Abstract
Mortality and morbidity after cardiac arrest remain high due to ischemia/reperfusion (I/R) injury causing multi-organ damages, even after successful return of spontaneous circulation. We previously generated H2O2-activatable antioxidant nanoparticles formulated with copolyoxalate containing vanillyl alcohol (PVAX) to prevent I/R injury. In this study, we examined whether PVAX could effectively reduce organ damages in a rat model of whole-body ischemia/reperfusion injury (WBIR). To induce a cardiac arrest, 70µl/100 g body weight of 1 mmol/l potassium chloride was administered via the jugular venous catheter. The animals in both the vehicle and PVAX-treated groups had similar baseline blood pressure. After 5.5 minutes of cardiac arrest, animals were resuscitated via intravenous epinephrine followed by chest compressions. PVAX or vehicle was injected after the spontaneous recovery of blood pressure was noted, followed by the same dose of second injection 10 minutes later. After 24 hours, multiple organs were harvested for pathological, biochemical, molecular analyses. No significant difference on the restoration of spontaneous circulation was observed between vehicle and PVAX groups. Analysis of organs harvested 24 hours post procedure showed that whole body I/R significantly increased reactive oxygen species (ROS) generation, inflammatory markers, and apoptosis in multiple organs (heart, brain, and kidney). PVAX treatment effectively blocked ROS generation, reduced the elevation of pro-inflammatory cytokines, and decreased apoptosis in these organs. Taken together, our results suggest that PVAX has potent protective effect against WBIR induced multi-organ injury, possibly by blocking ROS-mediated cell damage.
Introduction
Cardiac arrest is a major public health issue and the leading cause of out-of-hospital death worldwide. 1 Survivors of cardiac arrest still experience multiple medical problems related to post resuscitation complications. 1 Thus, much effort has been devoted to prevent or minimize the complications that occur post cardiac arrest to vital organs. 2,3 However, it remains to be a major challenge in the management of post cardiac arrest patients. 2 We designed a whole-body ischemia /reperfusion (WBIR) injury model to mimic the physiological responses of cardiac arrest and resuscitation. Ischemia/reperfusion (I/R) injuries are predominantly caused by oxidative stress, which triggers cell death and produces irreversible tissue damages in multiple organs in the process of rapid reperfusion to the ischemic tissues. 4 -6
Among various reactive oxygen species (ROS), superoxide anion (O2 ·−) is short-lived and is rapidly reduced to H2O2 either spontaneously or by superoxide dismutase. 3,7 In contrast, H2O2 has a longer biological lifespan and is easily diffusible intra- and inter-cellularly. 2 Massive accumulations of H2O2 is known as a critical pathogenic mechanism of I/R that releases pro-inflammatory cytokines and triggers apoptosis, thereby inducing tissue damage. 8,9 We therefore hypothesized that H2O2 could be a therapeutic target for alleviating WBIR injury.
We have previously developed a nanoparticle formulated with H2O2-responsive antioxidant copolyoxalates containing vanillyl alcohol, termed PVAX, that could serve as a nanotherapeutic agent for excessive ROS generative diseases. 10,11 PVAX releases vanillyl alcohol (VA) during its hydrolytic degradation. VA is one of phenolic compounds that are found in diverse plants; it’s an active pharmaceutical ingredient in Gastrodia elata Blume, an herbal agent that has been used for ischemic injury of the brain and heart. 12,13 In addition, the peroxalate ester backbone in PVAX has H2O2 scavenging qualities which impedes further production of ROS (Figure 1A). Our previous studies suggested that PVAX acts as an antioxidative, anti-inflammatory and anti-apoptotic agent in I/R injuries. 10,11 However, its therapeutic potential for WBIR injury in cardiac arrest and resuscitation models has not been evaluated. Since WBIR caused the integrative events of the entire body, we evaluated the impact of PVAX on WBIR induced multiple organ damage in rat model by blocking ROS-mediated damage.

Chemical property and assessment of H2O2-responsive PVAX nanoparticle. (A) chemical structure of PVAX, (B) the sensitivity of PVAX nanoparticle to H2O2 based on peroxalate chemiluminescence, (C) scavenging of H2O2 by PVAX nanoparticle.
