Abstract
Schizophrenia (SCZ) is a severe mental disorder with high morbidity and lifetime disability rates. Patients with SCZ have a higher risk of developing metabolic comorbidities such as obesity and diabetes mellitus, leading to increased mortality. Antipsychotics (APs), which are the mainstay in the treatment of SCZ, increase the risk of these metabolic perturbations. Despite extensive research, the mechanism underlying SCZ pathophysiology and associated metabolic comorbidities remains unclear. In recent years, gut microbiota (GMB) has been regarded as a ‘chamber of secrets’, particularly in the context of severe mental illnesses such as SCZ, depression, and bipolar disorder. In this scoping review, we aimed to investigate the underlying role of GMB in the pathophysiology of SCZ and metabolic alterations associated with APs. Furthermore, we also explored the therapeutic benefits of prebiotic and probiotic formulations in managing SCZ and AP-induced metabolic alterations. A systematic literature search yielded 46 studies from both preclinical and clinical settings that met inclusion criteria for qualitative synthesis. Preliminary evidence from preclinical and clinical studies indicates that GMB composition changes are associated with SCZ pathogenesis and AP-induced metabolic perturbations. Fecal microbiota transplantation from SCZ patients to mice has been shown to induce SCZ-like behavioral phenotypes, further supporting the plausible role of GMB in SCZ pathogenesis. This scoping review recapitulates the preclinical and clinical evidence suggesting the role of GMB in SCZ symptomatology and metabolic adverse effects associated with APs. Moreover, this scoping review also discusses the therapeutic potentials of prebiotic/probiotic formulations in improving SCZ symptoms and attenuating metabolic alterations related to APs.
Introduction
Schizophrenia (SCZ) is a severe mental illness affecting 20 million people worldwide. Compared to the general population, patients with SCZ have a two- to three-fold higher mortality rate and 10–25 years reduced life expectancy, mainly due to the increased incidence of cardio-metabolic comorbidities such as cardiovascular disease and Type 2 diabetes mellitus (T2DM).1–3 Impaired glucose homeostasis in patients within the first episode of psychosis (FEP) affirms an intrinsic risk for developing T2DM. Unfortuntately, antipsychotics (APs), which are the mainstay of treatment in SCZ spectrum disorders, have been found to further exacerbate this risk. 4 Despite their clinical efficacy, all APs, and particularly second-generation APs (SGAs), induce severe metabolic adverse effects, including weight gain, adiposity, insulin resistance, and dyslipidemia.5–7 Studies show that SGAs in acute and chronic treatment can disrupt glucose metabolism peripherally and centrally.8,9 Currently, the most common mechanistic insights behind AP-induced metabolic alterations (AIMA) include heterogenous neurotransmitter–receptor interactions, 10 impaired hypothalamic appetite-regulating pathways 11 and energy-sensing,12,13 altered glucose and lipid metabolism,5,14 and aberrant adipose tissue homeostasis (increased lipogenesis and inflammatory state). 15 However, despite years of research, it is still unclear how these mechanisms may interact to produce the metabolic perturbations associated with SCZ and AP medication.
The human gastrointestinal (GI) tract is considered the most diverse and densely populated microbial habitat. It contains approximately 100 trillion microorganisms, including bacteria, viruses, and yeast collectively called the ‘gut microbiota’ or ‘gut microbiome’ (GMB). The GMB encodes for more than three million genes, and orders more than the human genome (~23,000 genes). Major bacterial phyla comprising the GMB include Firmicutes, Bacteroidetes, Actinobacteria, Proteobacteria, Fusobacteria, and Verrucomicrobia. Firmicutes and Bacteroidetes represent 90% of the GMB. For the Firmicutes phylum, the major genera include Clostridium, Lactobacillus, Bacillus, Enterococcus, and Ruminococcus. Bacteroides and Prevotella are the major genera of phylum Bacteroidetes, while Actinobacteria phylum (Bifidobacterium genus) is a less abundant component of GMB. 16 Gut bacteria are essential for regulating digestion, immunity, and metabolic homeostasis.16,17 These bacteria play a pivotal role in the digestion, fermentation, and absorption of several nutrients and metabolites such as carbohydrates, lipids, protein, and amino acids while producing secondary metabolites such as short-chain fatty acids (SCFAs).16,18 Increased intestinal permeability and decreased epithelial integrity have been associated with an altered GMB. Similarly, the GMB has been shown to modulate blood-brain barrier (BBB) integrity.19,20
