Abstract
Objectives:
In the UK, nine different compounds are available as long-acting antipsychotic injections (LAIs). There are few clinical guidelines for determining which LAIs are most effective in specific patient groups. To measure the clinical effectiveness of LAIs we aimed to determine the now-established concept of antipsychotic discontinuation rates and measure Clinical Global Impression (CGI) outcomes.
Method:
The population (n was approximately 560,000) was a secondary care NHS adult mental health service in Lanarkshire, Scotland, UK. This was a retrospective, electronic case note search of LAI-naïve patients commenced on paliperidone palmitate (n = 31), risperidone long-acting injection (RLAI) (n = 102) or zuclopenthixol decanoate (n = 105), with an 18-month follow up. Kaplan–Meier survival statistics for discontinuation rates and hospital admission were calculated. CGI severity and improvement scores were retrospectively assigned by the investigating team.
Results:
Paliperidone palmitate performed less favourably than risperidone long-acting injection (RLAI) or zuclopenthixol decanoate. Paliperidone palmitate had higher discontinuation rates due to any cause, inefficacy and increased hospitalization risk. Paliperidone palmitate had the smallest proportion of patients assigned a clinically desirable CGI-I score of 1 (very much improved) or 2 (much improved).
Conclusions:
Paliperidone palmitate had less favourable discontinuation and CGI outcomes compared with RLAI and zuclopenthixol decanoate. This could not be adequately explained by patients in the paliperidone group being more chronically or severely unwell, nor by the presence of comorbidities such as alcohol or substance misuse, or by the use of lower mean dosages compared with RLAI or zuclopenthixol decanoate. We considered that prescribers are familiarizing themselves with paliperidone and outcomes may improve over time.
Introduction
Increasing numbers of second-generation antipsychotics have been made available in long-acting injectable (LAI) form, expanding the choice in the UK to nine different compounds. There are few recommendations or guidelines for prescribers in terms of which LAI to select [Kane and Garcia-Ribera, 2009; Llorca et al. 2013] and there is a dearth of head-to-head comparison trials available to bridge this gap [Covell et al. 2012; McEvoy et al. 2014; Fleischhacker, 2009]. First-episode psychosis [Kahn et al. 2008] and cost effectiveness [Achilla and McCrone, 2013] studies have reported mixed results generally favouring second-generation over first-generation antipsychotics. For chronic schizophrenia, the advantage of oral second-generation antipsychotics remains unclear [Leucht et al. 2009; Goff et al. 2005; Geddes et al. 2000]. The question of clinical effectiveness (combining measures of efficacy plus tolerability) remains for LAIs and whether the new second-generation LAIs offer superior clinical and cost effectiveness. Since 2003, three of the most frequently prescribed LAIs within our health board organization were paliperidone palmitate (paliperidone), risperidone long acting injection (RLAI) and zuclopenthixol decanoate (zuclopenthixol) and were chosen for evaluation. They provided a comparison of a well-established first-generation LAI (zuclopenthixol decanoate) against two second-generation LAIs. We aimed to measure clinical effectiveness using the established concept of antipsychotic discontinuation [McEvoy et al. 2014; Kahn et al. 2008; Goff et al. 2005]. We also aimed to examine improvement by assigning Clinical Global Impression (CGI) [Guy, 1976] scores (severity and improvement).
Methods
NHS Lanarkshire is a health board in Scotland serving a population of around 560,000 people, with a spread of deprivation and rural- and urban-based areas. Patients in contact with secondary-care mental health services have an electronic record on a document management system, Genisys (see http://www.genisystechnology.co.uk/) that has been used since 2002. This system has search functionality allowing keywords to be identified in all files held within the database. For this study, permission to search the database was granted from the local Caldicott Guardian and the study was registered with NHS Lanarkshire’s Clinical Quality Department. No other ethical issues related to patient care or confidentiality were identified due to the study’s retrospective, case or chart note nature.
The Genisys database was searched for generic and trade name of the three LAIs, [paliperidone (Xeplion®), risperidone (Risperdal® Consta®) and zuclopenthixol decanoate (Clopixol®)], as well as subtle variations if common spelling errors were noted. Importantly, this initial population was filtered to include only LAI-naïve patients. Records that contained insufficient information to allow analysis were excluded, as was one record for which the LAI being used was for challenging behaviour in a patient with moderate learning disability. No further exclusions were applied.
