Abstract
Background:
Vonoprazan, a novel acid-suppressive drug, is non-inferior to proton pump inhibitors (PPIs) for the management of gastric acid-related diseases. However, the safety of vonoprazan has not been systematically evaluated yet.
Objectives:
To elucidate the incidence and type of adverse events (AEs) in patients taking vonoprazan.
Design:
Systematic review and meta-analysis.
Data sources and methods:
PubMed, EMBASE, and Cochrane Library databases were searched for all studies reporting the safety of vonoprazan. The incidences of any AEs, drug-related AEs, serious AEs, AEs leading to drug discontinuation, and common AEs were pooled. Odds ratios (ORs) were calculated to compare the incidence of AEs between patients taking vonoprazan and PPIs.
Results:
Seventy-seven studies were included. The pooled incidences of any AEs, drug-related AEs, serious AEs, and AEs leading to drug discontinuation were 20, 7, 1, and 1%, respectively. The incidences of any AEs (OR = 0.96, p = 0.66), drug-related AEs (OR = 1.10, p = 0.44), serious AEs (OR = 1.14, p = 0.36), and AEs leading to drug discontinuation (OR = 1.09, p = 0.55) were not significantly different between patients taking vonoprazan and PPIs. In subgroup analyses, patients with peptic ulcer disease (PUD) had higher incidences of any AEs, serious AEs, and AEs leading to drug discontinuation than those with gastroesophageal reflux disease (GERD), Helicobacter pylori (H. pylori) infection, and artificial ulcer after gastric endoscopic submucosal dissection (ESD), but patients with H. pylori infection had a higher incidence of drug-related AEs than those with PUD, GERD, and artificial ulcer after gastric ESD. The incidence of AEs was higher in patients taking long-term use of vonoprazan than those taking short-term use of vonoprazan.
Conclusion:
Vonoprazan is well tolerated and shows similar safety compared to PPIs. The safety of vonoprazan may be primarily influenced by its indications and duration.
Registration:
PROSPERO CRD42022314982.
Graphical abstract

Introduction
Acid-suppressive agents are widely used for the management of various upper gastrointestinal diseases, such as peptic ulcer disease (PUD), gastroesophageal reflux disease (GERD), Helicobacter pylori (H. pylori) infection, artificial ulcers after gastric endoscopic submucosal dissection (ESD), and Zollinger–Ellison syndrome. Histamine-2 receptor antagonists, the first class of acid-suppressive agents, have limited efficacy and poor tolerance. 1 Subsequently, in the late 1980s, proton pump inhibitors (PPIs) were introduced into our clinical practice, which strengthened the efficacy of management of gastric acid-related disorders. 2 However, several gaps remained, such as insufficient inhibition of gastric acid, nocturnal acid breakthrough, poor compliance, slow onset of action, and drug efficacy by CYP2C19 gene polymorphisms.1,3 Vonoprazan, a new potassium-competitive acid blocker, has been approved for the management of acid-related diseases in Asia, South America, and some parts of North America. 4 It acts on the key enzyme H+/K+-ATPase in the final step of gastric acid secretion by parietal cells, thereby hindering the exchange of H+ and K+ and inhibiting gastric acid secretion. 5 Notably, vonoprazan has some advantages as compared to PPIs.1,6–9 First, its activation is independent of acidic environment. Second, its onset of action is rapid, because its peak concentration can be reached quickly after 2 h of oral administration. Third, its efficacy is not affected by meals. Fourth, it can inhibit both activated and resting proton pumps in the gastric parietal cells, and the first dose can achieve the maximum acid-suppression and maintain its stability. Its plasma elimination half-life time is 5.7–8.8 h. Fifth, it is mainly metabolized by hepatic CYP3A4/5 enzyme, but not affected by CYP2C19 gene polymorphisms. Taken together, vonoprazan seems to exert more potent and prolonged acid-suppressing effects than PPIs. 5 Current evidence has suggested that vonoprazan can be non-inferior to PPIs for the management of gastric acid-related disorders and more effective than PPIs for healing severe erosive esophagitis (EE) and eradicating H. pylori.10–15 Several practice guidelines have also recommended vonoprazan as the first-line choice for the management of EE and H. pylori infection.16,17 However, until now, the safety of vonoprazan has not been systematically evaluated yet. Therefore, we performed a systematic review and meta-analysis to elucidate the incidence and type of adverse events (AEs) in patients receiving vonoprazan, and to explore the risk factors affecting the safety of vonoprazan.
