Abstract
Background:
Non-adherence to antipsychotics in schizophrenia is associated with an increased risk of psychotic relapse and hospitalization, a risk that is reduced with the use of long-acting injectable (LAI) antipsychotics. Randomized clinical trials (RCTs) have demonstrated the efficacy of paliperidone palmitate 3-monthly (PP3M) for psychotic relapse prevention in schizophrenia, but it remains poorly documented among individuals treated in real-life settings who can benefit the most out of LAIs.
Objectives:
The objective of this study was to evaluate the effectiveness of PP3M in relapse prevention among patients with schizophrenia.
Methods:
This is a multicentre retrospective study conducted in four outpatients’ clinics across Canada. All consecutive patients with a main diagnosis of schizophrenia who initiated PP3M between June 2016 and March 2020 were included. The primary outcome was psychotic relapse, defined using broad and clinically relevant criteria.
Results:
Among 178 consecutive patients who were switched to PP3M, the 12-month relapse rate was 18.5% and the relapse-free survival probability was 0.788 (95% confidence interval [CI] = 0.725–0.856). Comorbid diagnoses of personality disorders and substance use disorders were associated with hazard rates (HRs) of 3.6 (95% CI = 1.8–7.3, p < 0.001) and 3.1 (95% CI = 1.6–6.2), respectively. Increased psychopathology severity was associated with an increased likelihood of relapse, while having a job or being in school was protective.
Conclusion:
These findings reinforce the necessity of conducting research in patients with comorbid psychiatric disorders who are typically underrepresented in RCTs, yet overrepresented in real-life settings, in order to better inform and guide clinical practice.
Keywords
Introduction
Long-acting injectable (LAI) antipsychotics, such as paliperidone palmitate 1-monthly (PP1M), are considered standard of care in the treatment of schizophrenia spectrum psychotic disorders as these treatments help overcome the barrier of poor treatment adherence, which is a frequent cause of relapse.1–3 Paliperidone palmitate is the only second-generation antipsychotic with a 3-monthly LAI formulation. Randomized controlled trials (RCTs) have shown that paliperidone palmitate 3-monthly (PP3M) significantly delays time to relapse versus placebo, while its efficacy is non-inferior to that of PP1M (i.e. relapse-free rates at 48 weeks of 91.2% and 90.0%, respectively).4,5 However, results from RCTs need to be interpreted cautiously before generalizing them to all patients. 6 Indeed, individuals included in RCTs are generally not representative of those treated in real-life settings, as they are usually not on community order treatment, are highly adherent to treatment, and are free of common psychiatric comorbidities such as substance use disorders and personality disorders. 7 Regarding patients with a dual diagnosis of schizophrenia and substance use disorders, evidence suggests an increased risk of relapse in those individuals, even when receiving LAI antipsychotics. 8
To address the limited ability of phase III RCTs to generate findings that can be generalized to patients commonly encountered in clinical practice, observational studies conducted in naturalistic settings are pivotal. These offer the advantages of capturing a wider range of patients by having less stringent inclusion/exclusion criteria, and/or by having less burdensome assessments, and/or by relying on information collected during routine follow-up. This latter allows data to be extracted directly from patients’ medical records without the need for consent, therefore allowing the inclusion of more difficult-to-treat individuals such as those typically prescribed LAIs.