Demographic and clinical variables
Information was obtained from the electronic patient records and is listed in Table 1. Coprescription with adjunctive oral antipsychotics was recorded if it was a regular (not an ‘as required’ or ‘prn’ prescription) and was present from examining clinical correspondence for greater than 50% of the time the patient was on the LAI of interest. Among our study population, no patients received clozapine to augment their LAI. The proportion and combined dosage of patients who received adjunctive oral antipsychotics was also considered a possible marker of LAI effectiveness, the hypothesis being that additional oral antipsychotics would be prescribed if the LAI was deemed ineffective as a sole agent; this is the subject of the accompanying paper. Duration of contact with psychiatric services and a record of clozapine being trialled before the prescription of the LAI of interest were considered putative markers of illness severity and treatment refractoriness.
Clinical and demographic details of patients commenced on long-acting injections.

BNF, British National Formulary; LAI, long-acting injectable; CI, confidence interval; APP, antipsychotic polypharmacy; RLAI, risperidone long-acting injectable.
Includes drug-induced psychoses.
For RLAI alcoholic hallucinosis (n = 2), avoidant personality disorder (n = 1). For zuclopenthixol decanoate schizotypal disorder (n = 1).
χ2 = 9.6, d.f. = 2, p = 0.008 (psychotic versus nonpsychotic disorders).
χ2 = 36.8, d.f. = 8, p < 0.001 (second-generation versus others).
χ2 = 5.9, d.f. = 4, p = 0.21.
F(2,91) = 1.6, p = 0.2.
Discontinuation, hospitalization and clozapine switch
The time to LAI treatment discontinuation within 18 months for any cause was the main outcome measure of interest and was regarded as a determinant of clinical effectiveness. This 18-month period was chosen as it is clinically regarded as a meaningful time to assess the effects of a first-prescribed LAI and provided comparison with the figures from the widely influential CATIE trial [Goff et al. 2005]. For medications discontinued before 18 months, the reason was established from the electronic records and if necessary, consultation between the authors and individual clinicians. Discontinuation was further categorized to inefficacy, adverse effects, or other reasons. Where more than one reason was listed, the most significant clinical reason by consensus decision among the authors was chosen for analysis. A further proxy marker of effectiveness is that of hospital admission. Theoretically, if an antipsychotic is considered clinically effective, in terms of preventing relapse of psychosis, be it oral or LAI, it should prolong the time before admission to psychiatric hospital is required [McEvoy et al. 2014; Kishimoto et al. 2013]. For each LAI, if a patient was admitted to an acute psychiatric unit during the 18 months follow up, the date of this admission was recorded and used to calculate the ‘time to admission outcome’. In a similar fashion, time to starting clozapine was also recorded, as a marker of treatment refractoriness prior to initiation of the LAI and also putatively towards LAI treatment failure due to emergent treatment refractoriness.
Clinical Global Impression
The clinical status of individual patients was assessed using the CGI score for severity and improvement. The patient records had CGI severity (CGI-S) scores assigned at the outset of treatment with the long-acting antipsychotic injection and then after 18 months or the date of discontinuation, whichever came sooner. The CGI-I was also assigned retrospectively, according to information within the clinical records. All CGI scoring was assigned by four of the authors, all clinical psychiatrists (MC, PS, SM and MB) working within the Health Authority. Individual patient records were assigned randomly to the investigators, to reduce possible inter-rater discrepancy. Throughout data collection, there were regular discussions and joint assessments to reduce this further. Raters involved clinically and directly with patients records were asked to give the CGI assignment to author MC who also repeated all CGI measures independently of the other three assessors. At the conclusion of data collection, our CGI scores suggested a strong degree of inter-rater correlation. CGI-I scores were aggregated into three categories, (A) = (1) very much improved and (2) much improved, (B) = (3) minimally improved and (4) no change, and finally category (C) = (5) minimally worse, (6) much worse and (7) very much worse. The reason being that all LAIs are powerful, long-term psychotropic medications with significant adverse effects and some are potentially costly compared with others. As clinicians, we should strive for the best gains (an ‘A’ rating) in terms of CGI improvement, that is, CGI-I (1) and (2) from interventions such as LAIs and cautiously review all other ‘improvement’ categories, particularly if harm is being done. We felt that a CGI-I of category (B) that was (3) minimally improved and (4) no change, was of low clinical utility as it reflected patients exposed to medications they had little control over, risk of adverse effects and the possibility of no significant clinical gain.