Materials and Methods
Synthesis and Characteristics of PVAX Nanoparticle
We synthesized PVAX using 1, 4-cyclohexanedimethanol (21.96 mmol), 4-vanillyl alcohol (VA, 5.49 mmol), and oxalyl chloride (27.45 mmol), then generated PVAX nanoparticles by an emulsion/solvent evaporation method as previously reported. 14,15 Briefly, PVAX in dichloromethane was added to 5 mL of 10% (w/v) polyvinyl alcohol (PVA) solution. The mixtures were sonicated and homogenized to form fine oil/water emulsion, which was then transferred to a 20-mL PVA (1% w/v) solution and homogenized for 1 minute. The suspension was then frozen in liquid nitrogen and lyophilized to produce free-flowing particles. The particles were then centrifuged and washed with deionized water 3 times to remove the residual PVA.
For the H2O2 sensitivity assay, we prepared double batches of rubrene-loaded PVAX nanoparticles and added them into H2O2 solutions with various concentrations as previously reported. 10,11 The chemiluminescent intensity was measured using a luminometer (Femtomaster FB12, Zylux Corp, Huntsville, AL). We also evaluated the H2O2 scavenging ability of 1 mg PVAX nanoparticles by 10 μmol/L H2O2 solution (1 mL), and checked the H2O2 concentration by the Amplex Red assay kit (Invitrogen, Carlsbad, CA).
Whole Body Ischemia and Reperfusion Model in Rat
The cardiac arrest and resuscitation procedure was modified from a clinically relevant rat model. 16 -18 Animals were gender and age-matched, and were randomly assigned to each group (Sham+vehicle, Sham+PVAX, WBIR+vehicle and WBIR+PVAX) at the beginning of the experiment. Sprague Dawley rats (9-10 weeks of age) were anesthetized with isoflurane and intubated as the mechanical ventilation started (parameters of ventilator were 75 beats/minute; tidal volume was 6ml/kg). The carotid artery was cannulated for inner pressure monitoring, with the internal jugular vein (IJV) for drug and fluid loading. The rat was stabilized for 5-10 minutes after cannulation. As for the cardiac arrest, ice-cold potassium-chloride (KCl) solution (1 M; 70µl/100 g body weight (BW)) was delivered via the IJV catheter. Cardiac arrest was verified by the rapid decrease in arterial blood pressure. All rats experienced cardiac arrest for 5.5 minutes. The endotracheal tube was detached from the ventilator and isoflurane administration was turned off during the cardiac arrest. For drug administration, the animal surgeon was blinded to what compound that was being administered.
Before the resuscitation, the endotracheal tube was reattached, and the ventilator was turned on (parameters of the ventilator as before). Epinephrine (20µg/100 g BW) was injected via the IJV catheter, accompanied with chest compressions at rates of about 300 compression/minute. After 24 hours, 5 organs (heart, lung, liver, brain, and kidney) were harvested for reverse transcription-polymerase chain reaction (RT-PCR), dihydroethidium (DHE) staining and terminal deoxynucleotidyl transferase UTP nick end labeling (TUNEL) staining. Eleven rats per group were analyzed following our protocol. All experimental procedures were approved by the Institutional Animal Care and Use Committee of Beth Israel Deaconess Medical Center.
Dihydroethidium (DHE) Staining for ROS Generation
Dihydroethidium (DHE) Staining was performed on fresh frozen tissue sections as described previously. 10,11 Frozen heart, lung, liver, brain, and kidney tissues in the optimal cutting temperature (OCT) compound were cut into 5-µm-thick sections and placed on a glass slide. Tissue sections were d incubated with 5 μM dihydroethidium (DHE, Sigma-Aldrich, St. Louis, MO) at 37 °C for 30 min in a humidified chamber protected from light. Then, 4’, 6-diamidino-2-phenylindole (DAPI, Molecular Probes, Eugene, OR) staining for nuclei was applied. Images were assessed using a confocal fluorescence microscope. DHE fluorescent images were visualized by excitation at 488 nm and emission at 610 nm to detect the oxidized DHE product ethidium. The amount of DHE positive cells as a proportion of the total number of cells was calculated. At least 10 high power fields (∼2,000 nuclei/field) were randomly selected and were counted.
