Abstract
Background:
Intestinal obstruction is a severe abdominal condition that can be triggered by various medications; however, the drugs most strongly and frequently linked to this adverse event (AE) remain insufficiently defined in the current medical literature.
Objectives:
This study sought to systematically identify and evaluate medications most strongly and consistently associated with intestinal obstruction by analyzing real-world evidence from a large pharmacovigilance database.
Design:
We conducted a retrospective, observational pharmacovigilance study.
Methods:
Reports submitted to the U.S. FDA Adverse Event Reporting System (FAERS) between January 2004 and June 2024 were analyzed. Disproportionality analyses using the Reporting Odds Ratio (ROR), Proportional Reporting Ratio (PRR), Empirical Bayesian Geometric Mean, and Information Component were conducted to identify significant safety signals associated with drug-induced intestinal obstruction.
Results:
Among 21,433,114 AE reports, 60,814 (0.28%) involved intestinal obstruction. Humira (10,356 cases) was the most frequently reported drug, followed by Remicade (2223 cases), Avastin (1580 cases), Vedolizumab (1385 cases), Clozaril (1229 cases), and Accutane (1088 cases). Disproportionality analysis revealed that the top five drugs with the highest ROR and PRR were Accutane, Teduglutide, Lonsurf, Avastin, and Lynparza. Among the top 50 drugs, 47 lacked clear labeling in their drug packaging.
Conclusion:
Our findings, derived from FAERS signal detection, identify drug-event pairs that warrant further clinical evaluation. These results should not be interpreted as evidence of causality. The high volume of reports associated with certain drugs may reflect usage patterns, underlying disease conditions, or reporting behaviors. Caution should be exercised in clinical translation.
Plain language summary
Intestinal obstruction is a serious emergency, meaning that the contents of the intestine cannot pass through normally. This may lead to severe pain, infection and even more dangerous complications. Apart from well-known causes such as tumors or hernias, certain medications may also lead to intestinal obstruction, but this point is often overlooked. At present, information on which drugs are likely to cause this problem and what the risks are is very limited, mainly coming from scattered case reports. Our research aims to systematically investigate drugs associated with the risk of intestinal obstruction. We utilized the Adverse Event Reporting System (FAERS) of the U.S. Food and Drug Administration (FDA), a large database that collects reports on drug side effects worldwide. By analyzing these real-world report data, we can identify which drugs are reported abnormally frequently along with intestinal obstruction, thereby suggesting a possible association between them. The results of this study will help doctors and patients have a clearer understanding of the potential risks of intestinal blockage that drugs may bring. By identifying high-risk drugs, clinicians can be more cautious when prescribing and formulating treatment plans for patients, especially those at risk of intestinal problems, thereby helping to prevent the occurrence of this serious complication and ensuring medication safety.
Introduction
Intestinal obstruction, a blockage that prevents the normal flow of intestinal contents, is a common and potentially life-threatening acute abdominal condition. 1 It is broadly classified as mechanical or functional and can lead to serious complications, including ischemia, perforation, and systemic infection, significantly impacting patients’ quality of life.2–7 In clinical practice, drugs are a significant, yet often underrecognized, contributing factor. The existing literature on drug-induced intestinal obstruction (DIIO) is limited, primarily consisting of case reports or small-scale studies, which lack the statistical power for systematic insights. While some reviews discuss drug effects on intestinal function, they offer limited analysis of DIIO-specific mechanisms and risk factors.
Clinical experience and preliminary evidence suggest several mechanisms for DIIO, including (1) inhibition of intestinal motility (e.g., by opioids and anticholinergics); (2) induction of intestinal stenosis through inflammation or fibrosis; and (3) alteration of intestinal secretions, leading to dried or viscous content.8–12 However, clinical trials, with their strict inclusion criteria and limited sample sizes, are inadequate for fully elucidating DIIO’s real-world complexity.
