Abstract
Background
Venous thromboembolism (VTE) comprises deep vein thrombosis (DVT) and pulmonary embolism (PE). Chronic thromboembolic pulmonary hypertension (CTEPH) typically arises from acute pulmonary embolism. The pathogenesis of them involves multiple risk factors such as genetic predisposition. However, the findings from these studies are not entirely consistent. This study aims to investigate the association between FGA rs6050 polymorphism and susceptibility to thrombotic diseases.
Methods
We searched PubMed, OVID, Web of Science, Academic Search Ultimate, CNKI, and Wan Fang database. To assess the strength of associations, we calculated pooled odds ratios (ORs) and 95% confidence intervals (CIs) in different genetic models. Additionally, subgroup analyses, sensitivity analysis, and assessment of publication bias were also carried out.
Results
A total of 11 studies, including 9 reported results on VTE (3856 individuals [1545 cases]) and 3 on CTEPH (761 participants [350 cases]), revealed a significant association between the rs6050 polymorphism and susceptibility to both VTE and CTEPH. The A allele was consistently linked to an elevated risk of VTE across all genetic models (allele, homozygote, heterozygote, recessive, and dominant model), while it was also associated with an increased risk of CTEPH under all genetic models excluding the recessive model. Furthermore, subgroup analysis among ethnic groups revealed a significant association between rs6050 polymorphisms and VTE in both Caucasians and Asians under all genetic models. In Africans, the association with VTE was only observed for rs6050 polymorphisms in dominant and heterozygous models.
Conclusions
The FGA rs6050 polymorphism is positively associated with susceptibility to VTE and CTEPH.
Keywords
Introduction
The condition of venous thromboembolism (VTE) encompasses both deep venous thrombosis (DVT) and pulmonary embolism (PE). 1 A deep-vein thrombosis (DVT) is a blood clot that typically forms in the deep veins of the legs but can also affect the deep veins of the arms, the splanchnic veins, and the cerebral veins. Pulmonary embolism (PE) is caused by thrombi that originate elsewhere, typically in the pelvic region or deep veins of the leg, traveling to and occluding the arteries of the lung. 2 Chronic thromboembolic pulmonary hypertension (CTEPH) primarily results from acute pulmonary embolism. 3
VTE ranks globally as the third most prevalent acute cardiovascular syndrome, following myocardial infarction and stroke. 4 In epidemiological studies, the annual incidence of PE ranged from 39 to 115 cases per 100,000 population, while the incidence of DVT was found to be between 53 and 162 cases per 100,000 population. 5 The prevalence of VTE has shown a consistent upward trend based on multiple studies, encompassing both Caucasian and Asian populations.6–8 But the presence of significant racial disparities and a strong genetic correlation have been observed in several studies on VTE.9–11 The development of VTE is influenced by a spectrum of risk factors that include both acquired and genetic elements. Risk factors for venous thromboembolism include advanced age, prolonged immobilization, surgery, fractures, use of oral contraceptives and hormone replacement therapy, pregnancy, puerperium, cancer and antiphospholipid syndrome. 12 In addition to these well-established risk factors for venous thrombosis, numerous gene mutations have been documented to be associated with an increased vulnerability to VTE. 13
The pathogenesis of CTEPH is thought to be attributed to pulmonary thromboembolism and subsequent pulmonary vascular remodeling. This condition is characterized by persistent occlusions in the pulmonary arteries, leading to a progressive elevation in pulmonary pressure and eventual right heart failure. 14 The prevalence of CTEPH following PE ranges from 0.5% to 9%. 15 As a long-term complication of PE, the prognosis of CTEPH is characterized by high mortality rates. 16 Numerous genetic mutations have been reported to be linked with susceptibility to CTEPH.17–20
Fibrinogen (Fg), also known as clotting factor I, plays a pivotal role in the coagulation process. Fibrinogen is an integral component of the clotting cascade and undergoes conversion into insoluble fibrin during clot formation, which is essential for the stabilization of the formed blood clot. Research has shown that the levels of fibrinogen are significantly associated with single nucleotide polymorphisms (SNPs) and haplotypes on the FGA gene. 21 The presence of genetic variation can lead to abnormal synthesis and functional irregularities of fibrinogen, thereby causing aberrant blood coagulation processes and increasing the risk of VTE. Furthermore, the FGA Thr312Ala (rs6050) polymorphism has been a focal point of investigation. Several studies have reported an increased risk of thrombotic disease in individuals with this specific polymorphism.20,22,23 Additionally, some studies have demonstrated an elevated risk of stroke associated with the rs6050 polymorphism, highlighting the broad implications of genetic variability in fibrinogen on cardiovascular health.24,25
Numerous studies have reported an association between the FGA Thr312Ala polymorphism and conditions such as deep vein thrombosis, acute pulmonary embolism, and CTEPH.20,22,23 However, conflicting evidence suggests that FGA polymorphism may not be linked to susceptibility to pulmonary embolism.26,27 The inconsistent and contradictory results from previous studies have generated controversy in this field. Furthermore, although PE is a component of VTE, it is a life-threatening condition that significantly contributes to cardiovascular-related mortality, warranting special attention. Additionally, the limited sample size of case-control studies on VTE and CTEPH necessitates comprehensive integration of various data. Lastly, considering variations in genetic risk factors across populations, conducting a meta-subgroup analysis involving different races and regions can unveil heterogeneity in the occurrence of VTE and CTEPH among diverse populations. 28 Therefore, we conducted a comprehensive meta-analysis to further elucidate the relationship between FGA rs6050 polymorphism and susceptibility to VTE and CTEPH, while also conducting a separate study focused on the association with PE.
