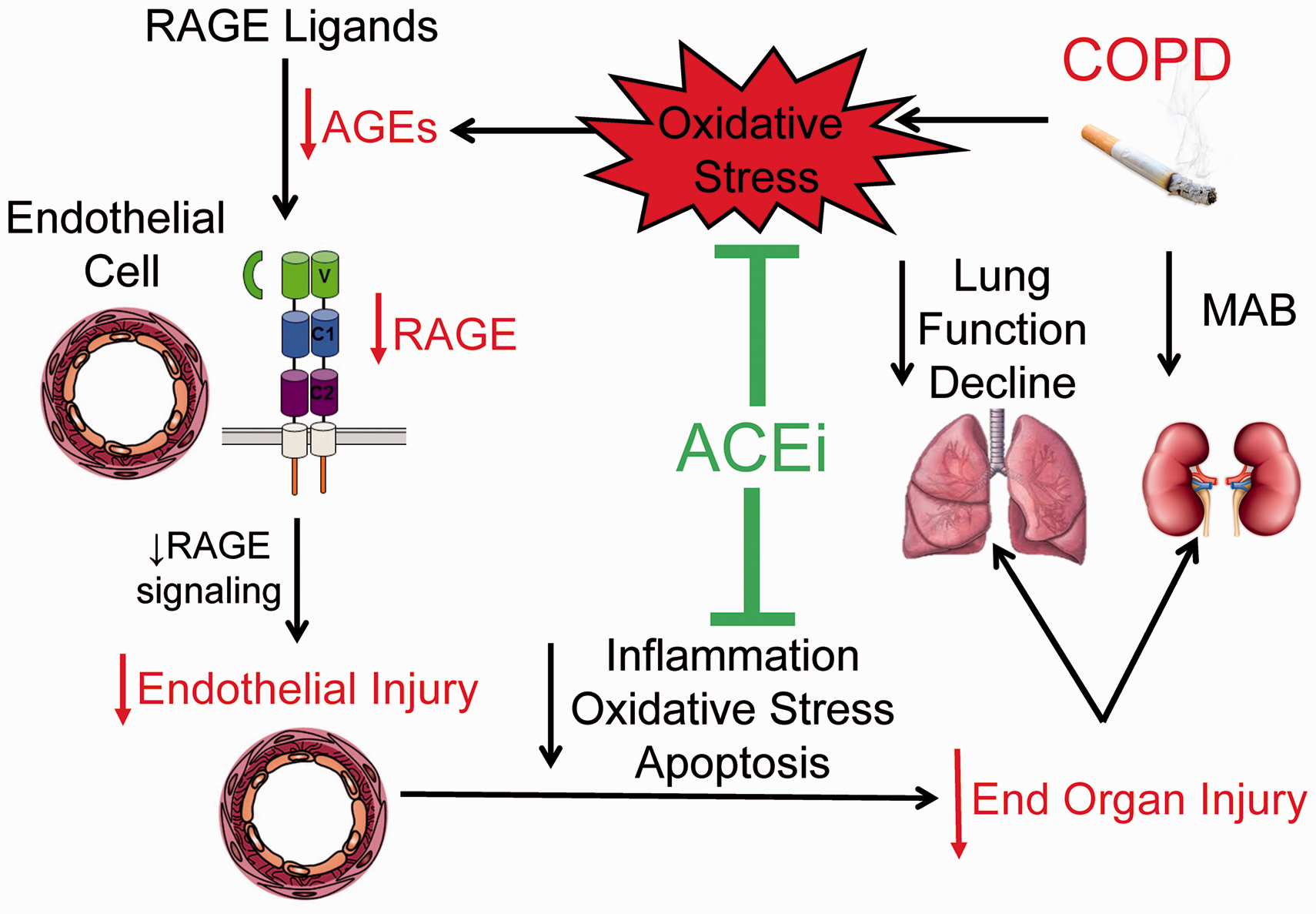
reported that enalapril reduced CS-induced COPD-like lung disease and chronic renal injury in CS-exposed mice by reducing pulmonary and renal oxidative stress levels, leading to reduced AGEs-RAGE signaling in pulmonary and renal ECs, leading in turn to reduced progression of pulmonary emphysema, small airway disease, and chronic renal injury (Fig. 4). Surprisingly, angiotensin-II levels were reduced in serum samples and lung homogenates from CS-exposed mice, and enalapril therapy increased angiotensin-II levels in these samples.