Therefore, a critical knowledge gap exists in understanding the scope and nature of DIIO. To address this, large-scale, systematic pharmacovigilance studies are essential. The FDA Adverse Event Reporting System (FAERS), a leading global database of post-market adverse events (AEs), provides a valuable resource for such investigation. Mining the FAERS database allows for the identification of signals and patterns of DIIO in a real-world context, which is crucial for drug safety surveillance and clinical practice optimization. 13
This study aims to bridge this gap by analyzing the FAERS database to identify drugs most frequently and strongly associated with intestinal obstruction, thereby aiding in the recognition and classification of DIIO risks to inform clinical decision-making.
Methods
This disproportionality analysis was conducted and reported in accordance with the recommendations of the REporting of A Disproportionality analysis for drUg Safety signal detection using individual case safety reports in PharmacoVigilance (READUS PV) statement.
Study design and reporting guidelines
This observational, retrospective pharmacovigilance study analyzed data from the FAERS database. The reporting of the study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14 In addition, the study followed the READUS-PV guidelines. 15 The completed STROBE and READUS-PV checklists are provided as Supplemental Files 1 and 2, respectively.
Data sources
This retrospective pharmacovigilance study utilizes the FAERS database (https://fis.fda.gov/extensions/FPD-QDE-FAERS/FPDQDE-FAERS.html). FAERS supports post-market surveillance for drugs and biologics, capturing AEs and medication errors reported to the FDA. Data in FAERS are voluntarily submitted by healthcare professionals and consumers. FAERS provides valuable information, including demographics, drug details, and clinical outcomes. 16
This retrospective pharmacovigilance analysis was conducted using the FAERS database. FAERS compiles reports on adverse drug events (ADEs), enabling signal detection and the quantification of drug-ADE associations.17,18 Updated quarterly, the FAERS database includes seven datasets: Demographics (DEMO), Drug Information (DRUG), Adverse Drug Reactions (REAC), Patient Outcomes (OUCT), Report Source (RPSR), Therapy Dates (THER), and Indications (INDI). Duplicate reports inevitably occur in FAERS due to the nature of data updates. The retrieval period was set from January 1, 2004, to June 30, 2024. FDA-recommended criteria were applied to remove duplicates and enhance result reliability.19–21 According to the FDA’s recommended deduplication reporting procedure, select the PRIMARY\_ID, CASE\_ID, and FDA\_DT fields from the DEMO table, and sort them in the order of CASE\_ID, FDA\_DT, and PRIMARY\_ID. For records with the same CASE\_ID, retain the one with the largest FDA\_DT value. In addition, for records with identical CASE\_ID and FDA\_DT, retain the one with the largest PRIMARY\_ID value.
After preprocessing, 17,956,653 DEMO reports, 65,083,962 DRUG cases, and 52,454,964 REAC records were obtained. This yielded 60,814 drug-related AE signals for intestinal obstruction (Figure 1).

Retrieval flowchart.
Study design (identification of target AE reports)
The FAERS database uses the Medical Dictionary for Regulatory Activities (MedDRA) to code ADR (Adverse Drug Reaction) information into standardized terms, called Preferred Terms (PTs). Standardized MedDRA Queries (SMQs) are tools within MedDRA, comprising PTs that represent similar medical conditions. They are designed to retrieve cases of interest from the MedDRA-coded database and enhance ADR signal detection and assessment. SMQs offer two search types to identify target cases: wide and narrow searches. Wide searches include all PTs potentially representing the condition, while narrow searches focus on PTs closely related to it.
To ensure specificity in identifying target AE reports, this study referenced MedDRA version 26.0 and used only PTs from the “Intestinal Obstruction (SMQ)” narrow search (see Supplemental Table 1 for PTs for the identification of intestinal obstruction AE cases). Reports were classified as target cases if any PTs from Supplemental Table 1 appeared in the “Patient Outcome” field.
Data mining algorithms and data analysis
Disproportionality analysis is a data mining method for quantitatively detecting ADR signals in large pharmacovigilance databases. Using the classic 2 × 2 contingency table (Table 1), disproportionality analysis assesses the difference between the frequencies of a target drug and AE, establishing their statistical association.
Two-by-two contingency table for disproportionality analyses.

a, Number of reports containing both the target drug and target adverse drug reaction; AEs, adverse events; b, number of reports containing other adverse drug reactions of the target drug; c, number of reports containing the target adverse drug reaction of other drugs; d, number of reports containing other drugs and other adverse drug reactions.