Terminal Deoxynucleotidyl Transferase UTP Nick End Labeling (TUNEL) Staining
To determine apoptotic cells, terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining was performed using an
Reverse Transcriptase-Polymerase Chain Reaction Analysis for mRNA Expression
The tissues of 5 organs were collected for molecular analysis after 24 hours WB I/R. Reverse transcriptase-polymerase chain reaction (RT-PCR) was performed as described previously. 10,11 mRNA expression levels were analyzed by RT-PCR with following specific primers: for tumor necrosis factor-α (TNF-α) sense, 5′-TAC TGA ACT TCG GGG TGA TTG GTCC-3′, TNF-α antisense, 5′-CAG CCT TGT CCC TTG AAG AGA ACC-3′; monocyte chemotactic protein-1 (MCP-1) sense, 5′-ACC ACA GTC CAT GCC ATC AC-3′, MCP-1 antisense, 5′-TTG AGG TGG TTG TGG AAAG-3′; 18 S sense, 5-GTT ATG GTT CCT TTG TCG CTC GCT C-3, 18 S anti-sense, 5-TCG GCC CGA GGT TAT CTA GAG TCA C-3.
Ribosomal 18 S primers acted as internal controls, and all RT-PCR signals were normalized to the 18 S signal of the corresponding RT product to eliminate the measurement error from uneven sample loading and to provide a semi quantitative measure of the relative changes in gene expression.
Statistical Analyses
Calculations and statistics were performed using GraphPad 5.0 software (GraphPad Software Inc., La Jolla, CA). Statistical analysis was carried out using the 1-way analysis of variance (ANOVA) and Bonferroni’s tests for post hoc differences between group means. Statistical significance was defined as P < 0.05. Results are presented as mean ± the standard error of the mean (SEM).
Results
H2O2-Responsiveness and H2O2-Scavenging Ability of PVAX
PVAX possesses peroxalate ester bonds and vanillyl alcohol (VA) covalently incorporated in its backbone (Figure 1A), which are able to perform peroxalate chemiluminescence reaction in the presence of H2O2 and fluorophore. The sensitivity of PVAX to H2O2 was evaluated based on peroxalate chemiluminescence. Two different batches of PVAX nanoparticles were loaded with rubrene as a fluorophore, and chemiluminescent nanoparticles were added into various H2O2 solutions (1, 2.5 and 5 μM). Both chemiluminescent nanoparticles showed a linear correlation between chemiluminescence intensity and H2O2 concentration (Figure 1B). The H2O2 scavenging ability of PVAX was assessed at 3 different time points (6, 12, and 24 hour). Addition of PVAX nanoparticles (1 mg/mL) significantly eliminated H2O2 in a time-dependent manner (Figure 1C).
Effects of PVAX on Mortality and Blood Pressure During WBIR
Schematic diagram of a cardiac arrest rat model and the experimental design is shown in Figure 2A and B, respectively. The first dose of PVAX (50ug/100ul) or 100ul PBS was injected after the spontaneous recovery of blood pressure (BP) was noted. The first injection was followed by the second injection (the same doses as the first time) 10 minutes later, before vein catheter was removed. During the procedure, initial BP and heart rate (HR), peak BP and HR, and end procedure BP and HR were recorded in detail for analysis later. Representative tracings of arterial blood pressure (ABP) of rats before, during, and after cardiac arrest and resuscitation are shown in Figure 2C. There was no statistical difference in the initial BP and HR between the vehicle group and PVAX group (Table 1). Since WBIR injury mainly occurred in the process of global reperfusion to ischemic tissues, PVAX was injected after the return of spontaneous circulation. Resuscitation caused significant increase of systolic and diastolic peak blood pressure, then fell to normal pressure in vehicle control. However, in the PVAX group, the increase of systolic BP (SBP) and diastolic BP (DBP) were not reach to significant difference during resuscitation. In addition, the increase of peak blood pressures in the PVAX group were significantly lower than those in the vehicle group (SBP 192.3 ± 9.8 vs 170.6 ± 17.2, P = 0.048; DBP 128.9 ± 6.1 vs 112.4 ± 6.7, P = 0.037) (Table 1). At the end procedure, there was no difference in the initial BP and HR between the vehicle group and PVAX group. Two rats in WBIR-vehicle group and 1 in WBIR-PVAX group died during the procedure, and thus 88% of rats (22/25) were successfully resuscitated after 5.5 minutes of cardiac arrest.