Materials and Methods
Literature Search
The databases of PubMed, OVID, Web of Science, Academic Search Ultimate, Chinese National Knowledge Infrastructure, and Wan Fang were systematically searched to collect eligible case-control studies, cohort studies and cross-sectional studies investigating the association between FGA rs6050 gene polymorphisms and the risk of VTE or CTEPH. The retrieval time was up to June 2024. In order to avoid missing any potentially relevant literature, references from the included articles were also traced and manually searched. The search terms were as follows: (“Fibrinogen” OR “Thr312Ala” OR “FGA” OR “Fib2” OR “AMYLD2” OR “rs6050” OR “Factor I”) AND (“Polymorphism” OR “Variation” OR “Diversity” OR “Genotype”) AND (“Venous Thromboembolism” OR “Thromboembolism” OR “Pulmonary Embolism” OR “Embolism” OR “Pulmonary Hypertension”).
Inclusion and Exclusion Criteria
The current meta-analysis implemented rigorous inclusion and exclusion criteria. The inclusion criteria of the search encompassed the following: research employing cohort, case-control, or cross-sectional designs; studies investigating the association between Fibrinogen Aα Thr312Ala gene polymorphisms and risks of VTE or CTEPH; the enrolled patients in the case group were confirmed by the diagnostic criteria of VTE or CTEPH; studies that provided genotype distributions; studies that provide sufficient data to calculate odds ratios (ORs) and 95% confidence intervals (CIs).
The exclusion criteria were primarily based on the following: studies lacking sufficient information regarding genotype distribution, studies lacking a healthy control group, reviews, meta-analyses, animal experiments, case reports, abstracts-only articles, unpublished studies, and data that had been duplicated in other studies.
Data Extraction
Two investigators independently screened the potential eligible literature and extracted data. First, the title and abstract of citations obtained through the search strategy were screened for eligibility. Next, the full text of all potentially eligible trials was retrieved and further evaluated to exclude studies not relevant to the topic. In case of disagreement, a third reviewer was consulted for a final decision. To acquire missing data, corresponding authors of included studies were contacted by email.
The data extracted from individual studies encompassed thrombotic disorder category, first author, year of publication, country, subject ethnicity, article type, number of cases, number of controls, source of control subjects, genotyping method, genotype distribution, and Hardy Weinberg Equilibrium (HWE) tests in the control group. HWE test is a method used to assess whether a sample has reached genetic equilibrium.
Quality Assessment
The Newcastle Ottawa Scale (NOS) was utilized for evaluating the quality of case-control studies, 29 while for cross-sectional studies, the criteria established by the Agency for Healthcare Research and Quality (AHRQ) were applied. 30 The quality assessment of the included studies was independently conducted by two investigators. In cases where disagreements arose and consensus could not be reached, a third party was consulted to make the final decision. Genotype distribution was examined using exact HWE through chi-square tests. A study is deemed high-quality if it achieves NOS scores ≥7 points or AHRQ scores ≥8 points, with a P-value for HWE exceeding the 0.05 threshold.
