Abstract
Acid suppression is the primary therapy for erosive esophagitis (EE). Although proton pump inhibitors (PPIs) are considered as the first-line medication for EE, 10–20% of patients with Los Angeles C and D grade EE do not gain complete mucosal healing and symptom control despite 8-week double-dose PPI treatment. Vonoprazan is a novel potassium-competitive acid blocker (P-CAB), which blocks the H+, K+-adenosine triphosphatase enzymes in a K+-competitive and reversible manner. Vonoprazan exhibits different pharmacological and pharmacokinetic profiles from conventional PPIs, and has a rapid, potent and sustained acid inhibitory effect. In this review, we summarized and discussed current evidence regarding the role of vonoprazan in terms of mucosal healing, maintaining remission and symptom relief for the management of EE, including the initial and maintenance treatment of EE, as well as for PPI-resistant EE patients. Safety concerns and cost-effectiveness analysis of vonoprazan were also mentioned in the article. As a potent and well-tolerated acid blocker, vonoprazan has the potential to become a novel option for the management of EE.
Introduction
Gastroesophageal reflux disease (GERD) is a common acid-related disease caused by the reflux of gastroduodenal contents entering into the esophagus or mouth. 1 It can be classified into non-erosive gastroesophageal reflux disease, erosive esophagitis (EE) and Barrett’s esophagus according to the endoscopic manifestations. GERD is currently one of the most prevalent gastrointestinal diseases in Western countries and affects 13% of the population worldwide. 2 Although it was once thought to be less common in Asian countries, the prevalence of GERD has now reached over 10% in Japan, China and India.3,4 GERD not only results in huge burden on health-related quality of life and work productivity, but also plays an important role in the occurrence of esophageal adenocarcinoma.5,6
Acid suppression has been considered as the mainstay treatment for EE, and proton pump inhibitors (PPIs) represent the drug of choice in the last several decades. In spite of the proven acid-suppressing efficacy for symptom relief as well as mucosal healing, 7 a few limitations related to PPIs have been reported lately. First of all, many early-generation PPIs show a delay in action onset, and several dose cycles may be needed that takes 3–5 days until maximal efficacy are fully reached. 8 Besides, PPIs may be inadequate to fully overcome nocturnal acid breakthrough, and a histamine H2 receptor antagonist (H2RA) is usually required before bedtime to achieve better therapeutic gain. 9 However, after a few weeks, tolerance does develop due to H2RA upregulation, resulting in reduced efficacy. 10 It has been well documented that the current PPI treatment may not be sufficient for EE, and 10–20% of patients with Los Angeles C and D (LA-C/D) grade EE do not gain complete mucosal healing and symptom control despite 8-week double-dose PPI therapies.11,12 As a result, there is unmet need in the management of EE.
Vonoprazan (TAK-438), a novel potassium-competitive acid blocker (P-CAB), received its first global approval in 2014 and was launched in 2015 for use as an acid suppressant. In the present review, we discussed and summarized the current evidence regarding the efficacy and safety of vonoprazan in the management of EE.
Vonoprazan, a new P-CAB
Both PPIs and P-CABs act on gastric H+, K+-adenosine triphosphatase (H+/K+-ATPase), which is responsible for the critical final step of the gastric acid section. 13 PPIs block the action of H+/K+ ATPase of active phase by irreversible covalent binding, acidic environment is needed for its activation. In contrast to PPIs, P-CABs inhibit the H+/K+ ATPase of both active and resting phase by reversible K+-competitive ionic binding independent of gastric acid activation. 14
Clinical development of P-CABs
Many candidate compounds categorized as P-CABs have been developed since 1980s. SCH28080 and linaprazan (AZD0865), which are imidazopyridine derivatives, are the first batch of prototype P-CABs. However, the clinical development of them was discontinued due to dose-related hepatotoxicity concerns or limited clinical benefit compared with PPIs in terms of mucosal healing and symptom relief.15–18 Revaprazan (YH1885), which is pyrimidine derivative, is the first P-CAB developed for clinical use, and is approved for the treatment of duodenal ulcer and gastric ulcer in South Korea.19,20 Revaprazan exhibited similar efficacy as conventional PPIs in terms of gastric acid control. 21 Vonoprazan (TAK-438), based on the pyrrole structure, is the second P-CAB introduced in clinical use. The efficacy and safety of vonoprazan for the treatment of EE has been reported in recent clinical trials, which is summarized and discussed later in the current review. Tegoprazan, which is based on the benzimidazole structure, is a latest P-CAB currently under development and has recently showed potential for the treatment of GERD and peptic ulcers.22,23
Pharmacological and pharmacokinetic profiles of vonoprazan
