Abstract
Background:
Prolonged or excessive use of acid suppressants may increase the risk of Clostridioides difficile infection (CDI) by altering the intestinal microecosystem. Vonoprazan, a novel potassium-competitive acid blocker, exhibits a faster and more sustained acid-suppressive effect than proton pump inhibitors (PPIs). Therefore, vonoprazan may have a greater impact on the gut microbiota, potentially resulting in CDI.
Objectives:
This study aimed to explore the potential relationship between acid suppressants and CDI by the Japan Adverse Drug Event Report (JADER) and the FDA Adverse Event Reporting System (FAERS) databases.
Design:
A retrospective analysis of the JADER and FAERS databases was examined by disproportionality analysis.
Methods:
We performed signal detection analyses of CDI induced by vonoprazan and PPIs using the JADER and FAERS databases. The association between acid suppressants and CDI was calculated using the reporting odds ratio (ROR) and corresponding 95% confidence interval (95% CI). When the lower limit of the 95% CI is exceeded by 1, the association is considered statistically significant.
Results:
In the JADER database, the ROR (95% CI) for vonoprazan and PPIs based on suspect drug reports was 15.84 (12.23–20.50) and 2.51 (1.92–3.28), respectively. In the FAERS database, the ROR (95% CI) for vonoprazan and PPIs based on primary and secondary suspect drug reports was 11.50 (6.36–20.82) and 1.42 (1.34–1.51), respectively. Subgroup analysis showed that elderly patients aged 60 years and older were more strongly associated with CDI. The ROR (95% CI) for vonoprazan and PPIs in patients aged 60 years and older in the JADER database was 15.35 (11.59–20.33) and 1.65 (1.14–2.39), respectively. Similarly, the ROR (95% CI) for vonoprazan and PPIs in the FAERS database was 12.56 (6.26–25.20) and 1.43 (1.31–1.57), respectively. Excluding the effect of Helicobacter pylori (H. pylori) infection, the use of acid suppressants was still associated with CDI.
Conclusion:
While signal detection analysis based on the JADER and FAERS databases could not establish causality, our study demonstrated that both vonoprazan and PPIs were significantly associated with CDI. Vonoprazan showed a stronger association with CDI in both databases.
Plain language summary
Introduction:
Vonoprazan is a new type of acid suppressant, which has a stronger effect on acid inhibition than traditional proton pump inhibitors (PPIs). Vonoprazan may have a greater impact on the gut microbiota, which may increase the risk of Clostridioides difficile infection (CDI). The FDA created the FDA Adverse Event Reporting System (FAERS) database to support the post-market surveillance program. The PMDA created the Japan Adverse Drug Reaction Event Report (JADER) database to specifically collect adverse reaction reports in Japan. To further understand the potential relationship between acid suppressants and CDI, this study was analyzed using the JADER and FAERS databases.
Methods:
This study analyzed cases of CDI reported after the use of acid suppressants in the JADER and FAERS databases.
Results:
The analysis revealed that vonoprazan and PPIs are significantly associated with CDI in both databases. Notably, vonoprazan exhibited a stronger association compared to PPIs. Subgroup analysis indicated that this association was more pronounced in elderly patients aged 60 years and older. Additionally, excluding the influence of Helicobacter pylori (H. pylori) did not diminish the association between acid suppressants and CDI.
Conclusion:
Although signal detection analysis based on the JADER and FAERS databases could not establish causality, the results showed that both vonoprazan and PPIs were significantly associated with CDI. Vonoprazan was also more strongly associated with CDI than PPIs, which could be a potential safety concern, and further clinical studies are needed to confirm this finding.
Keywords
Introduction
Clostridioides difficile infection (CDI) is a common and serious hospital-acquired infection. 1 Clostridioides difficile (formerly Clostridium difficile, C. difficile) is a gram-positive, anaerobic bacillus, which can cause intestinal inflammation and tissue damage, leading to serious symptoms such as serious diarrhea, gastrointestinal bleeding, and even pseudomembranous colitis.2,3 In 2011, an estimated 453,000 primary cases, 83,000 first recurrence cases, and 29,300 deaths were associated with CDI in the United States. 4 Due to healthcare prevention, cases of healthcare-associated infections declined from 2011 to 2017. 5 Recent epidemiological data shows that as the incidence of hospital-acquired CDI declined, the proportion of community-acquired CDI increased. 6
In addition to the known risk factors (such as the use of antibiotics, advanced age, hospitalization, and severe underlying disease), proton pump inhibitors (PPIs) and H2 receptor blockers (H2RAs) have been repeatedly shown to increase the risk of CDI.7,8 A meta-analysis of 23 studies including nearly 300,000 patients showed a 65% increase in the incidence of C. difficile-associated diarrhea in patients after using PPIs. 9 Another meta-analysis that included 67 studies came to a similar conclusion. It showed a significant correlation between PPI use and recurrent CDI as well. 10 In addition, another study has indicated that H2RAs were associated with CDI, but the risk is lower than that of PPIs. 11 These findings indicate that CDI is associated with acid suppressants, and the prevention of CDI should also pay special attention to the adverse reactions caused by acid suppressants.
