Abstract
Background:
The community of gut microbes is a key factor controlling the intestinal barrier that communicates with the nervous system through the gut–brain axis. Based on our clinical data showing that populations of
Methods:
Dextran sulfate sodium was used to induce colitis in rats, which then underwent an enema with
Results:
Colitis-related symptoms in rats were significantly relieved after
Conclusions:
The administration of
Introduction
Ulcerative colitis (UC) is a chronic and relapsing disease of the intestinal tract that substantially decreases patients’ quality of life. 1 In addition to diarrhea, abdominal pain, malnutrition and other gastroenterological abnormalities, psychiatric disorders are also associated with UC. 2 Dysbiosis of the gut microbiota was shown to induce mental health disorders through the gut–brain axis by modulating the inflammatory response, neuronal network or immunological regulation. 3 Clinical studies have indicated that the incidence of fatigue, anxiety, depression, and other psychiatric disorders is significantly higher in patients with UC than in healthy controls.4–6 Although the cause and effect of neurological disorders associated with UC are unclear, clinical trials have suggested that the remission of digestive diseases is often accompanied by the relief of mental health disorders.7,8
To date, the pathogenesis and etiology of UC are not clear and might involve genetic, environmental and immunological factors.9–11 As the intestinal mucosa is a complicated environment where the gut microbiome plays a vital role in intestinal functioning, dysbiosis of the gut microenvironment is closely related to the incidence of UC.12,13 The depletion of “anti-inflammatory” taxa has been observed in patients with UC, along with expansion of proinflammatory microorganisms. 14
Microbiota modulation for the treatment of colitis is a hot research topic,15,16 and various reports have proven that probiotic bacteria exert a satisfactory effect on repairing the intestinal barrier and can help reestablish intestinal homeostasis.
17
Recently, our clinical data revealed that the abundance of
However, researchers have not determined whether
Materials and methods
Fecal biodiversity analysis on an ulcerative patient
One male patient aged 52 years was diagnosed with UC and underwent fecal microbiota transplantation (FMT) using FMT capsules (patent NO. ZL201510304041.4). Colonoscopy was carried out to evaluate UC before and after FMT treatment. Stool samples were collected from this patient before and after FMT treatment and stored at −80°C until a further gut microbiota analysis using 16S rRNA sequencing. Stool samples were sent to G-Bio, Hangzhou, China for 16S rRNA sequencing and an analysis of the stool composition.
Roseburia intestinalis culture and identification
Morphology of R. intestinalis
The morphology of the
The sample was prepared for the SEM analysis as described below. The isolated and purified bacteria were inoculated in a shake flask containing 40 ml of BD BACTEC™ Lytic/10 Anaerobic/F Culture Vials liquid medium and cultured without shaking for 48 h. After culture, 500 μl of the bacterial solution were aspirated and centrifuged at 3000 rpm for 3 min. The supernatant was removed, and 800 μl of phosphate-buffered saline (PBS) were added to wash the pellet 2–3 times. Then, 1 ml of 2.5% glutaraldehyde was added to the precipitate, and the sample was mixed and incubated at 4°C overnight; the sample was centrifuged at 3000 rpm for 3 min, and the supernatant was removed. Gradient ethanol dehydration was performed with a 20%, 50%, 80%, and 100% ethanol, with dehydration for 10 min each, and centrifugation was performed at 3000 rpm for 3 min; then, 100% 2-methyl-2-propanol was added 2–3 times for washing, and the samples were observed using SEM.
For Gram staining, the bacterial suspension was fixed on glass slides using a flame. Crystal violet solution A was added and incubated for 2 min. The slide was washed with running water to remove the excess dye. Iodine-potassium iodide solution B was added and incubated for 5 min. The slide was washed with running water. Excess water was allowed to run off, and then the sections were immediately placed in absolute acetone. Four changes of absolute acetone were used for a total of 100 s. The sample was briefly washed with running water until the acetone was removed. Carbolfuchsin solution C was added and incubated for 1–11 min, followed by a brief wash with running water, and the sample was observed after drying.