Notable differences of vonoprazan in pharmacological and pharmacokinetic profiles from traditional PPIs have been demonstrated. Matsukawa J, et al. performed an animal experiment using primary cultured rabbit gastric glands with 2-h pre-incubation of either vonoprazan or lansoprazole, vonoprazan has a high level of accumulation in both the resting and the actively secreting oxyntic gastric glands while lansoprazole only accumulated in resting glands, which suggested that the effect of vonoprazan does not rely on the gastric acid activation. 24 Moreover, as shown in Japanese and UK phase I studies, 20 mg of vonoprazan resulted in almost the same maximum concentration as well as area under the curve from time 0 to 48 h (AUC 0–48 h) values under satiated and fasting conditions. 25 Another recent phase I study conducted in healthy male and female Caucasian subjects exhibited similar results. 26 Therefore, the absorption of vonoprazan is not affected by food intake, and it can be administered regardless of meal, leading to higher medication compliance than that of PPIs. Vonoprazan is lipophilic and weak bases characterized by a higher pKa value of 9.06 than that of PPIs (3.8–5.0), thus making it highly accumulated and protonated instantly in the acid space such as the parietal cells. 27 Meanwhile, vonoprazan is an acid-stable, water-soluble pyrrole and can be administered in immediate-release formulations, and therefore has a rapid absorption. As demonstrated in healthy subjects, rapid maximal plasma concentration was achieved after oral administration of vonoprazan with Tmax of up to 2 h as well as an estimated elimination half-life of up to 9 h, which was higher than that of conventional PPIs (1–2 h). 28 These characteristics lead to a rapid onset and prolonged action of its acid inhibitory effect starting from the first day of administration. This was supported by a crossover study to evaluate the acid-inhibitory effects of vonoprazan versus two control PPIs (esomeprazole and rabeprazole) in healthy adult male subjects. It was found that vonoprazan (20 mg) exhibited greater gastric pH 4 holding time ratio (HTR) than esomeprazole (20 mg) or rabeprazole (10 mg) on both days 1 and 7. 29 In addition, vonoprazan is metabolized in the liver mainly via CYP3A pathway of cytochrome P450, 30 which is distinct from PPIs whose metabolisms are mostly via CYP2C19 pathway. 31 Therefore, there is less influence of the CYP2C19 polymorphism on drug exposure of vonoprazan. An exploratory study conducted in healthy Japanese volunteers with mixed CYP2C19 genotypes found that pH 4 HTR and pH 5 HTR were higher with vonoprazan 20 mg than those with esomeprazole 20 mg irrespective of CYP2C19 status. 32
Effect of vonoprazan versus different PPIs on intragastric pH
The cure rate of GERD is largely dependent on maintaining an intragastric pH of 4 or greater. 33 The reported intragastric pH 4 HTR needed for GERD management is 83–91% (20–22 h/day). 33 According to the latest Chinese and American College of Gastroenterology (ACG) guideline for the management of GERD,34,35 GERD is initially treated with once-daily administration of a PPI (i.e. lansoprazole 30 mg, omeprazole 20 mg, rabeprazole 10 mg, esomeprazole 20 mg or pantoprazole 40 mg). Of note, these five PPIs are not equivalent in the inhibitory effect on intragastric pH, and different way of administration may result in different potency.25,28,29,32,36–46 As summarized in Table 1, a standard daily dose of a PPI may be insufficient to achieve the pH 4 HTR required for GERD management. On day 1 of dosing, the pH 4 HTR is approximately 22.6–50% with lansoprazole,38,39 24.7–65.1% with omeprazole,42,43 26.3–34.3% with rabeprazole,29,37 23.9% with esomeprazole 29 and 31% with pantoprazole, 39 respectively. After repeated dosing, the pH 4 HTR is approximately 42.3–63% with lansoprazole,38,39 46–90% with omeprazole,41–43 49–71.8% with rabeprazole,29,44,45 61.2–68% with esomeprazole,29,32 and 56% with pantoprazole, 39 respectively.
Effect of vonoprazan and different PPIs on intragastric pH.

HTRs, holding time ratios; n/a, not available; PPI, proton pump inhibitors.
Previous trials have demonstrated greater acid-inhibitory effect of vonoprazan 20 mg daily compared with a PPI of standard daily dosing. In a randomized open-label crossover study involving 20 healthy Japanese male adults, the pH 4 HTR of vonoprazan 20 mg daily was significantly higher than that of esomeprazole 20 mg daily or rabeprazole 10 mg daily on both days 1 and 7. In addition, the day 1 to day 7 ratio of pH 4 HTRs was greater with vonoprazan than that was with esomeprazole or rabeprazole (>0.8, 0.370, 0.393, respectively). 29 A recent phase I trial from U.S. subjects also exhibited the higher pH 4 HTR with vonoprazan 20 mg daily than that with lansoprazole 30 mg daily on day 1 and day 7 (62.4% versus 22.6%, 87.8% versus 42.3%, both p < 0.0001). 38 In a study involving 28 healthy Japanese volunteers, median pH 4 HTR of vonoprazan 20 mg daily was greater than that of esomeprazole 20 mg daily or twice daily (95% versus 68%, p < 0.001; 95% versus 91%, p = 0.019). 32 Since vonoprazan is a very potent and long-acting acid inhibitor, the proper comparison is probably a PPI with standard or higher doses administered twice daily (BID). More recently, Takeuchi T, et al conducted a crossover study to evaluate the acid-inhibitory effect of the standard dose of vonoprazan versus rabeprazole 10/20 mg BID in three cohorts of 10 healthy Japanese volunteers, and found that the pH 4 HTR of vonoprazan 20 mg daily was greater than those of rabeprazole 10 mg BID (88.4% versus 53.8%; p = 0.001) and rabeprazole 20 mg BID (95.0% versus 74.5%; p = 0.0015). 36 Thus, vonoprazan exerts a more potent and durable acid-inhibitory effect compared with 2–4 times the standard daily dose of rabeprazole administered in two divided doses.