Acid suppressants mainly include H2RAs, PPIs, and potassium-competitive acid blockers (P-CABs). These drugs are widely used in gastric acid-related diseases, such as peptic ulcer, gastroesophageal reflux disease (GERD), and Helicobacter pylori (H. pylori) infection.12–14 Vonoprazan, as a novel P-CAB, inhibits gastric acid secretion by competitively inhibiting the binding of proton pumps in the resting and active states to K+, thereby inhibiting the exchange of H+ and K+. 15 Our previous study compared the acid-inhibitory effects of various acid suppressants, the results showed that the acid-inhibitory effect of vonoprazan was significantly more effective than PPIs and H2RAs. 16 Compared with conventional acid suppressants, vonoprazan has a faster and more sustained acid-suppressing effect and has shown better clinical efficacy and tolerance. 17 However, some studies have shown that prolonged or excessive use of acid suppressants may alter the intestinal microecosystem and increase the risk of CDI in patients. 18 Currently, there is a lack of evidence regarding the relationship between novel P-CABs and CDI, and further studies are needed to explore their effects.
Spontaneous reporting systems are excellent resources for post-marketing drug safety monitoring, reflecting the realities of clinical practice. Identification of safety signals using a spontaneous adverse event (AE) reporting database may be an effective way to generate hypotheses about possible relationships between unknown or potential AEs and drugs. The FDA Adverse Event Reporting System (FAERS) is the world’s largest database of spontaneous AE reports. With nearly 19 million reports currently registered, it has extensive coverage and complete data that provide important information about drug safety and drug side effects. In comparison, the Japanese Adverse Drug Event Reporting (JADER) database has a small amount of data, but it is of great significance for collecting adverse reaction reports of locally marketed drugs in Japan. Although the data published by FAERS and JADER differ in nature, they can serve as complementary sources of information. 19
Therefore, this study aimed to explore the potential relationship between vonoprazan and CDI by FAERS and JADER databases. Further, we analyzed the occurrence characteristics of acid suppressants and CDI.
Methods
Data sources
The JADER data are publicly available and can be downloaded from the Pharmaceuticals and Medical Devices Agency (PMDA) website (https://www.pmda.go.jp/index.html). The JADER database consists of four data tables: DEMO, DRUG, REAC, and HIST. The ‘DEMO’ table contains basic patient demographic information such as gender, age, weight, height, reporting year, and reporter. The ‘DRUG’ table contains information about the drug’s generic name, trade name, dose, route of administration, start date of administration, and end date of administration. The ‘REAC’ table contains AE variable, including the name of the AE, the date of onset, and the outcome. The ‘HIST’ table contains the patient’s underlying disease information.
The FAERS data can be downloaded from the FDA website (http://www.fda.gov/). The FAERS database consists of seven data tables: DEMO, DRUG, REAC, OUTC, RPSR, INDI, and THER. The ‘DEMO’ table contains basic information such as gender, age, date of the AE, and country where the AE occurred. The ‘DRUG’ table contains information such as drug name, route of administration, and dosage. The ‘REAC’ table contains the name of AE. The ‘OUTC’ table contains information on patient outcomes. The ‘PRSR’ table contains information on the source of the event report. The ‘INDI’ table contains information about the patient’s indication. The ‘THER’ table contains information on the start and end dates of administration.
The JADER database for this study used data reported between April 2004 and September 2023. The FAERS database used data reported between January 2004 and September 2023.
