Abstract
Objective
This study examined whether the immunomodulator fingolimod (FTY720) could alleviate renal ischemia/reperfusion (I/R)-induced lung injury and explored the potential mechanisms.
Methods
Renal I/R was established in a rat model, and FTY720 (0.5, 1, or 2 mg/kg) was injected intraperitoneally after 15 minutes of ischemia. Pro-inflammatory cytokine levels, oxidative stress, apoptosis, and the mRNA expression of the sphingosine-1-phosphate (S1P)-related signaling pathway genes sphingosine kinase-1 (SphK1) and sphingosine kinase-2 were analyzed in lung tissue.
Results
Increased pro-inflammatory cytokine levels; decreased total superoxide dismutase, catalase, and glutathione peroxidase levels; increased apoptosis; and increased S1P lyase and SphK1 expression were observed following renal I/R. FTY720 reversed renal I/R-induced changes and effectively attenuated lung injury.
Conclusion
FTY720 protected against acute lung injury in rats subjected to renal I/R by decreasing pulmonary inflammation and apoptosis, increasing oxidative stress, and modulating S1P metabolism.
Keywords
Introduction
Renal ischemia/reperfusion (I/R) injury is most commonly associated with acute renal impairment, kidney transplantation, and/or extracorporeal shock wave lithotripsy. 1 , 2 Additionally, patients often present with distant organ damage following renal I/R. 3 This effect is partly mediated by the diminished exclusion of inflammatory cytokine release. Acute lung injury is the one of the major causes of death in patients with renal I/R injury.4,5
Multiple pathophysiological processes are involved in renal I/R-induced lung injury. Elevated levels of inflammatory mediators cause disturbances in microcirculation, increased capillary permeability, destruction of cell membranes and lysosomes, cell edema, and tissue damage leading to lung injury. 6 To date, there are no preventative therapies targeting lung injury caused by renal I/R. Furthermore, treatment strategies that maintain oxygenation capacity and pulmonary mechanics following renal I/R are limited.
Fingolimod (FTY720), a sphingosine-1-phosphate (S1P) receptor agonist, is a promising immunomodulator that could interfere with the pathogenesis of I/R-induced injury.7–9 FTY720 is a phosphorylation target for sphingosine kinase-2 (SphK2) that binds to S1P receptors 1 to 5. 10 The S1P signaling pathway is involved in various biological functions, including cell proliferation and survival, extracellular matrix assembly, endothelial cell barrier integrity, and immune cell trafficking.11–13 In animal models of lung injury induced by hindlimb I/R; lipopolysaccharide, paraquat, or bleomycin exposure; or lung transplantation, FTY720 treatment significantly decreased lung leak, inflammation, and endothelial cell apoptosis. 7 ,14–16 However, whether FTY720 has similar beneficial effects on renal I/R-induced lung injury remains unknown.
In the current study, we establish a rat model of lung injury induced by renal I/R and explored the impact of FTY720 treatment on key pathophysiological processes including inflammatory responses, oxidative stress, apoptosis, and S1P-related signaling.
Materials and methods
Experimental animals
Adult Sprague–Dawley rats (200–300 g) were obtained from the laboratory animal center of North Sichuan Medical College. Rats were housed individually in cages. All rats were housed under a 12-hour/12-hour light/dark cycle at 20 to 24°C with free access to water and food. The protocol of the study was approved by the Ethics Review Board for Animal Studies of North Sichuan Medical College (NSMC2018 45). The study complied with the guidelines for laboratory animal experiment of the National Institutes of Health.