Overall, vonoprazan displays a rapid, potent and sustained acid inhibitory effect, and seems to have the potential to become a new therapeutic option in the management of GERD from a clinical standpoint.
Role of vonoprazan for EE
The therapeutic target for EE is to relieve symptoms, heal and maintain remission of EE, prevent complications, and improve health-related quality of life. According to the latest Chinese and ACG guidelines,34,35 initial treatment and maintenance phases should be applied in the treatment for EE. Currently, P-CABs have shown fair evidence in both phases of EE treatment.
Initial treatment of EE
In a phase II randomized, double-blind, dose-ranging clinical trial, Japanese subjects with endoscopically confirmed EE (LA grades A–D) were randomized to receive 8-week therapy with a range of doses (5, 10, 20, 40 mg, once daily) of vonoprazan or lansoprazole (30 mg, once daily) to investigate the efficacy of mucosal healing. Remarkably, all doses of vonoprazan were non-inferior to lansoprazole for mucosal healing, and had comparable incidence of adverse events. Vonoprazan exhibited good tolerability even with the highest tested dose of 40 mg. 47 Following the phase II results, Ashida K et al. subsequently conducted a phase III clinical trial in Japan, in which EE patients were randomized to receive vonoprazan 20 mg or lansoprazole 30 mg once daily for 8 weeks. The non-inferiority of vonoprazan to lansoprazole in the healing of EE was verified (overall healing rate at 8 weeks with vonoprazan 20 mg and lansoprazole 30 mg: 99.0% versus 95.5%, P valuenon-inferiority < 0.0001). 48
We further conducted a phase III, randomized, double-blind, double-dummy, parallel-group study 49 across Asian countries including mainland China, Taiwan, Malaysia and South Korea. The 8-week EE healing rates in the vonoprazan (n = 238) and lansoprazole (n = 230) arms were 92.4% and 91.3%, respectively, which was consistent with the results reported in the previous phase III study in Japan. 48 It is worth noting that patients with LA grade C/D in the vonoprazan arm had numerically higher mucosal healing rate when compared with those in the lansoprazole arm in both Asian multicentre study and Japanese study, which indicated the superiority of vonoprazan in the patients with higher grade EE to some extent. Of note, most of reported studies used lansoprazole as the control arm to investigate the efficacy of vonoprazan for treating EE. Since different PPIs are not equivalent and the potency of PPIs for treating EE partly depends on the type, frequency, dose as well as duration of treatment, thus results of comparative studies comparing vonoprazan and PPIs may depend on choice of PPIs and the way of administration. A meta-analysis consisting of six randomized controlled trials for vonoprazan 20 mg and PPIs suggested that vonoprazan may be non-inferior to PPIs for the treatment of GERD, with risk ratios for efficacy and adverse events of 1.06 [95% confidence interval (CI): 0.99–1.13], 1.08 (95% CI: 0.96–1.22), respectively. Meanwhile, subgroup analysis demonstrated the superiority of vonoprazan to PPIs for those with severe EE. 50 Similar findings were shown in a Bayesian network meta-analysis. 51 As healing of EE is correlated with intragastric pH, the superiority of vonoprazan to PPIs for healing EE is mainly due to the superior acid suppression of vonoprazan, as represented by the greater gastric pH 4 HTR than PPIs in healthy subjects.29,32,36,38 Future direction should be comparing and determining the efficacy of vonoprazan 20 mg with a PPI of standard or higher doses administered once or twice daily for healing EE.
Besides mucosal healing, symptomatic relief in GERD was also a target to reach. As P-CABs can block gastric acid secretion rapidly, it can provide heartburn relief soon after starting initial treatment. A randomized, parallel-group, double-blind, comparative clinical study was conducted comparing vonoprazan and lansoprazole with respect to rapid heartburn relief. 52 In this study, heartburn was relieved significantly sooner with vonoprazan than with lansoprazole. Complete heartburn relief was achieved in 31.3% and 12.5% of patients on the first day of treatment with vonoprazan and lansoprazole. Of note, complete nocturnal heartburn relief was achieved in significantly more patients in the vonoprazan arm than in the lansoprazole arm (p < 0.01). Therefore, vonoprazan might show the advantage of nocturnal symptom control considering nighttime heartburn affects sleep in 79% of GERD patients 53 (Table 2).