Definitions of AEs and drugs of interest
This study defines AEs using the Preferred Term (PT) from standardized Medical Dictionary for Regulatory Activities/Japanese version (MedDRA/J 26.0). The Standardized MedDRA Queries (SMQ) are groupings of MedDRA terms, usually at the PT level, which are associated with adverse drug reactions. 20 We used nine PTs for CDI detection based on the SMQ ‘pseudomembranous colitis’ (SMQ code: 20000080). The ‘pseudomembranous colitis’ SMQ, including PTs clostridium difficile colitis (PT code: 10009657), gastroenteritis clostridial (PT code: 10017898), pseudomembranous colitis (PT code: 10037128), clostridium difficile infection (PT code: 10054236), clostridium colitis (PT code: 10058305), clostridium bacteremia (PT code: 10058852), clostridial infection (PT code: 10061043), clostridium test positive (PT code: 10070027), and clostridial sepsis (PT code: 10078496). We excluded the following terms not related to acid suppressant causing CDI: antibiotic-associated colitis (PT code: 10052815). 21 We investigated vonoprazan and PPIs (omeprazole, lansoprazole, rabeprazole, esomeprazole) approved in Japan. Each drug name was determined by using the generic name and trade name listed in the PMDA. In the JADER database, the role of drug in AEs was coded as suspected drug (S), concomitant drug (C), and interaction (I). In the FAERS database, the role of drug in AEs was coded as primary suspected drug (PS), second suspected drug (SS), concomitant drug (C), and interaction (I). This study analyzed the inclusion of reports labeled as ‘PS’ and ‘SS’, ‘C’ and ‘I’ in two-by-two combinations, respectively. 22 In this analysis, ‘S’, ‘PS’, and ‘SS’ were considered suspected CDI AEs, and ‘C’ and ‘I’ were considered possible CDI AEs.
Statistical analysis
In our study, we used disproportionality analysis to evaluate the potential relationship between acid suppressants and CDI by calculating the reporting odds ratio (ROR) and corresponding 95% confidence interval (95% CI). A 2 × 2 contingency table was used to calculate ROR and 95% CI (Supplemental Table S1). The association was considered to have a signal when the lower limit of the 95% CI of the calculated ROR >1 with the number of reports ⩾3. A higher ROR suggested a stronger association between acid suppressants and CDI.
To further explore the effects of different stratification regimens on the association between acid suppressors and CDI, we adjusted the ROR using a multivariate logistic regression model. Stratified by sex (female and male), age (<60, ⩾60 years), and indications (H. pylori infection and non-H. pylori infection). In age group stratification, we categorized 10–19, 20–29, 30–39, 40–49, and 50–59 as patients <60 years old, and for those labeled 60–69, 70–79, 80–89, 90–99, ⩾100, and the elderly, we categorized them as ⩾60 years old.
The time-to-onset (TTO) of CDI equals event onset date minus therapy start date. 23 To ensure the accuracy of the calculations, we removed input errors (AEs occurring earlier than treatment start), inaccurate date entries, and reports with missing specific data. For cases with multiple therapy start dates, we substituted the first therapy start date prior to event onset date into the formula for calculation. Statistical significance was defined as a p-value <0.05. Data analysis was performed using MySQL 8.0, Navicat Premium 16, and Microsoft Excel 2021. The ‘Methods’ section of this study is based on the information provided by the official website of FAERS and JADER databases, and with reference to this article describing signal detection using data mining. 24
Results
Descriptive analysis
From April 2004 to September 2023, there were 852,537 AEs reported in the JADER database. The total number of CDI cases associated with vonoprazan and PPIs were 91 and 254, respectively. The patients aged 60 years and above accounted for a greater proportion than those aged below 60 years. For vonoprazan reports, more cases were reported in 2019–2023 than in 2014–2018 (62.35% versus 37.65%). Vonoprazan and PPIs are used more in patients with H. pylori infection. In JADER, 61 CDI-associated cases of ‘S’ and 30 CDI-associated cases of ‘C’ were reported for vonoprazan, but no cases of ‘I’ were reported. In total, 44.49% of patients with PPIs-induced CDI recovered, 29.92% were recovering, and 7.48% of CDI patients died. A total of 29.67% and 32.97% of patients with CDI caused by vonoprazan recovered and were recovering, respectively, and 13.19% of CDI patients died. The clinical characteristics of CDI associated with vonoprazan and PPIs were described in Supplemental Table S2.
From January 2004 to September 2023, there were 16,626,371 AEs reported in the FAERS database. The total numbers of CDI cases associated with vonoprazan and PPIs were 26 and 3004, respectively. Similarly, a larger proportion of patients were aged 60 years and above than those aged below 60 years. The proportion of reports of vonoprazan associated with CDI also increased in the last 5 years. For PPIs reports, more cases were reported in 2014–2023 than in 2004–2013 (96.74% versus 3.26%). In FAERS database, vonoprazan and PPIs are used more in patients with GERD. For acid suppressant’s role in CDI reported by FAERS, vonoprazan was reported as ‘SS’ in 11 cases, ‘C’ in 15 cases, but no cases of ‘PS’ were reported. PPIs were reported as ‘PS’ in 369 cases, ‘SS’ in 782 cases, ‘C’ in 1838 cases, and ‘I’ in 15 cases. The clinical characteristics of CDI associated with vonoprazan and PPIs were described in Supplemental Table S3.