This study utilized four approaches for ADE signal mining, namely the Reporting Odds Ratio (ROR), Bayesian Confidence Propagation Neural Network (BCPNN), and Multi-Item Gamma Poisson Shrinker (MGPS) methods. The ROR method originated from the Lareb laboratory of the Netherlands Pharmacovigilance Centre, characterized by low bias and high sensitivity, and it helps reduce bias in infrequently reported events. Compared with ROR, the PRR stands out for its higher specificity. Currently, the BCPNN method represents a mature signal detection technology that has been adopted globally. The BCPNN can identify early signals even with sparse or incomplete data, and its detection results become more robust as report volume grows; nevertheless, it suffers from computational complexity and limited transparency. Moreover, the MGPS approach excels in identifying signals associated with rare AEs. Although there is no gold standard for signal detection methods, each has its own characteristics, with distinct advantages and limitations regarding applicability and feasibility in databases.22,23 A signal was defined by meeting the following criteria:
Lower limit of 95% Confidence Interval (CI) >1
Proportional Reporting Ratio (PRR) ⩾2
Chi-squared (χ2) ⩾4
Information component (IC025) >0
Empirical Bayesian Geometric Mean (EBGM) >1
EBGM 0.5 (EBGM05) >2
Table 1 displays the 2 × 2 table for disproportionality measurement, and Table 2 outlines the calculation formulas and thresholds. Higher values of the ROR and Bayesian Confidence Propagation Neural Network (BCPNN) indicate a stronger association between the target drug and ADE.11,24 R version 4.1.1 was used in this study for data collection, processing, and analysis.
Four major algorithms used for signal detection.

95% CI, 95% confidence interval; a, number of reports containing both the target drug and target adverse drug reaction; b, number of reports containing other adverse drug reaction of the target drug; c, number of reports containing the target adverse drug reaction of other drugs; d, number of reports containing other drugs and other adverse drug reactions; EBGM, empirical Bayesian geometric mean; EBGM05, the lower limit of 95% CI of EBGM; E(IC), the IC expectations; IC, information component; IC025, the lower limit of 95% CI of the IC; N, the number of reports; V(IC), the variance of IC; χ2, Chi-squared.
Results
Basic information on AE reports
From January 1, 2004, to June 30, 2024, a total of 21,433,114 AE reports were recorded in the FAERS database. By using the PTs from Supplemental Table 1, 60,814 reports were identified as related to the AE of interest. The annual distribution of AE reports is shown in Figure 2, with 2019 recording the highest number of reports (8.2%).

Number of reported cases of drug-induced bowel obstruction from Q1 2004 to Q2 2024.
Healthcare professionals were the primary source of reports, contributing 58.8% of the total (Table 3). The United States was the top reporting country, with 26,514 AE reports (43.6%) submitted (Table 3). Regarding patient demographics, female patients with DIIO accounted for 50.0%, outnumbering male patients (41.6%; Table 3). The age group 18–64.9 years had the highest number of patients (Table 3). Regarding patient outcomes, 58.9% of patients had prolonged hospitalization, and 8187 patients (13.5%) were reported to have died from DIIO (Table 3).
Clinical characteristics of reported drug-induced intestinal obstruction.

CA, China; CN, consumers; DE, Germany; F, female; FR, France; JP, Japan; M, male; US, United States.
Identification of potential culprits—List of drugs
Figure 3 summarizes the top 50 drugs associated with intestinal obstruction based on the frequency of AE reports. Table 4 shows the incidence and distribution of the top 15 DIIO (see Supplemental Table 2 for the statistics and distribution of the top 50 DIIO). Humira (10,356 cases) was the most frequently reported, followed by Remicade (2223 cases), Avastin (1580 cases), Vedolizumab (1385 cases), Clozaril (1229 cases), and Accutane (1088 cases). Among these 50 drugs, 23 were listed in their package inserts as potentially causing diarrhea, which may alleviate certain types of intestinal obstruction, though these drugs are not standard treatments for the condition. Only Creon, Enbrel, and OxyContin explicitly listed the association of intestinal obstruction in their package inserts, while the remaining 47 drugs did not mention intestinal obstruction as an adverse effect. In addition, 33 drugs were listed in their package inserts as potentially causing symptoms such as abdominal pain, bloating, nausea, and vomiting, which may suggest intestinal obstruction, while 17 drugs did not include any of these symptoms in their labeling.

