Abstract
Background:
Antipsychotic agents (APS) are widely used drugs to treat psychotic symptoms and can effectively reduce both positive and negative symptoms of schizophrenia. For decades, some studies suggested that there is a relationship between using APS and the risk of venous thromboembolism (VTE) and pulmonary embolism (PE). However, results remain inconclusive.
Method:
This review has been registered in International Prospective Register of Systematic Reviews (PROSPERO, ID: CDR42020155620). Relevant studies were identified among observational studies published up to 1 October 2019 in the databases MEDLINE, EMBASE, and Cochrane Library. Random or fixed-effects models were used to calculate the pooled odds ratio (OR).
Results:
In total, 28 observational studies were included. The results showed that compared with non-users, current APS users have significantly increased risks of VTE [OR 1.55 95% confidence interval (CI) 1.36, 1.76] and PE (OR 3.68, 95% CI 1.23, 11.05). Subgroup analyses suggested that new users were associated with a higher risk of VTE (OR 2.06, 95% CI 1.81, 2.35). For individual drugs, increased risk of VTE and PE was observed in taking haloperidol, risperidone, olanzapine, prochlorperazine but not in chlorpromazine, quetiapine or aripiprazole. However, careful interpretation is needed because of high heterogeneity among studies and scarce data.
Conclusion:
The present comprehensive meta-analysis further indicates a significantly increased risk of VTE and PE in current APS users compared with non-users. Subgroup analyses suggest that new users are more likely to develop VTE. However, due to significant heterogeneity among studies, conclusions should be considered with caution.
Introduction
Antipsychotic agents (APS) are a group of drugs with a wide range of indications used mainly for psychotic symptom treatment, and are recommended for treating schizophrenia, bipolar disorder, resistant depression, autism spectrum disorder, Tourette’s disorder
Many cases of sudden death caused by PE related to APS exposure have been reported for decades,8
–12 but the relationship between APS usage and risk of VTE and PE is still controversial. The first epidemiological study of APS associated with VTE risk was conducted by Walker
Thus, a comprehensive meta-analysis of all observational studies is needed to ascertain the correlation of APS with VTE and PE risk. Studies with recurrent VTE risk as the outcome were also evaluated in subgroup analysis. Furthermore, VTE risk associated with APS of different types, potencies, doses, usage durations, and individual APS exposure were also assessed to provide more information on clinical practice.
Methods
Data sources and searches
This review has been registered in PROSPERO (International Prospective Register of Systematic Reviews, ID: CDR42020155620) 29 and conducted by Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 30 and Meta-Analyses of Observational Studies in Epidemiology (MOOSE). 31 Articles published up to 1 October 2019 in the databases MEDLINE, EMBASE, and Cochrane Library were searched. The keywords used in the search were “antipsychotic agents,” “antipsychotic drugs,” “antipsychotics” AND “thrombosis,” “deep venous thrombosis,” “venous thrombosis,” “pulmonary embolism,” and the full electronic search strategy is in the supplemental materials. In addition, further relevant studies were found in the references of retrieved articles.
Eligible criteria and quality assessment
The following inclusion criteria were used to select eligible studies: (1) observational design, (2) APS users
Two authors (LYZ and XJ) independently selected the eligible studies following the criteria above and assessed the quality of included studies. As recommended by the Cochrane Collaborations, the Newcastle–Ottawa Scale (NOS) was used to assess the methodological quality of the included studies. A study with a below-average NOS score was defined as low quality, and a study with a NOS score at or above the average was defined as high quality.
Data extraction
Two authors (LYZ and XJ) extracted data independently, and discrepancies were resolved through discussion with the third author (XY). Data related to the characteristics and reported results of each study were extracted. The following data were extracted: study name, data source, study design, study population characteristics, total population, ascertainment of APS exposure, outcome, definition of outcome, VTE risk factors exclusion, controlled variables, raw data, unadjusted OR, RR or HR with corresponding 95% CIs and adjusted OR, RR or HR with corresponding 95% CIs in the analysis.