Methods
This meta-analysis was conducted according to the Preferred Reporting Items Systematic Reviews and Meta-Analyses (PRISMA) statement. The PRISMA checklist was shown in the Supplemental Material.
Registration
This meta-analysis was registered in PROSPERO with a registration number of CRD42022314982.
Literature search
Three major electronic databases (i.e., EMBASE, PubMed, and Cochrane Library) were searched. The following keywords were used: ‘Vonoprazan’, ‘Takecab’, and ‘TAK-438’. The last search was performed on 19 June 2022.
Study selection criteria
All eligible studies should evaluate the use of vonoprazan and report its safety data. Exclusion criteria were as follows: (1) duplicates; (2) guidelines, reviews, or meta-analyses; (3) case reports, comments, notes, letters, or protocols; (4) experimental or animal studies; (5) clinical trial registration alone; (6) the study population was healthy people; (7) vonoprazan was not used; and (8) the safety data could not be extracted. Publication language was not restricted.
Data extraction
Two researchers (WX and YS) independently extracted the following information from each included study: first author; publication year; type of publication; country; study design; enrollment period; sample size; indications for acid-suppressing agents; dose, frequency, and duration of vonoprazan, PPIs, or placebo; combined medications; total number and age range of patients taking vonoprazan, PPIs, or placebo; number of patients who experienced any AEs, drug-related AEs, serious AEs, AEs leading to drug discontinuation, and common AEs. Any AEs, drug-related AEs, serious AEs, and AEs leading to drug discontinuation are defined in accordance with the reports of each included study. Disagreement was resolved after discussion with another researcher (ZB).
Study quality assessment
The Cochrane Risk of Bias tool was used for assessing the quality of randomized controlled trials (RCTs) in terms of seven domains, including sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective outcome reporting, and other bias. The risk of bias is graded as ‘low risk’, ‘high risk’, and ‘uncertainty’.
The Newcastle–Ottawa Scale (NOS) was used for assessing the quality of cohort studies. It uses a semi-quantitative system to evaluate the quality of studies in the parts of selection, comparability, exposure, and outcomes. A total of nine stars is the highest score. A NOS score of 0–3, 4–6, and 7–9 represents low, moderate, and high quality, respectively.
The Johanna Briggs Institute Scale was used for assessing the quality of single-arm studies. 18 It includes 10 questions addressing the internal validity and risk of bias of case series, particularly confounding, selection, and information bias, in addition to the importance of clear reporting. If a study had a score of ⩽4, 5–6, and ⩾7, it would be of low, moderate, and high quality, respectively.
Outcomes
The primary outcome of this meta-analysis was the incidence of AEs, including any AEs, drug-related AEs, serious AEs, AEs leading to drug discontinuation, and common AEs, in patients taking vonoprazan.
The secondary outcome was the difference in the incidence of AEs between patients taking vonoprazan versus those taking PPIs.
Statistical analyses
Meta-analyses were performed using Stata software version 12.0 (Stata Corp, College Station, TX, USA), RStudio version 4.2.0 (R Foundation for Statistical Computing, Vienna, Austria), and Review Manager software version 5.4 (Cochrane Collaboration, Nordic Cochrane Centre, Copenhagen, Denmark). A random-effects model was employed. Dichotomous outcomes were expressed as odds ratios (ORs) with 95% confidence intervals (CIs). Cochrane Q test and the I2 statistics were employed to assess heterogeneity among studies, and p < 0.1 or I2 > 50% was considered as statistically significant heterogeneity. Publication bias was evaluated with the Eggers’ test, and p < 0.1 was considered as significant publication bias. Meta-regression analyses were employed to explore the sources of heterogeneity according to the pre-specified covariates, which included type of publication (Full-text versus Abstract versus Unpublished), study design (RCT versus non-RCT), sample size (>200 versus ⩽200), indications for vonoprazan (H. pylori infection versus GERD versus PUD versus Artificial ulcers after gastric ESD), duration of vonoprazan (>8 weeks versus ⩽8 weeks), ethnicity (Asian versus non-Asian), and age group (Adult versus Adolescent). If the number of included studies was ⩾2, subgroup analyses would be conducted according to the covariates mentioned above. In addition, we performed further subgroup analyses according to the duration of vonoprazan for the management of EE (short-term healing versus long-term maintenance) and PUD (short-term healing versus long-term prevention). p < 0.1 was considered as statistically significant interaction.