Experimental design and renal I/R model
Twenty-five rats were assigned randomly into five groups (n = 5 per group) as follows: sham-operated group (Sham group), renal I/R group (RIR group), renal I/R + FTY720 (0.5 mg/kg) group (FTY0.5 group), renal I/R + FTY720 (1 mg/kg) group (FTY1 group), and renal I/R + FTY720 (2 mg/kg) group (FTY2 group). The rat model of renal I/R was established successfully according to a previously described protocol. 17 Briefly, after an injection of sodium pentobarbital (50 mg/kg, i.p.), a midline laparotomy was performed, and the right kidney was removed. Then, a non-traumatic microvascular clamp was applied to occlude the left renal hilum and induce renal ischemia. Reperfusion was allowed after the clip was held for 45 minutes. The same laparotomy procedure was performed in the Sham group without removing the right kidney and clipping the left renal pedicle. Rats in the FTY0.5, FTY1, and FTY2 groups received peritoneal injections of 0.5, 1, and 2 mg/kg FTY720 (Cayman Chemical, Ann Arbor, MI, USA), respectively, 30 minutes before reperfusion, whereas animals in the Sham and RIR groups received normal saline. The wound was perfused with bupivacaine (0.25%) and closed with sterile suture. Rectal temperature was maintained at 36 to 37°C during surgery. All rats were housed individually after recovering from anesthesia. Finally, rats were euthanized with pentobarbital sodium (60 mg/kg i.p.) 6 hours post-reperfusion. Left renal tissue, bronchoalveolar lavage fluid (BALF), and lung tissue were harvested for examination.
Histopathologic examination of kidney
Renal samples were fixed in 10% neutral-buffered formalin for 24 hours, embedded in paraffin, and sectioned into 5-µm-thick slices. Subsequently, hematoxylin–eosin (H&E) staining was performed using the standard protocol, and the pathological alterations were assessed by a pathologist using a light microscope (Olympus, Tokyo, Japan) in a blinded manner. 4 Renal tissue was histopathologically examined for the degree of Bowman’s space enlargement, tubular cell necrosis, cell exfoliation in the lumen, vascular congestion, and intratubular cast formation. The level of each manifestation was graded according to the extent of change as follows: 0, no change; 1, <20% change; 2, 20% to 40% change; 3, 41% to 60% change; 4, 61% to 80% change; and 5, >80% change. The sum of all numerical scores in each group was taken as the total histopathological score.
Analysis of renal function
Serum cystatin C (Cys C), serum creatinine (Scr), and blood urea nitrogen (BUN) levels were measured using an autoanalyzer (Siemens, Munich, Germany).
Measurement of inflammatory mediator levels in BALF and the lungs
BALF was collected by perfusing the lungs with 4 mL of PBS through a tracheal cannula placed in each rat under anesthesia. The collection procedure was repeated three times. Briefly, the chest was opened after sacrifice. The trachea was isolated via blunt dissection after median sternotomy. A small-caliber tube was inserted into the airway. PBS was slowly infused bilaterally into the lungs. Then, BALF was withdrawn into the tubes. The percent fluid recovery exceeded 80%. BALF samples were centrifuged (1500 × g) for 10 min at 4°C. The concentrations of tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and IL-6 in BALF and lung tissue were measured using commercial enzyme-linked immunosorbent assay kits (Shanghai Zcibio Biotechnology Ltd., Shanghai, China).
Measurement of oxidative stress in the lungs
Lung tissue was harvested, homogenized, and centrifuged. Supernatant was collected, and the levels of total superoxide dismutase (T-SOD), glutathione peroxidase (GSH-PX), catalase (CAT), and malondialdehyde (MDA) were determined using corresponding diagnostic kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer s instructions.
Measurement of neutrophil infiltration in the lungs
A formalin-fixed left lower lung lobe tissue sample was prepared from each animal. Samples were dehydrated and embedded in Paraplast (Leica Microsystems GmbH, Wetzlar, Germany). The tissues were cut into 4-μm-thick sections and stained with H&E. Lung neutrophil infiltration was examined by a pathologist who was blinded to the group allocation under a light microscope.