To our knowledge, only a total of five naturalistic studies examining the use of PP3M have been published. First, Fernández-Miranda et al. 9 prospectively assessed the effectiveness of switching to PP3M 84 patients with severe schizophrenia who had been stabilized with PP1M for 2 years. Over the 2-year period following the switch to PP3M, a significant decrease in the number of psychiatric ward admission was observed in comparison with when patients were on PP1M (4.8% versus 10.7%). However, study consent was required, thereby introducing a potential selection bias as such a requirement often precludes the inclusion of less adherent patients, and psychiatric comorbidities were not reported, thereby limiting the ability to fully characterize the study population. Second, Garcia-Portilla et al. 10 prospectively evaluated PP3M effectiveness which, although a phase IIIb study, could be considered naturalistic as it was carried out in real-life settings. While the primary end point examined was symptomatic remission, they also observed decreased hospitalization rates during the 12-month treatment period with PP3M compared with the previous 12 months (4.6% versus 13.5%). Consent was again required for study participation and patients with psychiatric comorbidities, including substance use disorder, were excluded, thereby limiting the representativeness of their sample. Third, Li et al. 11 retrospectively compared 428 patients with non-affective disorders on PP3M with 1136 matched patients on PP1M using the Multi-State Medicaid Database in the United States. After an average follow-up period of around 15 months, they observed a significantly lower relapse rate (10.5% versus 15.7%) and better treatment adherence for patients on PP3M compared with PP1M. While such a study design allows for a high level of generalizability given the large population evaluated, the exclusive reliance on health claims data introduces several limitations, including information bias due to incomplete or inaccurate data. Fourth, Ferraro et al. 12 reported that 5 out of 14 patients (35.7%) switched from PP1M to PP3M had relapsed within the following year, a rate numerically higher than the 8.8% reported in Savitz et al. 5 pivotal trial, which they hypothesized could be due to the relatively high proportion of patients with substance use disorder in their sample (57.1%). While this study relied on information included in case notes, thereby avoiding the selection bias that may occur when requiring signed informed consent, its limited sample size limits the strength of the conclusions. A fifth study that was reported in two separate papers described the outcomes of a prospective cohort of consecutive patients switched from PP1M to PP3M. Wallman et al. 13 limited their analyses to patients with schizophrenia diagnoses only and found that only 8 patients (7.2%) among 111 had relapsed during the 2 years following PP3M initiation. Clark et al. 14 included in their analyses patients with diagnoses other than schizophrenia and examined factors associated with relapse. Among their enlarged sample of 186 patients initiated on PP3M, there were 20 relapses (10.8%) during the same 2-year period; they were more likely to occur in patients with a non-schizophrenia diagnosis, receiving polypharmacy or who had previously used clozapine. Although this study was conducted in a naturalistic setting and that there was no consent required, only primary psychiatric diagnoses were reported in both articles.
In sum, there are several limitations inherent to the naturalistic studies published so far which significantly hinder the ability to expand the obtained results to more complex patients who could nevertheless greatly benefit from the use of PP3M. Some of these limits could be addressed by conducting an observational study based on a large sample of patients in which the prevalence of psychiatric comorbidities is closer to what is encountered in clinical practice, notably by having less stringent inclusion/exclusion criteria.
To address these limitations, we herein retrospectively investigated the effectiveness of PP3M use in all patients switched from PP1M to PP3M during the observation period in four clinics representing real-life settings. Furthermore, we studied the role of a variety of clinical factors in predicting relapse in these patients, including psychiatric comorbidities.
Methods
Study design and population
This was a multicentre retrospective observational study carried out in four psychiatric outpatient clinics across Canada, including two first-episode psychosis (FEP) intervention programs. All consecutive patients treated at the four participating sites who initiated PP3M between 1 June 2016 and 1 March 2020 were identified through medical records. To obtain as representative a sample as possible, very broad inclusion criteria were used: being aged 18 or more; having a main diagnosis of schizophrenia spectrum psychotic disorder [based on Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5)], including schizophrenia, schizoaffective disorder, schizophreniform disorder, brief psychotic disorder, delusional disorder, unspecified psychosis; having received at least one dose of PP3M during the observation period. Hence, patients who received a single injection of PP3M during the study period were nevertheless included in this observational study to avoid a selection bias that could have resulted from excluding people for whom PP3M was discontinued early. There were no exclusion criteria.
Data collection
Data were retrospectively extracted from patients’ medical records by the research team at the time of first PP3M injection, at each following injection and, if applicable, at the time of PP3M discontinuation or at the end of the follow-up period, that is, 1 March 2021. Information collected at PP3M initiation included demographics (i.e. gender, date of birth, ethnicity, employment status, living arrangements), DSM-5 psychiatric diagnoses (primary and comorbid), duration of psychotic disorder, number and duration of psychiatric hospitalizations in the previous 2 years, community treatment order, criminal history (i.e. guilty of a criminal offense or unfit to stand trial), type of clinical setting (i.e. FEP program, assertive community treatment team, general outpatient psychiatric clinic, hospitalization ward, prison, other), pharmacological treatment history (i.e. total duration of exposure to antipsychotics, concomitant psychotropic medication, total duration of exposure to PP1M, last dose of PP1M received and date, number of consecutive injections at that dose before switching to PP3M), as well as psychopathology severity (Clinical Global Impression – Severity [CGI-S]). At each PP3M injection, the following information was collected: date of injection, PP3M dose received, reason for dose or interval change if applicable, any addition of oral antipsychotics (including treatment indication), any reported adverse event, and, if applicable, date of PP3M discontinuation as well as reason for cessation. Up until the end of data collection, the following data were retrieved: dates of admission/discharge of any psychiatric hospitalizations following PP3M initiation (date of data collection if ongoing) and reason(s) for the hospitalization.