Analysis
The results were tabulated using Microsoft Excel 2007, which was also used for the majority of statistical analysis. Histograms were created to assess the distribution of numerical data. Continuous data was reported as means with 95% confidence intervals (CIs) and compared using analysis of variance and two-tailed t tests as appropriate. Categorical data were analysed by way of chi-square tests, with significance levels requiring a p value of less than 0.05. Kaplan–Meier survival curves were computed using MedCalc statistical software for Microsoft Windows (http://www.medcalc.org), to calculate percentages, censored data and to demonstrate time to discontinuation, hospitalization or switch to clozapine. Overall differences within the three groups were assessed for significance using the log-rank test [Bland and Altman, 2004]. Differences between LAIs in terms of risk for discontinuation, hospitalization and time to clozapine were presented as hazard ratios (HR) with 95% CIs.
Results
A total of 651 patients were found with mention of at least 1 of the LAIs of interest. Of these patients, 394 were excluded as they had previously received another LAI. A further 18 patients were excluded due to insufficient information in their case record for data collection: RLAI (n = 3), zuclopenthixol decanoate (n = 15), χ2 = 10.6, d.f. = 2, the p value was equal to 0.005. The remaining 238 eligible patient records comprised paliperidone palmitate (n = 31), RLAI (n = 102) and zuclopenthixol decanoate (n = 105).
Table 1 shows pretreatment demographic and clinical features for the three LAI patient cohorts. There was a statistically significant increased proportion of female patients in the zuclopenthixol decanoate group at 54% compared with 32% for paliperidone palmitate and 29% for RLAI, χ2 = 14.3, d.f. = 2, p = 0.0008. Patients commenced on zuclopenthixol decanoate also tended to be older with a mean age of 42.4 years compared with 36.0 for paliperidone palmitate and 35.1 for RLAI (F(2,235) = 9.6, p = 0.001). Between the three LAIs there were no significant differences in length of contact with psychiatric services or the use of compulsory measures to provide treatment without the patients’ consent. There was a proportionally higher number of patients with a primary diagnosis of bipolar affective disorder who received treatment with paliperidone palmitate. Stratifying patients into psychotic and nonpsychotic disorders revealed a greater proportion of patients (86%) with psychotic disorder treated with RLAI, χ2 = 9.6, d.f. = 2, p = 0.008. A greater proportion of patients on paliperidone palmitate (61%) were coprescribed an antidepressant medication compared with RLAI (41%) and zuclopenthixol (42%), although this was not statistically significant, χ2 = 4.3, d.f. = 2, p = 0.12. Patients receiving RLAI were less likely to be prescribed a mood stabilizer: RLAI (11%), zuclopenthixol decanoate (27%), paliperidone palmitate (29%), χ2 = 10.5, d.f. = 2, p = 0.005. Almost half of zuclopenthixol decanoate patients were treated with at least one additional antipsychotic during the 18-month period, this approached statistical significance (χ2 = 5.7, d.f. = 2, p = 0.057). Expressed as a percentage of the maximum dose stated in the BNF, zuclopenthixol decanoate was used at lower doses than paliperidone palmitate or RLAI. Patients prescribed zuclopenthixol decanoate were also significantly less likely to have recorded comorbid alcohol misuse at 25%, χ2 = 9.1, d.f. = 2, p = 0.01; or drug misuse at 22%, χ2 = 8.2, d.f. = 2, p = 0.01. There were low numbers of patients having had previous trials of clozapine, therefore statistical testing was not performed.
Figure 1 demonstrates Kaplan–Meier survival curves calculated for discontinuation, hospitalization and clozapine switch. The statistics for this are shown on Table 2.

Kaplan–Meier graphs for discontinuation events.
Kaplan–Meier tests for discontinuation, hospitalization and clozapine switch.