Statistical analysis
Review Manager (RevMan) Version 5.2 software was utilized for all statistical analyses. The occurrence of VTE or PE in current APS users compared with unexposed individuals or past APS users was the outcome measure of this analysis. Adjusted ORs, RRs, or HRs with their corresponding 95% CIs were synthesized for analysis. Moreover, raw data were also used to calculate unadjusted ORs. Because the outcome of interest was rare, the OR, RR, and HR can be regarded as equivalent. The inverse variance method was used for combining studies, and both fixed and random-effects models were performed for pooling. Heterogeneity was assessed by Cochran’s Q chi-square test and
Results
Search results
The three databases were searched by using the keywords, and a total of 2275 potentially relevant articles were identified after duplicates were removed. Of all these articles, 2202 were excluded by the screening of titles and abstracts. Of these, 19 articles are conference abstracts, 116 articles are a letter or note, 352 are case reports, 685 are reviews, and 1030 articles are obviously not relevant. Thereafter, 73 articles were included in the full-text assessed. Of these, 45 studies were excluded because of the reasons reported in the diagram (Figure 1). Therefore, 28 studies13 –16,19,21 –28,33 –47 were eligible for inclusion and quality assessment.

Flow diagram of relevant studies identification.
Characteristics of included studies
Table 1 shows the characteristics of the 28 studies included in the meta-analysis. Of all the included studies, 17 were case-control studies,14,16,19,21,22,24,33 –35,37 –42,45,46 of which nine had a nested case-control design.14,16,19,21,40 –42,45,46 Eleven were cohort studies.13,15,23,25 –28,36,43,44,47 In terms of data sources, 20 studies identified patients from databases,13 –16,19,21,23,25,26,28,36,37,40 –47 four studies identified patients from hospital medical records,22,24,27,38 two used autopsy records,35,39 one study used both police records and hospital records, 34 and one used previous cohort study data. The patient populations of most of these studies were heterogeneous, and diagnostic categories were not restricted. In addition, two studies included dementia patients only,45,47 six studies focused on elderly patients,15,36,41,45 –47 and two studies focused on female APS users,23,46 of which one study included menopausal women 46 and the other study focused on pregnant women. 23 While the majority of included studies ascertained VTE and PE by diagnostic codes, six studies used PE diagnostic codes only,13,19,34,35,39,44 and 18 studies excluded or controlled mainly confounding factors of VTE and PE.14 –16,19,21,23,27,28,34,36,37,40 –43,45 –47 Three studies employed recurrent VTE as the outcome. One study is a re-analysis of the Leiden Thrombophilia Study (LETS), a case-control study on patients with venous thrombosis, rather than a case-control design directly for the purpose on the VTE risk of antipsychotics. Supplemental materials show the results of the NOS score that was used to assess the quality of observational studies. The mean score was 6.6 for the 28 studies.
Characteristics of studies.
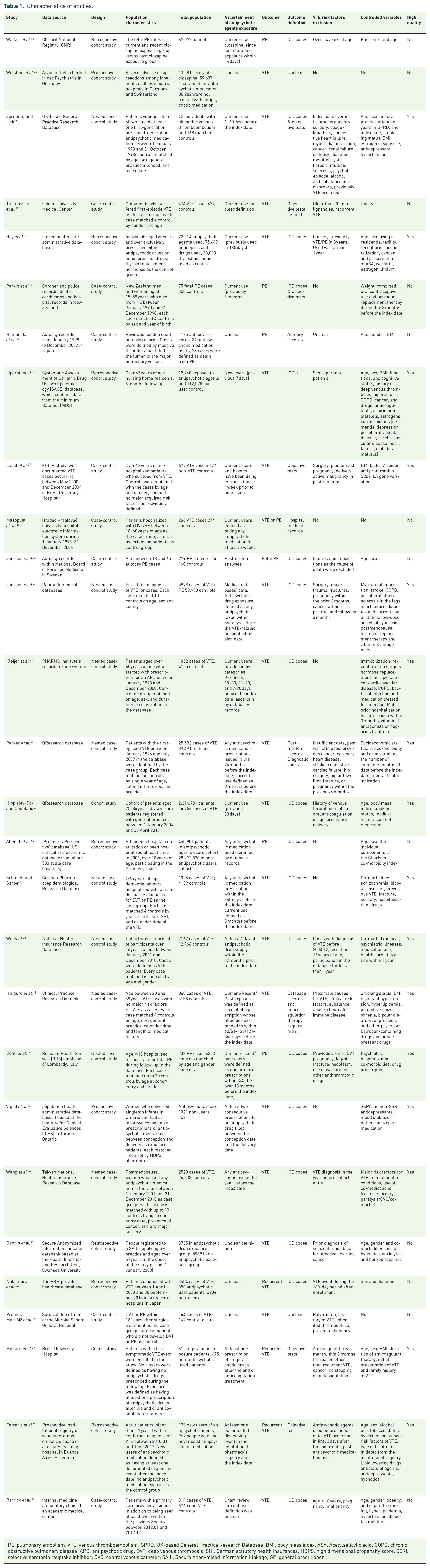