Animals and groups
Adult male Sprague-Dawley rats were purchased from the Experimental Animal Center, Institute of Field Surgery, Committee of Army Medical Center, and housed at 22°C on a 12:12-h light-dark cycle with free access to water and food. The rats were randomly assigned to the following groups (
Induction of colitis and R. intestinalis treatment
The animals were allowed to adapt to the environment for 1 week before the experiment. The whole experiment lasted for 17 days (Figure 3a). Specifically, 4% (w/v) DSS (M.W.: 36,000–50,000 Da, MP Biomedical, United States) in drinking water was used to induce acute colitis in rats for 7 days (DSS group), and the control group of rats received only regular sterile drinking water (control group). A freshly prepared DSS solution was administered every 2 days. Rats in the DSS + R.I group received 109 CFU/100 µl of
Behavioral tests
Behavioral tests were performed 24 h after the last treatment as described in a previous report.
23
After 1 week of DSS exposure with or without 7 days of
Open field test
Each rat was gently placed in a 100 × 100 × 45 cm square box. The bottom of the box was divided into 25 small squares, and the nine small center squares were defined as the central area and the other 16 squares as the peripheral area. Each rat was slowly placed into the box along the corner of the wall and allowed to explore the apparatus for 15 min. During the experiment, the chamber was kept quiet, and the box was cleaned after testing. Movements were digitally captured and analyzed using EthoVision XT video tracking software (Noldus Information Technology, Inc., Netherlands) to evaluate the rat’s ability to explore the center or surrounding area and the walking distance. 23
Tail suspension test
The TST involves a transparent square box with hooks. Approximately 1 cm of the rat’s tail was taped on the hook, and the rat was hung upside down 40 cm from the ground. The suspension lasted for 6 min; the first minute was considered the adaptive time (latency period), and the recording continued from the 2nd to the last minute. During the experiment, the room was kept quiet. Movements were digitally captured and analyzed using EthoVision XT video tracking software to assess the rats in a state of immobility. 23
Light/dark box
The LDB assay was performed as previously reported. 24 Rats were individually placed in a light and dark box apparatus, that is, a box (60 × 30 × 30 cm) divided into a dark area and a light area (30 × 30 cm each), and allowed to explore for 10 min. Rats were placed in the light area and allowed to move freely. The time spent in the dark side and the number of transitions between the light side and dark side were evaluated.
Hematoxylin-eosin staining (H&E)
Harris’ hematoxylin (YuanYe Biotechnology, Shanghai, China) was used to stain the samples for 5 min, which were then gently washed with water. Differentiation was carried out for 5 s with a 1% hydrochloric acid alcohol solution (1 ml), and the color of the slice was observed to change from blue to red. Then, after the sections were washed with tap water for 1.5 h, they were stained with 0.5% eosin (water-soluble, Aladdin Bio-Chem Technology, Shanghai, China) for 2 min. After the aforementioned staining process, the sections were dehydrated with different concentrations of ethanol and cleared with 100% xylene. Cover slips were mounted over the sections before they were assessed under a microscope (Carl Zeiss, Oberkochen, Germany).
Masson’s trichrome staining
After 5 min of staining with Harris’ hematoxylin, the sections were washed with running water. The samples were differentiated with 1% hydrochloric acid for 1 min and then rinsed for 1.5 h. Next, Ponceau (Aladdin Bio-Chem Technology, Shanghai, China) was added for 7 min, and distilled water was used for washing. The samples were treated with a 1% aqueous solution of phosphomolybdic acid (Fuguang Fine Chemical Research Institute, Tianjin, China) for approximately 5 min without washing with water and directly counterstained with an aniline blue (Aladdin Bio-Chem Technology, Shanghai, China) solution for 5 min. Finally, the sample was treated with 1% glacial acetic acid (Tianjin Tianli Chemical Reagent Co., Tianjin, China) for 1 min and shaken. After staining, the slices were dehydrated with different concentrations of ethanol and cleared with 100% xylene. Cover slips were mounted over the sections before they were assessed under a microscope.
Immunohistochemical staining
The histological sections were subjected to antigen retrieval, cooled for 2 h, treated with 0.3% (w/v) H2O2 for 15 min, blocked with PBS containing 5% (w/v) bovine serum and 0.1% (w/v) Triton X-100 for 15 min, and incubated with a goat anti-Iba-1 antibody (1:500, Abcam, United Kingdom), rabbit anti-Zo1 antibody (1:500, Abcam, United Kingdom) or mouse anti-GFAP antibody (1:500, Sigma-Aldrich, United States) overnight at 4°C. After the sections were washed, they were incubated with biotinylated secondary antibodies (1:200, Thermo Fisher Scientific, United States) for 1 h, followed by staining with the avidin–biotin–peroxidase complex (ABC, Vector Laboratories, United States). The diaminobenzidine (DAB) reaction was visualized from the immunoprecipitated product. After the aforementioned staining procedures, the sections were dehydrated with different concentrations of ethanol and cleared with 100% xylene. Cover slips were mounted over the sections, which were then assessed under a microscope (Carl Zeiss, Oberkochen, Germany).