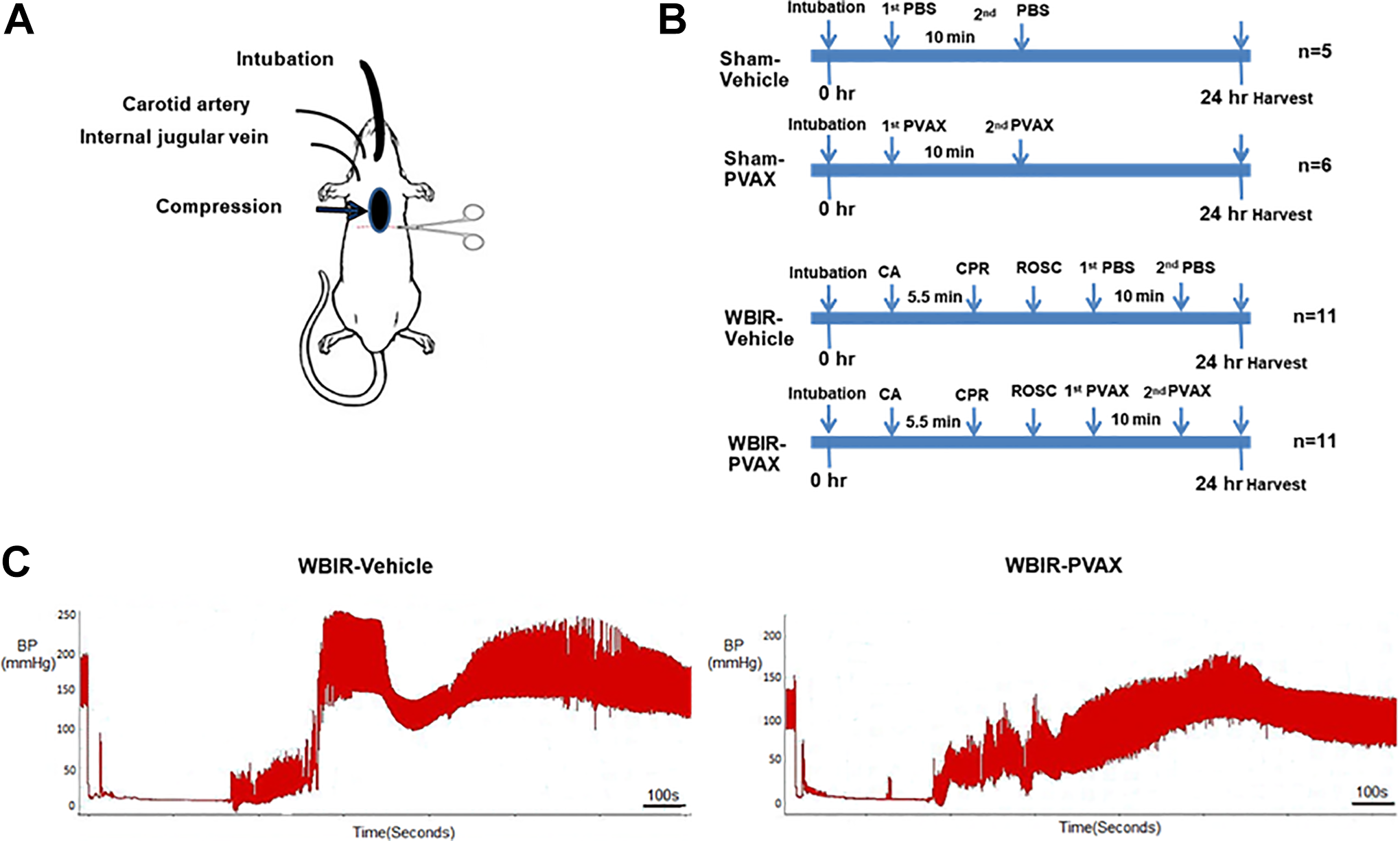
Cardiac arrest rat model and blood pressures monitoring during WBIR procedure. (A) a schematic diagram for cardiac arrest rat model, (B) the timeline for WBIR Procedure. WBIR, whole body ischemia/reperfusion. CA, cardiac arrest, CPR, cardiopulmonary resuscitation, ROSC, the return of spontaneous circulation, BP, blood pressure. (C) blood pressure curve during WBIR procedure in vehicle and PVAX groups.
Procedural and Invasive Monitoring Characteristics.

(N = 11 for each group).
WBIR, whole body ischemia/reperfusion; BW, body weight; mmHg, the millimeter of mercury; SBPi, initial systolic blood pressure; DBPi, initial diastolic blood pressure; HRi, initial heart rate; bpm, beats per minute; SBPp, peak systolic blood pressure; DBPp, peak diastolic blood pressure; HRp, peak heart rate; SBPe, systolic blood pressure at end procedure; DBPe, diastolic blood pressure at end procedure; HRe, heart rate at end procedure; CPR, cardiopulmonary resuscitation.