The brain connects with the gut via bidirectional communication involving neuroendocrine and neuro-immune pathways. These connecting pathways are closely regulated by the GMB and together form the microbiota-gut-brain (MGB) axis. The bidirectional communication of the MGB axis is such that changes in GMB composition can affect behaviors or behavioral perturbations may alter the GMB.19,21 The vagus nerve is a chief mediator of the bidirectional communication along the gut-brain axis through cholinergic activation of nicotinic receptors. 22 Sensory afferent neurons of the vagus nerve detect a diverse range of chemical and mechanical stimuli in the intestines and transmit messages to the nucleus tractus solitarius in the brainstem to initiate autonomic, endocrine, and behavioral responses.23,24 Working in the other direction, the vagus nerve also regulates gut functions including regional motility, secretion, permeability, and mucosal immune response which collectively can induce changes in GMB composition and activity. 25 Furthermore, a functioning GMB is also required for the development and regulation of the hypothalamic-pituitary-adrenal (HPA) axis. Altered GMB in early life plausibly impacts neuro-immuno-endocrine functions and predisposes an individual toward stress-related disorders in adulthood. 26 Increased gut permeability due to intestinal barrier impairment has been associated with elevated corticosterone levels in early life stress. 27 The neuro-immuno-endocrine functions of the MGB axis are regulated by GMB-derived molecules such as SCFAs, tryptophan metabolites,28,29 and secondary bile acids (e.g. deoxycholic acid, ursodeoxycholic acid, and lithocholic acid), which are derived from primary bile acids (cholic acid and chenodeoxycholic acid) primarily by gut bacteria. 30 The GMB is also capable of secreting and utilizing neurotransmitters and neuroactive compounds such as serotonin (5-HT), dopamine, norepinephrine, and γ-aminobutyric acid (GABA), which underly behavioral alterations.28,31–33 Likely, owing to these neuro-immuno-endocrine modulations (mainly HPA axis dysfunction), changes in GMB composition have been associated with severe mental illnesses such as major depression, 34 bipolar disorder, 35 and SCZ.36,37
Accumulating evidence suggests that GMB alteration largely associated with SCZ pathogenesis and AIMA. AP treatment causes dysbiotic (i.e. obesogenic) shifts in the GMB, including an increased abundance of Firmicutes and decreased abundance of Bacteroidetes phyla in rodents and human studies.22,38–41 In addition, changes in GMB composition have been associated with altered glutamate neurotransmission, 42 cognitive impairments in SCZ, and AP-induced increases in adiposity and alterations in glucose homeostasis and energy balance.43,44 Considering the potential role of GMB in the pathogenesis of SCZ and AIMA, several prebiotics and probiotics have been examined to improve disease symptoms45,46 and adverse metabolic effects associated with APs.47–50 The present scoping review synthesizes current clinical and preclinical evidence examining the role of the GMB in SCZ and AIMA, including the use of prebiotic/probiotic formulations as a therapeutic adjunct. Improved understanding of the links between GMB, SCZ, and AIMA and the therapeutic potential of prebiotic/probiotic formulations can provide further insight into the complex pathology of SCZ and holds important implications for improving metabolic outcomes in these patients.
Methods
The present study protocol was developed using the Joanna Briggs Institute’s scoping review methodological framework.51,52
Search strategy
A search strategy including three main concepts (illness/treatment, GMB, and metabolic alterations) was developed, discussed, and implemented by the authors (R.S., N.S., and E.S.). Ovid Medline, EMBASE, PsychINFO, EBSCO’s CINAHL, Scopus, and Google Scholar databases were searched from inception to December 2021. Specific search terms and syntax were adjusted as necessary for each database. The full search string is provided in Supplementary Table 1.
Screening and study selection
All search results were imported to Covidence for screening and automatic deduplication. Three authors (R.S., N.S., and E.S.) independently completed the title/abstract and full-text screening according to prespecified inclusion and exclusion criteria (Supplementary Table 2). Conflicts were resolved by discussion and consensus among the authors, and consultation with senior authors (S.M.A. and M.H.).
Relevant data extracted from each study were presented in tables with the following headings: publication (reference), study design (country of origin, study design, and experimental techniques), significant outcomes, type of prebiotic/probiotic used, and change in the GMB composition.
Results and discussion
Search results
Our search from all the databases and gray literature search yielded 724 results, which upon deduplication reduced to 400. These 400 studies were subjected to the title and abstract screening, which excluded 323 studies, and 73 were assessed for full-text screening. In total, 27 studies were further removed in full-text screening, while 46 studies (13 preclinical and 33 clinical) included in quantitative synthesis (Figure 1).