The top 50 drugs with the highest number of reported intestinal obstructions.
Statistics and distribution of the top 15 drug-induced intestinal obstructions.
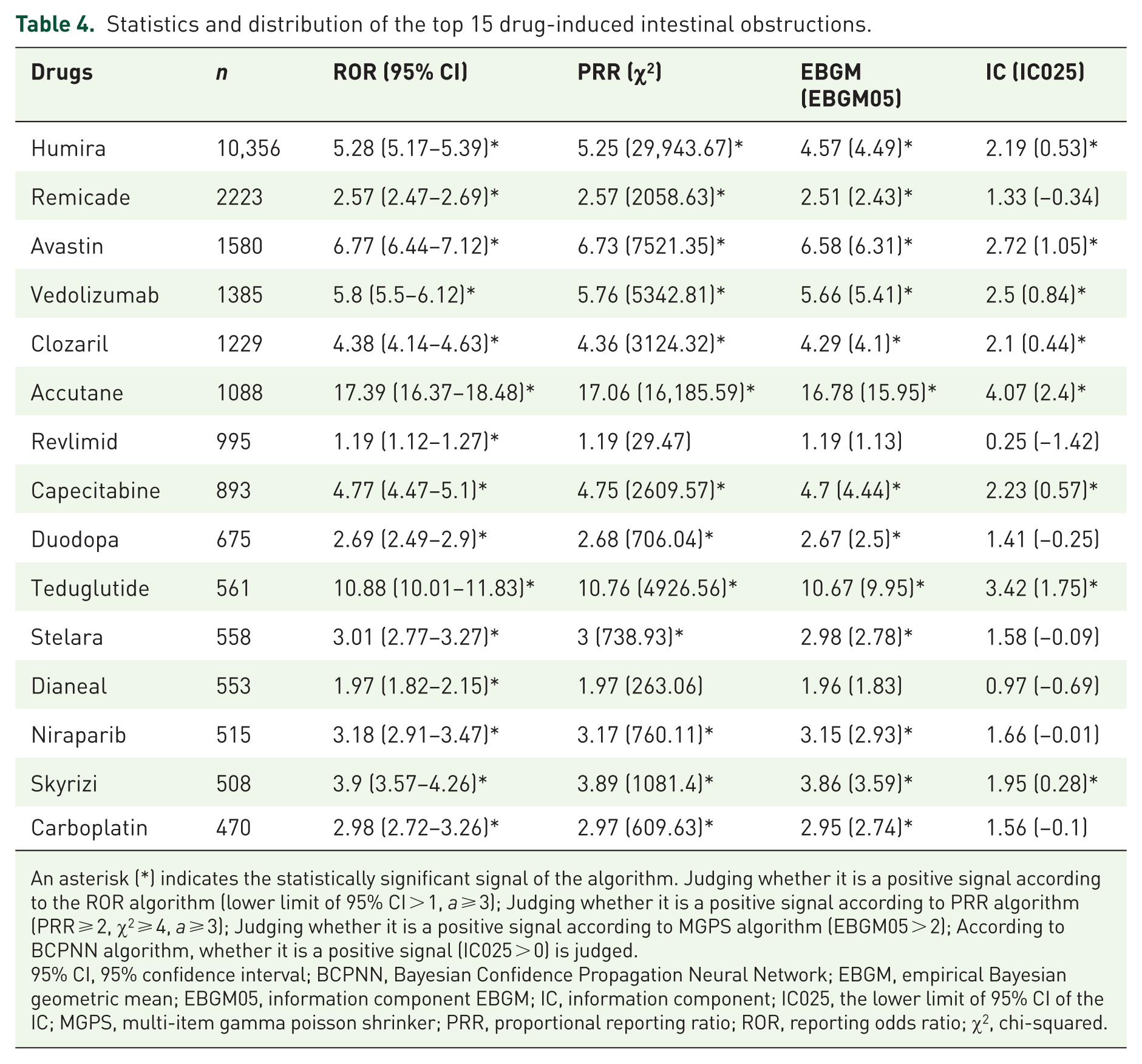
An asterisk (*) indicates the statistically significant signal of the algorithm. Judging whether it is a positive signal according to the ROR algorithm (lower limit of 95% CI > 1, a ⩾ 3); Judging whether it is a positive signal according to PRR algorithm (PRR ⩾ 2, χ2 ⩾ 4, a ⩾ 3); Judging whether it is a positive signal according to MGPS algorithm (EBGM05 > 2); According to BCPNN algorithm, whether it is a positive signal (IC025 > 0) is judged.