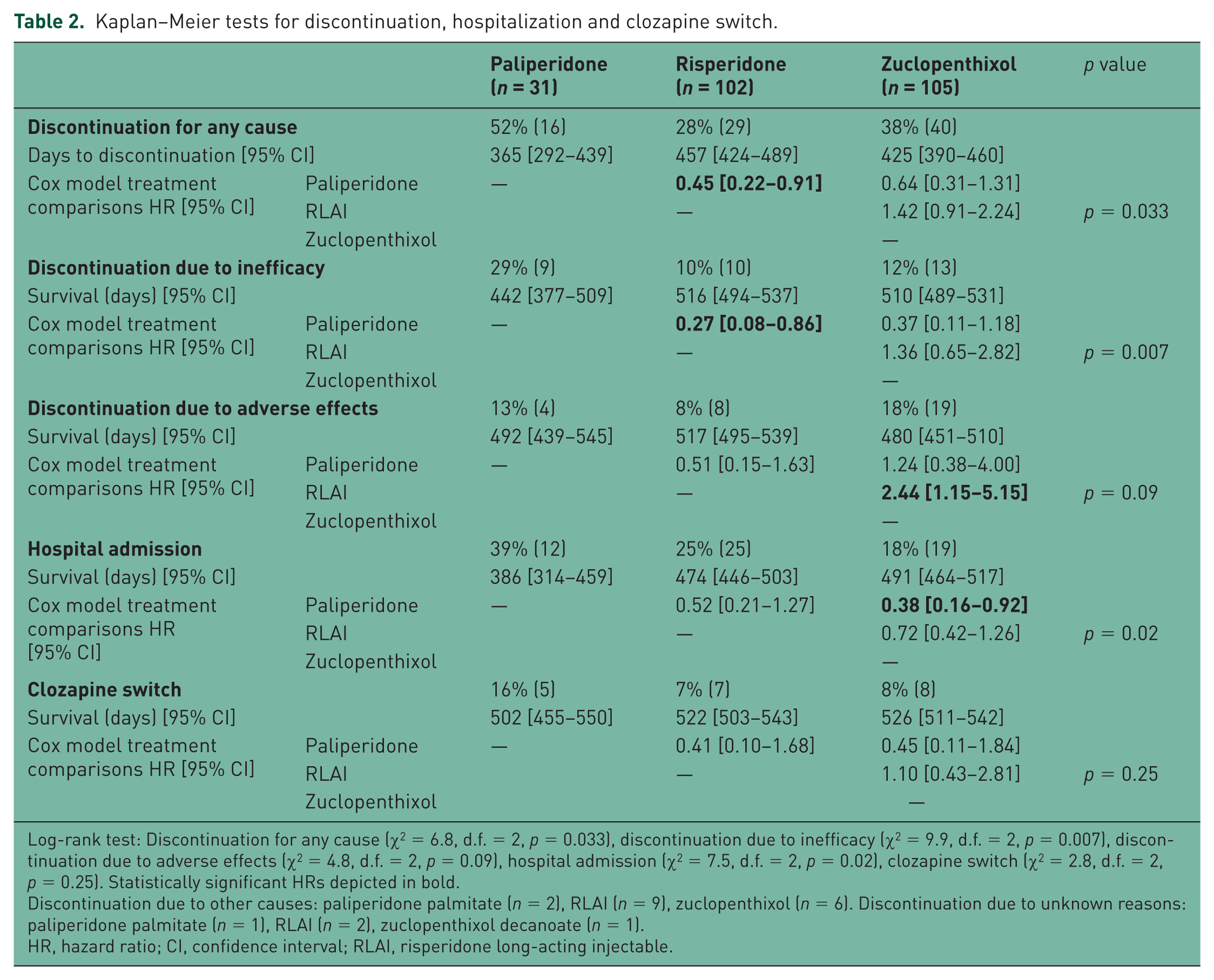
Log-rank test: Discontinuation for any cause (χ2 = 6.8, d.f. = 2, p = 0.033), discontinuation due to inefficacy (χ2 = 9.9, d.f. = 2, p = 0.007), discontinuation due to adverse effects (χ2 = 4.8, d.f. = 2, p = 0.09), hospital admission (χ2 = 7.5, d.f. = 2, p = 0.02), clozapine switch (χ2 = 2.8, d.f. = 2,p = 0.25). Statistically significant HRs depicted in bold.