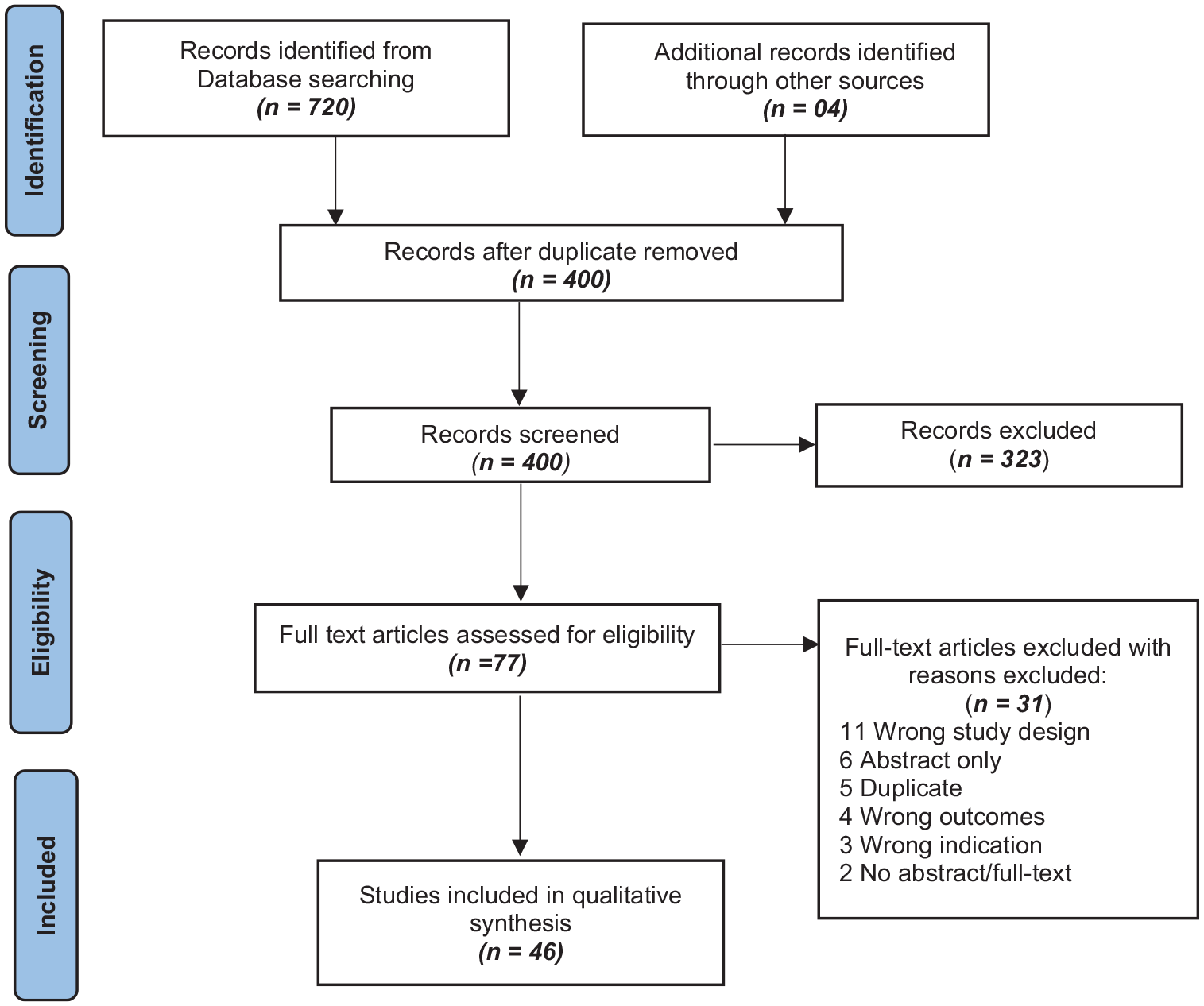
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flowchart.
Role of the GMB in SCZ and AIMA
Emerging evidence from preclinical and clinical studies suggests the putative role of the GMB in SCZ symptomatology22,36,53 and AIMA.22,44,54,55 The evidence supporting this line of thought is summarized in the following sections.
Preclinical evidence – GMB and SCZ (Table A)
Preclinical studies in mice suggest that the GMB is important in modulating behavioral phenotypes resembling clinical symptoms of SCZ, such as social isolation and cognitive impairment. In one study, GMB depletion in adolescent mice using antibiotics was associated with SCZ-like behavioral phenotype, an altered tryptophan metabolic pathway, and reduced brain derived neurotrophic factor (BDNF) expression in the adult brain. 56 Similar social and cognitive impairment has been observed in germ-free mice (GFM).22,56 Furthermore, fecal transplantation from SCZ patients to GFM has been shown to induce SCZ-like behavioral phenotypes, mainly psychomotor hyperactivity, learning and memory impairment, depression and anxiety-like behaviors, and increased startle response in the pre-pulse inhibition (PPI) test.42,57 These behavioral alterations were also associated with altered neurotransmitter levels, such as increased extracellular basal dopamine (in the prefrontal cortex), 5-HT (hippocampus), and elevation of the tryptophan degradation pathway (kynurenine-kynurenic acid pathway) in the brain and periphery, which play a pivotal role in SCZ pathogenesis. 57 Hippocampal glutamate hypofunction and disruptions in the glutamate-glutamine-GABA cycle are majorly associated with SCZ pathophysiology, mainly negative symptoms, cognitive decline, and heightened dopamine neurotransmission in mesocortical pathways.58,59 Related to this, mice which received fecal transplants from SCZ patients displayed decreased glutamate and increased glutamine and GABA levels in the hippocampus compared to mice that received fecal transplants from HCs.42,57 These behavioral and neurochemical phenotypes of SCZ were also associated with altered GMB composition. Mice that received the SCZ fecal transplant displayed an increased relative abundance of taxa such as Parabacteroides, Bacteroides, Clostridium, Odoribacter, and Fusobacterium, 57 as well as families, including Bacteroidaceae, Coriobacteriaceae, Prevotellaceae, and Veillonellaceae. 42 Together, this evidence suggests a possible role of the GMB in the pathogenesis of SCZ.