Assessment of cell apoptosis
Lung tissues were fixed with 4% paraformaldehyde, embedded in paraffin, and sectioned at 5 μm. After deparaffinization and rehydration, the sections were treated with 10 μg/mL protease K at 37°C for 25 minutes. Fresh terminal deoxynucleotidyl transferase-mediated dUDP nick-end labeling (TUNEL) reaction mixture was added onto the samples, which were then incubated at 37°C for 60 minutes. Apoptosis was measured using a TUNEL assay kit (Roche, Basel, Switzerland).
Measurement of mRNA expression of S1P-related signaling pathway genes
The right lower lung lobe tissue samples of each animal were homogenized. Total RNA was extracted using TRIzol reagent (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions. For real-time PCR, the total RNA (1 μg) from each sample was resuspended in 25 μL of reaction buffer, and cDNA was reverse-transcribed using a synthesis kit (Takara Biotechnology Co. Ltd., Dalian, China). The housekeeping gene β-actin was used for data normalization. Relative mRNA expression was measured using the 2−ΔΔCT method. The primers used for real-time PCR were as follows: S1P lyase (S1PL) forward primer, 5′-ATT GGC AAG GTG GCA TCT ACG CAT AT-3′; S1PL reverse primer, 5′-CCA GTT CTG ACT TGA GGA AGC GAG CA-3′; SphK1 forward primer, 5′-TCG TTC TGG TGC TGG TGC TGC TTC A-3′; sphingosine kinase-1 (SphK1) reverse primer, 5′-GGA AGG CAA CCA CTG GCA CAT GAA CC-3′; SphK2 forward primer, 5′-GGG TTG AGC CTG AGT GAG TGG GAA G-3′; SphK2 reverse primer, 5′-CGG CAG AGA AGA AGC GAG CAG TTG A-3′; β-actin forward primer, 5′-GAA GAT CAA GAT CAT TGC TCC T-3′; and β-actin reverse primer, 5′-TAC TCC TGC TTG CTG ATC CA-3′.
Statistical analysis
All statistical analyses were performed using SPSS 23.0 (IBM, Armonk, NY, USA). Continuous variables are presented as the mean ± SD. Statistical significance was evaluated using analysis of variance followed by Tukey’s multiple comparison test. P < 0.05 was considered significant.
Results
Effects of FTY720 on renal injury
Renal injury was assessed via histopathologic examination and assessments of renal function. Rats subjected to renal I/R exhibited significant structural damage compared with rats in the Sham group (Figure 1d and e) and Cys C (0.50 ± 0.02 mg/L vs. 0.26 ± 0.03 mg/L), Scr (60.75 ± 6.08 µmol/L vs. 39.33 ± 10.02 µmol/L), and BUN levels (10.90 ± 1.96 mmol/L vs. 6.94 ± 0.85 mmol/L) were significantly higher in the RIR group than in the Sham group (all P < 0.05; Figure 1a–c). However, treatment with 0.5 mg/kg FTY720 significantly decreased Cys C and Scr levels (0.27 ± 0.06 mg/L and 42.00 ± 4.47 µmol/L, respectively; both P < 0.05) and attenuated pathological injuries (Figure 1a, b, and f).

Serum levels of (a) Cys C), (b) Scr, and (c) BUN in the Sham, RIR, FTY0.5, FTY1, and FTY2 groups. Representative microscopic findings of the left kidney (d–h) after renal I/R in the Sham, RIR, FTY0.5, FTY1, and FTY2 groups, respectively (hematoxylin and eosin, ×200 magnification), including the enlargement of Bowman’s space (green arrow), necrosis and exfoliation of proximal tubular cells in the lumen (black arrow), dilated renal tubules and flattened cells (red arrow), and significantly reduced red blood cell counts in glomerular capillaries (blue arrow). (i) Histological injury scores for left kidney tissue. Data are presented as the mean ± SD. #P < 0.05 vs. the Sham group; *P < 0.05 vs. the RIR group; ##P < 0.01 vs. the Sham group; **P < 0.01 vs. the RIR group.