Discontinuation due to other causes: paliperidone palmitate (n = 2), RLAI (n = 9), zuclopenthixol (n = 6). Discontinuation due to unknown reasons: paliperidone palmitate (n = 1), RLAI (n = 2), zuclopenthixol decanoate (n = 1).
HR, hazard ratio; CI, confidence interval; RLAI, risperidone long-acting injectable.
Table 2 shows results for treatment discontinuation, hospitalization and clozapine switch. These results are derived from Kaplan–Meier survival analysis and refer to days to treatment discontinuation, hospitalization and clozapine switch. The log-rank chi-square test result is given to show overall differences within the groups and Cox model treatment comparisons using HRs with 95% CIs are given to show the differences between the LAIs using paliperidone as the comparator drug.
Treatment discontinuation for any cause differed between the LAIs and was highest in the paliperidone group. This was statistically significant when compared with RLAI [HR = 0.45 (95% CI 0.22–0.91), p = 0.03], but not when compared with zuclopenthixol decanoate.
Treatment discontinuation due to inefficacy also differed between the LAIs and was highest in the paliperidone group. This was statistically significant when compared with RLAI [HR = 0.27 (95% CI 0.08–0.86), p = 0.007], but not when compared with zuclopenthixol. Treatment discontinuation due to adverse effects did not differ significantly between the LAIs, although showed a trend (p = 0.09) with lowest discontinuation due to adverse effects associated with RLAI. The risk of hospital admission differed between the LAIs and was highest in the paliperidone group. This was statistically significant when compared with zuclopenthixol [HR = 0.38 (95% CI 0.16–0.91), p = 0.02], but not when compared with RLAI.
Table 3 depicts CGI scores for severity and improvement during treatment. The mean CGI at start of treatment was similar for all three LAIs (paliperidone = 4.5, RLAI = 4.6 and zuclopenthixol = 4.5, F(2,235) = 0.11, p = 0.89). The end-point CGI differed (higher for paliperidone compared with RLAI and zuclopenthixol) but this failed to achieve statistical significance (F(2,235) = 2.4, p = 0.096). Analysis of variance did not show statistically significant differences for mean CGI-I scores (F(2,235) = 1.8, p = 0.18). However, following stratification of CGI-I into aggregated groups, (A) = (1) and (2), (B) = (3) and (4), and (C) = (5), (6) and (7), paliperidone was associated with the lowest proportion of patients being assigned CGI-I category (A) (1, very much improved, or 2, much improved); χ2 = 11.0, d.f. = 4, p = 0.026.
Clinical Global Impression severity and improvement outcomes.

RLAI, risperidone long-acting injectable; CGI-S, Clinical Global Impression score severity; CGI-I, clinical global impression score improvement; CI, Confidence Interval; *CGI groups tested, (A) versus (B & C combined) (χ2 = 11.8, d.f. = 4, p = 0.02).
Discussion
The likelihood of discontinuation due to any cause, discontinuation due to perceived clinical inefficacy and psychiatric hospitalization was highest for paliperidone. The likelihood of discontinuation due to adverse effects was similar for all three LAIs. Compared with RLAI and zuclopenthixol, treatment with paliperidone palmitate was associated with the smallest proportion of patients assigned a desirable CGI-I score of 1 (very much improved) or 2 (much improved). We consider why paliperidone was associated with these unfavourable outcomes.
Clinical and demographic descriptions
There were fewer men prescribed zuclopenthixol compared with paliperidone and RLAI, of interest, given its association with reduction in aggression [Haessler et al. 2009], which is commoner in men. Patients on zuclopenthixol were older than those taking RLAI or paliperidone palmitate. This may be a reflection of the higher proportion of women, and a tendency for women to develop psychotic illnesses at an older age [Castle et al. 1998]. Paliperidone was prescribed in a significantly higher proportion of individuals with bipolar affective disorder than either of the other LAIs, in keeping with evidence to its effectiveness as an antimanic treatment [Vieta et al. 2010]. This may also be linked to the significantly higher coprescription of antidepressants in patients receiving paliperidone. The lower rates of comorbid alcohol and substance misuse with zuclopenthixol in our study may have influenced a more favourable outcome whilst for paliperidone, higher rates of alcohol and substance misuse may have influenced a more unfavourable outcome, although this may not be the case as similar comorbidity was seen with RLAI. Our comorbidity rates were of interest and inconsistent with a randomized 6-month trial showing lower severity of alcohol and substance misuse following RLAI treatment [Rubio et al. 2006].