Preclinical evidence – GMB and AIMA (Table 1B)
Rodent models have also been instrumental in characterizing AIMA and associated changes in the GMB. For example, in one study, olanzapine treatment in male and female rats increased body weight and adiposity, decreased locomotion, and increased the expression of interleukin (IL)-6 in adipose tissue and plasma levels of IL-8, IL-1 β, leptin, and free fatty acids.39,40 Olanzapine also increased fatty acid synthase (FAS) expression in the liver and CD68 (a macrophage marker, which causes the release of proinflammatory cytokines from adipocytes) expression in adipose tissue. These metabolic alterations were associated with increased abundance of Firmicutes and decreased abundance of Bacteroidetes, which was reversed in rats receiving olanzapine with antibiotic cocktail treatment.39,40 In line with these findings, olanzapine treatment in female C57BL/6 J GFM shifted the GMB to an obesogenic profile characterized by increased abundance of phyla Firmicutes, Proteobacteria, and decreased Bacteroidetes. Olanzapine also exhibited antimicrobial activities against common enteric bacteria such as Escherichia coli and Enterococcus faecalis. 38
Preclinical findings showing the role of GMB in SCZ and AIMA.

5-HT, serotonin; AIMA, AP-induced metabolic alterations; CD68, cluster of differentiation 68; FT-GFM, fecal transplanted germ-free mice; GABA, γ-aminobutyric acid; GFM, germ-free mice; GMB, gut microbiome/gut microbiota; HC, healthy controls; HDL, high-density lipoprotein; HFD, high-fat diet; IL, interleukin; KEGG, Kyoto Encyclopedia of Genes and Genomes; LDL, low-density lipoprotein; PPI, pre-pulse inhibition; SCZ, schizophrenia; SREBP, sterol regulatory element-binding protein.
Similarly, risperidone treatment in female wild-type C57BL/6 J mice has been shown to cause significant weight gain, associated with decreased energy expenditure and altered GMB composition, with a notable increase in the relative abundance of Firmicutes versus Bacteroidetes. Although risperidone-induced weight gain was not attenuated by antibiotic treatment, risperidone differentially inhibited the growth of anaerobes in cultured gut microbes. 62 Recently, metformin treatment was found to reverse olanzapine-induced metabolic alterations (increased body weight, serum glucose, triglycerides, LDL, and decreased HDL levels) and gut dysbiosis (increase in the Firmicutes: Bacteroidetes ratio) in rats. 61 Taken together, these preclinical findings further support the interplay of the GMB and AIMA.
Clinical evidence – GMB and SCZ (Table 2)
A plethora of evidence from human studies supports the involvement of the GMB in SCZ pathogenesis and metabolic alterations.22,36,53 A dysregulated MGB axis may explain the role of the GMB in SCZ pathogenesis, presenting as altered inflammatory processes and immune responses in SCZ patients. 22 Microbial translocation across the gastrointestinal tract (GIT) barrier, mediated by soluble CD14 (sCD14) and LPS-binding proteins (LBP), causes low-grade inflammation and immune reaction. Patients with SCZ exhibit altered patterns of these serological bacteria translocation markers including increased sCD14 seropositivity, and a positive correlation between levels of LBP and increased BMI. These alterations could be related to GI integrity and function.56,63 A magnetic resonance spectroscopy study in individuals with high and ultra-high risk for psychosis found altered GMB composition and higher choline levels in the cortex, which is a marker of membrane dysfunction. This suggests a role of gut dysbiosis-associated membrane dysfunction in SCZ. 64 GMB alterations in SCZ patients have also been associated with increased gut glutamate synthase activity and IgA levels, linking the GMB and immune response with SCZ pathophysiology. 65 Some recent studies have also reported gut dysbiosis in patients experiencing their first episode of SCZ.60,66,67 For example, Zhu et al. 60 found that the GMB of SCZ patients had greater alpha and beta diversity at the genus and microbial gene level and comprised more genes than healthy controls (HCs). These alterations in GMB composition were associated with SCFAs synthesis, metabolism of tryptophan and other neurotransmitters, such as GABA and glutamate. 60 Zhang et al. 67 investigated the GMB of AP-naïve patients with first-episode SCZ. They found an increased abundance of potentially pathogenic bacteria belonging to phylum Proteobacteria, genus Romboutsia and decreased abundance of genera Lachnospiraceae and Faecalibacterium, essential for SCFA production.67,68 SCZ patients also showed increased serum and fecal concentrations of SCFAs, which was negatively correlated with cognitive function. Patients with higher circulating SCFAs showed immune activation evidenced by increased CRP, IL-6, soluble IL-2R, and sCD14 in Zhu et al.60,69
Clinical findings showing the role of GMB in SCZ.