Western blotting
Proteins were extracted from the brains and intestinal tissues using RIPA lysis buffer (KeyGen Biotech, Nanjing, China) containing phenylmethylsulfonyl fluoride (Biosharp Life Sciences, Hefei, China) and a phosphatase inhibitor cocktail (MedChemExpress, Shanghai, China). The total protein concentration was determined with a BCA Protein Assay Kit (KeyGen Biotech, Nanjing, China). For western blotting, the samples (30 mg protein) were separated on 10% (w/v) acrylamide SDS-PAGE gels and then transferred to a nitrocellulose membrane (Pall Corporation, United States). The membrane was then blocked with 5% (w/v) BSA (Gentihold, Beijing, China) before an overnight incubation with goat anti-Iba-1 (1:500, Abcam), mouse anti-GFAP (1:500, Sigma-Aldrich), rabbit anti-ZO1 (1:500, Abcam), and mouse anti-β-actin (1:500, Santa Cruz Biotechnology, United States) primary antibodies at 4°C. After three washes for 10 min with Tris-buffered saline containing 0.1% (v/v) Tween 20 (TBST), the nitrocellulose membranes were incubated with a fluorescent dye-labeled secondary antibody (1:15000; LI-COR Biosciences, United States) for 60 min at room temperature. After the incubation with the appropriate secondary antibodies, the membranes were analyzed and quantified using an Odyssey CLx Imager and Image Studio software (LI-COR Biosciences).
RNA extraction and qRT-PCR
Total RNA was extracted from the brains and intestinal tissues using TRIzol reagent (Thermo Fisher Scientific, United States) according to the manufacturer’s protocol. The RNA concentration was quantified using ultraviolet spectrophotometry at 260/280 nm (Nanodrop 2000, Thermo Fisher Scientific, United States). The PrimeScript First-Strand cDNA Synthesis Kit (Takara Bio, Japan) was used to reverse transcribe cDNAs. PCR was performed in an Mx3000P qPCR System (Agilent Technologies, United States) with the corresponding primers (Takara Bio, Japan) and SYBR Green PCR master mix (Takara Bio, Japan). The primer sequences were as follows: il-17a (sense primer: 5' TCAACCGTTCCACTTCACCC 3', anti-sense primer: 5' ACTTCTCAGGCTCCCTCTTCAG 3'), il-6 (sense primer: 5' CAACGATGATGCACTTGCAGA 3', anti-sense primer: 5' CTCCAGGTAGCTATGGTACTCCAGA 3').
Enzyme-linked immunosorbent assay
The changes in the levels of cytokines and neurotransmitters in the serum, colon and intestine were determined using ELISAs. Blood samples were collected, stored at 4°C overnight and centrifuged at 3000
Analysis of the gut microbiota using 16S rRNA sequencing
Rat stool samples were collected daily by stimulating the anus and were stored at −80°C. The stool samples and FMT bacterial fluid were sent together to Sangon Biotech (Shanghai, China) for 16S rRNA sequencing with the Illumina MiSeq platform. The biodiversity of the microbiota was analyzed by performing principal component analysis and clustering analysis.
