Abstract
Medication-overuse headache (MOH) is a debilitating condition in which frequent and prolonged use of medication for the acute treatment of pain results in the worsening of the headache. The purpose of this paper is to review the most recent literature on MOH and discuss future avenues for research. MOH accounts for a substantial share of the global burden of disease. Prevalence is often reported as 1–2% but can be as high as 7% overall, with higher proportions among women and in those with a low socioeconomic position. Management consists of withdrawing pain medication, focusing on prophylactic and nonmedical treatments, and limiting acute symptomatic medication. Stress reduction and lifestyle interventions may support the change towards rational pain medication use. Support, follow up, and education are needed to help patients through the detoxification period. There is fertile ground for research in MOH epidemiology, pathophysiology, and neuroimaging. Randomized and long-term follow-up studies on MOH treatment protocols are needed. Further focused research could be of major importance for global health.
Introduction
Headache is one of the most common health complaints. Of 301 acute and chronic diseases tracked by the Global Burden of Disease (GBD) studies, two headache forms are ranked among those with the highest prevalence: tension-type headache (TTH) and migraine [Global Burden of Disease Study 2013 Collaborators, 2015].
Medication-overuse headache (MOH) is another headache form that ranks high in the most recently published GBD report. It is a debilitating condition in which frequent and prolonged use of medication for the acute treatment of pain results in worsening of the headache. It is not as prevalent as TTH or migraine but it is very disabling. The GBD studies use a metric called years of life lived with disability (YLDs), which estimates years of life lived with any short-term or long-term health loss [Murray and Lopez, 2013; Institute for Health Metrics and Evaluation, 2013]. Among all causes of global YLDs, MOH ranks 18th [Global Burden of Disease Study 2013 Collaborators, 2015].
The personal impact of MOH has been shown to be greater than that of migraine or TTH in several European countries. Compared with those who have migraine or TTH, people with MOH are more likely to report adverse effects of headache on education, career, earnings, social acceptance, and feeling of control over their headaches. Those with MOH also report more lost days for productive work, housework, and social activities [Steiner et al. 2014]. MOH is among the most costly of neurologic diseases [Russell and Lundkvist, 2012] and is the most costly among headache disorders [Linde et al. 2012]. Very importantly, the high burden of MOH to the individual and to society is unnecessary because it is preventable and largely treatable.
The purpose of this perspective review is to discuss results of recent research and highlight important new areas of general interest to the study of MOH.
Diagnosing MOH
MOH was first described in the 1950s [Boes and Capobianco, 2005]. There are as many as 40 different terms for MOH, including rebound headache, drug-induced headache, drug dependence headache, and medication-misuse headache [Westergaard et al. 2014b]. The current diagnostic criteria, arrived at by consensus among experts from the International Headache Society, are listed in the third edition of the International Classification of Headache Disorders (ICHD-3 beta) (Table 1) [Headache Classification Committee of the International Headache Society, 2013].
International Classification of Headache Disorders (ICHD-3 beta) diagnostic criteria for medication-overuse headache and its subtypes.

ASA, acetylsalicylic acid; NSAID, nonsteroidal anti-inflammatory drug.
(Summarized from: Headache Classification Committee of the International Headache Society [2013].)
The specific characteristics of headache pain are important for the diagnosis of the primary underlying headache (e.g. migraine, TTH, cluster headache), but for the diagnosis of MOH, the most important parameter is number of headache days: 15 or more days per month.
The second criterion involves overuse of acute pain medication for more than a three-month period. ‘Overuse’ is assessed in terms of the number of medication days as opposed to doses. The cut-off levels for overuse are 10 or 15 medication days per month depending on the type of drug or combinations of different drugs.
It is important to ask patients about over-the-counter (OTC) pain medication use, often taken on top of prescribed pain medications. Patients may not report medication overuse when they believe their level of intake does not qualify as an ‘overdose’.
The third criterion requires the exclusion of other headache diagnoses. This requires a thorough history, physical examination, and in some cases, ancillary tests.
The new ICHD-3 beta criteria do not require a trial of medication withdrawal and subsequent improvement in order to establish that medication overuse is the cause of the headache. It is possible for a patient to be diagnosed with more than one headache disorder. For example, a patient can be diagnosed as having chronic migraine, and when there is associated medication overuse, the diagnosis of MOH should also be applied [Headache Classification Committee of the International Headache Society, 2013].