Effects of FTY720 on pro-inflammatory cytokine production in BALF and lung tissue
Significantly higher IL-6 (27.66 ± 1.75 pg/mL vs. 14.17 ± 0.82 pg/mL), IL-1β (12.75 ± 0.52 pg/mL vs. 10.85 ± 0.91 pg/mL), and TNF-α levels (110.93 ± 5.48 pg/mL vs. 93.19 ± 3.65 pg/mL) were observed in BALF from the RIR group than in BALF from the Sham group (all P < 0.01; Figure 2a–c). However, IL-6, IL-1β, and TNF-α levels were significantly decreased in the FTY0.5 group (16.77 ± 1.07, 11.56 ± 0.38, and 94.65 ± 1.82 pg/mL, respectively, all P < 0.05; Figure 2a–c). Similarly, the RIR group displayed significantly higher IL-6 (39.22 ± 2.98 pg/mL vs. 21.51 ± 1.22 pg/mL) IL-1β (13.45 ± 0.55 pg/mL vs. 11.71 ± 0.22 pg/mL), and TNF-α levels (103.43 ± 6.23 pg/mL vs. 89.30 ± 5.65 pg/mL) in lung tissue than the Sham group (all P < 0.01; Figure 3a–c). By contrast, FTY720 at 0.5 and 1 mg/kg significantly suppressed the elevated levels of IL-6 (31.02 ± 1.94 and 29.64 ± 1.36 pg/mL, respectively), IL-1β (11.95 ± 0.23 and 12.07 ± 0.15 pg/mL, respectively), and TNF-α (92.55 ± 3.38 and 92.01 ± 3.94 pg/mL, respectively; all P < 0.05; Figure 3a–c).