Statistical analysis
SPSS statistics software 17.0 (SPSS, United States) was used for all quantitative analyses. Data are presented as the means ± SE of the number (
Results
Changes in the gut microbiome in patient with FMT treatment
A colonoscopy examination was used to detect the remission of colonic ulcers in a patient with UC after the FMT treatment (Figure 1). We also analyzed the microbiome composition of stool samples from this patient before and after the FMT treatment, and the results revealed that the population of
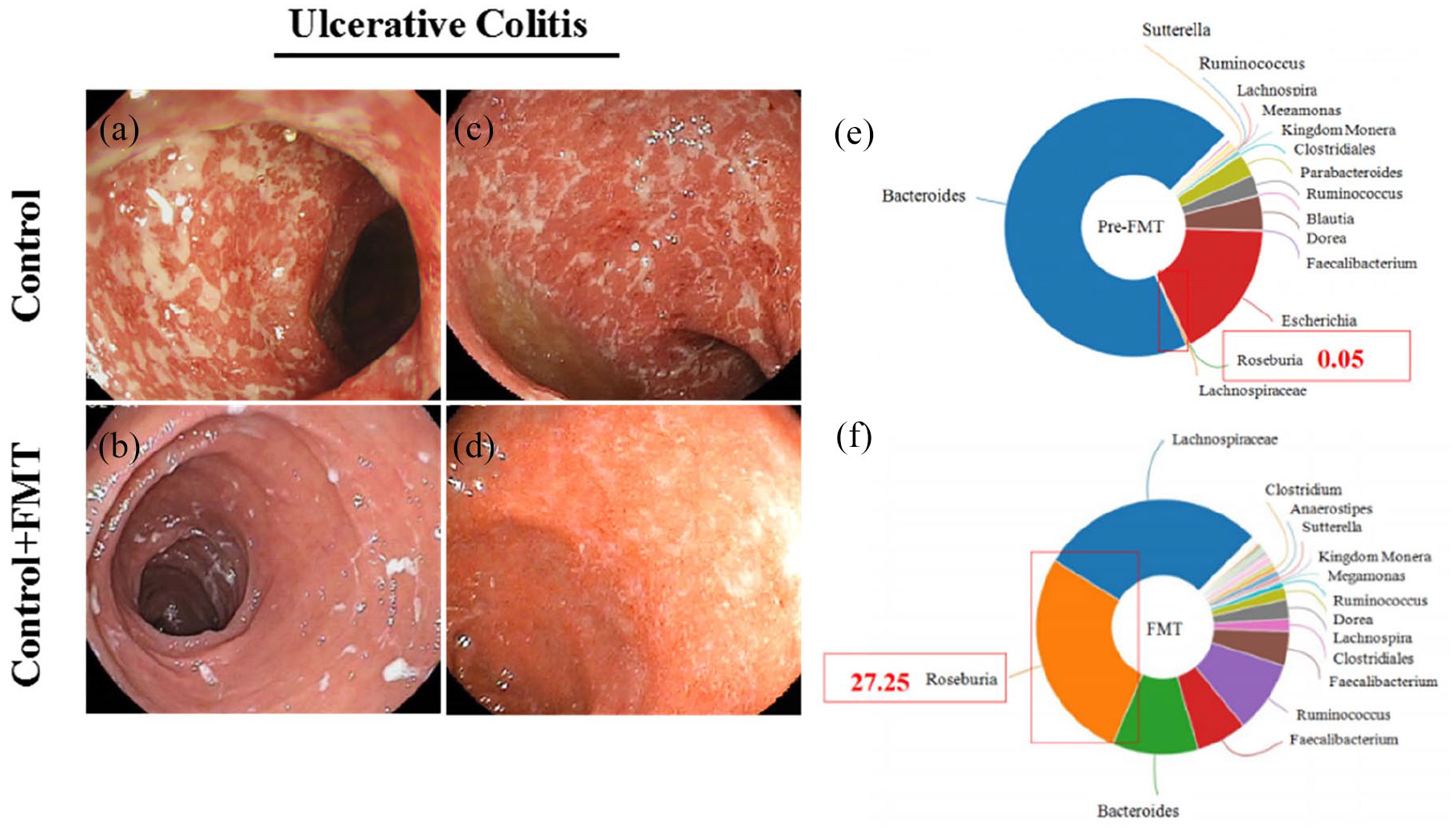
Comparison of colonoscopy pictures and intestinal bacterial composition of a patient receiving FMT. (a)–(d) Contrast colonoscopy photos of the same patient with UC after FMT treatment. (a) and (c) Colonoscopy pictures before FMT treatment, with diffuse and multiple ulcers on the colonic mucosa, as well as blurred blood vessels, a fragile texture, bleeding, and purulent secretions. The images shown in (b) and (d) were captured after FMT treatment, showing ulcer remission with pseudopolyps and a smoother mucosal layer. (e) Diagram of the composition of the fecal microbiota when the patient had not received FMT; (f) diagram of the composition of the fecal microbiota after the patient received FMT.
Identification and characterization of R. intestinalis

(a) Gram staining results showing that
Identification of Roseburia intestinalis.