Effects of PVAX on ROS Generation and Apoptosis After WBIR
The increased generation of ROS has been suggested as a major contributor to the pathogenetic mechanisms during I/R injury. To determine whether PVAX successfully blocks ROS generation as an antioxidant agent in multiple organs after 24 hours of WBIR, DHE staining served as an indicator of ROS generation. 19,20 As shown in Figure 3, WBIR induced large amount of the ROS production as observed in percentage of DHE-positive cells in all 5 organs selected especially in the brain. PVAX treatment effectively decreased the number of DHE-positive cells in 5 organs selected.
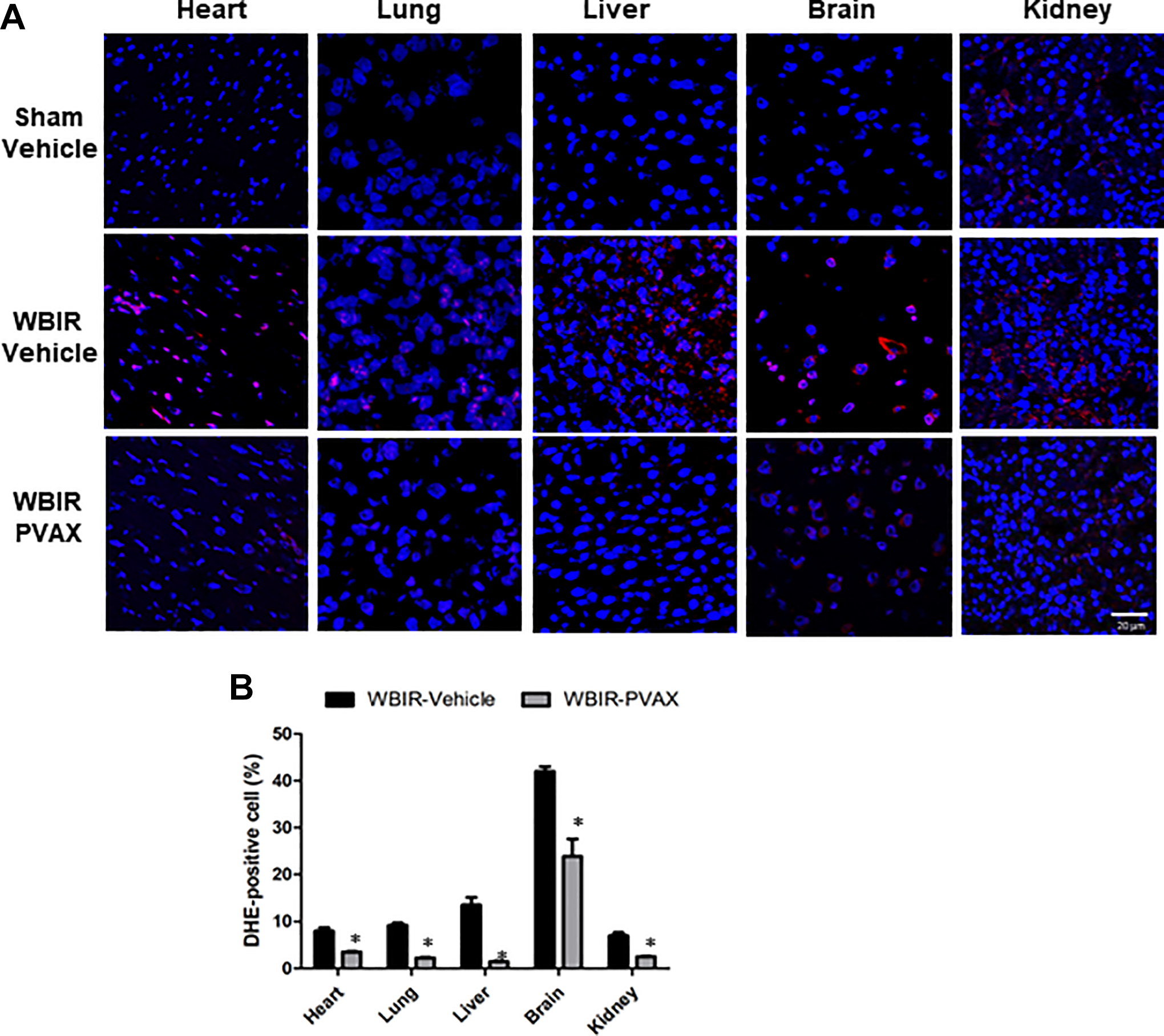
Effects of PVAX on ROS generation in multiple organs after WBIR. (A) Representative image of DHE fluorescent staining in heart, lung, liver, and brain, and kidney after WBIR. DHE, red; nuclei, blue. (B) Quantification of DHE-positive nuclei in heart, lung, liver, brain, and kidney after WBIR *, P < 0.05 vs. WBIR-vehicle.