Summary of vonoprazan on the initial treatment of EE.
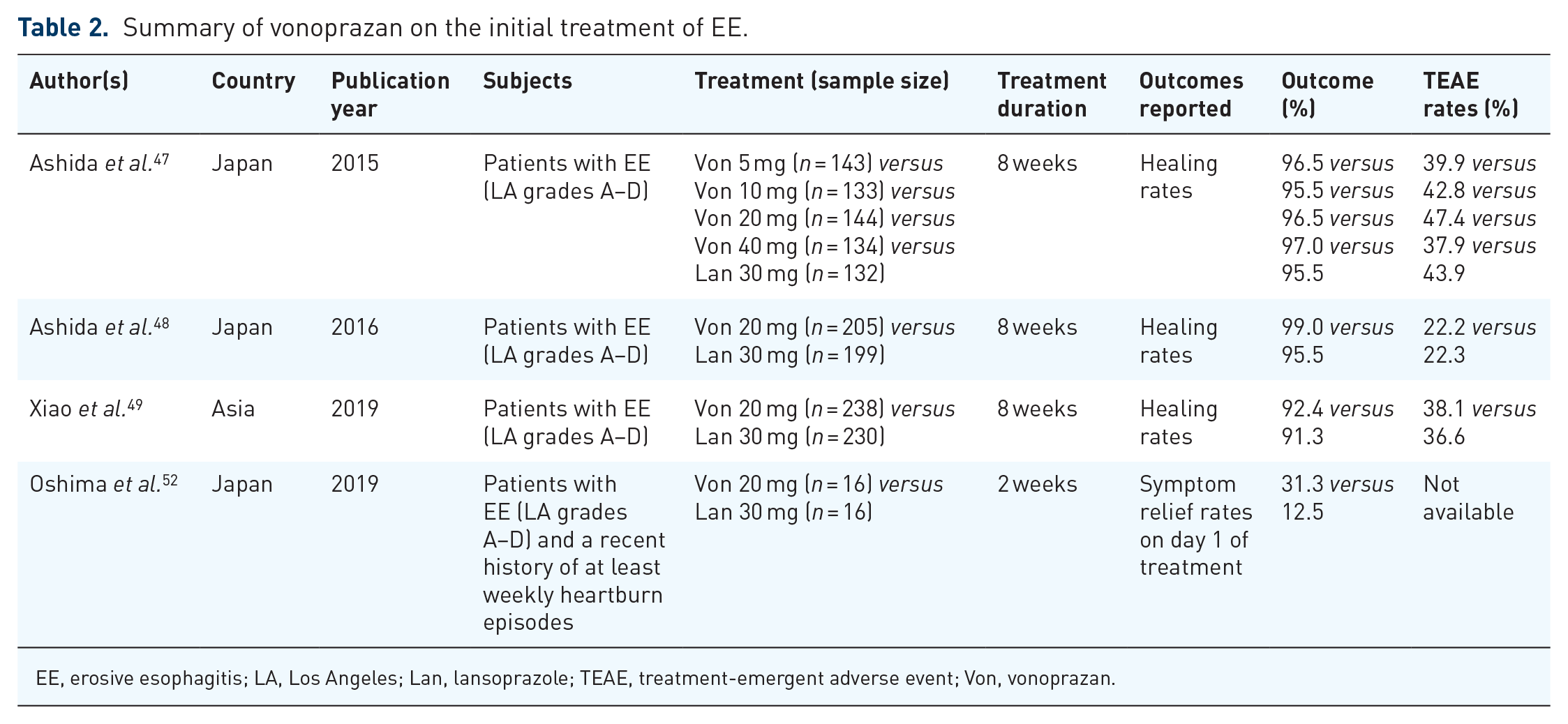
EE, erosive esophagitis; LA, Los Angeles; Lan, lansoprazole; TEAE, treatment-emergent adverse event; Von, vonoprazan.
Maintenance treatment of EE
The efficacy and safety of vonoprazan versus PPIs for the maintenance treatment of EE have also been explored. The abovementioned phase III study in Japan by Ashida et al. 48 consisted of a second sequential long-term maintenance phase, in which those who achieved healed EE during the initial treatment were randomized to receive 52-week maintenance therapy of either vonoprazan 10 or 20 mg. It turned out that EE recurred in less than 10% of patients treated with vonoprazan 10 mg or 20 mg. Besides, vonoprazan was well tolerated for up to 52 weeks, and the most common treatment-emergent adverse event of vonoprazan was nasopharyngitis. 48 Another randomized, double-blind, parallel-group, phase III clinical study in Japan compared the efficacy of vonoprazan and lansoprazole for maintenance therapy of 24 weeks for EE. The recurrence rates of EE were 16.8%, 5.1% and 2.0% with lansoprazole 15 mg, vonoprazan 10 mg and vonoprazan 20 mg, respectively, indicating the non-inferiority of vonoprazan to lansoprazole in maintaining remission of healed EE (both p valuenon-inferiority < 0.0001). 54 However, the patients in the lansoprazole arm received only 15 mg per day, which would undermine the potency of the study. We recently conducted a phase III, double-blind, parallel-group, multicenter study 55 in non-Japanese Asian patients with healed EE. The EE recurrence rates during the 24 weeks were 13.8%, 14.0% and 25.0% in the vonoprazan 10-mg, vonoprazan 20-mg and lansoprazole 15-mg group, respectively. Both doses of vonoprazan showed superiority to lansoprazole in preventing recurrences of EE during 24-week maintenance therapy (Table 3).