29
The increased angiotensin-II levels that were detected in enalapril-treated and CS-exposed mice likely reflected improved pulmonary EC viability and increased capacity to synthesize ACE-I in enalapril-treated mice as these cells are the major source of this enzyme.
186
ACEi switch off oxidative stress-AGEs-RAGE signaling in lungs and kidneys to limit end-organ injury in CS-exposed mice. ACEi are known to have anti-oxidant properties. Treating CS-exposed mice with an ACEi reduces oxidative stress levels in the lungs and kidneys, which reduces the AGEs burden in the lungs and kidneys. This in turn, reduces RAGE activation and expression. Reduced RAGE signaling reduces pulmonary and renal endothelial dysfunction and injury, and thereby limits disease progression in the lungs and kidneys of CS-exposed mice.
29
Prospective, randomized, placebo-controlled clinical trials of ACEi in COPD patients have not been conducted. Nevertheless, results of several observational and retrospective studies of COPD patients and smokers suggest that ACEi may have efficacy as novel disease-modifying therapies for COPD patients. These studies have linked ACEi use to a trend towards lower mortality rates in patients with COPD on oxygen therapy. 187 Treatment with ACEi for indications other than COPD was associated with decreased mortality in COPD patients hospitalized for COPD exacerbations 188 and a reduced risk of pneumonia in COPD patients. 189 Peterson et al. reported that baseline use of ACEi in smokers without COPD was associated with a slower rate of FEV1 decline and a reduction in incident COPD. 190 In the latter study, this protective effect on FEV1 decline in smokers was not observed in individuals taking ARB, beta blockers, calcium channel blockers, statins, insulin, or oral hypoglycemic agents. In a time-matched nested case-control study of two population-based retrospective cohorts, it was shown that both ACEi and ARB, especially when used in combination, reduced COPD hospitalization and total mortality. 191
ARBs also improve endothelial dysfunction and reduce tissue oxidative stress levels in murine models of diabetes. 185 The efficacy of the ARB losartan, was tested in CS-exposed mice and showed that the ARB reduced TGF-β signaling, lung oxidative stress levels, alveolar septal cell apoptosis, pulmonary inflammation, emphysema development, and airway fibrosis. 146 The authors postulated that the protective effects of losartan in CS-exposed lungs were due to losartan’s effects on inhibiting TGF-β signaling in the lung. In another study of mice with pancreatic elastase-induced emphysema, another ARB (irbesartan), decreased emphysema development and improved lung compliance and running distance in mice when compared with vehicle-treated control mice. 192 However, a limitation of these studies was that the effects of ARB therapy on EC function and oxidative stress levels in the lungs (and other organs) were not studied.
There has been only one randomized, placebo-controlled clinical trial evaluating the therapeutic efficacy of an ARB in COPD patients. In this trial, four months of treatment with irbesartan was tested in a cohort of 60 COPD patients selected for having a FEV1 < 50% of predicted, and without obvious cardiovascular disease that would have necessitated the administration of an ACEi or ARB. 193 Irbesartan did not have a significant effect on spirometry results or respiratory muscle strength (the latter was the primary endpoint of the study), but reduced total lung capacity and the mean hematocrit values, raising the possibility that ARBs have beneficial effects in COPD patients. However, it is not clear whether these COPD patients had endothelial dysfunction or injury, and additional trials of ARBs in larger cohorts of COPD patients with the vascular phenotype are needed.
In these prior studies, the effects of ACEi and ARBs on different COPD phenotypes, including components of the vascular COPD phenotype, were not examined. However, a more recent study linked ACEi use to slowing of the progression of emphysema. Parikh et al. assessed ACEi use in the Multi Ethnic Study of Atherosclerosis (MESA) lung study of individuals in the general population aged 45–84 years who had no clinical evidence of cardiovascular disease. 194 The study participants were followed over a ten-year observation time-frame and percent emphysema was measured using high-resolution computed tomography (HRCT) scans and related to medication use. Baseline use of an ACEi or ARB, especially when prescribed at the full doses, was protective against the progression of HRCT scan-determined emphysema measured over the ten-year observation time-frame, especially among former smokers. The results of Parikh et al. are consistent with the finding that enalapril prevented the progression of pulmonary emphysema in CS-exposed mice. 29 Parikh et al. suggested that ARBs and ACEi inhibit the renin-angiotensin system to antagonize TGF-β signaling thereby reducing the progression of airspace enlargement. 194 However, endothelial dysfunction, and levels of oxidative-stress, AGEs, and RAGE were not measured by Parikh et al.