Ischemia/reperfusion injury has been linked to the generation of ROS, which in turn lead to tissue damage through induction of apoptosis. 10 We further examined whether PVAX effectively reduced WBIR-induced apoptosis in multiple organs. As shown in Figure 4, the percentage of apoptotic cells markedly increased in all 5 organs after WBIR, especially in the liver. PVAX treatment significantly decreased apoptotic cells in 5 organs (the lowest was shown in the brain). These results indicate that PVAX successfully reduced WBIR-induced ROS overproduction and apoptosis.
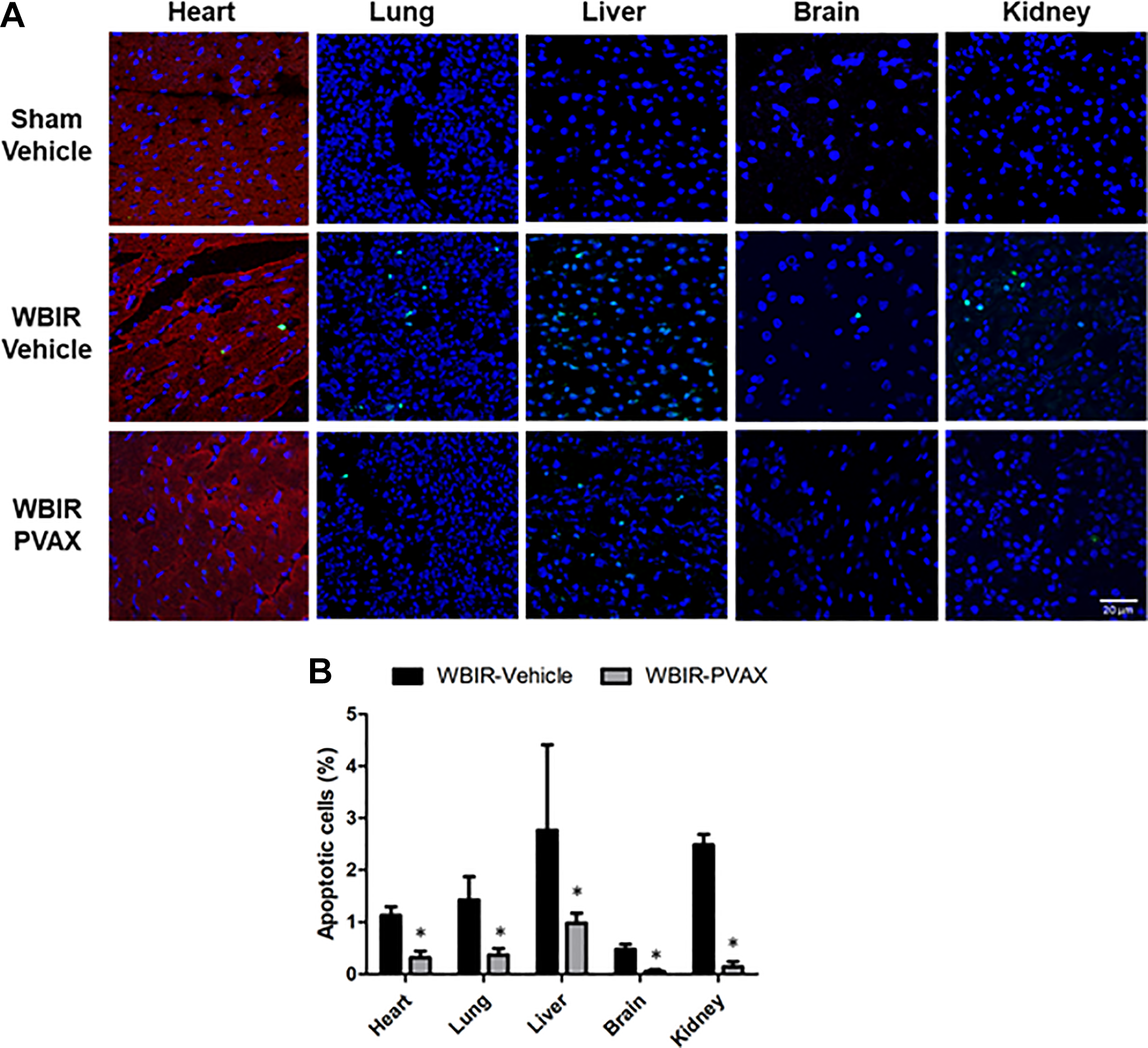
Effects of PVAX on apoptosis in multiple organs after WBIR. (A) Representative image of TUNEL fluorescent staining in heart, lung, liver, and brain, and kidney after WBIR. TUNEL, green; nuclei, blue; α-actinin, red. (B) Quantification of TUNEL-positive nuclei in heart, lung, liver, and brain, and kidney after WBIR. *, P < 0.05 vs. WBIR-vehicle.