Outcomes
The main outcome variable was relapse, which was a priori defined as an increase of psychotic symptoms requiring either (1) stopping PP3M in favor of another antipsychotic due to a relapse, (2) increasing PP3M dose or shortening the injection interval by more than 2 weeks, (3) supplementing with an oral antipsychotic due to psychotic symptoms, and (4) a psychiatric hospitalization. This broad definition of relapse was favored to ensure that the outcome captured could more accurately reflect symptoms exacerbation interfering with patients’ functioning, that is, not limited only to those requiring hospitalization. 15 Secondary outcomes were also used to facilitate comparisons with previous studies: (1) psychiatric hospitalization only; (2) PP3M discontinuation, which was defined as not receiving PP3M for 4 months (3-month regular dose interval + 1 month) due to either relapse, insufficient efficacy, side effects or patient’s decision. Time to discontinuation was calculated from PP3M initiation to date of discontinuation or to loss to follow-up.
Statistical analyses
Patients’ demographic and clinical characteristics as well as treatment-related factors are presented using descriptive statistics. Time to relapse was calculated using the Kaplan–Meier estimator and represented in Kaplan–Meier curves. Associations between independent variables and time to relapse were examined using Cox proportional hazard models. Analyses included all information gathered during the 12-month period following PP3M initiation. Multivariate analyses were also carried out; Cox models were adjusted for potential confounding factors that were identified using the causal directed acyclic graph (DAG) approach. With this knowledge-driven method, confounding factors are selected by drawing DAGs representing causal relationships between variables of interest and the outcome. Unlike data-driven methods for confounders selection, such as forward and backward stepwise regression, which introduce significant bias and lead to erroneous confidence intervals, the DAG approach avoids the adjustment for non-confounding variables such as colliders and mediators. 16 Statistical analyses were performed with R version 4.0.4 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patients’ characteristics
A total of 178 patients were included in this study; their characteristics are presented in Table 1. The sample was primarily male (83.7%) and Caucasian (73.0%), with a mean age of 33.9 years. At PP3M initiation, the most frequent diagnosis was schizophrenia (62.9%), and the average duration of illness was 4.6 years. A majority of patients (54.5%) had at least one psychiatric comorbidity, among which substance use disorder and personality disorder were the most frequent (34.3% and 18.5%, respectively). In the 2 years prior to PP3M initiation, 50.6% of the patients had been hospitalized at least once for psychiatric reasons and mean CGI-S was 3.1 at PP3M initiation. At the time of the first PP3M injection, 72 patients (40.4%) were treated in a FEP program, 47 (26.4%) in a general outpatient psychiatric clinic, and 19 (10.7%) in an assertive community treatment team.
Patients’ characteristics.

ADHD, attention-deficit/hyperactivity disorder; CGI-S, Clinical Global Impression–Severity; SD, standard deviation.
Treatment characteristics
Treatment characteristics are detailed in Table 2. Patients had been treated with PP1M for an average of 2.4 years and the most frequent last doses received were 100 and 150 mg (35.6% and 35.1%, respectively). PP3M was initiated at doses of 350 and 525 mg for 36.5% and 39.9% of the patients, respectively. The dosage switch from PP1M to PP3M was made in agreement with the 3.5 ratio recommended by the product monograph for 90.8% of the patients.
Treatment characteristics.

mg, milligram; PP1M, paliperidone palmitate once-monthly; PP3M, paliperidone palmitate 3-monthly; SD, standard deviation.