Disproportionality analysis
For the JADER database, the ROR (95% CI) of the ‘S’ analysis for vonoprazan and PPIs as a whole was 15.84 (12.23–20.50) and 2.51 (1.92–3.28), respectively. The ROR (95% CI) of ‘S’ analysis for omeprazole, lansoprazole, rabeprazole, and esomeprazole was 2.94 (1.58–5.47), 2.55 (1.76–3.71), 4.10 (2.42–6.95), and 3.63 (1.17–11.32), respectively. The lower limit of 95% CI of ‘S’ for acid suppressants were >1, and thus the AE signal was detected. The ROR (95% CI) of the [‘C’ + ‘I’] analysis for vonoprazan and PPIs as a whole was 1.64 (1.14–2.35) and 1.00 (0.87–1.16), respectively. The lower limit of 95% CI of [‘C’ + ‘I’] for vonoprazan was >1, and a signal was detected (Figure 1).
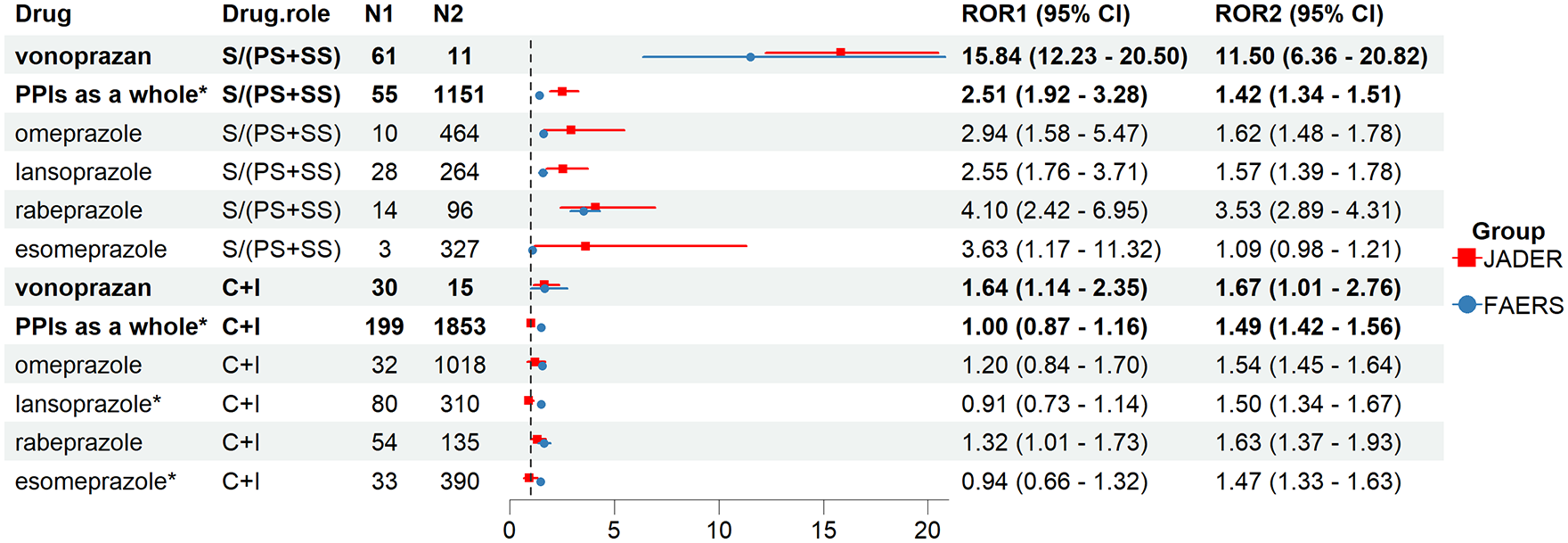
Number of reports and the ROR based on the role of acid suppressants reported for Clostridioides difficile infection in JADER and FAERS database. N1 and ROR1 (95% CI) represent the number of reports and the ROR in JADER database. N2 and ROR2 (95% CI) represent the number of reports and the ROR in FAERS database.
For the FAERS database, we find similar results. The ROR (95% CI) of the [‘PS’ + ‘SS’] analysis for vonoprazan and PPIs as a whole was 11.50 (6.36–20.82) and 1.42 (1.34–1.51), respectively. The ROR (95% CI) of the [‘PS’ + ‘SS’] analysis for omeprazole, lansoprazole, rabeprazole, and esomeprazole was 1.62 (1.48–1.78), 1.57 (1.39–1.78), 3.53 (2.89–4.31), and 1.09 (0.98–1.21), respectively. With the exception of esomeprazole, the lower limit of 95% CI of [‘PS + SS’] for the other acid suppressants were >1, and the AE signal was detected. The ROR (95% CI) of the [‘C’ + ‘I’] analysis for vonoprazan and PPIs as a whole was 1.67 (1.01–2.76) and 1.49 (1.42–1.56), respectively. The lower limit of 95% CI of [‘C’ + ‘I’] for vonoprazan and PPIs as a whole were >1, and the AE signal was detected (Figure 1).