The NCBI database was searched and the first three results were compared. According to the strain identification results, the strain used in this study shares the closest genetic relationship with
R. intestinalis improved health in colitis rats
During the period of DSS challenge, the body weight of rats with colitis in both the DSS and DSS + R.I groups was significantly decreased compared with the control group (Figure 3). When DSS stimulation was stopped, rats showed a recovery in body weight and the change in the DSS + R.I group was more rapid than in the DSS group. The stool samples from each group also presented different characteristics. Feces from the DSS group appeared watery and bloody, but the stool samples from the

Health conditions of rats and colon histology. (a) Experimental design. (b) Body weight change in each group. (c) Disease activity index (DAI) scores of each group. (d) Fecal traits of each group. Stool samples were collected from each group on the 17th day. (e) The length of colon from rats in each group. (f) Comparison of colon length in each group of rats. (g) Time spent by rats in the center area during the open field test (OFT). (h) Effects of
R. intestinalis has ameliorated depressive-like behaviors in colitis rats
Rats were subjected to behavioral tests at the end of treatment to evaluate whether the remission of colitis was accompanied by neuroprotective effects. We evaluated the locomotor activity of the rats using the OFT, TST and LDB assays, and significant differences were observed in all groups (Figure 3).
Specifically, the OFT, TST and LDB assays showed that the administration of
The recovery of intestinal function induced by R. intestinalis
We next carried out 16S ribosomal RNA sequencing to assess the diversity and composition of the gut microbiota in rats from different groups. A heatmap of the operational taxonomic unit profiles showed variation before (DSS S) and after (DSS E) DSS challenge, while the abundance of the fecal microbiota from the rats in the DSS + R.I group showed insignificant changes before and after

Effects of
We performed histological assays to examine rats subjected to different treatments to further investigate the effect of

Representative images of changes in the intestinal morphology of each group. (a) Representative images of H&E-stained colon sections from rats with different treatments after 7 days. (b) Representative images of Masson’s trichrome-stained colon sections from rats with different treatments after 7 days. Treatment with
We examined the mucosal integrity and tight junctions of colon tissues from the rats in different groups by performing IHC assays and western blotting to further investigate the restoration of GI function mediated by the

The expression of molecules related to the gut barrier. The astrocyte phenotype was assessed by performing GFAP IHC in the colon. Representative image of IHC staining for (a) Zo-1 and (b) GFAP in colonic mucosal epithelial cells of the control group, DSS group and DSS + R.I group. (c) Levels of the Zo-1 and GFAP proteins were analyzed using western blotting.