Statistical Analysis
Hardy-Weinberg equilibrium (HWE) tests of each study were conducted in our analysis for evaluation of included populations. The association between rs6050 polymorphisms and susceptibility to VTE or CTEPH was evaluated by calculating odds ratios (ORs) and their corresponding 95% confidence intervals (CIs) using a fixed or random-effect model based on the quantification of the heterogeneity. The range of I2 is between 0 and 100, representing the extent of heterogeneity observed among studies. If there was no statistical heterogeneity observed among the results (I2 statistic <50%), the fixed-effect model (Mantel-Haenszel method) would be employed for meta-analysis. If statistical heterogeneity existed (I2 statistic >50%), an investigation into the source of heterogeneity would be conducted, and the random effect model (Inverse Variance Method) would be adopted. Subgroup analyses were also performed according to ethnicity. The overall and subgroup analyses were both performed in five genetic models: allele model (T vs A), homozygous model (TT vs AA), heterozygote model (TT vs AT), dominant model (TT vs AT + AA), and recessive model (AA vs AT + TT). We thought it was statistically significant between rs6050 polymorphism with VTE or CTEPH if P < 0.05. Sensitivity analyses were also carried out to examine the stability of synthetic results. Furthermore, publication bias was investigated through the calculation of Egger's test and the construction of Begg's funnel plot. In cases where publication bias was detected in Egger's test (with p-value ≤0.05), we utilized the trim-and-fill method to identify and correct for funnel plot asymmetry arisingfrom publication bias. All statistical analyses were performed by using Stata version 18.0 (Stata Corporation).
This meta-analysis was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and adhered to the recommendations outlined in the PROSPERO database for systematic review and meta-analysis protocols.
Results
Study Characteristics
A total of 1262 studies were initially identified through a systematic search. After eliminating 79 duplicated articles, 1183 studies remained for screening. Upon reviewing the titles and abstracts, 1143 studies were excluded as they were either animal experiments or unrelated to the topic under investigation. Subsequently, a detailed full-text review of the remaining 40 studies resulted in the exclusion of another 29 studies for various reasons: 12 were excluded due to being meta-analyses, review articles, or case reports; while 17 were excluded because of insufficient data or duplication of previous findings. Ultimately, a total of 11 studies met the inclusion criteria and were included in the current meta-analysis.20,22,23,26,31–37
Among the 11 studies, 9 studies involving 1545 cases and 2311 controls focused on VTE (including 4 studies involving 325 cases and 526 controls focused on PE) and 3 studies involving 350 cases and 411 controls focused on CTEPH. In terms of VTE, three studies were based on the Caucasian population, five were based on the Asian population, and two studies were based on the African population. In the subset of research investigating the rs6050 polymorphism in relation to PE, one study focused on the Caucasian population, while three studies were conducted on the Asian population. As for CTEPH, two studies were conducted on the Caucasian population and one study was conducted on the Asian population.
It should be mentioned that Laura J. Rasmussen-Torvik's study 32 included a mixed population, providing specific genotype distributions for both the Caucasian and African populations. Due to the intricate nature of genotypic distribution among different racial groups, genotype distribution of independent ethnicity was included in the ethno-based stratified subgroup analysis. Figure 1 illustrates the flowchart of selection and inclusion process. Table 1 summarizes the general characteristics of all included studies. Table 2 presents the detailed rs6050 polymorphism genotype distributions and the P-value of HWE in each control group. According to the NOS quality evaluation system, the quality of the 10 case-control studies were further evaluated in detail. For the cross-sectional study, the AHRQ system was utilized. Detailed information can be found in Table 3 and Table 4.

Flow diagram of selection and inclusion process.
Characteristics of Included Studies.

Year, publication year. Abbreviations: VTE, venous thromboembolism; PE, pulmonary embolism; CTEPH, chronic thromboembolic pulmonary hypertension; PCR, polymerase chain reaction.
Genotype Distribution and HWE.

Abbreviations: VTE, venous thromboembolism; PE, pulmonary embolism; CTEPH, chronic thromboembolic pulmonary hypertension.
*P value for Hardy–Weinberg equilibrium test in controls.
NOS Quality Evaluation Form.

①
AHRQ Quality Evaluation Form for the Cross-Sectional Study (Mustafa Çörtük 2023 20 ).

Quantitative Synthesis
Meta-Analysis of Association Between rs6050 Polymorphisms and VTE Susceptibility
Nine studies22,23,26,32–37 addressing the association between rs6050 and VTE were included in the analyses, comprising five studies from China, two studies from the USA, one study from the UK, and one study from France. These studies involved a sample size of 1545 VTE cases and 2311 controls. The pooled results demonstrated that rs6050 polymorphism was significantly related to the risk of VTE in the allele model (OR = 1.49, 95%:1.23–1.80, P < 0.001; I2 = 67.4%, P = 0.001) (Figure 2A), homozygote model (OR = 1.89, 95%:1.33–2.67, P < 0.001; I2 = 51.5%, P = 0.029) (Figure 2B), heterozygote model (OR = 1.45, 95%:1.25–1.67, P < 0.001; I2 = 47.9%, P = 0.045) (Figure 2C), dominant model (OR = 1.52, 95%:1.33–1.74, P < 0.001; I2 = 49.1%, P = 0.039) (Figure 2D) and recessive model (OR = 1.54, 95%:1.26–1.88, P < 0.001; I2 = 45.2%, P = 0.058) (Figure 2E).