95% CI, 95% confidence interval; BCPNN, Bayesian Confidence Propagation Neural Network; EBGM, empirical Bayesian geometric mean; EBGM05, information component EBGM; IC, information component; IC025, the lower limit of 95% CI of the IC; MGPS, multi-item gamma poisson shrinker; PRR, proportional reporting ratio; ROR, reporting odds ratio; χ2, chi-squared.
Ranking of drugs by reporting frequency
Among the 50 drugs, we further assessed them using four different algorithms: ROR, PRR, EBGM, and Information Component (IC). Drugs that met all four algorithmic criteria were considered strong signals, resulting in the identification of 15 strong signals. These 15 drugs were ranked by signal strength (ROR values) in descending order of ROR value, as shown in Figure 4. The top five drugs in this ranking were Accutane, Teduglutide, Lonsurf, Avastin, and Lynparza, showing a high degree of consistency across the four signal detection methods. Among these five drugs, only Teduglutide explicitly lists the risk of intestinal obstruction in its package insert. The package insert for Accutane did not mention any symptoms suggestive of intestinal obstruction.
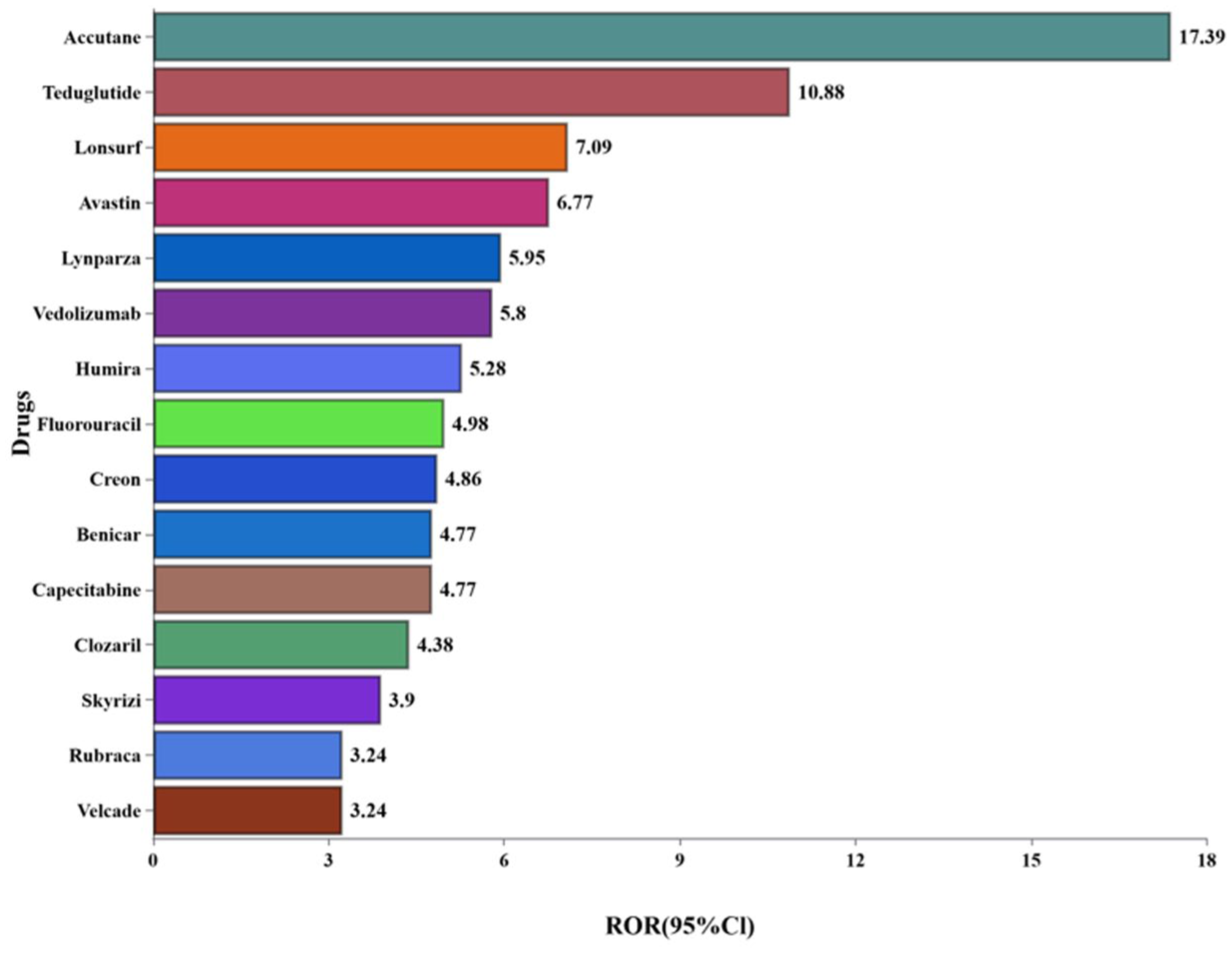
Fifteen drugs with strong signals.
Discussion
This study provides a comprehensive pharmacovigilance analysis of DIIO using the FAERS database. We not only identified drugs most strongly associated with DIIO but also, for the first time in a large-scale systematic review, highlighted the critical gap between strong real-world safety signals and the information in drug package inserts. We described the clinical characteristics of these AEs and, crucially, identified the drugs most strongly associated with DIIO. Many of these drugs did not list intestinal obstruction as a potential link in their package inserts, and their association with this AE remains underrecognized. DIIO is a significant AE in drug therapy, as it can cause severe harm, including death. In this study, we found that DIIO reports peaked in 2019 (8.2%), suggesting growing clinical awareness of this adverse reaction. However, a significant knowledge gap regarding this link remains. Healthcare professionals were the