PE, pulmonary embolism; VTE, venous thromboembolism; GPRD, UK-based General Practice Research Database; BMI, body mass index; ASA, Acetylsalicylic acid; COPD, chronic obstructive pulmonary disease; APD, antipsychotic drug; DVT, deep venous thrombosis; SHI, German statutory health insurances; HDPS, high dimensional propensity score; SSRI, selective serotonin reuptake inhibitor; CVC, central venous catheter; SAIL, Secure Anonymised Information Linkage; GP, general practitioner
Main results
Figure 2 shows the ORs of VTE and PE risks with APS used in individual studies and summary estimates. Twenty-one studies (22 estimates) provided data eligible for the risk of VTE analysis, showing that compared with non-users, current APS use can significantly increase the risk of VTE by about 50% (OR 1.55 95% CIs 1.36, 1.76). However, a high heterogeneity was observed (

Forest plot of the association between current antipsychotic agents (APS) used and risk of venous thromboembolism (VTE) and pulmonary embolism (PE).
Suitable data were extracted from four studies for PE risk estimates and showed that current APS use also increased the risk of PE (OR 3.68, 95% CIs 1.23, 11.05), but the heterogeneity was significantly high (
Subgroup meta-analysis
The results of the subgroup meta-analyses are shown in Table 2. The risk of recurrent VTE was increased for APS users based on three studies’ data analysis (OR 1.62 95% CIs 1.18, 2.24), and no heterogeneity was observed (
Association between antipsychotic agent use and VTE and/or PE in subgroup meta-analysis.

APS, antipsychotic agents; FGA, first-generation antipsychotic agents; PE, pulmonary embolism; SGA, second-generation antipsychotic agents; VTE, venous thromboembolism.
In the subgroup of meta-analyses concerning categories of APS, using FGA (OR 1.47, 95% CIs 1.21, 1.78) and SGA (OR 1.62, 95% CIs 1.28, 2.05) both significantly increased the risk of VTE. A similar association was observed for combination use of two types of APS (OR 2.01, 95% CIs 1.47, 2.75). When studies were grouped by usage duration, new users were more vulnerable to VTE (OR 2.06 95% CIs 1.81, 2.35) than continuing users (OR 1.29, 95% CIs 1.04, 1.61). Significantly increased risk of VTE was found in high-potency (OR 1.31, 95% CIs 1.22, 1.41) but not in low-potency (OR 1.65, CIs 95% 0.99, 2.77) APS. Association of the dose of APS use with VTE risk was also assessed. The results showed the risk of VTE was significantly higher in both high-dose (OR 1.86, 95% CIs 1.12, 3.09) and low-dose (OR 1.45, 95% CIs 1.11, 1.90) APS. For the association between individual drugs and VTE and/or PE risks, only a few studies provided analyses. Increased risk of VTE and PE was observed for haloperidol (OR 1.64, 95% CIs 1.20, 2.23), risperidone (OR 1.63, 95% CIs 1.16, 2.31), olanzapine (OR 1.63, 95% CIs 1.12, 2.37), and prochlorperazine (OR 1.90, 95% CIs 1.06, 3.40). A significant association was not found for chlorpromazine (OR 1.36, 95% CIs 0.98, 1.87), quetiapine (OR 1.61, 95% CIs 0.57, 4.55) or aripiprazole (OR 2.79, 95% CIs 0.31, 25.37). However, substantial heterogeneity was found for most of these analyses. Further details of subgroup analyses of individual studies and summary estimates are shown in Figure 3.