Bronchoalveolar lavage fluid levels of IL-6 (a), IL-1β (b), and TNF-α (c) in the Sham, RIR, FTY0.5, FTY1, and FTY2 groups. Data are presented as means ± SD. ##P < 0.01 vs. the Sham group; **P < 0.01 vs. the RIR group; *P < 0.05 vs. the RIR.

Lung levels of IL-6 (a), IL-1β (b), and TNF-α (c) in the Sham, RIR, FTY0.5, FTY1, and FTY2 groups. Data are presented as the mean ± SD. ##P < 0.01 vs. the Sham group; **P < 0.01 vs. the RIR group; *P < 0.05 vs. the RIR group.
Effects of FTY720 on oxidative stress in the lungs
The RIR group exhibited significantly lower T-SOD (46.85 ± 2.11 U/mg protein vs. 52.95 ± 0.71 U/mg protein), CAT (6.77 ± 0.33 U/mg protein vs. 8.85 ± 0.41 U/mg protein), and GSH-PX levels (81.29 ± 2.99 U/mg protein vs. 119.47 ± 7.40 U/mg protein) and notably higher MDA levels (0.92 ± 0.04 U/mg protein vs. 0.75 ± 0.09 U/mg protein) in lung tissue than the Sham group (all P < 0.01; Figure 4a–d). These changes were reversed in the FTY0.5 group (T-SOD, 49.39 ± 1.83 U/mg protein; CAT, 7.73 ± 0.72 U/mg protein; GSH-PX, 105.68 ± 7.18 U/mg protein; MDA, 0.84 ± 0.04 U/mg protein; all P < 0.05; Figure 4a–d)
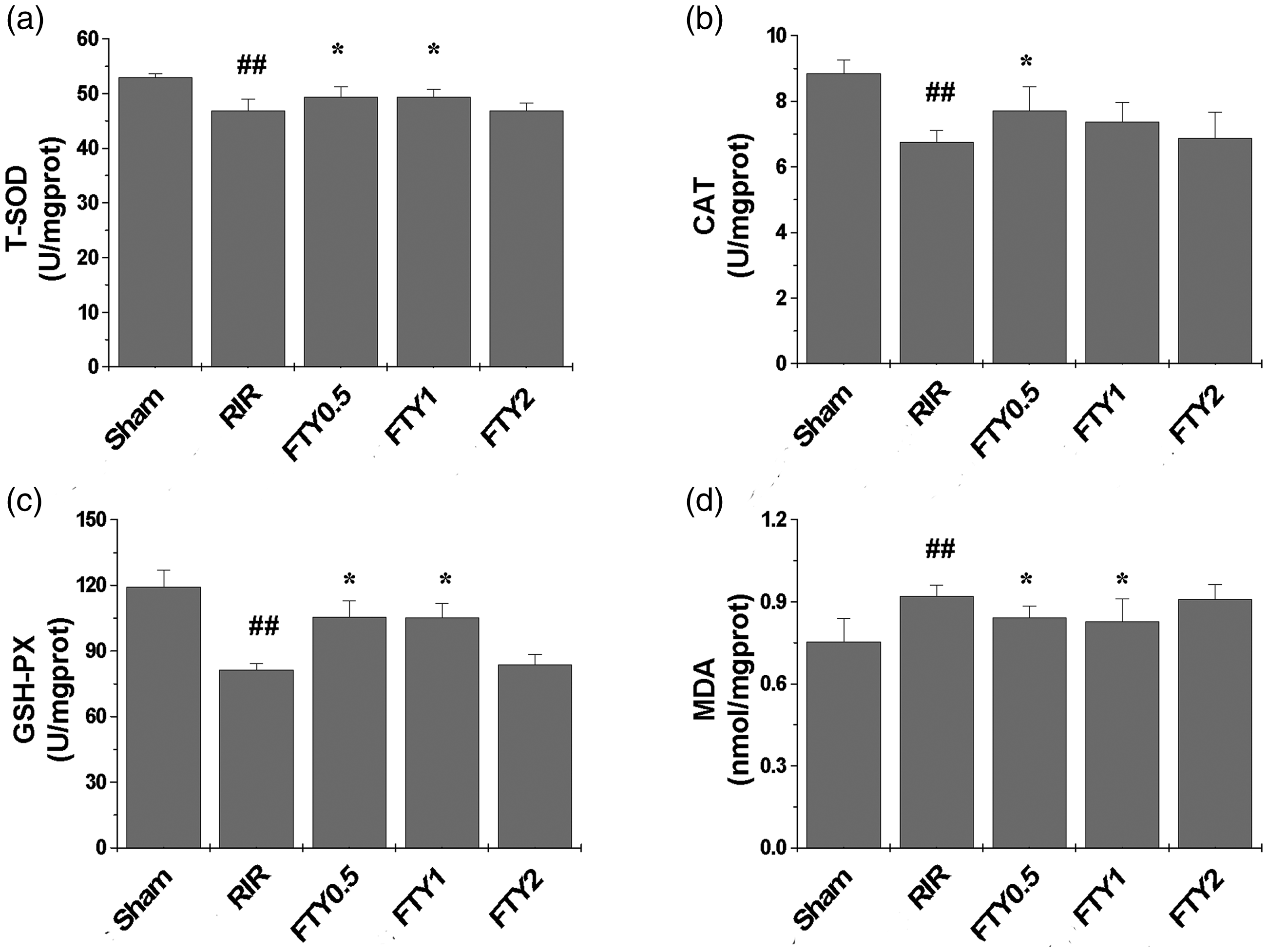
Lung levels of T-SOD (a), CAT (b), GSH-PX (c), and MDA (d) in the Sham, RIR, FTY0.5, FTY1, and FTY2 groups. Data are presented as the mean ± SD. ##P < 0.01 vs. the Sham group; *P < 0.05 vs. the RIR group.