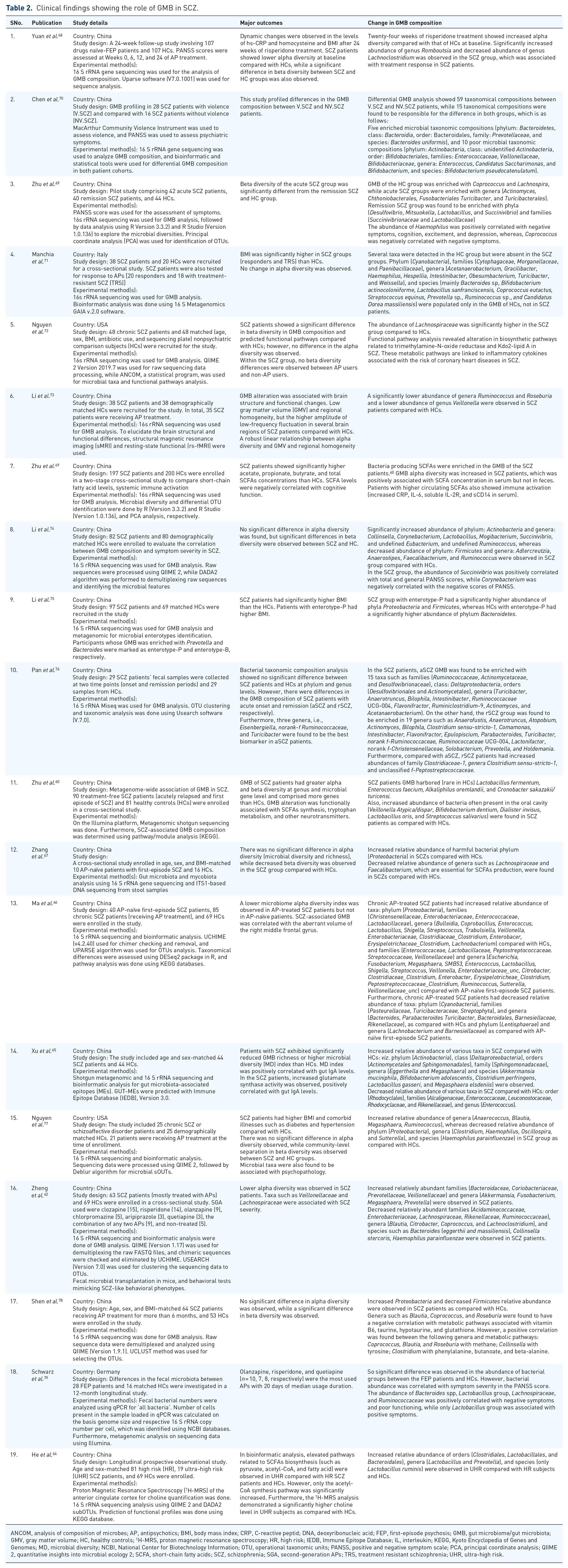
ANCOM, analysis of composition of microbes; AP, antipsychotics; BMI, body mass index; CRP, C-reactive peptid; DNA, deoxyribonucleic acid; FEP, first-episode psychosis; GMB, gut microbiome/gut microbiota; GMV, gray matter volume; HC, healthy controls; 1 H-MRS, proton magnetic resonance spectroscopy; HR, high risk; IEDB, Immune Epitope Database; IL, interleukin; KEGG, Kyoto Encyclopedia of Genes and Genomes; MD, microbial diversity; NCBI, National Center for Biotechnology Information; OTU, operational taxonomic units; PANSS, positive and negative symptom scale; PCA, principal coordinate analysis; QIIME 2, quantitative insights into microbial ecology 2; SCFA, short-chain fatty acids; SCZ, schizophrenia; SGA, second-generation APs; TRS, treatment resistant schizophrenia; UHR, ultra-high risk.