Forest plot of individual studies and summary estimates of subgroup analyses on recurrent venous thromboembolism (VTE), study design, high-quality study, study population, types of antipsychotic agents (APS), potency, dose, and duration of APS use, individual APS.
Sensitivity analysis and publication bias
Sensitivity analysis was performed by eliminating each study in turn. The pooled ORs of association between APS exposure and VTE risks were not altered after omitting each individual study in turn. However, for APS and PE risks, the ORs were altered after eliminating three individual studies19,34,35 in turn, indicating that the result was not stable. The funnel plot was used to assess the publication bias, and the results were shown in Figure 4. Visual inspection of the funnel plot showed obvious asymmetry. Therefore, the publication bias needs to be considered.
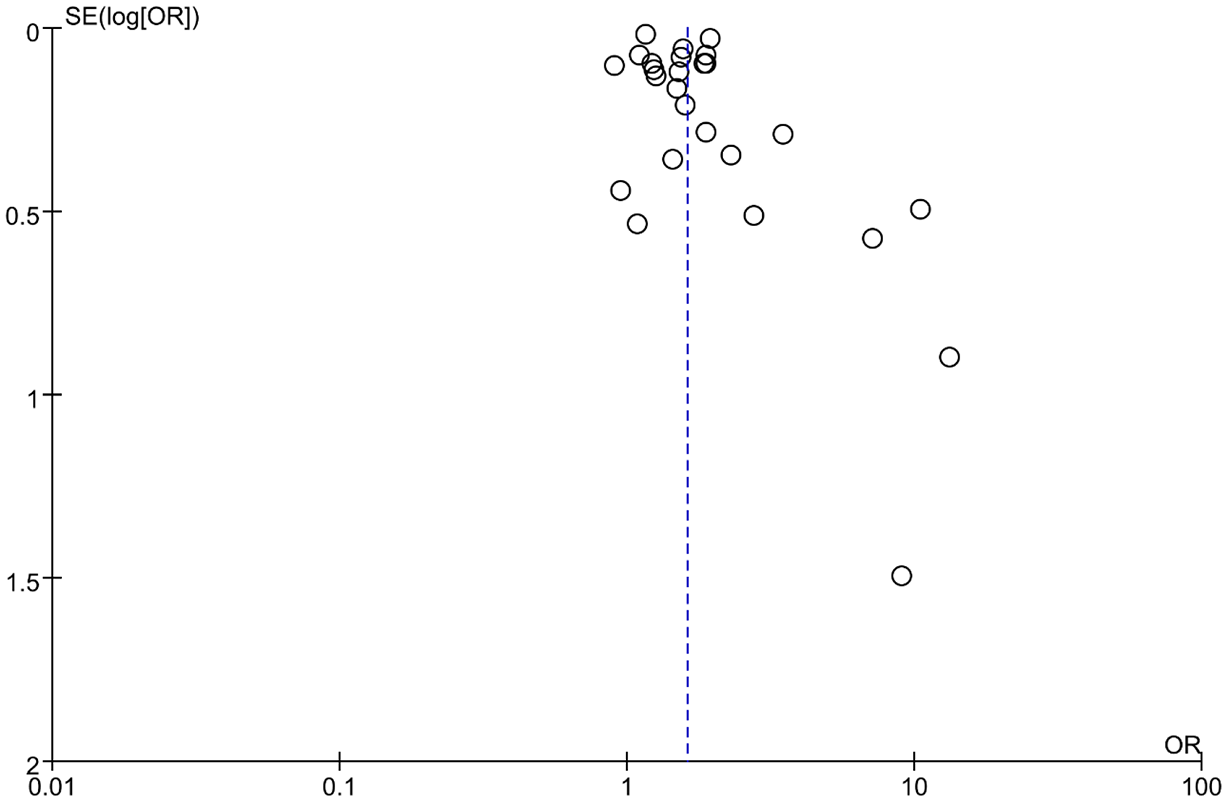
Funnel plot on the publication bias of all included studies.