primary source of reports, contributing 58.8%, possibly reflecting their greater concern about this AE in clinical practice. Regarding patient outcomes, the mortality rate for intestinal obstruction was 13.5%, highlighting the significant health links of DIIO, especially since 58.9% of patients experienced prolonged hospital stays.
The clinical characteristics of DIIO showed that female patients (50.0%) were more likely to experience this AE than male patients (41.6%), with the highest number of reports coming from the 18–64.9 years age group. These patterns in gender and age distribution suggest that certain drugs may pose a higher risk for specific patient populations. Although many drugs did not explicitly list intestinal obstruction as a risk in their package inserts, we identified several drugs closely associated with this reaction, such as certain antibiotics, anesthetics, and anticancer drugs. Previous studies have shown that some drugs can induce intestinal obstruction by affecting smooth muscle function, inhibiting gut motility, or altering the gut microbiome.11,12 Our findings strongly corroborate and extend these known mechanisms to a broader drug landscape. Thus, DIIO may be an underestimated clinical concern. Geographically, the United States accounted for 43.6% of the total reports, likely due to the widespread use of the FAERS database and well-established reporting mechanisms, highlighting differences in pharmacovigilance practices across countries.
We further analyzed the top 50 drugs associated with DIIO in the FAERS database, evaluating their package insert labeling and signal strength. Using four signal detection algorithms—ROR, PRR, EBGM, and IC—we identified 15 drugs with strong signals. The top five drugs, ranked by signal strength in descending order of ROR, were Accutane, Teduglutide, Lonsurf, Avastin, and Lynparza. Although these drugs were strongly associated with intestinal obstruction, only Teduglutide explicitly listed this risk in its package insert, while the others did not. This discrepancy underscores a potential lag in regulatory labeling and highlights the value of proactive pharmacovigilance in identifying post-market risks.