The signals of vonoprazan and PPIs in different CDI events (such as Clostridium difficile colitis, Pseudomembranous colitis, CDI, Gastroenteritis clostridial) are shown in Figures 2 and 3. Among the ‘S’ reports in the JADER database, vonoprazan had a strong signal in C. difficile colitis and CDIs, and rabeprazole had a strong signal in CDIs. Among the [‘PS’ + ‘SS’] reports in the FAERS database, vonoprazan had a strong signal in C. difficile colitis. In Supplemental Table S4, we added the number of cases and signal for each PT in the FAERS and JADER databases.

Signaling profile of vonoprazan and PPIs in different Clostridioides difficile infection events based on suspect drug reports in JADER and FAERS database.

Signaling profile of vonoprazan and PPIs in different Clostridioides difficile infection events based on concomitant drug and interaction reports in JADER and FAERS database.
Subgroup analysis
After excluding lack of information on age and gender, 766,719 reports in JADER database and 9,568,984 reports in FAERS database were included in subgroup analysis. As shown in Figure 4, the association between acid suppressants and CDI was assessed by gender (female, male), age (<60 and ⩾60 years), and indications (H. pylori infection and non-H. pylori infection) based on the ‘S’ in the JADER database and the [‘PS + SS’] in the FAERS database, respectively. For the gender subgroup analysis, we found in the JADER database that male patients using PPIs were more likely to develop CDI than females. Whereas in the FAERS database, female patients using PPIs were more likely to develop CDI. For the age subgroup analysis, we found that CDI reports were more common in people aged 60 years and older. For disease subgroup analysis, patients with H. pylori infection were more likely to develop CDI. When H. pylori infection was excluded, that is, not in combination with antibiotics, acid suppressants were still associated with CDI. Similar results were obtained based on subgroup analyses of [‘C’ + ‘I’] in the JADER and FAERS databases (Figure 5). When a PT that tested positive for clostridium was excluded, data from eight PTs were included to assess the relationship between acid suppressants and CDI. The results of the signals labeled as ‘S’, [‘PS’ + ‘SS’], and [‘C’ + ‘I’] in the JADER and FAERS databases were generally consistent with the initial results (Supplemental Table S5).