Outcomes
As displayed in Table 3, 33 patients (18.5%) had relapses according to our a priori definition and 19 patients (10.7%) discontinued PP3M during the 12 months following its initiation. Among the 19 discontinuations, 7 were due to side effects, 4 to insufficient response, 3 to the patient’s decision, 1 to relapse, and 4 due to other considerations. The survival function for relapse highlights that relapses were evenly distributed during the study period (see Figure 1). The relapse-free survival probability at 12 months was 0.788 (95% confidence interval [CI] = 0.725–0.856). Similarly, 12-month discontinuation-free survival probability was 0.863 (95% CI = 0.806–0.924). As for psychiatric hospitalizations, 17 patients (9.6%) were hospitalized for psychiatric reasons, translating into an hospitalization-free survival probability at 12 months of 0.889 (95% CI = 0.840–0.940).
Patients outcomes at 12 months and over the total follow-up period.

CI, confidence interval.
Last relapse occured after 864 days.
Last treatment discontinuation occured after 775 days.
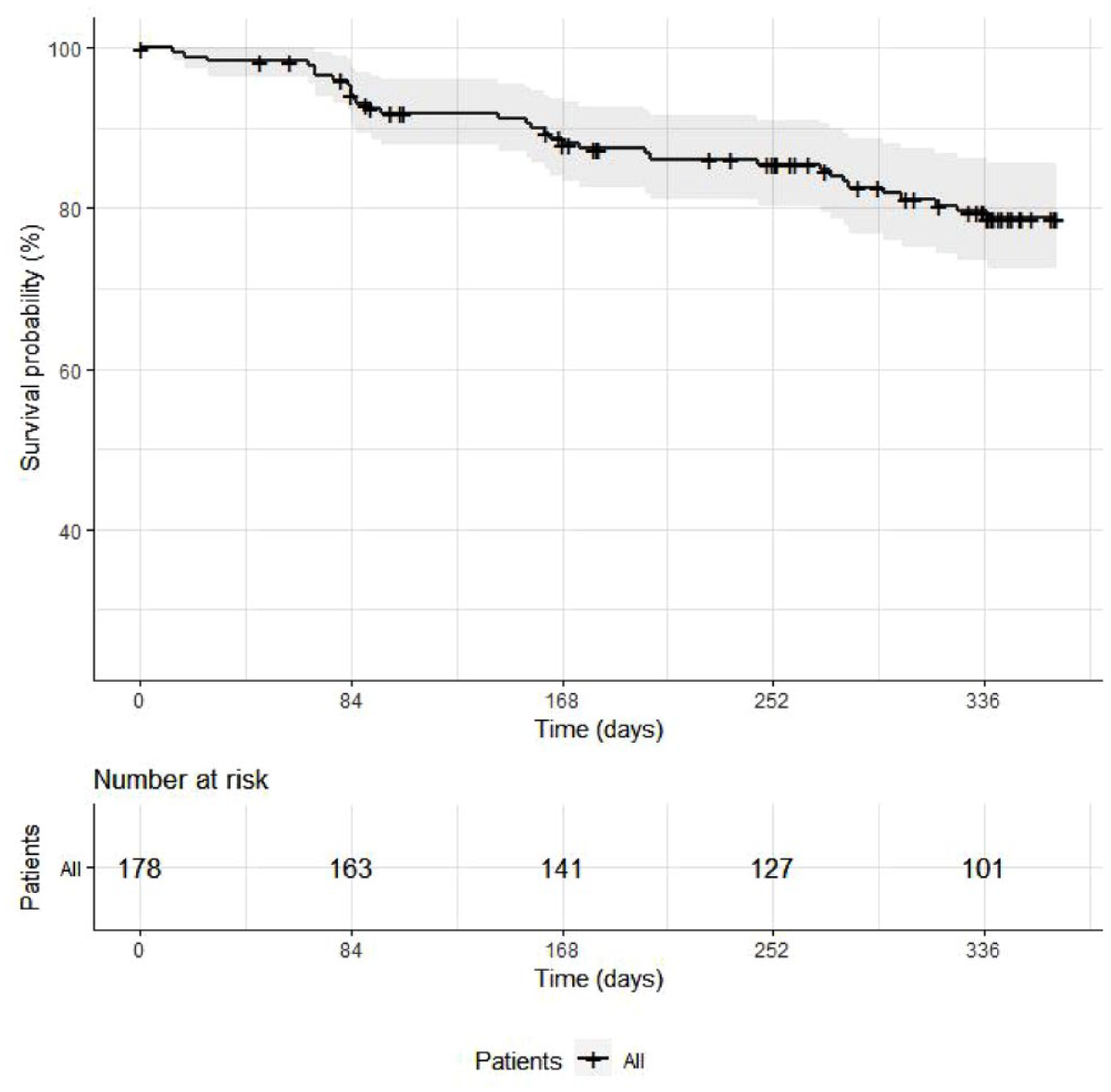
Kaplan–Meier curve of the relapse-free survival function at 12 months.