Humira, the most frequently reported drug (10,356 cases), does not explicitly list the risk of intestinal obstruction in its package insert. Previous studies suggest that immunosuppressive drugs like Humira may indirectly increase the risk of intestinal obstruction by affecting the gut immune system and weakening gut motility. 25 Similarly, immunosuppressive agents like Remicade and Vedolizumab have also been closely associated with DIIO. 26 These drugs may alter gut motility by inhibiting inflammation or modifying gut immune function, thereby increasing the risk of intestinal obstruction.
To strengthen the biological plausibility of our findings, we delve deeper into the mechanistic links of the top-signal drugs. For instance, the strongest signal from Accutane (isotretinoin) is mechanistically intriguing. Beyond its known association with inflammatory bowel disease (IBD), recent evidence suggests it can directly alter the gut microbiome composition and impair the intestinal mucosal barrier, potentially leading to a subclinical, chronic inflammation that predisposes patients to functional obstruction and adhesion formation.27,28 This provides a plausible pathway beyond the traditional motility-centric view of DIIO.
Teduglutide and Lynparza warrant particular attention due to their drug signals. Teduglutide, used to treat short bowel syndrome, enhances nutrient absorption and improves intestinal function. However, its potential effect on gut motility could lead to intestinal obstruction. 29 Its primary mechanism, the potent stimulation of intestinal mucosal growth, can paradoxically lead to mechanical obstruction through hyperplasia and polyposis, a risk that is explicitly acknowledged in its label but may be under-appreciated in clinical practice. 30 Notably, Teduglutide’s package insert explicitly lists the risk of intestinal obstruction, alerting clinicians to this AE. By contrast, Lynparza, a PARP inhibitor, has less direct mention of intestinal obstruction but remains relevant, as chemotherapy drugs are often linked to gut dysfunction, and cancer patients are at higher risk of obstruction due to chemotherapy-induced intestinal damage. 31 For anticancer drugs like Avastin, the mechanisms are often multifactorial, involving treatment-induced peritoneal carcinomatosis, colonic strictures from bevacizumab-related inflammation, or direct cytotoxic effects on enteric neurons and glial cells. 32
Although Accutane does not list intestinal obstruction as a risk in its package insert, studies suggest it may induce obstruction by altering the gut microbiome and impairing gut barrier function. 27 This finding, combined with the strong signals for other drugs, has direct implications for clinical monitoring and potential labeling updates. We recommend that for drugs with strong DIIO signals like Accutane, Lynparza, and Lonsurf, regulatory agencies and manufacturers consider updating the “Warnings and Precautions” section of the package insert. Clinicians prescribing these agents should maintain a higher index of suspicion for DIIO, especially when patients present with nonspecific gastrointestinal symptoms like abdominal pain, bloating, nausea, and vomiting. In addition, the study found that 33 drugs listed symptoms like abdominal pain, bloating, nausea, and vomiting, which suggest intestinal obstruction, while 17 drugs did not list any of these symptoms, highlighting labeling differences regarding DIIO.
In conclusion, our large-scale pharmacovigilance study establishes that DIIO is a significant and under-recognized clinical threat. We provide a systematically prioritized list of high-risk drugs—including Accutane, Lynparza, and Lonsurf—whose strong association with DIIO is not reflected in their current package inserts. The clear take-home message for clinicians is the imperative for heightened vigilance when prescribing these agents, particularly when patients report nonspecific gastrointestinal symptoms. For researchers and regulators, our findings deliver an actionable agenda: the proposed mechanistic links for top-signal drugs demand urgent validation in targeted preclinical and epidemiological studies, and the identified labeling gaps warrant formal regulatory review. Ultimately, translating these pharmacovigilance signals into validated clinical knowledge and updated safety information is the crucial next step to mitigate the substantial morbidity and mortality associated with DIIO.
Limitations
Despite identifying valuable signals regarding the association between various drugs and intestinal obstruction, this study has several limitations inherent to its data source and methodology.