Results
Study selection and characteristics
Initially, 1514 papers were identified. Finally, 77 studies were included (Figure 1).10–15,19–89 The characteristics of included studies were listed in Table 1. These included studies which were published between 2015 and 2022. Seventy-four studies were conducted in Asia,10–15,19–22,24–28,30–53,55–89 and three in non-Asian countries.23,29,54 Thirty-four studies were RCTs,10–14,19–23,32–35,38,42–44,46,52,54,55,57,60,61,63,72,74,77,80,83,85,86,89 24 cohort studies,15,24,27,31,36,37,41,47,50,51,53,56,59,62,64–67,71,75,79,81,84,87 and 19 single-arm studies.25,26,28–30,39,40,45,48,49,58,68–70,73,76,78,82,88 The indications for vonoprazan included H. pylori infection (n = 49),11,15,19,22–27,29,30,33,34,36,37,39–41,45–47,50–53,56,58,59,62–67,71–75,77–82,84,85,87,89 GERD (n = 18),10,14,20,21,28,38,43,44,48,49,54,57,60,61,68–70,86 PUD (n = 6),12,13,32,42,55,76 and artificial ulcers after gastric ESD (n = 4).31,35,83,88 The sample size ranged from 12 to 2715. The duration of vonoprazan ranged from 1 to 104 weeks.

Flow chart of study selection.
Characteristics of included studies.
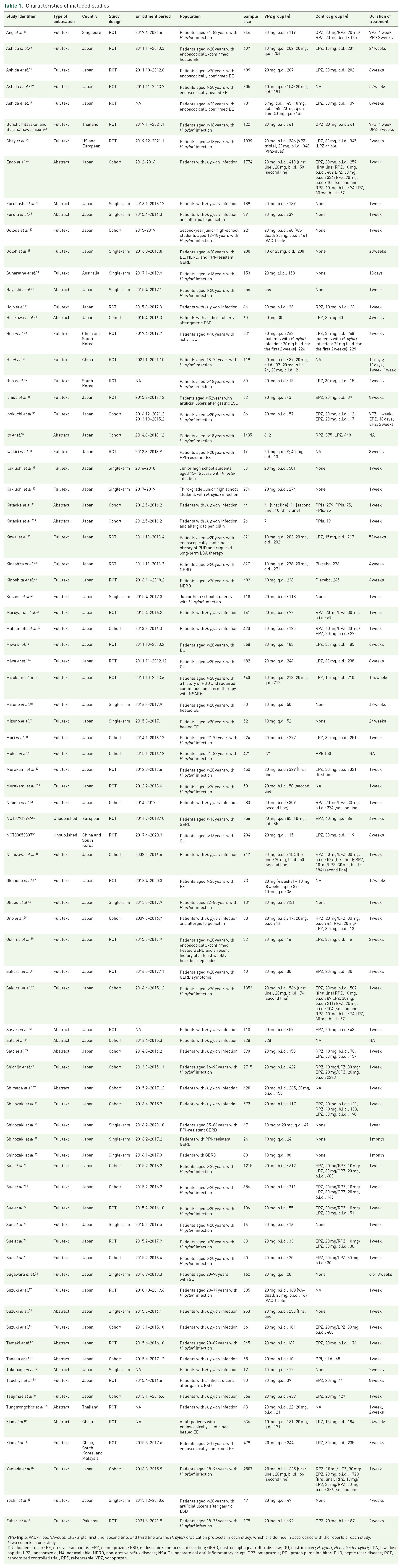
VPZ-triple, VAC-triple, VA-dual, LPZ-triple, first line, second line, and third line are the H. pylori eradication protocols in each study, which are defined in accordance with the reports of each study.
Two cohorts in one study.