Forest plots for association between rs6050 polymorphism and VTE in different genetic models.
We also conducted subgroup analyses according to ethnicity. In the Caucasian group, a significant association was observed between the rs6050 mutation and the risk of VTE in allele (OR = 1.32, 95%:1.15–1.51, P < 0.001; I2 = 0.0%, P = 0.697), homozygote (OR = 1.65, 95%:1.11–2.44, P = 0.012; I2 = 25.1%, P = 0.263), heterozygote model (OR = 1.36, 95%:1.13–1.65, P = 0.001; I2 = 0.0%, P = 0.375), dominant (OR = 1.40, 95%:1.17–1.66, P < 0.001; I2 = 0.0%, P = 0.649), and recessive model (OR = 1.44, 95%:1.04–1.99, P = 0.028; I2 = 51.2%, P = 0.129). Similarly, an evident association between the rs6050 mutation and VTE risk was observed in the Asian group in allele (OR = 1.97, 95%:1.26–3.08, P = 0.003; I2 = 81.5%, P < 0.001), homozygote (OR = 2.74, 95%:1.39–5.40, P = 0.004; I2 = 62.6%, P = 0.030), heterozygote (OR = 1.57, 95%:1.18–2.07, P = 0.002; I2 = 69.5%, P = 0.011), dominant (OR = 1.81, 95%:1.39–2.36, P < 0.001; I2 = 71.6%, P = 0.007), and recessive model (OR = 1.93, 95%:1.44–2.59, P < 0.001; I2 = 29.5%, P = 0.225). In African group, a significant association was also discovered between rs6050 mutation and VTE risk in dominant model (OR = 1.55, 95%:1.07–2.24, P = 0.021; I2 = 22.7%, P = 0.255) and heterozygote model (OR = 1.67, 95%:1.13–2.47, P = 0.009; I2 = 19.2%, P = 0.266). It can be concluded that while allele (OR = 1.21, 95%:0.93–1.57, P = 0.149; I2 = 0.0%, P = 0.483), homozygote (OR = 1.19, 95%:0.67–2.11, P = 0.565; I2 = 0.0%, P = 0.515), and recessive model(OR = 0.88, 95%:0.52–1.51, P = 0.644; I2 = 0.0%, P = 0.919) showed an insignificant correlation between the FGA rs6050 polymorphism and the susceptibility of VTE in African population, other genetic models indicated a significant correlation (Supplementary Figure S1).
Meta-Analysis of Association Between rs6050 Polymorphisms and PE Susceptibility
Four studies23,26,35,37 addressing the correlation between rs6050 and PE were included in the analyses, with three conducted in China and one in the UK, encompassing a total of 325 PE cases and 526 controls.
The pooled results demonstrated that rs6050 polymorphism was significantly related to the risk of PE in the allele model (OR = 2.09, 95%:1.15–3.79, P = 0.015; I2 = 85.3%, P < 0.001) (Figure 3A), dominant model (OR = 2.04, 95%:1.01–4.14, P = 0.047; I2 = 76.0%, P = 0.006) (Figure 3B), recessive model (OR = 2.13, 95%:1.41–3.22, P < 0.001; I2 = 46.6%, P = 0.132) (Figure 3C), and homozygote model (OR = 2.92, 95%:1.15–7.44, P = 0.024; I2 = 68.6%, P = 0.023) (Figure 3D) excluding the heterozygote model (OR = 1.81, 95%:0.87–3.78, P = 0.111; I2 = 75.2%, P = 0.007) (Figure 3E).

Forest plots for association between rs6050 polymorphism and PE in different genetic models.
Meta-Analysis of Association Between rs6050 Polymorphisms and CTEPH Susceptibility
Three studies20,26,31 addressing the correlation between rs6050 and CTEPH were included in the analyses, with one study conducted in China, one in the UK, and one in Turkey, encompassing a total of 350 CTEPH cases and 411 controls. The pooled results demonstrated that rs6050 polymorphism was significantly related to the risk of CTEPH in the allele model (OR = 1.47, 95%:1.19–1.82, P < 0.001; I2 = 0.0%, P = 0.502) (Figure 4A), dominant model (OR = 2.35, 95%:1.27–4.37, P = 0.007; I2 = 59.8%, P = 0.083) (Figure 4B), heterozygote model (OR = 2.57, 95%:1.18–5.57, P = 0.017; I2 = 71.7%, P = 0.029) (Figure 4C), and homozygote model (OR = 2.28, 95%:1.35–3.83, P = 0.002; I2 = 0.0%, P = 0.602) (Figure 4D) excluding the recessive model (OR = 1.04, 95%:0.39–2.79, P = 0.932; I2 = 76.5%, P = 0.014) (Figure 4E).