Subgroup analysis of acid suppressants-related Clostridioides difficile infection based on suspect drug reports in JADER and FAERS database.
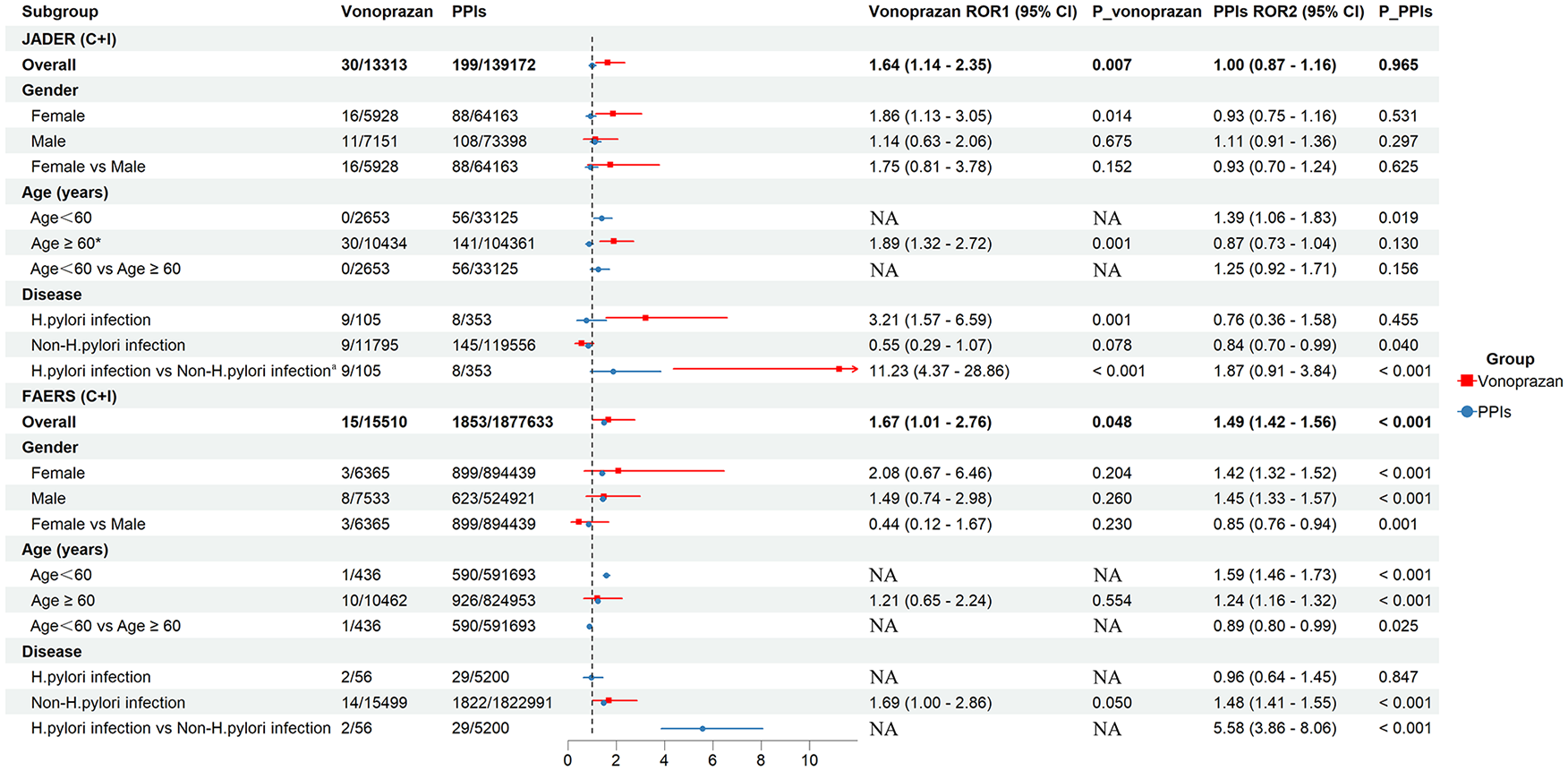
Subgroup analysis of acid suppressants-related Clostridioides difficile infection based on concomitant drug and interaction reports in JADER and FAERS database.
TTO analysis
We collected the TTO of vonoprazan- and PPIs-associated CDI from the JADER and FAERS databases, and the results are shown in Figure 6. The TTO analysis showed that the median TTO of CDI associated with vonoprazan was 4.5 [interquartile range (IQR): 1.75–11] days and that associated with PPIs was 25 (IQR: 7–62) days in the JADER database. The TTO analysis showed that the median TTO of CDI associated with vonoprazan was 14 (IQR: 5–63) days and that associated with PPIs was 26 (IQR: 8–64) days in the FAERS database. PPIs occurred about the same time in both databases, while the TTO of vonoprazan-associated CDI was about 10 days later in the FAERS database than in the JADER database.

TTO of acid suppressants-related CDI. (a) Vonoprazan associated with CDI in JADER database. (b) Vonoprazan associated with CDI in FAERS database. (c) PPIs associated with CDI in JADER database. (d) PPIs associated with CDI in FAERS database.
Discussion
In this study, we detected the safety signal for the induction of CDI by vonoprazan and PPIs using the JADER and FAERS databases. To our knowledge, this is the first study to explore the association between acid suppressants and CDI using spontaneous adverse reaction database reports. Overall, we observed that vonoprazan had a more significant association with CDI than PPIs in both JADER and FAERS databases. This means that vonoprazan may be more likely to cause CDI than PPIs in the real world.