Effects of FTY720 on apoptosis in the lungs
The extent of apoptosis was significantly increased in the RIR group compared with the findings in the Sham group (apoptotic area: 10.89 ± 0.46 vs. 5.83 ± 0.66; all P < 0.01; Figure 5a, b, and f). However, this increase in apoptosis was reversed in all rats receiving FTY720 (apoptotic area: 6.06 ± 0.93, 4.45 ± 0.58, and 4.56 ± 1.15 in the FTY0.5, FTY1, and FTY2 groups, respectively, all P < 0.01; Figure 5c–f).

Apoptosis of lung cells in the (a) Sham, (b) RIR, (c) FTY0.5, (d) FTY1, and (e) FTY2 groups (×400 magnification). (f) The positive apoptotic area. Data are presented as the mean ± SD. ##P < 0.01 vs. the Sham group; **P < 0.01 vs. the RIR group.
Effects of FTY720 on the mRNA expression of S1P-related signaling pathway genes
The mRNA expression of S1PL (2.94 ± 0.83 vs. 1.01 ± 0.13), SphK1 (4.01 ± 0.73 vs. 1.02 ± 0.21), and SphK2 (2.64 ± 0.84 vs. 1.11 ± 0.43) was significantly higher in the RIR group than in the Sham group (all P < 0.05; Figure 6a–c). However, in the FTY0.5 and FTY1 groups, S1PL (1.36 ± 0.27 and 1.58 ± 0.52, respectively), SphK1 (1.50 ± 0.29 and 1.88 ± 0.31, respectively) and SphK2 mRNA expression (1.24 ± 0.30 and 1.43 ± 0.44, respectively) was significantly downregulated versus that in the Sham group (all P < 0.05; Figure 6a–c).