Discussion
The aim of this meta-analysis is to ascertain the risk of VTE and PE among users of APS compared with non-users. Cohort and case-control studies were included in the review. Except for two studies with unclear definition,22,25 most of the studies identified VTE and PE by International Classification of Diseases codes, medical records, and objective tests, which can result in reliable VTE and PE diagnoses. The results of this study indicate that the risk of both VTE and PE is higher for APS users than non-users, with a pooled OR estimate of 1.55 (95% CIs 1.36, 1.76) for VTE and 3.68 (95% CIs 1.23, 11.05) for PE. For individual drugs, increased risk of VTE and PE was observed in taking haloperidol, risperidone, olanzapine, prochlorperazine, but not in chlorpromazine, quetiapine, or aripiprazole.
This finding is consistent with previously published meta-analyses that found correlations between APS use and VTE risk.17,18,20 However, compared with the most recent meta-analysis, 20 nine additional studies were identified in this review.13,21 –28 Our subgroup analysis also suggests that APS exposure increases the risk of VTE recurrence. This finding further implicates APS as an independent factor contributing to VTE. Subgroup analysis of cohort, case-control and high-quality studies also showed that VTE risk significantly increased in APS users, which strengthens the confidence of the results. However, the results still need to be interpreted with care because of high heterogeneity among the included studies. Our finding that APS use increased risk of PE is also consistent with previous studies, though the strength of this finding is limited by high heterogeneity among included studies and wide CIs.19,20 The present sensitivity analysis also suggested that this result is not stable, which indicates that the result of high risk of PE is not reliable.
As far as we know, this is the first meta-analysis of pooled comprehensive data based on five studies about APS new users and VTE risk reported by observational studies, indicating that VTE risk appears to be greater in new users.16,21,42,45,46 This result suggests that APS users in the initial stage are more vulnerable to VTE. Several possible explanations might be involved in this result. Continuing APS users were not observed to have as high a risk for VTE as new APS users, indicating that tolerance is increased with longer APS exposure. Another possible explanation is that some psychotic symptoms are related to VTE. Increased risk for VTE among patients with concurrent depressive, bipolar, and schizophrenic disorders was reported that implied psychiatric symptoms might take part in the process of VTE risk. 48 Psychiatric symptoms relieved after APS treatment may partially explain the higher risk in new APS users. In addition, the VTE test usually was arranged in the first few days of medication treatment, potentially leading to a biased overestimation of VTE risk in the initial stage. Interestingly, a similar finding was reported in a meta-analysis about APS exposure and myocardial infarction (MI) risk. 49 A common pathway might contribute to both MI and VTE in APS users.
FGA use and SGA use were both related to the risk of VTE, and the effect sizes are quite similar. Compared with the most recent review, three additional studies for evaluating VTE with FGA21,40,45 and two additional studies with SGA21,45 were included, and a consistent result was observed. The study conducted by Ballard
The mechanism of VTE risk and exposure to APS remains unclear. Psychotic disorders and APS might both play a role in the occurrence of VTE. Possible mechanisms that have been reported include immobilization, metabolic syndrome, antiphospholipid antibody increases, and hyperprolactinemia. Immobilization leads to circulatory stasis, which as one of Virchow’s triad is a well-known risk factor contributing to VTE. Many kinds of APS have been indicated to have a side effect of sedation, which can cause immobilization.
5
Also, symptoms of mental illness, such as negative symptoms of schizophrenia, dementia, and major depressive disorder, could lead to social withdrawal and immobilization. There is a significantly increased risk of weight gain involved in using a variety of APS, especially SGA, a relationship that has been known for decades. Metabolic syndrome and obesity could contribute to increased platelet activity
Antiphospholipid syndrome is frequently observed as an increase in anticardiolipin antibodies (aCL) and most commonly manifests as VTE, potentially relating to increased VTE risk among APS users. A significant positive relationship between serum IgM aCL and serum clozapine level was shown in a clinical trial,
53
but the opposite result was reported in another one.
56
Hyperprolactinemia is another underlying reason for APS use being related to high risk of VTE. It has been reported that hyperprolactinemia may contribute to coagulation disorder.
57
In addition, a positive correlation was observed between prolactin levels and some coagulation activation markers in male patients taking APS but not in females.