Anti-inflammatory effect of R. intestinalis on the central nervous system of rats with colitis
As behavioral tests showed that
The ELISA of rat serum samples showed that both IL-6 and IL-17 were upregulated in the rats with colitis, but decreased in rats that underwent the
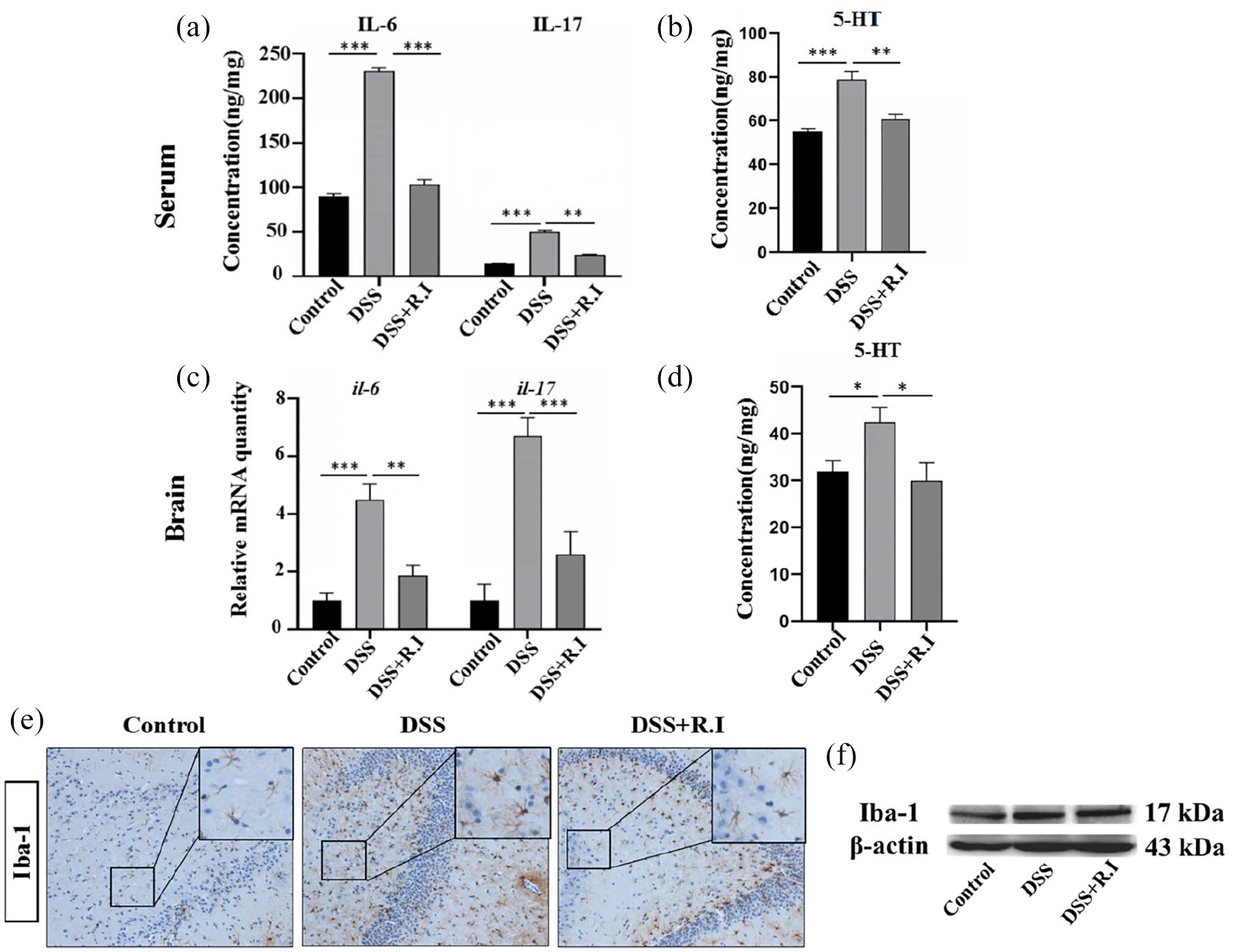
The effects of
The dorsal motor nucleus of the vagus nerve and the hypothalamus regulate the intestinal nervous and immune systems through nerves or neuroendocrine pathways, and their functions may also be affected by neuroinflammation due to the activation of microglia. Therefore, we examined the activation of microglia in the hypothalamus by performing Iba-1 IHC and western blotting. After the DSS challenge, microglial cells were activated and presented remarkably enlarged cellular bodies and hyper-ramified and thick processes; however, these alterations were alleviated in the DSS + R.I group (Figure 7). The quantification of Iba-1 was also confirmed through western blotting (Figure 7).
Discussion
Although the etiology and pathogenesis of UC have not been confirmed, the connection between gut microbiota dysbiosis and intestinal malfunction has been verified in many preclinical and clinical studies.13,29–31 In this study, we found that the abundance of
FMT is regarded as an effective method for treating intractable inflammatory bowel diseases. Our group has been using this approach for a long time to treat patients with UC in clinical practice and has achieved desirable results. Moreover, colitis remission correlates with the relief of mental health disorders through the microbiota–gut–brain axis.23,32,33 Based on our findings in clinical patients with UC, we hypothesized that exogenous supplementation with
In this study, we constructed a colitis model in rats by administering DSS as a representative platform to study the effect of
Based on our results,
We also investigated behavioral changes in rats to study whether
Furthermore, the changes in the levels of IL-6 and IL-17 in both serum and brain also suggests an effect of
In conclusion, this study is the first to report the therapeutic and protective effects of
Footnotes
Acknowledgements
Not applicable.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Innovation Project, the Military Scientific Committee of the People’s Liberation Army of China (17-163-12-ZT-002-060-01); Medical Science and Technology Pilot Project for Youth Investigators, the Military Scientific Committee of the People’s Liberation Army of China (16QNP098); and the Frontier Innovation Capability Cultivation Program of Army Medical Center of PLA (2019CXJSB008).
Research ethics and patient consent
The study protocol was approved by the Department of Gastroenterology, Daping Hospital (Medical Research Ethics 2014. Number 17), and all subjects provided written informed consent prior to participating in the study.