mRNA expression of S1PL (a), SphK1 (b), and SphK2 (c) in lung tissue in the Sham, RIR, FTY0.5, FTY1, and FTY2 groups. Data are presented as the mean ± SD. #P < 0.05 vs. the Sham group; *P < 0.05 vs. the RIR group.
Effects of FTY720 on neutrophil infiltration in the lungs
Neutrophil infiltration in the lungs was significantly increased in the RIR group compared with that in the Sham group (9.58 ± 1.36 vs. 6.67 ± 1.03; P < 0.01; Figure 7a, 7b, and 7f). This increase was attenuated in the FTY0.5, FTY1, and FTY2 groups (5.92 ± 0.80, 6.25 ± 1.04, and 6.93 ± 0.83, respectively; P < 0.01; Figure 7c–f), although no significant differences in neutrophil infiltration were identified among these three groups.
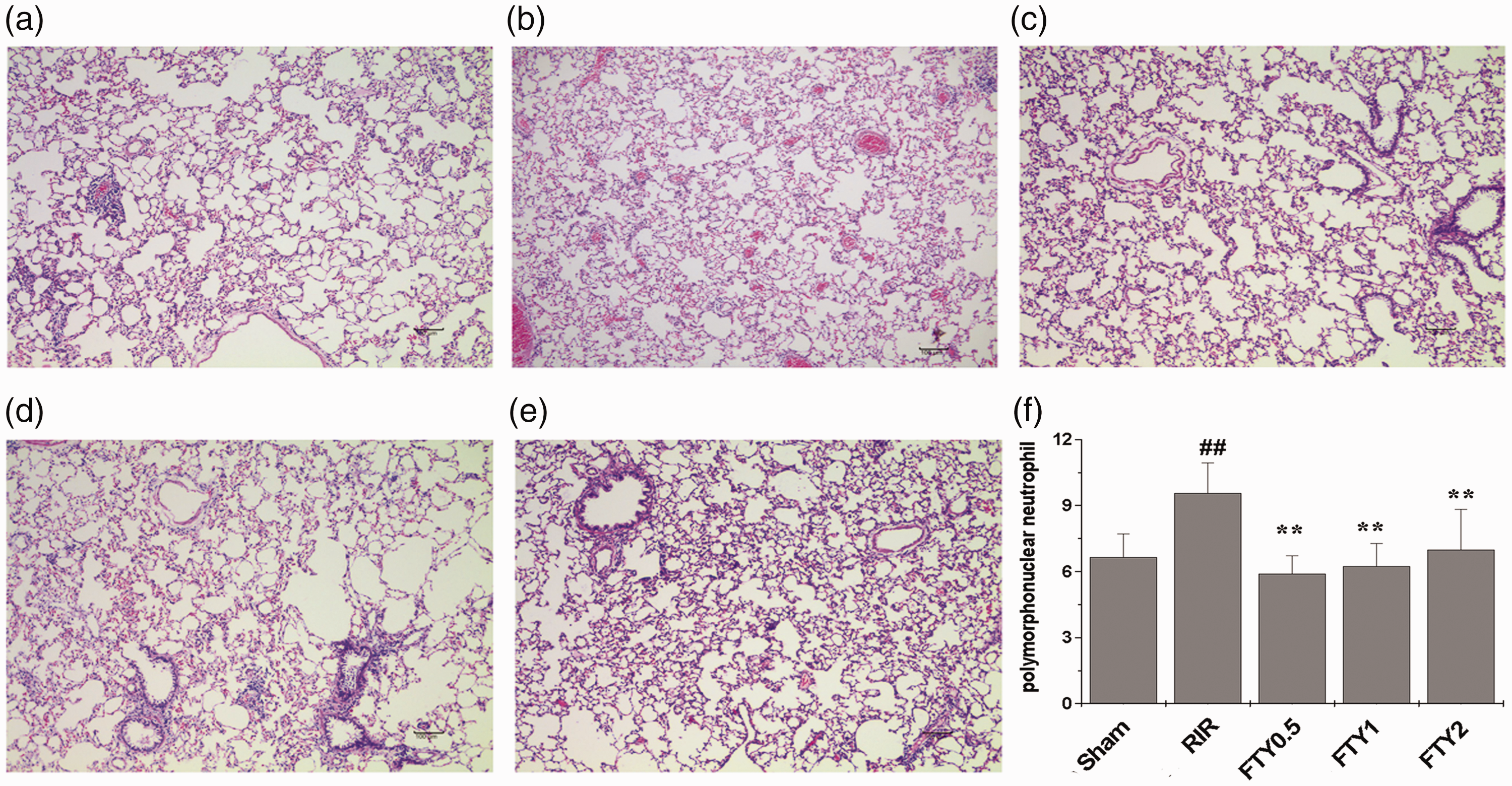
Neutrophil infiltration in lungs in the Sham, RIR, FTY0.5, FTY1, and FTY2 groups (×200 magnification). Data are presented as the mean ± SD. ##P < 0.01 vs. the Sham group; **P < 0.01 vs. the RIR group.
Discussion
The current study evaluated the effect of FTY720 on renal I/R-induced lung injury in a rat model. Our findings confirmed that the activation of multiple responses, including inflammation, oxidative stress, and apoptosis, triggered a sequence of events leading to acute lung injury. FTY720 effectively attenuated lung injury caused by renal I/R by inhibiting inflammatory cytokine production and neutrophil infiltration, suppressing apoptosis and oxidative stress, and modulating S1P metabolism.