Predicting factors
Characteristics for patients who experienced a relapse during the first 12 months following PP3M initiation, compared with those who did not, are presented in Table 4. Hazard ratios (HR) were also estimated using the Cox proportional model. In these univariate analyses, comorbid personality disorder (HR = 3.6, 95% CI = 1.8–7.3, p value < 0.001), substance use disorder (HR = 3.1, 95% CI = 1.6–6.2, p value = 0.001), CGI-S score at baseline (HR = 1.4, 95% CI = 1.0–2.0, p value = 0.027) and being on welfare (HR = 2.1, 95% CI = 1.0–4.3, p value = 0.038) were associated with reduced survival times. A same trend, although not statistically significant, was observed for having been hospitalized at least once in the 2 years prior to PP3M initiation (HR = 1.9, 95% CI = 0.94–3.9, p value = 0.075). On the contrary, being at work and/or at school was associated with an increased survival time (HR = 0.32, 95% CI = 0.12–0.83, p value = 0.020).
Comparison of patients and treatment characteristics based on relapse at 12 months.
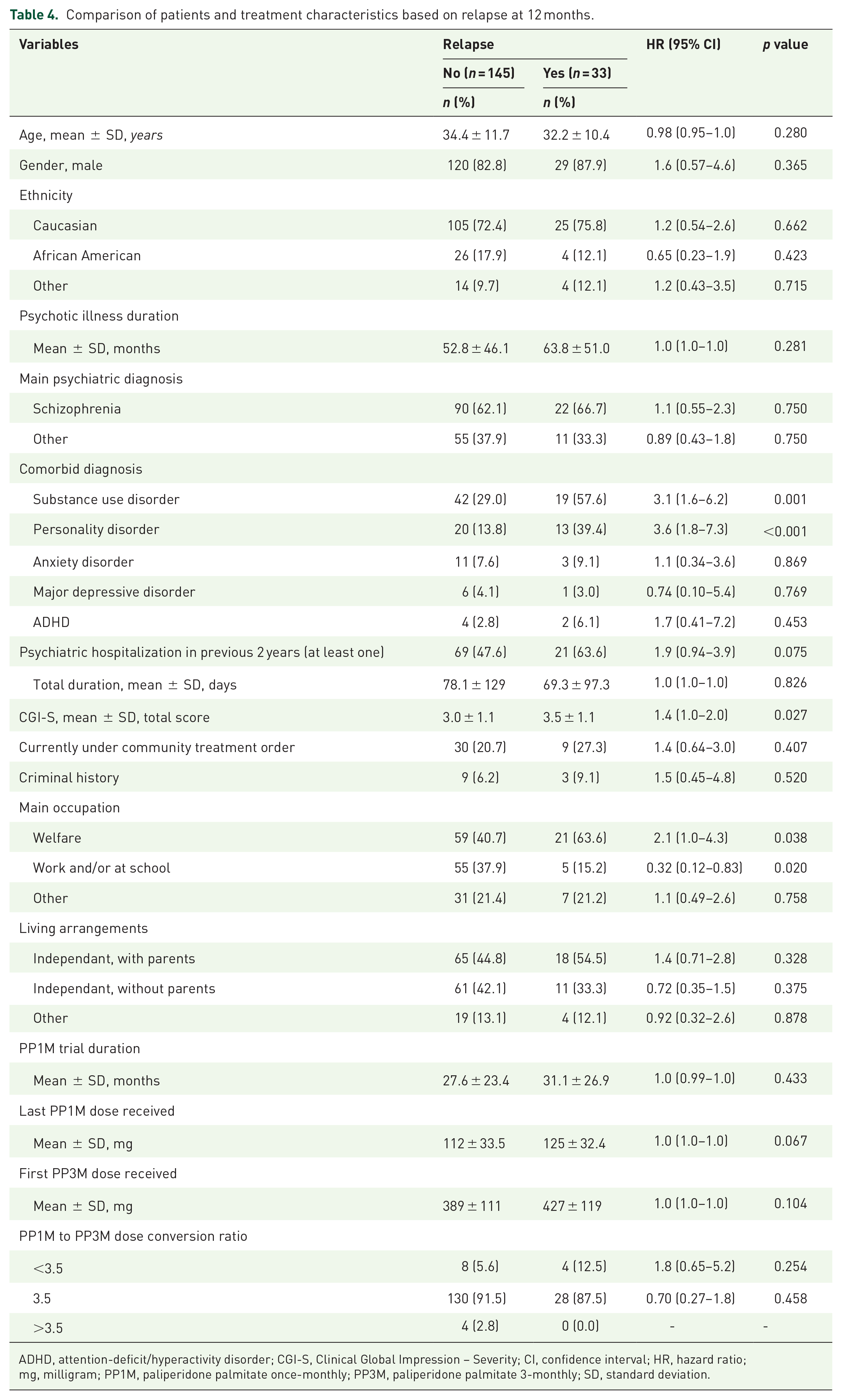
ADHD, attention-deficit/hyperactivity disorder; CGI-S, Clinical Global Impression – Severity; CI, confidence interval; HR, hazard ratio; mg, milligram; PP1M, paliperidone palmitate once-monthly; PP3M, paliperidone palmitate 3-monthly; SD, standard deviation.
To further examine the association between personality disorder as well as substance use disorder and relapses in patients receiving PP3M, a multivariate Cox proportional model was constructed based on the DAG approach. In addition to these two variables, gender and age were thus also included. The associations previously observed remained statistically significant for both personality disorder [adjusted HR (aHR) = 3.1, 95% CI = 2.3–4.2, p value < 0.0001] and substance use disorder (aHR = 2.2, 95% CI = 1.2–3.9, p value = 0.007). Kaplan–Meier curves were also estimated to illustrate the associations between personality disorder as well as substance use disorder and relapse at 12 months (see Figure 2). Patients with both personality disorder and substance use disorder had a significantly