Furthermore, one study found that AP-treated patients with SCZ showed an increased relative abundance of phylum Proteobacteria and decreased abundance of phylum Cyanobacteria compared with HCs, and descreased abundance of phylum Lentisphaerae compared with AP-naïve first-episode SCZ patients. These GMB alterations were associated with abnormal right middle frontal gyrus volume. 66 Another study which examined the association of the GMB with brain structural and functional changes found that low gray matter volume (GMV) and regional homogeneity and high amplitude of low-frequency fluctuation were observed in several brain regions of SCZ patients compared with HCs. These patients had lower abundance of genera Ruminococcus and Roseburia, and lower abundance of genus Veillonella. 73
Differential abundance of microbial taxa also appears to be associated with varying psychopathology of SCZ including (1) an inverse relation between abundance of family Ruminococcaceae and severity of negative symptoms, (2) a positive relation between abundance of genus Bacteroides and severity of depressive symptoms, and (3) a positive relation between phylum Verrucomicrobia and self-reported mental well-being. 77 Lower alpha diversity was observed in SCZ patients receiving SGAs, while changes in abundance of taxa such as Veillonellaceae and Lachnospiraceae were associated with SCZ severity.42,72 Another study has reported the difference in GMB composition among the acute SCZ and remission SCZ patients. The remission SCZ group had enriched phyla (Desulfovibrio, Mitsuokella, Lactobacillus, and Succinivibrio). Furthermore, the abundance of Haemophilus was positively correlated with negative symptoms, cognition, excitement, and depression, whereas Coprococcus was found to be negatively correlated with negative symptoms. 69 In line with this, Pan et al. 76 have reported that compared with acute SCZ, remission SCZ patients had increased abundances of family Clostridiaceae-1 and genus Clostridium sensu-stricto-1 and unclassified family Peptostreptococcaceae. 76 Moreover, there was positive correlation between Succinivibrio abundance and total and general Positive and Negative Symptom Scale (PANSS) scores, while Corynebacterium was negatively correlated with the negative scores of PANSS. 74
The increased relative abundance of Proteobacteria and decreased relative abundance of Firmicutes was also observed in SCZ patients receiving APs. The abundance of some genera (such as Blautia, Coprococcus, and Roseburia) was positively correlated with specific metabolic pathways (vitamin B6, taurine, and hypotaurine). In contrast, the abundance of some genera was negatively correlated with metabolic pathways of tyrosine (Collinsella), phenylalanine, and beta-alanine (Clostridium). 78 Furthermore, SCZ patients with or without violent behavior had different GMB profiles, which are highly enriched (phylum Bacteroidetes) and poorly enriched composition (phylum Actinobacteria). 70 Hence, in the light of the evidence presented, the GMB has a noticeable association with SCZ symptoms and pathophysiology.
Clinical evidence – GMB and AIMA (Table 3)
Chronic AP-treated SCZ patients with metabolic comorbidities, including diabetes, weight gain, and hypertension, have also shown altered GMB composition. For example, in one study, first-episode SCZ patients were found to have decreased abundance of spp. such as Bifidobacterium, E. coli, and Lactobacillus as compared with HCs. Twenty-four weeks of risperidone treatment significantly increased body weight, BMI, serum glucose and lipids, antioxidant markers (i.e. superoxide dismutase), and high-sensitivity C-reactive peptide (hs-CRP). These metabolic changes were accompanied by an increased abundance of Bifidobacterium spp. and E. coli, and a decreased abundance of Lactobaccillus spp. and the Clostridium coccoide group compared with baseline. Change in the abundance of Bifidobacterium was associated with a change in body weight. 80 In another study, adolescents receiving risperidone treatment were found to have increased BMI, a lower Bacteroidetes: Firmicutes ratio, and increased abundance of Proteobacteria and Actinobacteria and decreased abundance of Verrucomicrobia compared with an AP-naïve psychiatric control group. 41
Clinical findings showing role of GMB in AIMA.

AIMA, AP-induced metabolic alterations; AP, antipsychotic; BMI, body mass index; DNA, deoxyribonucleic acid; GMB, gut microbiome/gut microbiota; HC, healthy controls; HDL, high-density lipoprotein; HOMA-IR, Homeostatic Model Assessment for Insulin Resistance; KEGG, Kyoto Encyclopedia of Genes and Genomes; LDL, low-density lipoprotein; QIIME, Quantitative Insights Into Microbial Ecology; SCZ, schizophrenia; SGA, second-generation antipsychotics; SOD, superoxide dismutase.
Another study reported that risperidone treatment for 24 weeks caused a significant change in the abundance of Bacteroidetes, Christensenellaceae, Enterobacteriaceae, and Proteobacteria, which was associated with the changes in metabolic parameters such as increased BMI, HOMA-IR, and serum lipids. 81 On the contrary, 6 weeks of olanzapine treatment in an observational study caused significant weight gain but did not change the GMB composition in SCZ patients. 54 In a nutshell, the GMB could be regarded as a potential player in metabolic perturbations associated with AP treatment.