For many people, daily use of medication for a chronic pain indication, for example arthritis, back pain, postoperative pain, or cancer, does not lead to headache. However, a person with a pre-existing headache disorder who develops chronic headache with frequent intake of pain medication (for whatever indication) must be assessed for MOH. If a patient has MOH, increasing symptomatic pain medication will not help. In contrast, it could maintain a disabling chronic headache that is refractory to both pharmacological and nonpharmacological treatment.
Population prevalence of MOH
The publication of GBD 2013 boosted efforts in studying MOH epidemiology. Prevalence was estimated for 188 countries based on best available data. It is estimated that 63 million people worldwide have MOH [GBD 2013 Collaborators, 2015].
MOH is difficult to study epidemiologically [Steiner, 2014]. Fortunately, there has been an increased focus on producing methodologically sound prevalence estimates for all headache disorders, including MOH [Steiner, 2004; Stovner et al. 2014; Ahmed et al. 2014]. In the last 10 years, there has been a remarkable increase in MOH population-based studies but there is still a lack of studies from many regions [Steiner et al. 2010].
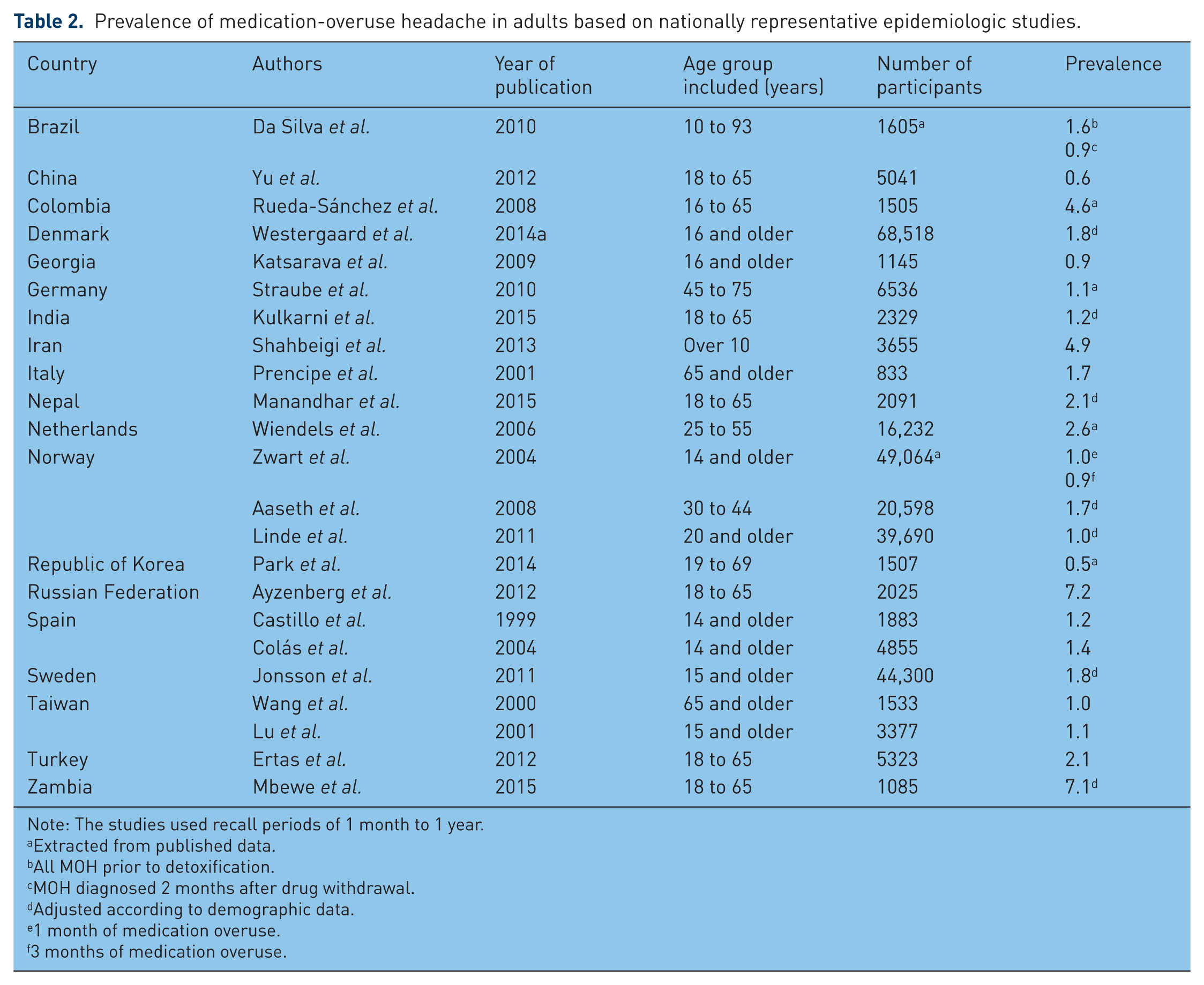
Prevalence estimates based on nationally representative data from 16 countries (Brazil, China, Colombia, Georgia, Germany, Iran, Italy, Republic of Korea, Netherlands, Norway, Russian Federation, Spain, Sweden, Turkey, Taiwan and the United States) have been summarized in a systematic review [Westergaard et al. 2014b]. Preliminary data from the Republic of Korea reported at a headache congress by Chu and colleagues [Chu et al. 2011] have since been published [Park et al. 2014], and there are four additional studies from Denmark [Westergaard et al. 2014a], India [Kulkarni et al. 2015], Nepal [Manandhar et al. 2015] and Zambia [Mbewe et al. 2015].