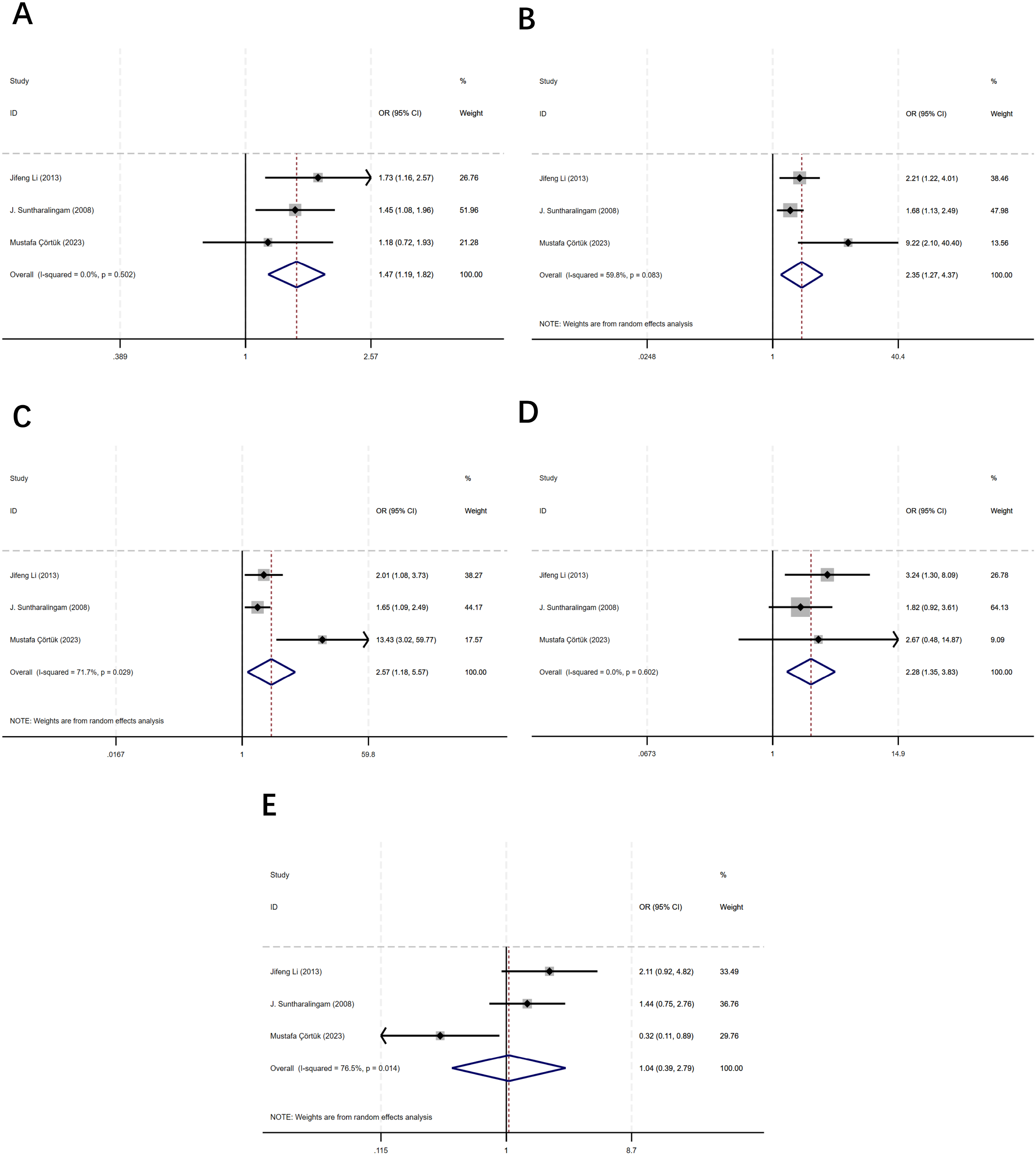
Forest plots for association between rs6050 polymorphism and CTEPH in different genetic models.