DU, duodenal ulcer; EE, erosive esophagitis; EPZ, esomeprazole; ESD, endoscopic submucosal dissection; GERD, gastroesophageal reflux disease; GU, gastric ulcer; H. pylori, Helicobacter pylori; LDA, low-dose aspirin; LPZ, lansoprazole; NA, not available; NERD, non-erosive reflux disease; NSAIDs, nonsteroidal anti-inflammatory drugs; OPZ, omeprazole; PPI, proton pump inhibitor; PUD, peptic ulcer disease; RCT, randomized controlled trial; RPZ, rabeprazole; VPZ, vonoprazan.
Study quality
Quality of RCTs, cohort studies, and single-arm studies were summarized in Supplemental Figure 1, Supplemental Table 1, and Supplemental Table 2, respectively. Most of the included studies were of moderate and high quality.
Incidence of AEs in patients taking vonoprazan
Any AEs
Forty-four studies explored the incidence of any AEs in patients taking vonoprazan (Figure 2(a)). The pooled incidence of any AEs was 20% (95% CI = 13–29%) with significant heterogeneity (I2 = 98.9%; p < 0.01). Meta-regression analyses demonstrated that the heterogeneity might be attributed to the study design, sample size, and duration of vonoprazan (Supplemental Table 3). No significant publication bias was detected (p = 0.483).

Forest plots of the incidences of any AEs (a) and drug-related AEs (b) in patients taking vonoprazan.
Eight of the 44 studies reported the severity of any AEs. The pooled incidences of mild, moderate, and severe AEs were 86, 11, and 2%, respectively (Figure 2(a)).
In subgroup analyses, the pooled incidence of any AEs was 17, 20, 47, and 2% in patients taking vonoprazan for the management of H. pylori infection, GERD, PUD, and artificial ulcers after gastric ESD, respectively (pinteraction < 0.01); 40 and 68% in those for short-term healing of EE and long-term maintenance of healed EE, respectively (pinteraction = 0.01); 30 and 85% in those for short-term healing of PUD and long-term secondary prevention of PUD, respectively (pinteraction < 0.01); 43 and 17% in those for a duration of >8 weeks and ⩽8 weeks, respectively (pinteraction = 0.04); 22 and 13% in Asian and non-Asian patients taking vonoprazan, respectively (pinteraction = 0.42); and 28 and 21% in adult and adolescent patients taking vonoprazan, respectively (pinteraction = 0.33) (Figure 2(a)).
Drug-related AEs
Thirty-one studies explored the incidence of drug-related AEs in patients taking vonoprazan (Figure 2(b)). Meta-analysis demonstrated that the pooled incidence of drug-related AEs was 7% (95% CI = 5–9%) with significant heterogeneity (I2 = 88.8%; p < 0.01). Meta-regression analyses did not find any source of heterogeneity (Supplemental Table 3). No significant publication bias was detected (p = 0.987).
In subgroup analyses, the pooled incidence of drug-related AEs was 10, 5, 7, and 0% in patients taking vonoprazan for the management of H. pylori infection, GERD, PUD, and artificial ulcers after gastric ESD, respectively (pinteraction < 0.01); 7 and 9% in those for short-term healing of EE and long-term maintenance of healed EE, respectively (pinteraction = 0.44); 4 and 17% in those for short-term healing of PUD and long-term secondary prevention of PUD, respectively (pinteraction = 0.01); 7 and 6% in those for a duration of >8 weeks and ⩽8 weeks, respectively (pinteraction = 0.87); and 7 and 7% in adult and adolescent patients taking vonoprazan, respectively (pinteraction = 0.83) (Figure 2(b)).
Serious AEs
Thirty-five studies explored the incidence of serious AEs in patients taking vonoprazan (Figure 3(a)). Meta-analysis demonstrated that the pooled incidence of serious AEs was 1% (95% CI = 0–2%) with significant heterogeneity (I2 = 90.8%; p < 0.01). Meta-regression analyses demonstrated that the heterogeneity might be attributed to the indications for vonoprazan and duration of vonoprazan (Supplemental Table 3). No significant publication bias was detected (p = 0.544).

Forest plots of the incidences of serious AEs (a) and AEs leading to drug discontinuation (b) in patients taking vonoprazan.