Data from the USA [Scher et al. 2010] were derived from a case-control study of adults randomly selected for a telephone interview. Overall population prevalence was not estimated but authors described MOH prevalence as 32% among 206 cases with chronic headache. Results of all other nationally representative epidemiologic studies among adults are summarized in Table 2.
Prevalence of medication-overuse headache in adults based on nationally representative epidemiologic studies.
Note: The studies used recall periods of 1 month to 1 year.
Extracted from published data.
All MOH prior to detoxification.
MOH diagnosed 2 months after drug withdrawal.
Adjusted according to demographic data.
1 month of medication overuse.
3 months of medication overuse.
Studies with sex-specific data reported higher prevalence among women, with female:male ratios ranging from about 2:1 to 5:1. However, there was a notable exception in the study from Iran where the prevalence among females (4.8%) was slightly lower than the prevalence among males (5.1%) [Shahbeigi et al. 2013].
Studies from four countries in Europe (Denmark, Norway, Spain, Sweden) showed that MOH is most prevalent among middle-aged people in their 30s to 50s [Westergaard et al. 2014b; Aaseth et al. 2008; Zwart et al. 2004; Colás et al. 2004; Jonsson et al. 2011]. However, the study from India showed the opposite trend [Kulkarni et al. 2015]. In Nepal, the oldest age group (56–65 years) had the highest prevalence [Manandhar et al. 2015] while in Zambia, prevalence increased with age, going as high as 23.8% in the oldest age group [Mbewe et al. 2015].
The Zambian study showed a large difference in prevalence between rural and urban areas (2.1% and 14.5%, respectively) which the authors attributed to very limited access to OTC medication in rural areas [Mbewe et al. 2015]. However, there was a slight (nonsignificant) rural preponderance observed in India (1.5% rural
Large Scandinavian studies from Norway and Sweden showed higher prevalence among those with lower socioeconomic position [Hagen et al. 2012; Jonsson et al. 2011]. Danish data showed highest prevalence among those on social welfare (11.0%), early pensioners (7.5%) and people receiving sickness benefits (6.0%) [Westergaard et al. 2014a].
There are differences among ethnic groups within several European countries, with migrants having a higher prevalence in Denmark [Westergaard et al. 2014a], Germany [Kavuk et al. 2006], the Netherlands [Wiendels et al. 2006], and Sweden [Jonsson et al. 2011]. The reasons for these differences are not known and may be genetic, cultural, or socioeconomic; or possibly related to differences in accessing health care, the stress of migration, and communication difficulties.
Chiappedi and Balottin [Chiappedi and Balottin, 2014], and Gelfand and Goadsby [Gelfand and Goadsby, 2014] recently reviewed the literature on MOH in children and adolescents and showed that population-based epidemiologic studies are sparse (with data available only from Norway, Taiwan, and the US) but MOH is commonly encountered in the clinical setting.
Commonly overused medications
OTC analgesics are the most overused headache medications in both population- and clinic-based studies. In specialist care settings, a larger proportion of patients overuse centrally acting drugs [Kristoffersen and Lundqvist, 2014].