Summary of vonoprazan on the maintenance treatment of EE.
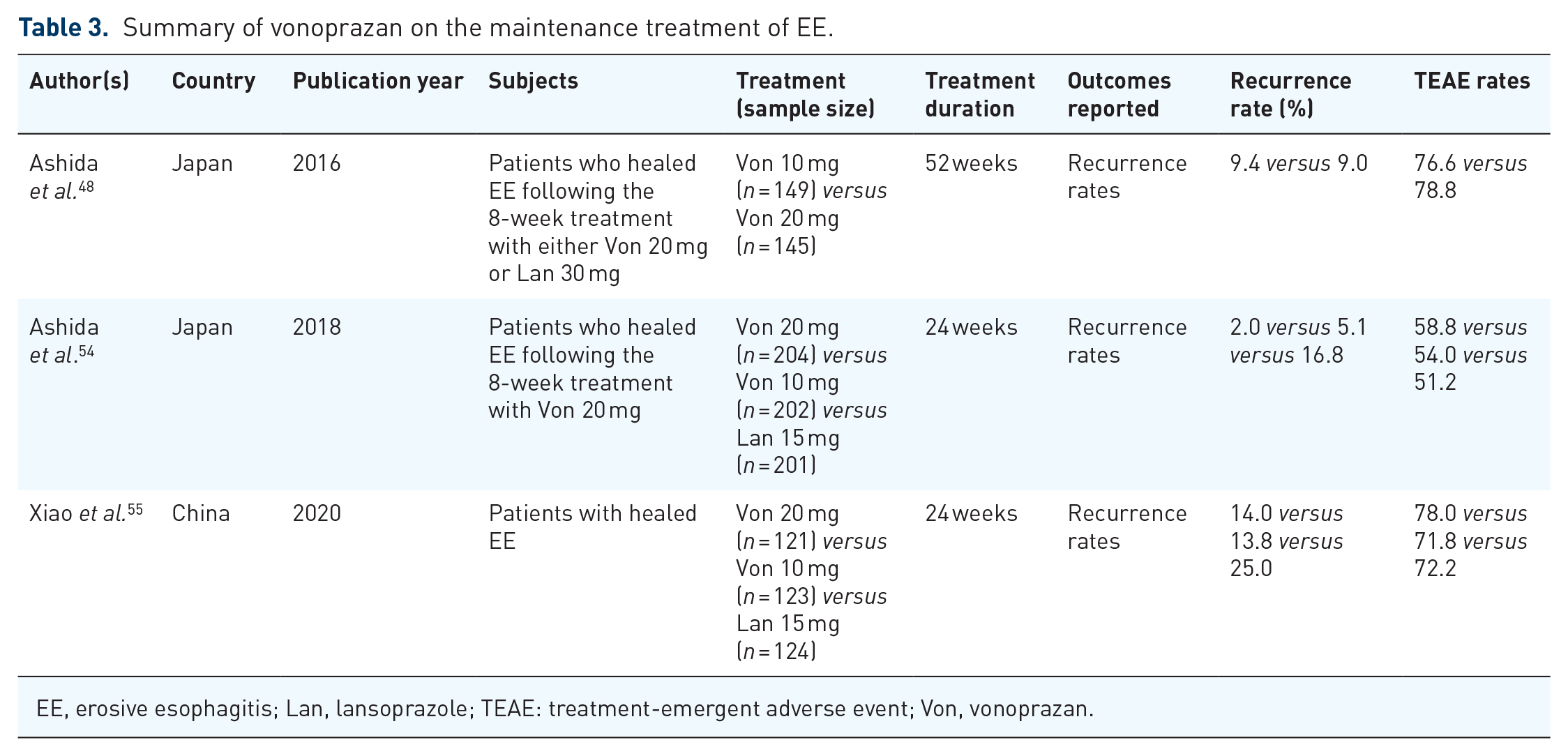
EE, erosive esophagitis; Lan, lansoprazole; TEAE: treatment-emergent adverse event; Von, vonoprazan.