In subgroup analyses, the pooled incidence of serious AEs was 0, 0, 4, and 0% in patients taking vonoprazan for the management of H. pylori infection, GERD, PUD, and artificial ulcers after gastric ESD, respectively (pinteraction = 0.07); 1 and 3% in those for short-term healing of EE and long-term maintenance of healed EE, respectively (pinteraction = 0.01); 1 and 13% in those for short-term healing of PUD and long-term secondary prevention of PUD, respectively (pinteraction < 0.01); 3 and 0% in those for a duration of >8 weeks and ⩽8 weeks, respectively (pinteraction = 0.02); 1 and 1% in Asian and non-Asian patients taking vonoprazan, respectively (pinteraction = 0.94); and 1 and 0% in adult and adolescent patients taking vonoprazan, respectively (pinteraction < 0.01) (Figure 3(a)).
AEs leading to drug discontinuation
Forty-seven studies explored the incidence of AEs leading to drug discontinuation in patients taking vonoprazan (Figure 3(b)). Meta-analysis demonstrated that the pooled incidence of AEs leading to drug discontinuation was 1% (95% CI = 0–1%) with significant heterogeneity (I2 = 83.0%; p < 0.01). Meta-regression analyses demonstrated that the heterogeneity might be attributed to the duration of vonoprazan (Supplemental Table 3). No significant publication bias was detected (p = 0.578).
In subgroup analyses, the pooled incidence of AEs leading to drug discontinuation was 0, 2, 3, and 0% in patients taking vonoprazan for the management of H. pylori infection, GERD, PUD, and artificial ulcers after gastric ESD, respectively (pinteraction < 0.01); 2 and 6% in those for short-term healing of EE and long-term maintenance of healed EE, respectively (pinteraction = 0.02); 1 and 8% in those for short-term healing of PUD and long-term secondary prevention of PUD, respectively (pinteraction < 0.01); 5 and 0% in those for a duration of >8 weeks and ⩽8 weeks, respectively (pinteraction < 0.01); 1 and 2% in Asian and non-Asian patients taking vonoprazan, respectively (pinteraction = 0.72); and 1 and 1% in adult and adolescent patients taking vonoprazan, respectively (pinteraction = 0.13) (Figure 3(b)).
Common AEs
Meta-analyses demonstrated that increased serum gastrin level was the most common AE (19%, 95% CI = 3–44%), followed by increased pepsinogen I level (17%, 95% CI = 2–43%), nasopharyngitis (14%, 95% CI = 7–22%), bloating (9%, 95% CI = 4–15%), loose stools (8%, 95% CI = 4–15%), diarrhea (7%, 95% CI = 4–9%), heart burn (6%, 95% CI = 3–9%), abdominal pain (5%, 95% CI = 2–9%), and upper respiratory tract infection (5%, 95% CI = 3–8%) (Table 2).
Results of meta-analyses regarding common AEs.

AE, adverse event; CI, confidence interval.
Difference in the incidence of AEs between patients taking vonoprazan versus PPIs
Any AEs
Twenty-six studies compared the incidence of any AEs between patients taking vonoprazan versus PPIs (Supplemental Figure 2). Meta-analysis demonstrated that the incidence of any AEs was statistically similar between patients taking vonoprazan versus PPIs (OR = 0.96, 95% CI = 0.80–1.15, p = 0.66) with significant heterogeneity (I2 = 69.0%; p < 0.01). Meta-regression analyses demonstrated that the heterogeneity might be attributed to the type of publication and study design (Supplemental Table 3). No significant publication bias was detected (p = 0.768).
Drug-related AEs
Twenty studies compared the incidence of drug-related AEs between patients taking vonoprazan versus PPIs (Supplemental Figure 3). Meta-analysis demonstrated that the incidence of drug-related AEs was statistically similar between patients taking vonoprazan versus PPIs (OR = 1.10, 95% CI = 0.87–1.39, p = 0.52) with significant heterogeneity (I2 = 42.0%; p = 0.04). Meta-regression analyses did not find any source of heterogeneity (Supplemental Table 3). No significant publication bias was detected (p = 0.472).