Sensitivity Analysis
We conducted sensitivity analysis to evaluate the stability of the results in the association between rs6050 polymorphism and the risk of VTE and CTEPH by removing one study at each round of the analysis. As it is shown in Supplementary Figures S2-4, removing each study respectively from the meta-analysis did not change the initial result. It suggests that this meta-analysis provides more reliable evidence to prove the association between the rs6050 and VTE or CTEPH.
Publication Bias
We utilized Egger's (Supplementary Figures S5-7) and Begg's tests (Supplementary Figures S8-10) to assess the presence of publication bias for each genetic model. The results of Egger's test revealed that all models had P-values above the threshold (P > 0.05), except for the heterozygote model of PE (P = 0.019), the allele model of VTE (P = 0.032), the dominant model of VTE (P= 0.002), and the heterozygote model of VTE (P = 0.006), as well as the dominant model of CTEPH (P = 0.029). These specific models exhibited indications of publication bias. To further validate our findings, a trim-and-fill analysis was conducted on the five models that displayed publication bias in Egger's test. The symmetry observed in funnel plots in Supplementary Figure S11 after the trim-and-fill adjustment indicates that our study results remain robust. Thus, despite signs of publication bias observed in certain specific models, our study maintains its reliability and validity after adjustment.
Discussion
VTE is a significant health concern encompassing both DVT and PE.4,38 DVT is characterized by abnormal blood clot formation in deep veins, commonly observed in the lower extremities. PE is predominantly believed to occur due to detachment of a blood clot from the deep veins of the lower extremities, leading to subsequent blockage of the pulmonary artery. Despite their pathological connection, DVT and PE exhibit significant differences in terms of clinical presentation and treatment strategies. 39 Moreover, owing to its diverse clinical manifestations and potential lethality, PE can rapidly induce hemodynamic changes resulting in acute right heart failure or even death; thus necessitating urgent research attention on PE that deserves special consideration. It should be noted that several studies investigating rs6050 gene polymorphism did not differentiate between different types of VTE which may impact research accuracy. Therefore, our meta-analysis specifically focuses on VTE while separately discussing studies related to PE. CTEPH is a chronic condition that arises following PE, typically as a consequence of acute PE. 3 It manifests as persistent pulmonary hypertension and has the potential to culminate in right ventricular failure and mortality.
FGA gene encodes the alpha chain of fibrinogen, a crucial component involved in blood clotting. SNPs within the coding sequence of FGA gene can result in structural and functional alterations of the protein, which are associated with various clinical manifestations.22,40,41 Significantly, the FGA AαThr312Ala (T/A, rs6050) variant in the coding region of fibrinogen has been extensively investigated and closely associated with the onset of VTE and CTEPH.20,23 A recent exome-wide association study involving 14723 cases and 334315 controls identified sixteen genes associated with VTE through protein-coding variants. 42 Our meta-analysis reports the association of one of the polymorphisms (rs6050) in these genes with VTE susceptibility.
Numerous studies have documented the prevalence of the rs6050 polymorphism across diverse ethnicities and geographical regions. A large-scale candidate gene association study including various ethnic groups reported the T allele frequency of 0.761 in Europeans (with the A allele at 0.239) and 0.641 in Africans (with the A allele at 0.359). In Asians, the T allele frequency was noted to be 0.553 (A allele at 0.447), and in Hispanics, it was 0.732 (A allele at 0.268). 43 A research conducted among Iranian women indicated that the FGA rs6050 polymorphism is relatively common, with the wild-type T allele and mutant A allele frequencies at 59% and 41% in recurrent pregnancy loss (RPL) patients, and 55.3% and 44.7%, respectively, in the control group. 44 Additionally, Rasmussen-Torvik et al determined that the frequency of the FGA rs6050 Ala allele is approximately 0.24 in the general white population, which increases to 0.28 in patients with VTE. 32 These findings underscore the global distribution of the rs6050 Thr312Ala polymorphism and stress the significance of examining this polymorphism across different ethnicities and geographical areas.
Our findings of the current meta-analysis of VTE suggest that individuals carrying the A variant allele of rs6050 have an elevated risk of developing VTE across diverse populations, including those of Asian, Caucasian, and African. According to our meta-analysis of PE, we demonstrated that the A variant allele could serve as a risk factor for PE, which nearly doubles the risk of PE. However, due to the limited number of included studies and small sample sizes, we did not conduct a subgroup analysis based on ethnicity. According to our meta-analysis of CTEPH, we demonstrated that the A variant allele could serve as a risk factor for CTEPH among the population of Asian, Caucasian and Turkish.
In conclusion, allelic gene A serves as a significant risk factor in the development of VTE (including PE) and CTEPH. Following sensitivity analysis, the final pooled results demonstrate high quality and reliability.