Results of comparative studies comparing vonoprazan and PPIs may depend on choice of PPIs and the way of administration. Miwa H et al tried to compare the maintenance efficacy of vonoprazan versus different PPIs by a Bayesian network meta-analysis, which combines both direct and indirect evidence of 22 randomized controlled trials, and found that the maintenance effect of vonoprazan 10 mg was superior to esomeprazole 10 mg and omeprazole 10 mg, with odds ratios of 13.92 (95% CI 1.70–114.21), 9.23 (95% CI 1.17–68.72), respectively. No superiority of vonoprazan 10 mg was demonstrated to rabeprazole 10 mg as well as lansoprazole 15 mg. Vonoprazan 20 mg showed superior maintenance effect to all of the above PPIs. 56 Further direct head-to-head comparison trials are necessary to compare the efficacy of vonoprazan with different PPIs for the maintenance treatment of EE.
PPI-resistant EE
Around 10–15% of patients of EE do not achieve complete mucosal healing and symptom control despite 8-week double-dose PPI therapies,11,12 which may be attribute to the lack of sufficient acid inhibition, persistent non-acid reflux, reflux sensitivity as well as non-GERD causes. 3 A study using esophageal impedance-pH testing demonstrated that pathological reflux cannot be completely decreased even on twice-daily PPI treatment, especially during nocturnal period, 57 while vonoprazan exhibits its acid inhibitory effect for both daytime and nighttime. There is inter-patient variability in efficacy of PPIs due to CYP2C19 metabolism, which is distinct from vonoprazan as it is metabolized mainly via CYP3A pathway. The action of PPIs relies on the gastric acid activation and requires mealtime dosing to ensure adequate effect, leading to poor compliance with PPIs. One study found less than 50% of patients to be compliant with once-daily PPI use by 3 months. 58 The absorption of vonoprazan is not affected by food intake and can be administered regardless of meal, thus resulting in better compliance.
The first randomized, double-blind, multicenter study evaluating the efficacy of vonoprazan in patients with PPI-resistant EE was reported in 2017. 59 A total of 19 patients, who had EE despite previous PPIs therapy were randomly assigned to the vonoprazan 20 mg and 40 mg arm. It was showed that both dosage of vonoprazan could inhibit gastric acid secretion over a 24-h period with a significant increase in gastric pH 4 HTR, and could completely heal mucosal erosions at 8 weeks in more than 60% of PPI-resistant EE patients. 59 Hoshino S et al performed a prospective study among 24 patients with PPI-resistant EE. The study consisted of a 4-week initial treatment phase of vonoprazan 20 mg followed by an 8-week maintenance phase of vonoprazan 10 mg for those who healed EE after the initial phase. It was found that esophageal mucosal breaks were treated successfully in 21 (87.5%) out of 24 patients, and the frequency scale for symptoms of GERD (FSSG) score was also significantly relieved at 4 weeks after the initial phase. Moreover, the mucosal healing was maintained in 16 (76.2%) out of 21 patients during the second phase of the study. 60 To investigate the efficacy of long-term maintenance therapy with vonoprazan for PPI-resistant EE, the same team subsequently conducted a 52-week maintenance study with 10 mg vonoprazan among 16 patients with PPI-resistant EE who achieved remission from the above preceding study. They found that endoscopic remission was maintained at 52 weeks in 93.8% of patients with PPI-resistant EE. No significant change of the FSSG score was observed at 52 weeks, indicating the symptom relief also maintained. 61 Similar results were also found in other trials62–64 (Table 4). Therefore, vonoprazan may exhibit a potential role for fulfilling the unmet need in terms of treating PPI-resistant EE. However, all of these trials were lack of a placebo or active control arm and with a small sample size. Large-scale, well-designed randomized controlled trials (with a placebo or active control arm) are necessary to verify the role of vonoprazan for patients with PPI-resistant EE.
Summary of vonoprazan for the PPI-resistant EE.

EE, erosive esophagitis; FSSG, Frequency Scale for the Symptoms of GERD; HTR, holding time ratio; Lan, lansoprazole; PPI, proton pump inhibitor; TEAE, treatment-emergent adverse event; Von, vonoprazan.
Vonoprazan in different populations
To date, almost all clinical trials of vonoprazan so far available have been conducted in Asian populations while there are fewer clinical data in non-Asian patients. It is reported that the severity of EE, level of esophageal acid exposure as well as the genetic polymorphism affecting hepatic metabolism of drugs is different between Asians and non-Asians,65,66 raising the possibility that the efficacy of vonoprazan may differ between them.