Serious AEs
Twenty-one studies compared the incidence of serious AEs between patients taking vonoprazan versus PPIs (Supplemental Figure 4). Meta-analysis demonstrated that the incidence of serious AEs was statistically similar between patients taking vonoprazan versus PPIs (OR = 1.14, 95% CI = 0.87–1.49, p = 0.36). There was no statistically significant heterogeneity (I2 = 0.0%; p = 0.93). No significant publication bias was detected (p = 0.579).
AEs leading to drug discontinuation
Thirty-one studies compared the incidence of AEs leading to drug discontinuation between patients taking vonoprazan versus PPIs (Supplemental Figure 5). Meta-analysis demonstrated that the incidence of AEs leading to drug discontinuation was statistically similar between patients taking vonoprazan versus PPIs (OR = 1.09, 95% CI = 0.82–1.45, p = 0.55). There was no statistically significant heterogeneity (I2 = 5.0%; p = 0.39). No significant publication bias was detected (p = 0.395).
Common AEs
Meta-analyses demonstrated that the incidences of increased serum gastrin (OR = 6.25, 95% CI = 4.08–9.59, p < 0.01) and pepsinogen I levels (OR = 3.49, 95% CI = 2.40–5.10, p < 0.01) were significantly higher in patients taking vonoprazan than those taking PPIs. There was no statistically significant heterogeneity (I2 = 0%; p = 0.45 and I2 = 0%; p = 0.66, respectively). The incidence of diarrhea was significantly lower in patients taking vonoprazan than those taking PPIs (OR = 0.73, 95% CI = 0.57–0.94, p = 0.32) with significant heterogeneity (I2 = 57.7%; p < 0.01). The incidences of other common AEs were statistically similar between patients taking vonoprazan and PPIs (Figure 4).

Difference in the incidence of common AEs between patients taking vonoprazan versus PPIs.
Discussion
Our study has systematically reviewed the currently available evidence regarding the safety profile of vonoprazan, and found that the pooled incidences of any AEs, drug-related AEs, serious AEs, and AEs leading to drug discontinuation were 20, 7, 1, and 1%, respectively; nearly all the reported AEs were mild; and its safety was similar to that of PPIs.
Our meta-analysis has some features as compared to previous ones, as follows. First, previous meta-analyses explored the safety of vonoprazan for a certain indication.90–94 By comparison, our meta-analysis more comprehensively evaluated the safety of vonoprazan across multiple indications, including H. pylori infection, GERD, PUD, and artificial ulcers after gastric ESD. Second, previous meta-analyses only explored AEs leading to drug discontinuation and/or any AEs.90–94 By comparison, our meta-analyses also evaluated drug-related and serious AEs. Third, our meta-analysis, but not previous meta-analyses,90–94 evaluated a broader spectrum of common AEs and various severity of any AEs in patients taking vonoprazan.
Due to the lack of relevant data, we were not able to directly analyze the risk factors affecting the safety of vonoprazan. However, we performed subgroup analyses to evaluate the incidence of AEs in patients taking vonoprazan according to various conditions. First, patients with PUD had higher incidences of any AEs, serious AEs, and AEs leading to drug discontinuation than those with GERD, H. pylori infection, and artificial ulcer after gastric ESD. This might be because PUD is more complex and severe than other disease conditions, and requires a longer duration of vonoprazan. 95 Specifically, we included both studies regarding short-term healing of PUD and long-term secondary prevention of non-steroidal anti-inflammatory drugs or aspirin-induced PUD into the same subgroup. In short-term healing of PUD studies, some patients with PUD had concomitant H. pylori infection which needs to be eradicated by vonoprazan combined with other drugs.32,55 In long-term secondary prevention of PUD studies, patients required a longer duration of vonoprazan.13,42 Second, patients with H. pylori infection had a higher incidence of drug-related AEs than those with PUD, GERD, and artificial ulcer after gastric ESD. This was mainly because multiple drugs, including antibiotics, bismuth, and probiotics, should be added along with vonoprazan for H. pylori eradication.23,40 Third, it is readily understood that the incidence of AEs was higher in patients taking long-term (>8 weeks) use of vonoprazan than those taking short-term (⩽8 weeks) use of vonoprazan. This finding was also observed in patients taking vonoprazan for the management of EE and PUD. Fourth, the race of the population taking vonoprazan (i.e., Asian and non-Asian) might not influence the risk of AEs. Recently, a population pharmacokinetic model evaluated the impact of the population from Asia and Europe on vonoprazan exposure, and also showed that the population effect on vonoprazan exposure was limited. 96 Fifth, the incidence of AEs also seemed to be similar between adult and adolescent patients taking vonoprazan, which suggested that vonoprazan can be safe and well tolerated in both adults and adolescents. However, this finding required further validation in adolescents.