Probiotic/prebiotic formulations as therapeutic adjuncts in SCZ and AIMA
Probiotics are live organisms, mainly bacteria, that exert therapeutic benefits when taken in adequate quantities. Prebiotics are the complex and indigestible food material (such as oligosaccharides), which upon fermentation in the colon, produces metabolites (such as SCFAs) that nourish the GMB and promote health benefits. When probiotics and prebiotics are taken together, they are termed ‘synbiotics’; when they are intended to produce therapeutic benefits in patients suffering from mental illnesses, they are referred as ‘psychobiotics’.83,84
Preclinical evidence (Table 4A)
The present review includes five preclinical studies investigating the preventive role of prebiotic/probiotic formulations against AP-induced gut dysbiosis, metabolic alterations, and dysregulated expression of different neurotransmitter receptors in rodents.43,48–50 Two studies reported protective effects of the probiotic formulation VSL#3 (a freeze-dried mixture of Bifidobacterium spp., Lactobacillus spp., and Streptococcus thermophilus) in rats 48 and in mice 50 treated with olanzapine. Specifically, VSL#3 reversed olanzapine-induced metabolic alterations (increased body weight, fasting glucose, serum total cholesterol and triglycerides, and decreased HDL levels) in Wistar rats. 48 VSL#3 treatment restored olanzapine-induced GMB disarrangement (decreased Bacteroidetes, Akkermansia muciniphila, Enterobacteriaceae, and increased Firmicutes). VSL#3 also restored the olanzapine-induced oxidative stress and inflammatory aberrations in mice. 50 Recently, a probiotic formulation containing Akkermansia muciniphilasub was found to reverse olanzapine-induced metabolic alterations, mainly increased serum markers (insulin, total cholesterol, triglycerides), systemic inflammation (increased serum IL-6, and TNF-α levels), gluconeogenesis and adipocyte deposition markers in the liver, and insulin resistance. 85 A different group used the prebiotic formulation BimunoTM galacto-oligosaccharides (B-GOS®) to attenuate olanzapine-induced weight gain in female Sprague Dawley rats. In addition to preventing weight gain, B-GOS® treatment reduced mRNA expression of 5-HT2A receptors, and increased expression of GluN1 protein and GluN2A mRNA (glutamate receptor subunits) in the cortex. B-GOS® treatment also increased the abundance of Bifidobacteria spp. and decreased Coprococcus spp., Escherichia/Shigella spp., Oscillibacter spp., Clostridium, Coccoides spp., Rosbeuria Intestinalis Cluster, and Clostridium XVIII cluster. 43 In a separate study, B-GOS® and sodium acetate (a prebiotic) treatment increased and decreased plasma acetate levels in control and olanzapine-treated rats, respectively. These changes were associated with a decrease in brain histone deacetylase (HDAC) and histone acetyltransferase (HAT) activities in the cortex and hippocampus. 49 Recently, these findings suggest a preventive role of prebiotic/probiotics in olanzapine-induced weight gain and gut dysbiosis.
Effect of probiotic/prebiotic in SCZ and AIMA..

5-HT, serotonin; AIMA, AP-induced metabolic alterations; ALT, Alanine transaminase; APs, Antipsychotics; AST, Aspartate transaminase; BDNF, Brain derived neurotrophic factor; BMI, Body mass index; BPRS, Brief Psychiatric Rating Scale; CFU, colony-forming units; CRP, C-reactive peptide; DNA, deoxyribonucleic acid; ELISA, Enzyme-Linked Immunosorbent Assay; FEP, first episode of psychosis; GluN1, Glutamate [NMDA] receptor subunit zeta-1; GMB, Gut microbiome/gut microbiota; HADS, Hospital Anxiety and Depression Scale; HAT, histone acetyltransferase; HDAC, histone deacetylase; HDL, High-density lipoprotein; HFD, high-fat diet; HOMA-IR, Homeostatic Model Assessment for Insulin Resistance; IL, Interleukin; MCP-1, Monocyte Chemoattractant Protein-1; PANSS, Positive and Negative Symptom Scale; PBS, phosphate buffer saline; PEPCK, phosphoenolpyruvate carboxykinase; RANTES, Regulated upon Activation; RT-PCR, real-time polymerase chain reaction; qPCR, quantitative polymerase chain reaction; Normal T Cell Expressed and Presumably Secreted; RCT, Randomized controlled trial; SCZ, Schizophrenia; SGA, second-generation APs; SD, Sprague dawley; TAC, total antioxidant capacity; TNF-α, Tumor-necrosis factor alpha; TRANCE, TNF-related activation-induced cytokine
Clinical evidence (Table 4B)
In the last decade, several clinical studies have been conducted to explore the therapeutic potentials of prebiotics/probiotics in SCZ and AIMA. In human studies, a probiotic formulation containing Lactobacillus rhamnosus strain GG and Bifidobacterium animalis subsp. lactis Bb12 has been used in SCZ patients in three different clinical trials (list studies). Although probiotic treatment did not affect the total PANSS scores,46,92,91 one study found that it decreased both positive and negative symptom severity, with a greater effect for the former. 46 Other effects of probiotic treatment in these patients included less difficulty in bowel movement, 91 decreased inflammatory factors (acute-phase reactant von Willebrand), increased BDNF, T-cell specific protein RANTES and macrophage inflammatory protein 1 beta, 91 and reduced Candida albicans IgG levels. 46 These findings support the immunomodulatory effects of probiotics in SCZ patients. In a different study, a probiotic formulation containing Bifidobacterium breve A-1 was found to improve Hospital Anxiety and Depression Scale (HADS) and PANSS scores in SCZ patients. This was accompanied by increased levels of IL-22 and TNF-related activation-induced cytokines and an increased relative abundance of Parabacteroides. 45 More recently, probiotic supplementation (combination of Bifidobacterium, Lactobacillus, and Enterococcus) was shown to attenuate olanzapine-induced weight gain at the 4-week mark, although this was not sustained after 8 and 12 weeks of treatment. 47