Effect of PVAX on Pro-Inflammatory Cytokines Expression After WBIR
The inflammatory response to ischemia/reperfusion (IR)-induced organ damage may subsequently lead to a systemic inflammatory response with multiple organ failure. 21 To determine whether PVAX effectively reduced inflammation in multiple organs after 24 hours of WBIR, the expression of the pro-inflammatory cytokines (MCP-1 and TNF-α) was evaluated. As shown in Figures 5A and B, the MCP-1 expression significantly increased in the vehicle group of WBIR in 4 organs, excluding the kidney, particularly in the liver and brain. After PVAX treatment the MCP-1 expression significantly reduced in the 4 organs mentioned above, especially in the brain and heart. The TNF-α expression also significantly increased in all 5 organs selected, notably in the lung and liver, after WBIR; however, the PVAX treatment reduced TNF-α expression in the 4 organs, excluding the kidney, as shown in TNF-α expression in Figures 5C and D.

Effect of PVAX on Pro-inflammatory cytokines expression in multiple organs after WBIR. (A) Representative images of mRNA levels of MCP-1 in heart, lung, liver, brain and kidney after WBIR (B) Representative images of mRNA levels of TNF-α in heart, lung, liver, brain and kidney after WBIR (C) Quantification of MCP-1 level in heart, lung, liver, brain and kidney after WBIR. *, P < 0.05 versus vehicle. †#, P > 0.05 versus vs. WBIR-vehicle. (D) Quantification of TNF-α level in heart, lung, liver, brain and kidney after WBIR. *, P < 0.05 vs. vehicle. †, P > 0.05 vs. WBIR-vehicle.
Discussion
In this study, we created a novel animal model to demonstrate that WBIR induced by cardiac arrest would lead to damages in multiple organs. Animals that underwent WBIR showed increased ROS production, inflammation and cell death in organs such as brain, heart, lung, liver and kidney, compared to those that underwent sham procedure. We found that PVAX treatment significantly reduced WBIR-induced elevation of ROS, apoptosis, and the expression of 2 pro-inflammatory cytokines (TNF-α and MCP-1) in multiple organs 24 hours post-WBIR. These results suggest that PVAX treatment could have therapeutic benefits for WBIR-induced organ injury.
PVAX, an H2O2-responsive antioxidant polymer, has peroxalate ester bonds that can rapidly react with H2O2, while releasing vanillyl alcohol (VA), both of which exert anti-oxidative effects. 14,15 Our previous research showed that VA suppresses the expression of pro-inflammatory cytokines, and that PVAX has anti-apoptotic roles by inhibiting the H2O2 stimulated apoptotic cascade. 10,15 These properties support the use of PVAX as a therapeutic approach in WBIR injuries, in which multiple organs were injured by ROS generation, along with increased inflammation and apoptosis during global reperfusion to ischemic tissues after the return of spontaneous circulation.
Among ROS, H2O2 is relatively stable and has a relatively longer biological lifespan. H2O2 is also known to be easily diffusible within and between cells. 22 H2O2 has been shown to be essential for cellular signaling for normal physiological cellular functions at a very low physiological level. 23,24 However, overproduction of H2O2 is one of the critical pathogeneses of oxidative stress-induced tissue damage in I/R by releasing pro-inflammatory cytokines and triggering apoptosis. In WBIR, this process is initially triggered by partial restoration of organ perfusion in cardiopulmonary resuscitation, and further amplified by global reperfusion after the return of spontaneous circulation. 25 By targeting H2O2, PVAX can deliver VA to decrease ROS generation at the site of WBIR injury.