58
The other possible mechanisms include hyperhomocysteinemia, inflammatory process
This analysis has some limitations that should be considered. The most noticeable one is the high heterogeneity among included studies, which is similar to the previous meta-analysis.18,20 The heterogeneity can be observed in various aspects. First, the populations in each study are quite different. As reported above, VTE risk could be increased in first-episode drug-naïve schizophrenia patients. Hence the different study populations could result in different baseline VTE risks. Second, the definition of current use is inconsistent. We collected data on current APS exposure rather than recent or past exposure to mitigate heterogeneity caused by different durations of APS exposure. However, the definition of current exposure also was not consistent among studies; thus, VTE risk in APS users might be underestimated since greater risk was observed for short-term APS use. In addition, for some subgroup analyses, different definitions of subgroup exposure among included studies contributed to significant heterogeneity. For instance, for the subgroup analysis of the dose group, three studies defined a high dose as a chlorpromazine-equivalent dose over 100 mg.14,16,21 One study defined low-dose and high-dose group by chlorpromazine-equivalent dose ⩽150 mg, >300 mg.
46
Another one considered the low-dose group as ⩽25% of the maximum recommended in British National Formulary, and high dose as maximum and over.
42
It is worth noting that the present study directly combined the data in high and low-dose groups of these five publications that might result in statistical bias. Third, the inconsistent nature of non-users leads to different interpretations of the findings in each cohort study, which can also result in heterogeneity. For example, Ray
This comprehensive systematic review provides more information and strengthens the evidence on APS use and VTE risk for guideline developers, policymakers, and clinicians. In addition, the subgroup analyses draw clinician attention to an initial period of APS use and provide clues into the risk of individual APS. Therefore, the necessary physical health monitoring is needed to provide for psychiatric patients prescribed continuing antipsychotic medication in clinical practice. For example, the Two-level DVT Wells score assessment, D-dimer test and the proximal leg vein ultrasound scan can be considered for these patients, especially new antipsychotic drug users, in order to comprehensively evaluate the risk of VTE and PE. 62
Conclusion
The comprehensive meta-analysis indicates that both the significantly increased VTE and PE risk were found in current APS users compared with non-users. Subgroup analyses suggest that new users are more likely to develop VTE. Using haloperidol, risperidone, olanzapine, or prochlorperazine significantly increased the risk for VTE, but not chlorpromazine, quetiapine, or aripiprazole. However, high heterogeneity among studies and scarce data for individual APS calls for cautious interpretation of the findings from this review. Further studies could focus on specific populations of APS users and VTE risk of individual APS. In addition, more studies listing only PE as the outcome need to be conducted to provide more evidence based in clinical practice.
Supplemental Material
sj-pdf-1-tpp-10.1177_2045125320982720 – Supplemental material for Current antipsychotic agent use and risk of venous thromboembolism and pulmonary embolism: a systematic review and meta-analysis of observational studies
Supplemental material, sj-pdf-1-tpp-10.1177_2045125320982720 for Current antipsychotic agent use and risk of venous thromboembolism and pulmonary embolism: a systematic review and meta-analysis of observational studies by Yinzhao Liu, Jun Xu, Kacey Fang, Yue Xu, Ju Gao, Chao Zhou, Xiaowei Tang, Xinyu Fang, Jiu Chen, Chunming Xie, Fuquan Zhang, Xiangrong Zhang and Congjie Wang in Therapeutic Advances in Psychopharmacology
Footnotes
Author Contributions
Xiangrong Zhang, Congjie Wang, and Yinzhao Liu contributed to the design of the study. Yinzhao Liu, Jun Xu, Yue Xu, Ju Gao, Chao Zhou, Xiaowei Tang, Jiu Chen, Chunming Xie, Fuquan Zhang managed the data collection, analysis, and interpretation. Kacey Fang checked the data analysis and interpretation. Yinzhao Liu drafted the manuscript. Xiangrong Zhang, Congjie Wang and Kacey Fang contributed to revising the manuscript. All authors have approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key Research and Development Program of China (No. 2018YFC1314300 and 2016YFC1307002), the National Natural Science Foundation of China (81971255 and 81571314), Social Development Foundation of Jiangsu Province, China (No. BE2019610) and Jiangsu Provincial Medical Talent project (ZDRCA2016075).
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