A recently reported phase I trial from U.S. subjects showed that vonoprazan (20 mg daily) provided higher proportions of 24-h period with intragastric pH > 4 than lansoprazole (30 mg daily) on day 1 and day 7 (62.4% versus 22.6%, 87.8% versus 42.3%, both p < 0.0001). 38 Comparison of data from Japanese and European in phase I trials showed no clinically significant differences in pharmacodynamic properties of vonoprazan between Japanese and European healthy volunteers, suggesting translatability of data between these populations.25,28 Lately, Scarpignato, C et al conducted a population pharmacokinetic analysis to identify the factors that could affect drug exposure in population subgroups. The authors found that clinical covariates such as race, disease status, age and weight had a limited impact on vonoprazan exposure or safety. The limited impact of race suggests that efficacy and safety data for vonoprazan are translatable between Asian populations and non-Asian populations. 67
Efficacy of vonoprazan in patients with H. pylori infection or EE should be fully evaluated in different populations. Vonoprazan-based triple therapy has demonstrated higher eradication rates than lansoprazole-based triple therapy in both Asian 68 and, lately, in US and European studies (pHalcon-HP, NCT04167670). Currently, clinical trials of vonoprazan for the treatment of EE in the United States and Europe are underway and worth expecting.
Cost-effectiveness analysis of vonoprazan
‘Top-down’ strategy using vonoprazan and ‘step-up’ strategy using PPIs as initial treatment followed by a switch to vonoprazan for PPI non-responders has been compared to clarify the cost-effectiveness. A cost-utility analysis using a Markov simulation model found that the expected costs of the vonoprazan-, rabeprazole- and esomeprazole-first strategies were ¥36,194, ¥41,105 and ¥76,719, respectively. Meanwhile, the quality-adjusted life years gains for vonoprazan-first strategy compared with the rabeprazole- and esomeprazole-first strategies were 0.003 and 0.014, respectively. Subgroup analysis according to the severity of EE exhibited similar results. The findings indicated the superiority of the ‘top-down’ strategy with vonoprazan over PPI-first strategies in terms of cost-effectiveness, regardless of the severity of EE. 69 Similar results were shown in another cost-effectiveness analysis comparing vonoprazan and lansoprazole as the initial treatment for EE. 70 With regard to the long-term strategies with vonoprazan for the management of EE, a cost-utility analysis using a health state transition model was performed to compare the cost-effectiveness of the following four different strategies, including intermittent P-CAB using vonoprazan, intermittent PPI using lansoprazole, maintenance P-CAB using vonoprazan and maintenance PPI using lansoprazole. The cost-effectiveness ratios (Yen/days without EE) showed that the intermittent P-CAB strategy was the most cost-effective, with the least days for which medication were required and least physician visits. 71 It is worth noting that data of pharmacoeconomic evaluation in one country cannot be translated to another region, due to the many differences in the National Health Systems. Therefore, more cost-effectiveness studies are encouraged to be performed in multi-centers across different regions in the future. Future direction should be fully exploring the cost-effectiveness of both initial and long-term treatment options with vonoprazan for EE.
Safety profile of vonoprazan
The safety of a new drug is always a concern for clinicians; thus, the safety profile of vonoprazan has been investigated throughout the course of its clinical development. In phase I studies, vonoprazan at all tested doses was safe and well tolerated in healthy male volunteers, as compared with placebo or PPIs.25,28,29 During the phase II and III studies, the common adverse events of vonoprazan were nasopharyngitis, diarrhea, as well as dyspepsia with mild to moderate degree and well tolerated.47,48 Comparable incidence rates of adverse events were observed between vonoprazan (22.2%–47.4%) and lansoprazole (22.3%–43.9%).47–49 The incidence of adverse events of vonoprazan in the maintenance treatment of up to 52 weeks was higher than that in short-term treatment, 48 although the common adverse events were similar. In a phase III study comparing vonoprazan and lansoprazole for preventing ulcer recurrence during long-term non-steroidal anti-inflammatory drug therapy, the safety profiles of vonoprazan of both 10 mg and 20 mg doses were similar to that of lansoprazole 15 mg over 104 weeks of therapy. Adverse events for which a causal relationship could not be eliminated occurred in less than 20% of patients in each group, and no new safety issue was identified. 72
Liver function abnormality is one of the most concerned issues for developing new drugs. Since the clinical development of previous P-CABs, such as SCH28080 and linaprazan (AZD0865), was stopped because of severe dose-related hepatotoxicity, it is worth noting whether vonoprazan would cause hepatotoxicity. As demonstrated in reported trials,28,29,47,48 both short-term and long-term administration of vonoprazan did not cause clinical meaningfully abnormal differences of alanine transaminase level and aspartate transaminase level, probably due to the different chemical structure from other P-CABs.