First, the FAERS database, as a spontaneous reporting system, is inherently subject to reporting biases and underreporting, which may lead to either overestimation or underestimation of true associations. A key limitation is the inability to account for drug utilization patterns. The reporting frequency is influenced by a drug’s market availability, prescription volume, and duration of use, rather than solely its inherent risk. Consequently, a commonly prescribed drug may generate more reports simply due to higher exposure, potentially inflating its signal strength. Furthermore, the majority of FAERS reports originate from the United States, potentially limiting the global generalizability of our findings and introducing regional drug use disparities.
Second, imperfections in the data mining process, such as input errors and incomplete reports, may introduce analytical bias. In addition, efforts to eliminate duplicate records using event_dt, age, gender, and reporter_country resulted in the discarding of a considerable number of reports due to incomplete data. Thus, methods for duplicate report removal require further investigation.
Third, this analysis did not sufficiently account for the underlying diseases of patients, which is a profound limitation for certain drug classes. Some medications examined may be predominantly used in patients with predisposing conditions, thus confounding the association between the drug and the AE. A prime example is drugs like Humira (adalimumab) and Remicade (infliximab), which are prescribed for Crohn’s disease and other forms of IBD. Since intestinal obstruction (particularly from strictures) is a well-known complication of IBD itself, the strong signals we detected for these drugs are profoundly confounded. It is challenging to discern whether the reported obstruction is attributable to the drug’s mechanism, the natural history of the severe underlying disease, or a combination of both. This underscores that our findings for such agents should be interpreted not as evidence of causation, but as a flag for a complex clinical scenario requiring careful patient management.
Finally, the FAERS database and the disproportionality analysis method do not allow for causal inference, so the observed signals should be interpreted as hypothesis-generating. The detected associations may be confounded by concomitant medications, protopathic bias, or the natural history of the patient’s underlying disease. Further research, particularly analytical epidemiological studies (e.g., cohort or case–control studies) designed to control for these confounders, is needed to validate these findings and explore any potential causal relationships.
Conclusion
Our findings, derived from FAERS signal detection, identify drug-event pairs that warrant further clinical evaluation. These results should not be interpreted as evidence of causality. The high volume of reports associated with certain drugs may reflect usage patterns, underlying disease conditions, or reporting behaviors. Caution should be exercised in clinical translation.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251414836 – Supplemental material for Drug-induced intestinal obstruction: insights from the FDA Adverse Event Reporting System
Supplemental material, sj-docx-1-taw-10.1177_20420986251414836 for Drug-induced intestinal obstruction: insights from the FDA Adverse Event Reporting System by He Li, Yingjie Li, Lu Liu, Bing Zeng, Sihui Su, Jianjian Liu, Chun Pan and Tianlong Li in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251414836 – Supplemental material for Drug-induced intestinal obstruction: insights from the FDA Adverse Event Reporting System
Supplemental material, sj-docx-2-taw-10.1177_20420986251414836 for Drug-induced intestinal obstruction: insights from the FDA Adverse Event Reporting System by He Li, Yingjie Li, Lu Liu, Bing Zeng, Sihui Su, Jianjian Liu, Chun Pan and Tianlong Li in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-4-taw-10.1177_20420986251414836 – Supplemental material for Drug-induced intestinal obstruction: insights from the FDA Adverse Event Reporting System
Supplemental material, sj-docx-4-taw-10.1177_20420986251414836 for Drug-induced intestinal obstruction: insights from the FDA Adverse Event Reporting System by He Li, Yingjie Li, Lu Liu, Bing Zeng, Sihui Su, Jianjian Liu, Chun Pan and Tianlong Li in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-3-taw-10.1177_20420986251414836 – Supplemental material for Drug-induced intestinal obstruction: insights from the FDA Adverse Event Reporting System
Supplemental material, sj-xlsx-3-taw-10.1177_20420986251414836 for Drug-induced intestinal obstruction: insights from the FDA Adverse Event Reporting System by He Li, Yingjie Li, Lu Liu, Bing Zeng, Sihui Su, Jianjian Liu, Chun Pan and Tianlong Li in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