Several hypotheses can elucidate the mechanism underlying the relation between Thr312Ala (A/T) polymorphism and VTE. Studies have shown that the Thr312Ala polymorphism significantly influences clot structure and rigidity by modulating FXIII-induced α-chain crosslinking.45,46 Notably, the presence of the Ala312 allele potentially enhances FXIII's crosslinking efficacy, thereby promoting the formation of a thick and resilient fibrin network, which may contribute to an increased predisposition to thrombotic diseases. 46 Interestingly, although the Ala312 variant has been identified as a risk factor for VTE, no association was found between rs6050 and fibrinogen level. 47 Uitte et al found that the linkage disequilibrium (LD) was detected between the rs6050 polymorphism and the FGG-H2 haplotype of the fibrinogen gamma gene, which is associated with reduced levels of FGG prime(γ’). 48 Therefore, the rs6050 polymorphism may indirectly facilitate the occurrence of VTE by affecting the FGG gene.
The controversy surrounding the rs6050 polymorphism and its association with PE is indeed a subject of heated debate. Our meta-analysis has shown that the Ala allele is a potential risk factor for PE. Previous meta-analysis have shown that the rs6050 polymorphism is associated with ischemic stroke and the reason for this may be the functional role of rs6050 on fibrinclot formation. 49 Carter et al not only identified the rs6050 mutation as a risk factor for PE, 23 but also discovered a significant interaction between the rs6050 polymorphism and post-stroke mortality in patients with atrial fibrillation. 25 Altogether, these findings supported that the rs6050 polymorphism could influence clot stability, thereby increasing the risk of clot migration from lower limb veins to pulmonary arteries. This is consistent with the results of our meta-analysis. FGA Thr312Ala might affect the stability of blood clots by increasing the brittleness of fibrin clots, making them more likely to rupture under the influence of blood flow and migrate into the pulmonary artery, increasing the likelihood of pulmonary embolism. 50 However, Li JF and Le Gal G contend that this variation in peptide sequence does not influence the risk of PE.26,34 This is not consistent with the results of our meta-analysis. A possible reason for this could be that not all patients in Le Gal G's study conducted thoracic investigations, and thus we cannot exclude the possibility that some of them had asymptomatic pulmonary embolism, potentially biasing the results. Additionally, Li JF's study was limited to an Asian population, so the different results might be explained by possible genetic variations among individuals from different ethnic or geographic origins. Similarly, Zeng et al suggest that rs6050 may act as a protective factor of ischemic stroke by down-regulating fibrinogen levels in serum, which contrasts with our conclusion. 51 They consider rs6050 as a possible “candidate gene” responsible for cerebral hemorrhage (CH). However, when analyzing rs6050 alone, there is no significant association with sporadic CH. Therefore, rs6050 may not be a functional CH site, but may only be found in LD status. Moreover, the small sample size may also be a reason. It is important to conduct larger-scale randomized controlled trials (RCTs) and eliminate the effects of LD.
Our meta-analysis also reveals that the Ala allele of rs6050 is a risk factor for CTEPH. The Ala allele leads to increased cross-linking of α-chains, which can result in thicker fibers, impede physiological thrombolysis, and thus increase susceptibility for CTEPH development. 52 Given the significant association of the Thr312Ala polymorphism with both PE and CTEPH found in our meta-analysis, the potential of this genetic variant as a biomarker for distinguishing between CTEPH and PE is highly debated. However, in the majority of studies concerning VTE, CTEPH is frequently not assessed. The sample size we included is too small. Moreover, although the rs6050 genetic variant could potentially influence thrombus formation, thereby contributing to the development of CTEPH, it is important to note that some cases of CTEPH are not thrombus-related. We have not addressed CTEPH cases caused by non-thrombotic factors. Future research with larger sample sizes from RCTs is needed to explore the relationship between non-thrombotic CTEPH and genetic factors.
Although we found no significant racial differences in our study, previous research has investigated the association between rs6050 variations and VTE risk in different populations. A genome-wide association study (GWAS) and a small candidate gene association study in populations of European ancestry have demonstrated a positive association between the Ala allele of rs6050 and D-dimer levels, which is a known biomarker for thrombosis. 53 Additionally, Weng et al confirmed the association between D-dimer and FGA rs6050 in African Americans. 43 It is also noteworthy that D-dimer levels were higher in African Americans and lower in Asians. 54 These findings suggest that the relationship between the rs6050 polymorphism and VTE risk may not be consistent across all populations, indicating the need for further research to understand how this genetic variant affects VTE risk in different ethnic groups.