Future studies examining prospective changes in alcohol and drug misuse following LAI treatment will be of significant clinical interest. There were no significant differences in previous treatment with clozapine, important as these individuals would be considered treatment resistant and improvement with any LAI would be harder to achieve. Interestingly, RLAI which performed favourably across discontinuation and CGI measures had a higher proportion of patients previously treated with clozapine. Time to clozapine initiation did not differ between LAIs, suggesting that all three groups had similar proportions of treatment-refractory patients, consistent with the consensus that between 20% and 30% of patients with schizophrenia require clozapine therapy [Correll et al. 2009].
Treatment discontinuation and other markers of effectiveness
52% of patients started on paliperidone palmitate discontinued treatment before 18 months, higher than both RLAI (28%) and zuclopenthixol (38%). The reason for discontinuation of paliperidone palmitate in the majority of cases was inefficacy. This is consistent with the pattern recorded in the CGI severity and improvement subscales. There is a notable step in the Kaplan–Meier (discontinuation due to inefficacy) curve at the 100-day mark. Clinical experience suggests that beneficial effects of LAIs emerge after three doses. For paliperidone, this being 100 days, suggested prescribers allowed sufficient time to elapse before making discontinuation decisions regarding inefficacy. Discontinuation due to adverse effects showed no significant differences between the LAIs. However, 50% of those discontinuing zuclopenthixol did so due to adverse effects, more than either paliperidone (25%) or RLAI (28%). This was in keeping with the propensity for first-generation antipsychotic drugs, such as zuclopenthixol, to cause poorly tolerated sedation and dopaminergic side effects including bradykinesia and rigidity. Proportionally more patients on paliperidone (39%) required admission during the 18-month follow up and median time to admission was shortest by approximately 100 days. Admission rates are of particular interest given that over the period of introduction of paliperidone (2010 onwards), acute psychiatric admissions beds in the health authority reduced by one third; the assumption being that admission threshold to hospital prior to 2010 was lower and would be reflected in increased admission rates for RLAI and zuclopenthixol, which was not the case. Hospital admission has significant implications for patients and additional financial cost to healthcare systems. Aside from small differences in primary diagnosis and age, there were no other differences between LAI cohorts that explained different discontinuation rates.
Assessment of illness severity using Clinical Global Impression
The mean starting severity of illness was similar between the LAIs, between moderately (where CGI-S was 4) and severely (where CGI-S was 5) unwell. However, there were differences at the end of treatment or 18 months. Patients receiving paliperidone had the highest mean endpoint CGI-S scores (3.5), a change of 1 point on the CGI-S from start of treatment, with more favourable outcomes in the RLAI (mean CGI-S was 3.0) and zuclopenthixol (mean CGI-S was 2.9) cohorts. Our study showed CGI-S outcomes at 18 months or discontinuation in keeping with the EUFEST study [Kahn et al. 2008], where patients achieved end-point severity outcomes at 12 months of borderline ill (where CGI-S was 2) to mildly ill (where CGI-S was 3). This was encouraging, as our patients were unlikely to be in their first episode of psychosis and were more likely to have shown significant past contact with psychiatric services (over two thirds having contact greater than three years, indicating generally more chronic illness). When mean CGI-I scores were used, there were no statistically significant differences between LAIs. Forming the CGI-S ratings into categories (A, B and C) suggested that compared with zuclopenthixol or RLAI, paliperidone was associated with a significantly lower proportion of patients assigned to the clinically desirable category A (where CGI-I was 1 or 2). These less favourable CGI-S and improvement category outcomes for paliperidone may be a reflection of the lower proportion of patients with a primary psychotic illness suggesting that LAIs bring about greater improvements in psychotic rather than nonpsychotic disorders.