In recent years, acid suppressants-induced CDI have been a major concern. In a large pharmacoepidemiologic cohort study, increasing acid inhibition levels were associated with increased risks of hospital-acquired CDI. 25 Moreover, patients receiving long-term PPIs therapy had an increased risk of CDI compared to patients receiving short-term PPIs treatment. 25 Vonoprazan has a pKa of up to 9.3, which can immediately ionize in an acidic environment and rapidly accumulate in acidic secretory canaliculi, with a powerful acid suppression effect. 26 The half-life can be up to 9 h, which can continuously inhibit gastric acid secretion for a long time. 27 Previous research has shown that the acid inhibition effect of vonoprazan is significantly more effective than PPIs. The proportion of intragastric pH >4 at 24 h was nearly 3-fold higher with vonoprazan than lansoprazole after a single dose and nearly 2-fold higher after continuous administration up to day 7. 28 The potent and long-lasting acid inhibition effect of vonoprazan will limit the bactericidal effect of gastric acid on oral bacteria, and cause vertical translocation of intragastric bacteria to colonize the distal small intestine, causing gut microbiome dysbiosis, thereby increasing the risk of CDI. 29
Colonization resistance to C. difficile is not attributed to a single microbiome, but rather it is the result of multiple microbiota members interacting in an environment-dependent manner. 30 Studies have shown that increasing the abundance of Escherichia and Streptococcus makes mice susceptible to CDI colonization, whereas Porphyromonadaceae, Lachnospiraceae, Lactobacillus, and Alistipes played a protective role.30–32 Also in another study, it was found that PPIs increased the abundance of Enterococcaceae and Streptococcaceae, which were associated with CDI. 33 One study compared the effects of vonoprazan and lansoprazole on the gut microbiome of H. pylori-negative healthy individuals, the results showed that vonoprazan administration significantly increased the abundance of Streptococcus and significantly decreased the abundance of Lachnospiraceae. 34 This may indicate that the application of vonoprazan caused alterations in the gut microbiome, leading to the loss of colonization resistance to C. difficile and causing CDI. In addition, we previously conducted studies on the effects of vonoprazan on the human gut microbiota and found that vonoprazan significantly decreased the abundance of Coprococcus and Bifidobacterium, which are bacteria known to be associated with CDI. 35
The results of this study are inconsistent with the findings of two previous retrospective studies. Although previous studies found that vonoprazan and PPIs use was positively associated with CDI, the association between vonoprazan and CDI is not beyond that of PPIs use.36,37 However, previous findings have contradicted the effects of vonoprazan on the gut microbiome. 34 In this study, CDI signals were detected for both vonoprazan and PPIs, and the results of the ROR for the suspected drug calculated from the two databases showed that the ROR for vonoprazan was significantly greater than that of PPIs. However, there was no significant difference between the ROR for vonoprazan and PPIs when used as a concomitant or interaction drug. In addition, in the subgroup analysis of non-H. pylori infections, the results of ROR for suspected drugs calculated from both databases showed that the risk of CDI induced by vonoprazan was higher than that of PPIs. This study collected data from the JADER and FAERS databases, which contain spontaneous reports from multiple sources. Compared with previous studies, this study was able to reflect the situation of the population more broadly, rather than being limited to a single healthcare organization. This observed difference may be due to different data sources. In addition, our study had a relatively large sample size, which is more representative compared to previous studies. This variability needs more clinical studies to discuss the differences between vonoprazan and PPIs in inducing CDI to better understand the impact of vonoprazan on the gut microbiome and the risk of CDI.
In the age subgroup analysis, our study found that people aged 60 years and older were more likely to develop CDI. Studies have found that patients aged >65 years have a 5–10-fold increased risk of CDI, compared with patients <65 years. The older people are an important risk factor not only for CDI itself, but also for adverse clinical outcomes (including severity and mortality). 1 The elderly people are also more susceptible to CDI due to decreased immune capacity and gastrointestinal resistance as well. Therefore, we recommend that when using acid suppressants in elderly patients, they should be used according to the indications and avoid unnecessary overuse to reduce the occurrence of CDI.38,39 In the gender subgroup analysis, male patients using PPIs had a higher risk of developing CDI than females in the JADER database. In the FAERS database, it was found that female patients were more likely to develop CDI than male patients. Similar findings were found in this study examining antibiotics and CDI based on the FAERS database, with female patients representing 61% of CDI patients. 21 The FAERS and JADER data may yield different due to regional and ethnic differences. Further studies are needed to explore the relationship between CDI and gender as a factor.
Gastric acid has a protective effect on the human body. Under normal conditions, gastric pH is 1–2, and it is difficult for bacteria other than H. pylori to survive in the gastric juice or gastric mucosa. This acidic environment provides non-specific protection for the digestive tract. 40 Clostridium difficile is a gram-positive anaerobic bacillus. Spores are the dormant body that resists the adverse external environment, and they will ‘germinate’ when the conditions are suitable. The vegetative form of C. difficile survives in conditions of gastric pH >4, and the number of proliferations depends on the proportion of time in the day that pH >4. The use of acid suppressants effectively reduces gastric acid secretion and increases intragastric pH, which leads to the proliferation of spores and their conversion into a vegetative form of C. difficile.41,42 Compared to PPIs, vonoprazan was able to more significantly inhibit gastric acid secretion and maintain intragastric pH >4 for a longer percentage of time, suggesting that vonoprazan may have a stronger association with CDI. 43 In addition, the use of acid suppressants causes the gastric to empty more slowly, which encourages the growth of C. difficile spores. Long-term use of acid suppressants may also lead to the overgrowth of intestinal bacteria, which will stimulate the secretion of a large amount of bile acids, thus accelerating the growth of C. difficile spores, and leading to the development of CDI. 44 On the other hand, PPIs directly regulate gene expression levels of C. difficile-associated toxins. When C. difficile is exposed to PPIs, the maximum level of expression of toxin-associated genes can reach approximately 120-fold the corresponding normal level. 45 The mechanism by which acid suppressants cause CDI has not been fully elucidated. Therefore, further studies are necessary to verify the relationship between acid suppressants and CDI, and to explore the underlying biological mechanisms.