reduced relapse-free survival probability [0.392 (95% CI = 0.203–0.757)] than patients who did not suffer from any of these two comorbidities [0.890 (95% CI = 0.828–0.957)]. At 12 months, relapse rates were 9.7% (10/103), 23.8% (10/42), 28.6% (4/14), and 47.4% (9/19) for patients without substance use disorder nor personality disorder, with substance use disorder only, with personality disorder only, and with both comorbidities, respectively.

Kaplan–Meier curves of the relapse-free survival functions at 12 months depending on comorbid personality and substance use disorders.
Additional univariate analyses using the Cox proportional model were performed for the two secondary outcomes. Regarding psychiatric hospitalizations, the same association patterns with independent variables that were observed for relapses were found, albeit there were minor variations in terms of effect size and level of significance. As for PP3M treatment discontinuation, substance use disorder, personality disorder, and psychiatric hospitalizations in the 2 years prior to PP3M were associated with reduced discontinuation-free survival probabilities, and a similar trend was observed for criminal history.
Discussion
In this retrospective chart review of 178 consecutive patients with a schizophrenia spectrum psychotic disorder diagnosis who switched from PP1M to PP3M, 18.5% experienced a relapse at 12 months. Furthermore, we found that these rates varied strongly according to the presence or not of comorbid psychiatric diagnoses. Indeed, the 12-month relapse rate was 9.7% in patients with neither a comorbid diagnosis of substance use disorder nor personality disorder, 23.8% in those with only substance use disorder, 28.6% in those with only personality disorder, and 47.4% in those with both comorbidities concurrently. The very broad inclusion criteria, the absence of exclusion criteria, and the systematic sampling strategy of consecutive cases ensure the generalizability of the results to the population in which LAIs are typically used. In point of fact, this sample is comparable to what is found in routine psychiatric practice.