Cardiac resuscitation after cardiac arrest is often followed by major health consequences as multiple organ injuries occur mainly by the overproduction of ROS as well as increased inflammation and cell death. 4 The American Heart Association Guidelines for Cardiopulmonary resuscitation and Emergency Cardiovascular Care emphasizes that “effective post-cardiac arrest care consists of identification and treatment of the precipitating cause of cardiac arrest combined with the assessment and mitigation of ischemia-reperfusion injury to multiple organ systems.” 26 Post-resuscitation mortality is often attributed to cerebral and/or myocardial dysfunction, which is a pathological state resulted from WBIR. 27 In our study, animals treated with PVAX showed lowered peak arterial blood pressure. There was an early drop in peak BP after PVAX treatment, followed by a steady return to the end BP, and the BP remained stable afterward. However, there are other compounding variables that could affect the acute blood pressure response, such as anesthesia, In this experimental model. Thus, extrapolating acute BP observation to ultimate cardioprotection may be misleading in this model. In terms of inflammation and apoptosis, after PVAX treatment, both the brain and the heart of the WBIR animals showed markedly lower TNF-α and MCP-1 expression and apoptotic activities compared with vehicle treated group, which suggest potent anti-inflammatory and anti-apoptotic effects of PVAX. The decreased inflammation and apoptosis might contribute to the improved heart and cerebral function as suggested by other studies. 28,29
Despite the fact that liver has dual blood supply through the hepatic artery and portal system to minimize the damage from I/R, many patients who survived from cardiac arrest still suffer from hypoxic hepatitis post resuscitation.
30,31
Lung injury is another common complication after cardiac arrest and CPR. Acute pulmonary edema is often observed after the return of spontaneous circulation and its severity is associated with long-term survival and neurologic outcomes.
32
In this study, we found that ROS production and inflammation were both significantly higher in the livers and the lungs in animals 24 hours after WBIR compared with those of the sham group. The elevated ROS production and inflammation observed was consequently correlated with upregulation of apoptotic activities in those animals. However, PVAX, designed to inhibit ROS production, significantly reduced the inflammation and apoptotic activities in the livers and the lungs. However, there were noticeable inconsistencies between pro-inflammatory markers, and ROS production and apoptosis in the kidney. DHE and TUNEL staining showed a significant rise of ROS production and apoptotic activity in kidneys post-WBIR, which were effectively inhibited by PVAX treatment. However, when comparing pro-inflammatory markers in the kidney, there was no significant difference between the sham group and the 2 WBIR groups (with or without PVAX treatment). We speculate that kidneys might be less sensitive to WBIR compared to other organs. In clinical settings, less than half of the survivors of cardiac arrest will develop acute kidney injury, most of which had independent major risk factors such as older age, higher epinephrine dose, cumulative fluid balance and presence of shock, rather than WBIR.
6
Acute kidney injury is therefore not deemed as an independent factor for prognostic outcome of CPR.
6
Previous studies in kidney injury after cardiac arrest used at least 8 minutes for the duration of the ischemia (much longer than the 5.5 minutes we used in this study).
16,33
During WBIR in the cardiac arrest mouse model, Burne-Taney and colleagues reported that 10 minutes of WBIR is required to cause a reproducible renal tubular injury and an elevation in inflammation, and at least 30 minutes of renal blood flow cessation is needed to create similar physiological response in the isolated renal artery clamping model.
16
More studies are needed to better understand this phenomenon. One limitation of this study was the lack of long-term organ function evaluation. Thus, the overall clinical implication of PVAX as a treatment modality during cardiac resuscitation needs more investigation. However, improvement in inflammation and apoptosis are indicative of better organ functions
Many drugs are merely effective due to their lack of targeted delivery to the diseased area. To solve this problem, targeted drug delivery with nanotechnology has emerged as a method for effective therapy. 28,34 Nanotechnology is a powerful method because it can modify or imitate biological activities based on various purposes, allowing efficient and on-demand release of drugs to the targeted area, maximizing the therapeutic effects of the drug while minimizing undesired off-target side effects. 15,35,36
Conclusion
In conclusion, we found that ROS generation, pro-inflammatory cytokines, and apoptosis were considerably increased despite effective return of spontaneous circulation after 24 hours of WBIR. Our nanoparticle, PVAX, targeting the areas with H2O2 over production, exerted a potent suppressive effect on ROS induced inflammation and apoptosis in multiple organs. Therefore, PVAX could have a therapeutic potential for alleviating WBIR injury.
Footnotes
Author Contributions
Ruijian Li, MD, and Sang Jae Rhee, MD, are contributed equally to the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from the National Institutes of Health R44DK103389-01 (P.M.K.), the Brain Research Program of the National Research Foundation NRF-2017M3C7A104 4819, South Korea (C.G.S.) and Shandong Provincial Government-sponsored oversea Scholarship ZR2015HM039, China (R.L.).