The present meta-analysis has several potential limitations that warrant discussion. Firstly, the analysis was limited by the inclusion of only 11 case-control or cross-sectional studies, which not only hindered stratified or subgroup analyses but also potentially increased a risk of false-positive findings due to the relatively small sample size. Secondly, there was significant heterogeneity among the studies regarding PE and CTEPH. This heterogeneity could be attributed to various factors, including differences in participant recruitment, criteria for case and control selection, and diagnostic methodologies. Moreover, confounding factors such as gender, age, history of traumatic surgery, and tumor history might have influenced the results’ heterogeneity. It is worth noting that VTE is a complex multifactorial disease with an incidence closely associated with age. Recent studies have emphasized that fibrinogen function evolves with age.55,56 However, due to limitations in the original research materials used for this meta-analysis, subgroup analyses based on aging stratification were not performed. Given these constraints and considering both inherent limitations of included studies and our analysis approach, further RCTs are essential to validate our findings conclusively. Such trials would play a crucial role in developing personalized prevention and treatment strategies for VTE and CTEPH in future research. Finally, LD is a crucial factor in assessing the genetic architecture of complex traits and diseases. LD could exist between the FGA rs6050 variant and other genetic loci, which could affect the expression and function of the FGA gene. Literature has mentioned the existence of linkage disequilibrium between rs6050 and FGG genes, suggesting that the association of the rs6050 polymorphism might be affected by linkage with FGG. 48 This highlights the need for future research to systematically investigate LD patterns around this variant within the FGA gene. LD and whole genome sequencing analyses will not only refine our understanding of the genetic basis of VTE, but will also provide valuable insights into the functional impact of FGA gene variations.
Despite certain limitations, this meta-analysis still has several luminescent spots. It makes notable contributions as the pioneering study in exploring the ambiguous link between FGA gene polymorphisms and the risk of VTE and CTEPH. So far, this is the first known meta-analysis study aimed to clarify the vague correlation between rs6050 gene polymorphisms and the risk of VTE and CTEPH. By shedding light on the correlation between FGA gene polymorphisms and susceptibility to VTE and CTEPH, this research enables healthcare professionals to better assess individual patients’ genetic profiles. This advancement paves the way for personalized prevention and treatment strategies. Elucidating the relationship between FGA rs6050 gene polymorphisms and VTE/CTEPH may facilitate the early identification of individuals at elevated risk prior to the manifestation of clinical symptoms, thereby contributing to a more proactive approach in disease management. However, given the observed low relative risks and considerable heterogeneity across studies, it is prudent to interpret this variant with caution. Further research is crucial in order to solidify these preliminary findings. This entails conducting studies with larger sample sizes that encompass a wider range of ethnic groups while adhering to stringent methodological protocols. In parallel, experimental research is essential to uncover the underlying mechanisms that validate the clinical implications of FGA rs6050 gene polymorphisms, to ensure that these genetic insights translate into tangible benefits for patient care.
Conclusions
The current meta-analysis concluded that the rs6050 polymorphism of FGA significantly elevates the risk of VTE and CTEPH. This locus shows potential for its application in future clinical genetic diagnostics and therapeutic approaches. However, additional case-control studies with high quality are necessary to bolster the findings of this meta-analysis.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296251314476 - Supplemental material for Association of Fibrinogen Aα Thr312Ala (rs6050) Polymorphism with Venous Thrombosis and Chronic Thromboembolic Pulmonary Hypertension: A Meta-Analysis
Supplemental material, sj-pdf-1-cat-10.1177_10760296251314476 for Association of Fibrinogen Aα Thr312Ala (rs6050) Polymorphism with Venous Thrombosis and Chronic Thromboembolic Pulmonary Hypertension: A Meta-Analysis by Han Cheng, Haozhe Yang, Yantong Zhang, Zhanxu Wei, Lei Xia and Jing Yang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent Statements
Not applicable.
Funding
This work was supported by grants from National Natural Science Foundation of China(82200353), Jiangsu Province Double Innovation Doctoral Program(JSSCBS20221948), Suzhou Gusu Health Talent Program((2022)043), Suzhou Gusu Health Talent Plan Talent Research Project(GSWS2022014), Suzhou Science and Technology Innovation Policy Funding Project, Jiangsu Provincial Higher Education University Student Innovation and Entrepreneurship Training Program(202410285087Z), The Multi-center Clinical Research Project for Major Diseases in Suzhou(Grant Number: DZXYJ202302) and “Bo Xi” Talent Casting Plan of the First Affiliated Hospital of Soochow University. The funders had no roles in study design,data collection and analysis,decision to publish,or preparation of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