Increased serum gastrin and pepsinogen I levels might be the most common AEs, and their incidences were significantly higher in patients taking vonoprazan than those taking PPIs. They are believed to be a consequence of the potent gastric acid antisecretory effect of vonoprazan, 13 which is similar to PPIs,10,14,32,42 but they do not cause any harmful effects on gastric mucosa.13,21 The VISION trial evaluated the effect of vonoprazan on gastric mucosal tissue and its long-term safety. The four-year interim analysis found significantly higher levels of serum gastrin and pepsinogen I in the vonoprazan group than the lansoprazole group. Additionally, a case of foveolar-type adenoma developed in the vonoprazan group, and a case of oxyntic gland adenoma in the lansoprazole group. Histologically, hyperplasia of parietal cells and G cells was more frequently detected in the vonoprazan group. There was no neoplastic proliferation of endocrine cells in either group. 97 However, a case of foveolar-type gastric adenocarcinoma has recently been reported in a male patient with EE after a 156-week maintenance therapy of vonoprazan 10 mg daily. 98 In addition, gastric mucosal redness, ‘stardust gastric mucosa’, and white globe appearance can be observed in patients taking vonoprazan,99–101 but they are not of major clinical significance. Other common AEs were infrequent, and their incidences were similar to that of PPIs.
It has been reported that long-term use of PPIs may also lead to clostridium difficile infection, osteoporosis, vitamin B12 deficiency, hypomagnesemia, and fundic gland polyp, 102 but they have never been found in our meta-analysis. Certainly, such AEs in patients receiving long-term use of vonoprazan deserves further observations.
PPIs exposure is associated with increased risk of infection, decompensation, and death in cirrhosis and that of progression to renal insufficiency in chronic kidney disease.103–105 However, to date, no studies have reported that vonoprazan would cause such AEs. Indeed, patients with cirrhosis and chronic kidney disease were often excluded from our included studies, failing to evaluate the safety of vonoprazan in such patients. In future, prospective studies are needed to evaluate the safety of vonoprazan in patients with cirrhosis and chronic kidney diseases.
Our meta-analysis has some limitations as follows. First, a proportion of included studies were single-arm without control groups. Second, 18 studies were published as abstracts and 2 were unpublished clinical trials. Third, these included studies were mainly conducted in Japan. Fourth, we did not clearly discriminate which AEs were caused by PPIs/vonoprazan or antibiotics during H. pylori eradication therapy. Fifth, the heterogeneity among studies was statistically significant in most of our meta-analyses. Sixth, combined medication and underlying comorbidities, which might influence the safety of vonoprazan, could not be sufficiently evaluated in subgroup analyses.
Conclusion
Based on the current systematic review and meta-analysis, vonoprazan often has similar safety compared to PPIs, and rarely causes serious AEs and drug discontinuation. It seems that vonoprazan has a favorable safety profile in adolescent patients and non-Asian population. Furthermore, the safety of vonoprazan may be influenced by its indications and duration. However, more high-quality studies are necessary in future to validate these findings and to explore the risk factors affecting the safety of vonoprazan.
Supplemental Material
sj-doc-1-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-doc-1-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-7-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-docx-7-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-8-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-docx-8-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-9-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-docx-9-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-2-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-jpg-2-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-3-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-jpg-3-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-4-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-jpg-4-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-5-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-jpg-5-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-6-tag-10.1177_17562848231167858 – Supplemental material for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis
Supplemental material, sj-jpg-6-tag-10.1177_17562848231167858 for Incidence and type of adverse events in patients taking vonoprazan: A systematic review and meta-analysis by Wentao Xu, Zhaohui Bai, Yiyang Shang, Jing Wang, Yujun Wong and Xingshun Qi in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