Prebiotic formulations have also been investigated for modifying the GMB in SCZ patients receiving SGA treatment. For example, one study found that potato starch (resistant starch), a prebiotic, increased the abundance of Actinobacteria. 90 Currently, a prospective study using a combination of probiotics (combination of Bifidobacterium, Lactobacillus, and Enterococcus) and prebiotic (dietary fiber – maltodextrin and Perfect dietary fiber drink) formulations in 100 patients receiving SGAs with >10% weight gain is underway. 87 This study will prove insightful in understanding the role of probiotic and prebiotic supplementation in SCZ patients including its effect on weight gain, metabolic syndrome parameters, appetite, and GMB composition. 87
Conclusion
The GMB has been regarded as a ‘chamber of secrets’, particularly in severe mental illness and the associated metabolic comorbidities. Based on evidence from human and rodent studies, the GMB may serve as a mechanistic link between metabolic dysfunction and cognitive decline in SCZ given its involvement in energy metabolism and glucose regulation, as well as the programming of social behaviors and cognition, all of which are impaired in SCZ. GMB alterations also correspond to SCZ symptomatology clinically and can induce SCZ-like behavioral and neurochemical phenotypes when fecal microbiota from SCZ patients is transplanted into rodents. The major characteristics of gut dysbiosis are increased systemic and enteric inflammatory cytokine levels; disrupted gut membrane and BBB integrity and permeability; altered neurotransmitter (mainly dopamine, GABA, 5-HT, glutamate) biosynthesis, activity, and metabolism; and decreased SCFA synthesis. These alterations have been associated with SCZ pathophysiology and AIMA (as summarized in Figure 2), and therefore, the GMB should be a point of consideration in current psychopharmacotherapy. Early GMB characterization in a large group of patients with SCZ, both pre- and post-AP treatment, could help in understanding the mechanisms underlying these findings and aid in the discovery of biomarkers and inform future therapeutics. Furthermore, psychobiotic adjuncts could be of great interest in improving psychiatric symptoms and minimizing severe metabolic adverse effects associated with AP use.

Schematic representation of mechanistic insights showing involvement of GMB in SCZ and AP-related metabolic alterations.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253221096525 – Supplemental material for Gut microbiome in schizophrenia and antipsychotic-induced metabolic alterations: a scoping review
Supplemental material, sj-docx-1-tpp-10.1177_20451253221096525 for Gut microbiome in schizophrenia and antipsychotic-induced metabolic alterations: a scoping review by Raghunath Singh, Nicolette Stogios, Emily Smith, Jiwon Lee, Kateryna Maksyutynsk, Emily Au, David C. Wright, Giada De Palma, Ariel Graff-Guerrero, Philip Gerretsen, Daniel J. Müller, Gary Remington, Margaret Hahn and Sri Mahavir Agarwal in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253221096525 – Supplemental material for Gut microbiome in schizophrenia and antipsychotic-induced metabolic alterations: a scoping review
Supplemental material, sj-docx-2-tpp-10.1177_20451253221096525 for Gut microbiome in schizophrenia and antipsychotic-induced metabolic alterations: a scoping review by Raghunath Singh, Nicolette Stogios, Emily Smith, Jiwon Lee, Kateryna Maksyutynsk, Emily Au, David C. Wright, Giada De Palma, Ariel Graff-Guerrero, Philip Gerretsen, Daniel J. Müller, Gary Remington, Margaret Hahn and Sri Mahavir Agarwal in Therapeutic Advances in Psychopharmacology
Footnotes
Ethical Approval/Patient consent
Not applicable
Availability of data and materials
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RS is supported by Bebensee Schizophrenia Research Fellowship, by the Ian Douglas Bebensee Foundation, Toronto, Canada. JL is supported by the Ontario Graduate Scholarship. MH holds the Meighen Family Chair in Psychosis Prevention and the Cardy Schizophrenia Research Chair and is funded by operating grants from CIHR (grant nos. PJT-201610 and PJT-201909), the Danish Diabetes Academy, a Steno Diabetes Centre Fellowship, the BBDC, and the PSI Foundation. SMA is supported by in part by an Academic Scholars Award from the Department of Psychiatry, University of Toronto and the CAMH Discovery Fund, and has operating grants from the Canadian Institutes of Health Research (PJT-201909), PSI foundation, the CAMH Discovery Fund.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MH has received consulting fees from Alkermes. The authors have no other conflicts to declare.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