Although there may be differences in patient’s background in the JADER and FAERS databases, such as ethnicity, social background, and medical conditions, our findings show that the signal of CDI caused by vonoprazan is similar in both databases. All reports in the JADER database come from Japan, while 70% of reports in the FAERS database come from the United States. 19 As vonoprazan is currently available only in Asian countries such as Japan, it results in a lower number of cases in the FAERS database. However, in terms of CDI caused by PPIs, the signal intensity in the JADER database was higher than that in the FAERS database, possibly because PPIs are affected by CYP2C19 gene polymorphisms. Previous studies have shown that the risk of infection during PPIs therapy is altered by the functional status of CYP2C19. 46 For Asian populations, the CYP2C19 poor metabolizer is more frequent, while CYP2C19 ultra-rapid metabolizer is more frequent in Caucasian populations. This may indicate a higher correlation between PPIs and CDI in Asian populations. 47 This result should be interpreted with caution due to the relatively small amount of data in this study.
Limitations
Nonetheless, this study has several limitations. First, this study found an association, rather than a causal relationship, between acid suppressants and CDI. Analyses using the JADER and FAERS databases were not able to analyze the ‘risk’ of AEs, and could only point to ‘potential complications of AEs’ using ROR. 48 Second, voluntary reporting is not limited to health care professionals, consumers are also candidates for reporting AEs. Unfortunately, consumers have limited medical expertise and are unlikely to make the correct associations. 49 In addition, the association between drugs and adverse reactions is confounded by concomitant drugs and comorbidities. Drugs associated with CDI, such as antibiotics, are used in combination as concomitant drugs. However, the information on concomitant drugs in the two databases was insufficient to analyze due to the different focus of the reporter. In our study, we excluded the confounding factor of antibiotic combination by analyzing data from disease subgroups. Finally, because the spontaneous reporting system is voluntary, it is not possible to capture all AEs that occur, which may lead to exaggerated or underestimated results. Also, these data are from observational studies, not randomized controlled trial analyses, and reporting rates may be biased.
Conclusion
In this study, we analyzed the safety signals between vonoprazan and PPIs in causing CDI from the JADER and FAERS databases. The results showed that there was a significant association between vonoprazan and PPIs use and CDI. Moreover, vonoprazan was more strongly associated with CDI compared to PPIs. This may be a potential safety concern. Subgroup analysis showed that older people aged over 60 years were more likely to develop CDI. Even when patients with H. pylori infection were excluded, the use of vonoprazan and PPIs remained associated with CDI. Due to the limitations of the spontaneous reporting system, we recommend that further clinical studies demonstrate the mechanism and association of vonoprazan-induced CDI in future drug safety evaluations.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241260211 – Supplemental material for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System
Supplemental material, sj-docx-1-taw-10.1177_20420986241260211 for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System by Mengling Ouyang, Shupeng Zou, Qian Cheng, Xuan Shi, Yazheng Zhao and Minghui Sun in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986241260211 – Supplemental material for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System
Supplemental material, sj-docx-2-taw-10.1177_20420986241260211 for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System by Mengling Ouyang, Shupeng Zou, Qian Cheng, Xuan Shi, Yazheng Zhao and Minghui Sun in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986241260211 – Supplemental material for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System
Supplemental material, sj-docx-3-taw-10.1177_20420986241260211 for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System by Mengling Ouyang, Shupeng Zou, Qian Cheng, Xuan Shi, Yazheng Zhao and Minghui Sun in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-4-taw-10.1177_20420986241260211 – Supplemental material for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System
Supplemental material, sj-docx-4-taw-10.1177_20420986241260211 for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System by Mengling Ouyang, Shupeng Zou, Qian Cheng, Xuan Shi, Yazheng Zhao and Minghui Sun in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-5-taw-10.1177_20420986241260211 – Supplemental material for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System
Supplemental material, sj-docx-5-taw-10.1177_20420986241260211 for Vonoprazan-associated Clostridioides difficile infection: an analysis of the Japanese Adverse Drug Event Report and the FDA Adverse Event Reporting System by Mengling Ouyang, Shupeng Zou, Qian Cheng, Xuan Shi, Yazheng Zhao and Minghui Sun in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors are grateful to Department of Pharmacy, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology for providing administrative and technical support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