Limitations
Given the retrospective nature of this study, there is reliance on quality documentation of clinical findings. In most cases, it was possible to make a reasoned judgment about patient details and illness severity. Accurately measuring the titration rate of the LAI dose was not possible, partly due to the wide variety of dosing strategies for zuclopenthixol decanoate (one to four weekly), and variable quality of prescription change recording. Potentially, incorrect titration and low immediate prediscontinuation dose may arguably be of greater importance for paliperidone palmitate where less favourable effectiveness was shown. However, the high median doses of paliperidone palmitate suggest that it was used in proposed therapeutic [Scottish Medicine Consortium, 2011] and higher doses than RLAI zuclopenthixol decanoate. In some electronic case records, there was a dearth of information and these patients were excluded (as outlined in methods). There is also a risk that descriptions about illness severity were not conveyed accurately in the records available, but this was not unique to one particular LAI and thus should not bias findings towards one particular LAI. However, this may influence the magnitude of effect detected. The CGI was originally designed to be used prospectively, but has been used retrospectively by ourselves [Shajahan et al. 2008, 2010], and others [Barbee et al. 2004; Centorrino et al. 2005] for similar studies; it may not have the ability to define changes in specific symptoms, however is arguably more applicable to routine clinical practice and carries more meaning for patients and clinicians. The consistent findings between CGI results and objective markers such as discontinuation or hospitalization suggested that efforts to minimize inter-rater differences were successful. Time taken to hospitalization, although a useful marker of effectiveness, is at risk of influence from other variables, such as a patient’s social and family support, that are unrelated to the LAI medication. However, we feel it can be reasonably assumed within our free-to-access National Health Service, that these factors (social and family support) were distributed across all three LAI cohorts and consequently unlikely to bias results.
The effectiveness of paliperidone palmitate
Our study was a noninterventional, naturalistic study examining prescribing patterns and outcomes for the three most-prescribed LAIs in the local health board. Such naturalistic studies describe a broad range of patients and give outcome data that can be helpful in routine clinical practice [Centorrino et al. 2005]. Our findings suggest that for our main outcome measure, all cause discontinuation; paliperidone palmitate did not perform as favourably as RLAI or zuclopenthixol decanoate. In addition, paliperidone was associated with a higher risk of hospital admission compared with RLAI and was not associated with desired levels of CGI-I scores (1) and (2). Aside from higher rates of discontinuation due to adverse effects in the zuclopenthixol decanoate group, which did not reach statistical significance, there was little to distinguish RLAI and zuclopenthixol decanoate in terms of clinical effectiveness. The relatively unfavourable outcomes with paliperidone cannot be explained by evidence that the patients in that group were more severely or chronically ill to start with or more likely to be treatment refractory. There was evidence that patients on paliperidone and RLAI had a greater proportion of comorbid alcohol and drug misuse (almost double the proportion compared with that recorded for zuclopenthixol decanoate). This still does not adequately explain the difference in effectiveness between paliperidone and RLAI. We had previously seen mixed and some unfavourable discontinuation rates with RLAI compared with zuclopenthixol five years after the introduction of RLAI to our locality [Shajahan et al. 2008]. Five years later, there is little to differentiate zuclopenthixol from RLAI across the measures of effectiveness that we examined. It is possible that there were unknown factors resulting in the unfavourable outcomes for the relatively new paliperidone. It is possible that a similar pattern will be seen over time with improved effectiveness, as clinicians grasp a fuller understanding of patients most likely to benefit from paliperidone. This study requires replication with a greater number of patients commenced on paliperidone. It is possible that we have not arrived at the correct dosing structure or schedule for paliperidone.
As stated in a recent editorial in JAMA Psychiatry ‘…the need to integrate specific knowledge, general knowledge, and sometimes best hunches in clinical care of individual patients’ [Carpenter and Buchanan, 2015] may be something that evolves after physicians are given time to become familiar with a medication. Further open trials with a broad range of patients may help us decide whether there are true differences in clinical effectiveness between these LAIs. Meanwhile, we welcome the introduction of new LAIs to provide a wider range of treatments available for patients.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
All the authors are employed by the NHS. MT has received hospitality and fees from Janssen; Lundbeck; Otsuka; and Roche in the last 3 years.