Compared with conventional PPIs, greater increase in serum gastrin level was observed following the treatment of vonoprazan and returned to the baseline level after the cease of treatment, which may be attributed to stronger gastric acid inhibition. As demonstrated in the phase III trial, 48 the mean serum gastrin level increased after 8-week initial treatment of vonoprazan and lansoprazole, while more remarkable in vonoprazan group (1.5 times higher than in lansoprazole group). Longer treatment duration caused further increase in serum gastrin in vonoprazan arm during 52-week maintenance treatment. A recent study comparing vonoprazan 10 mg and 20 mg daily with lansoprazole 15 mg and 30 mg daily for a 4-year maintenance therapy of EE found significantly higher level of serum gastrin in vonoprazan than in lansoprazole group. 73
Gastric histologic changes have been documented with vonoprazan. Hypergastrinemia is considered as one of the risk factors of gastric neuroendocrine tumors; therefore, cautions should be brought during the long-term treatment with vonoprazan although currently no relevant effects on gastric neuroendocrine cells were reported. A greater hyperplasia of parietal cells and G cells was found with vonoprazan than with lansoprazole, as observed in the abovementioned 4-year study. 73 Small white protrusions called ‘stardust’ gastric mucosa, which was represented histologically by a mucus pool within a dilated duct surrounded by flattened glandular epithelium, appeared more frequently in the stomachs of patients receiving vonoprazan than in those not (4.9% versus 0.2%, p < 0.001). 74 Vonoprazan-associated gastric mucosa redness, represented histologically by inflammatory cell infiltration, parietal cell protrusions and oxyntic gland dilatation, was reported in four cases treated with vonoprazan and disappeared after drug discontinuation. 75 White globe appearance, which is a novel endoscopic marker useful in the diagnosis of early gastric cancer, was observed in the noncancerous stomach of a 68-year-old woman after 1-year administration of vonoprazan and then markedly reduced after changing to esomeprazole treatment. 76 The first case of foveolar-type gastric adenocarcinoma was recently reported in a man with EE after 156-week maintenance therapy of vonoprazan 10 mg daily. 77 Although the clinical relevance of these alteration remains unclear, a careful regular check of possible gastric histologic changes is necessary, especially during long-term administration.
Potential drug-to-drug interactions with vonoprazan have come into notice lately. It is reported that vonoprazan is metabolized in the liver mainly driven by CYP3A via cytochrome P450 pathway 30 ; thus, it is conceivable that there is interaction of vonoprazan with other drugs undergoing metabolism through this pathway. For instance, clarithromycin, a recognized strong CYP3A inhibitor, is a common component of the triple therapy for the eradication of H. pylori. A phase I study demonstrated that the triple therapy with vonoprazan–amoxicillin–clarithromycin increased the plasma concentration of both vonoprazan and clarithromycin while no changes were observed for amoxicillin, indicating a interference of the metabolism of vonoprazan and clarithromycin, probably via inhibition of CYP3A. 78 A recent study presented in Digestive Disease Week 2022 demonstrated that the plasma exposure of midazolam, a sensitive CYP3A substrate, increased following the coadministration of vonoprazan, as reflected by 1.9-fold increases in Cmax and AUC values. 79 These data suggest that plasma concentration of other drugs metabolized via CYP3A pathway may increase when administrated concomitantly with vonoprazan, thus lower doses with a narrow therapeutic index should be used. Another study used physiologically based pharmacokinetic modeling to evaluate the impact of moderate and strong CYP3A inducer on vonoprazan exposure, and found that the concentration of vonoprazan decreased by up to 81% when coadministrated with rifampin, a strong CYP3A inducer, and by up to 54% when coadministrated with efavirenz, a moderate CYP3A inducer. That is to say, vonoprazan exposure would decrease when administrated concomitantly with moderate-to-strong CYP3A inducers. Therefore, coadministration of vonoprazan with moderate-to-strong CYP3A inducers should be avoided. 80 In addition, there is potential drug-to-drug interaction of vonoprazan with drugs independent of CYP3A genetic polymorphism. It is reported that the concentration of tacrolimus increases with the co-administration of vonoprazan in kidney transplant recipients, which is not affected by the CYP3A genotype. 81 Drug-to-drug interactions with vonoprazan need further study.
In terms of other adverse events, there were case reports of severe hypomagnesemia due to use of vonoprazan, which improved after drug discontinuation.82,83 Changes in the gut microbiome have also been documented with vonoprazan. 84 Therefore, the long-term safety and tolerability of vonoprazan are yet to be confirmed.
Conclusions
As a novel P-CAB with different pharmacological and pharmacokinetic profiles, vonoprazan displays several benefits over PPIs, such as rapid onset of action, more potent and sustained acid suppression. From a clinical perspective, the non-inferiority of vonoprazan over conventional PPIs for the management of EE has been proven in current clinical trials to date. Therefore, vonoprazan might have the potential to become a novel option for the management of EE, especially for those with higher grade of EE and PPI-resistant EE patients. Of note, results of comparative studies comparing vonoprazan and PPIs may depend on choice of PPIs and the way of administration. Future direction should determine the efficacy of vonoprazan with a PPI of standard or higher doses administered once or twice daily for treating EE. Clinical trials of vonoprazan for the treatment of EE in the non-Asian populations are currently underway and worth expecting. Importantly, long-term safety profile of vonoprazan should be carefully considered as its indications will be further expanded. Future studies are required to explore the optimized strategies and cost-effectiveness of vonoprazan-based therapy, such as optimal dose, therapeutic duration, discontinuation, etc.