It has been suggested that among different COPD phenotypes, patients with the “vascular COPD phenotype” characterized by endothelial dysfunction (detectable by measuring MAB) might benefit the most from ACEi therapy treatment with respect to limiting progression of chronic lung pathologies and loss of lung function.66,195 However, no study to date has examined the efficacy of either ACEi or ARBs in COPD patients with the vascular phenotype which may represent a significant proportion (∼ 24%) of patients with stable COPD. 47 A major advantage in targeting endothelial dysfunction or injury in COPD patients with ACEi or ARBs is that it may limit the progression of not only emphysema, but also some common COPD co-morbidities, including chronic renal injury, cardiovascular disease, PAH, and possibly also muscle wasting, and osteoporosis. It is noteworthy that ACEi and ARBs are off-patent and have been tested in large numbers of individuals and have well established safety profiles. Thus, repurposing these cardiovascular drugs to limit disease progression in COPD obviates the need for expensive new drug development. The evidence to date provides a strong rationale for large randomized clinical trials testing the efficacy of ACEi and ARB in limiting the progression of COPD in patients with the vascular phenotype.
Conclusions and future directions
Endothelial injury is present in a significant subset of COPD patients (∼ 24%), as assessed by the presence of MAB. 47 Data from human and animal studies indicate that endothelial dysfunction and injury contribute not only to the genesis and progression of pulmonary lesions in COPD (especially emphysema development), but may also contribute to some of the common co-morbidities reported in COPD patients including PAH, atherosclerosis, and chronic renal injury. Recent studies link the pathways triggered by oxidative stress (e.g. lipid peroxidation and AGEs-RAGE signaling), reduced expression of anti-oxidant pathways (NRF2), perturbations in the generation of vaso-active mediators within the vasculature, and increased ER stress and the UPR to CS-induced endothelial injury and end-organ damage. Drugs that were developed to inhibit the renin-angiotensin-aldosterone system (ACEi and ARB) are widely used to treat other diseases characterized by endothelial injury (such as diabetes mellitus) to limit endothelial injury and the progression of end-organ injury. ACEi and ARB limit COPD progression in CS-exposed mice, and this may be linked, in part, to their capacity to reduce activation of the oxidative stress-AGEs-RAGE pathway in CS-exposed ECs. Observational and retrospective human studies support the notion that ACEi and ARB may have efficacy in limiting COPD progression in humans. However, only one clinical trial of an ARB in COPD patients has been conducted, but endothelial dysfunction was neither a recruitment criterion, nor a study endpoint. Targeting the perturbation in vascular levels of vasoactive mediators with endothelin-1 receptor antagonists and prostacyclin analogs has efficacy in patients with iPAH, and these therapies may have potential to reduce the endothelial dysfunction and injury that contributes not only to PAH, but also to other components of the vascular COPD phenotype (emphysema, chronic renal injury, and atherosclerosis).
Future studies should also assess the extent to which endothelial injury and dysfunction underlie COPD co-morbidities other than PAH, atherosclerosis, and chronic renal injury. Prospective, randomized, placebo-controlled clinical trials are needed to assess whether COPD patients with the “vascular COPD phenotype” characterized by generalized endothelial dysfunction (detected by measuring MAB) might benefit the most from ACEi or ARB therapy with respect to limiting progression of endothelial injury, loss of lung function, and injury to other organs. Future studies should determine whether routine screening of COPD patients for MAB has utility in the phenotyping and/or management of COPD patients. MAB should also be prospectively evaluated as a non-invasive and inexpensive biomarker to monitor responses to new therapies that target the endothelium.
2017 Grover Conference Series
This review article is part of the 2017 Grover Conference Series. The American Thoracic Society and the conference organizing committee gratefully acknowledge the educational grants provided for the support of this conference by Actelion Pharmaceuticals US, Inc., Gilead Sciences, Inc., and United Therapeutics Corporation. Additionally, the American Thoracic Society is grateful for the support of the Grover Conference by the American Heart Association, the Cardiovascular Medical Research and Education Fund, and the National Institutes of Health.