Inflammatory cytokines including TNF-α, IL-6, and IL-1β play critical roles in the recruitment and activation of inflammatory cells in response to acute lung injury. 18 TNF-α plays a key role in mitochondrial toxicity, apoptosis, neutrophil activation, and necrotic cell death in I/R-mediated lung injury. 19 IL-6 is regarded as an early biomarker of tissue damage reflecting the accumulation of neutrophils in the lungs. IL-6 and TNF-α further stimulate inflammatory cytokine release, resulting in an inflammatory cascade. Prior research revealed that cytokines and inflammatory pathways are responsible for the pathogenesis of multiple-organ failure following abdominal aortic aneurysm repair. 20 Lower-extremity I/R-induced lung injury stimulated IL-1β, which is also an important mediator in I/R. In our model of renal I/R-induced lung injury, the levels of these inflammatory cytokines were significantly elevated in BALF and lung tissue, supporting the histological findings of alveolar capillary membrane disruption, interstitial edema, and neutrophil infiltration. The present study found that the S1P receptor agonist FTY720 significantly curbed the production of TNF-α, IL-6, and IL-1β in BALF and lung tissues, which was consistent with previous studies on FTY720 in the treatment of lung injury. 15 FTY720 may prevent cytokine-mediated acute inflammatory responses in renal I/R-induced lung injury.
The lungs are vulnerable to oxidative stress because of their unique structure and function. 21 A previous study reported that MDA levels in BALF and lung tissue were significantly elevated after aortic I/R injury. 22 Our results suggested that accumulated MDA contributes to renal I/R-induced lung injury, which is consistent with previous findings. 23 In the present study, decreased MDA levels and increased T-SOD, CAT, and GSH-PX levels were observed after FTY720 treatment, indicating that FTY720 effectively elicits a protective response to oxidative stress.
Apoptosis following I/R is equally important in the development of lung injury. 24 Previous studies on I/R injury during lung transplantation found that the mode of pulmonary epithelial cell death upon reperfusion after a moderate duration (6 or 12 hours) of cold ischemic preservation was dominated by apoptosis. 25 Conversely, apoptosis was clearly observed in lung tissue after 45 minutes of ischemia followed by 6 hours of reperfusion. Previous findings indicated that apoptosis in the alveolar epithelium is evident only after reperfusion, and the degree of apoptosis increases with longer periods of reperfusion, in line with the findings of the current study. 26 The binding of S1P to its receptors may inhibit apoptosis. 27 Therefore, it was not surprising that FTY720 reduced pulmonary cell apoptosis.
It is noteworthy that FTY720 may have therapeutic limitations despite its beneficial effects because it induces the ubiquitination and proteasomal degradation of S1P1, leading to increased endothelial permeability after prolonged exposure. 10 This may explain why higher concentrations failed to enhance the protective effects of FTY720 and potentially exerted detrimental effects on vascular permeability. Indeed, several recent animal studies observed detrimental effects on vascular permeability when FTY720 was applied at higher concentrations and for longer periods, leading to tissue edema and worsening bleomycin-induced lung injury and ventilator-induced lung injury.14,28,29 This experiment indicated that renal I/R caused kidney injury, and this damage was attenuated by FTY720 (0.5 mg/kg). However, higher concentrations of FTY720 (1 and 2 mg/kg) did not aggravate renal damage.
Our study had several limitations. First, FTY720 is one of several S1P receptor agonists, and the effects of other S1P receptor agonists on renal I/R-induced lung injury were not evaluated. Second, FTY720 is clinically administered in an oral form, and the bioavailability of the active agent is affected by first-pass metabolism; therefore, the appropriate FTY720 dosage to elicit protective effects needs to be further evaluated. In addition, in this experimental study, we performed FTY720 pretreatment to ensure a sufficient dose and effect. Posttreatment with the treatment might better mimic the clinical setting. Finally, the long-term effects of FTY720 on lung injury caused by renal I/R remain unknown because of the short follow-up period of this study. Overall, the present study suggests that FTY720 at a low concentration (0.5 mg/kg) alleviates renal I/R-induced lung injury in rats by attenuating inflammation, oxidative stress, and apoptosis and modulating S1P metabolism. These observations indicate that FTY720 may be a promising treatment for acute lung injury after renal I/R.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was funded by Foundation of Education Department of Sichuan Province (No. 17ZA0185) and the Research and Development Program of North Sichuan Medical College (No. CBY15-A-ZD04).