The 12-month overall relapse rate herein reported is greater than that obtained in previous RCTs. Indeed, Berwaerts et al. 4 and Savitz et al. 5 observed relapse rates of 8.8% and 8.1% within 12 months of PP3M treatment, respectively. While these rates may seem significantly lower than the one obtained in the present study, several aspects need to be taken into account when comparing those findings. First and foremost, study populations were different; notably, patients included in these two RCTs did not suffer from any comorbid psychiatric diagnoses, and they did not have any history of substance dependence for at least 6 months prior to screening. These are two major considerations given that our results suggest that comorbid psychiatric diagnoses, more specifically substance use disorder and personality disorder, are associated with an increased risk of relapse during PP3M treatment; it is noteworthy that in the present study patients with neither substance use disorder nor personality disorder had a relapse rate (9.7%) very close to that reported in these RCTs. Furthermore, patients in this present study were younger (mean age = 34 years, versus 37–39 years), resulting from the participation of two FEP clinics, and they were mostly men (84%, versus 51–74%). Although these two factors did not reach statistical significance as predictors of relapse, they can nevertheless be hypothesized to impact in some way the present results. Second, relapse criteria used in these RCTs were more restrictive than those employed in the present study, therefore limiting any direct comparisons. Notably, PP3M dosage increase as well as oral antipsychotic supplementation due to psychotic symptoms deterioration were considered as a relapse in our study.
Other naturalistic studies have been conducted, but as previously discussed, there are many issues that hinder their comparability with the present findings.9–11 The study with the most comparable methodological design to the present one is that reported by Wallman et al. 13 and Clark et al. 14 in which relapse rates of 7.2% and 10.8% were found during the 2 years following PP3M initiation, which is much lower than the relapse rate herein reported. A first explanation for these lower rates than that obtained here are the different definitions of relapse used. Indeed, in this previous study, a relapse was considered as such if it resulted in a step-up in clinical care, that is, referral to home-treatment teams or hospitalization due to psychiatric deterioration. In contrast with the present study, the step-up in care definition did not include PP3M dosage increase nor oral antipsychotic supplementation. A second explanation is the difference in recruitment. In the present study, two of the four participating sites were FEP clinics, which translates into a younger population (mean age = 34 years, versus 43–45 years). This is an important consideration given that Ferraro et al. 12 found a very high relapse rate of 35.7% within a sample of FEP patients, a population in which comorbid psychiatric diagnoses are the norm rather than the exception. Unfortunately, rates of comorbid diagnoses were not reported by Wallman et al. 13 nor by Clark et al., 14 making it impossible to compare their population with that of the present study on that crucial aspect.
The association with substance use disorder was not surprising since this is already recognized as a very strong predictor of relapse.8,17,18 The association between hospitalization during the last 2 years and relapse is neither surprising as this may just reflect a greater clinical instability. The association with personality disorder though has not been as thoroughly studied, yet there have been previous reports finding such a relationship. 19
The present results should be interpreted keeping in mind the following limitations. First, the retrospective and non-systematic nature of the observations limits the thoroughness of the information that could be collected. On the other hand, this ensures ecological validity, as this reflects routine clinical practice. Second, the definition of relapse is not consensual and was herein defined using very broad criteria. These criteria were deemed clinically significant, and, again, reflect routine clinical practice. Third, information about patients’ comorbidities relied on clinical diagnoses, which may lead to their underestimation as comorbidities are generally underreported. However, the relatively high prevalence of substance use disorder and personality disorder in the present study suggest that this might not be a major issue.
Conclusion
Although there are differences regarding the assessments performed and the definition of relapse, the present results may suggest that the outcomes of switching patients from PP1M to PP3M in everyday clinical practice may be less favorable than those reported in RCTs, in which more complex patients are underrepresented. 7 Moreover, substance use disorder and personality disorder were both associated with an increased risk of relapse in individuals receiving PP3M, suggesting that these patients may require special clinical attention when being switched from PP1M to PP3M. These two groups of disorders are frequently comorbid to schizophrenia spectrum psychotic disorder and, in addition with medication non-adherence, often motivate the use of LAI antipsychotics. Thus, the fact that these patients are generally not included in RCTs must be taken into account by clinicians before directly translating the results of these studies into their clinical practice. In this regard, naturalistic studies play a crucial role in providing valuable information about real-world outcomes.