Paracetamol is the single most commonly overused drug. This does not necessarily mean that it has a greater propensity to cause MOH; rather it could simply reflect how it is the most often purchased analgesic for headache.
There are country/regional differences for ergotamine. Its use has declined in the US and Europe, but it remains among the most overused medications in Latin America [Shand et al. 2015].
In the US, data from 1990 to 2005 showed a decrease in overuse of ergot compounds, acetylsalicylic acid, paracetamol, butalbital, and opioids, but an increase in overuse of triptans and nonsteroidal anti-inflammatory drugs [Meskunas et al. 2006]. A more recent study in US emergency departments spanning 2001 to 2010 noted an increase in use of several opioids [Mazer-Amirshahi et al. 2014].
Danish registry data showed that one in three people with MOH were dispensed an opioid at least once in a year. There were no significant differences in the prevalence of self-reported cancer among those with and without chronic headache, suggesting that opioids were likely being taken for noncancer pain [Westergaard et al. 2015].
Opioids are not recommended for headache treatment [Bendtsen et al. 2012]. Canadian and US-based reviews showed that there is weak evidence for tramadol efficacy in migraine [Orr et al. 2015; Kelley and Tepper, 2012] and it is not indicated for TTH. Prolonged opioid use is associated with progression or transformation from episodic to chronic migraine [Bigal et al. 2008]. Opioid overuse headache, a subclass of MOH, is very difficult to treat and often requires hospital admission. It is possible that MOH and opioid-induced hyperalgesia share the same pathophysiology and mechanism [Johnson et al. 2013].
There are several prescription drug registry studies on triptan overuse from European countries. The earliest was done only 2 years after the release of sumatriptan [Gaist et al. 1996]. The most recent studies are from the Netherlands [Dekker et al. 2011], Sweden [Von Euler et al. 2014], Denmark [Westergaard et al. 2015], and France [Braunstein et al. 2015]. These studies are consistent in showing how a small number of people account for a large share of triptan purchases. In France, for example, 2.3% of users purchased 20% of the total volume dispensed.
In 2008, triptans became available OTC in Sweden. Analysis of data on triptan purchases from 1991 to 2011 showed that the amount of dispensed prescriptions remained stable while OTC sales increased slowly to 11% of total expenditure [Von Euler et al. 2014]. There are no data on whether OTC availability of triptans increase MOH prevalence, but it is a legitimate concern [Tfelt-Hansen and Steiner, 2007] and could be a focus for future pharmacoepidemiologic studies.
MOH, perceived stress, and lifestyle
There are surprisingly few studies on the relationship between perceived stress and MOH despite the clear relationship between stress and headache [Yokoyama et al. 2009; Stensland et al. 2013; Lipton et al. 2014; Schramm et al. 2015].
Danish studies showed that young adults (aged 25–44 years) with headache who have limited coping resources are more likely to use analgesics when faced with stress [Koushede et al. 2011, 2012]. Population-based data confirmed the graded association between MOH and increasing levels of perceived stress [Westergaard et al. 2016].
There appears to be a relationship between lifestyle factors (smoking, obesity, physical inactivity) and MOH [Wiendels et al. 2006; Straube et al. 2010; Hagen et al. 2012; Westergaard et al. 2016]. Cross-sectional studies cannot show causation, and it is not possible to say whether an unhealthy lifestyle somehow leads to the development of MOH. However, it is important to screen for lifestyle factors among people with MOH, and it is possible that change in these modifiable factors could support change towards rational pain medication use.
Treatment goals and dilemmas
It can be argued that increasing medication use could be a patient’s response to increasing pain, and that the medication is not the cause of the worsening headache. Patients themselves might be averse to hearing that the ‘indispensable medication’ [Jonsson et al. 2013] could be the reason for their pain. However, near-daily use of acute pain medication without relief of headache suggests that the treatment is ineffective, if not harmful, and a new approach is indicated.
Several key points in MOH treatment are outlined in Table 3. Most cases of MOH can be managed in primary care. A follow-up study in the primary care setting in Norway compared two groups of patients: one group with MOH received brief intervention (BI), essentially simple advice and education on MOH; the other group did not. The BI group, whose headache days were reduced from 25 to 17, on average, was able to cut down medication intake from 24 to 13 days. The control group did not experience any change in headache days or medication days (25 and 22 days, respectively) at follow up [Kristoffersen et al. 2015].
Key points in the treatment of medication-overuse headache.
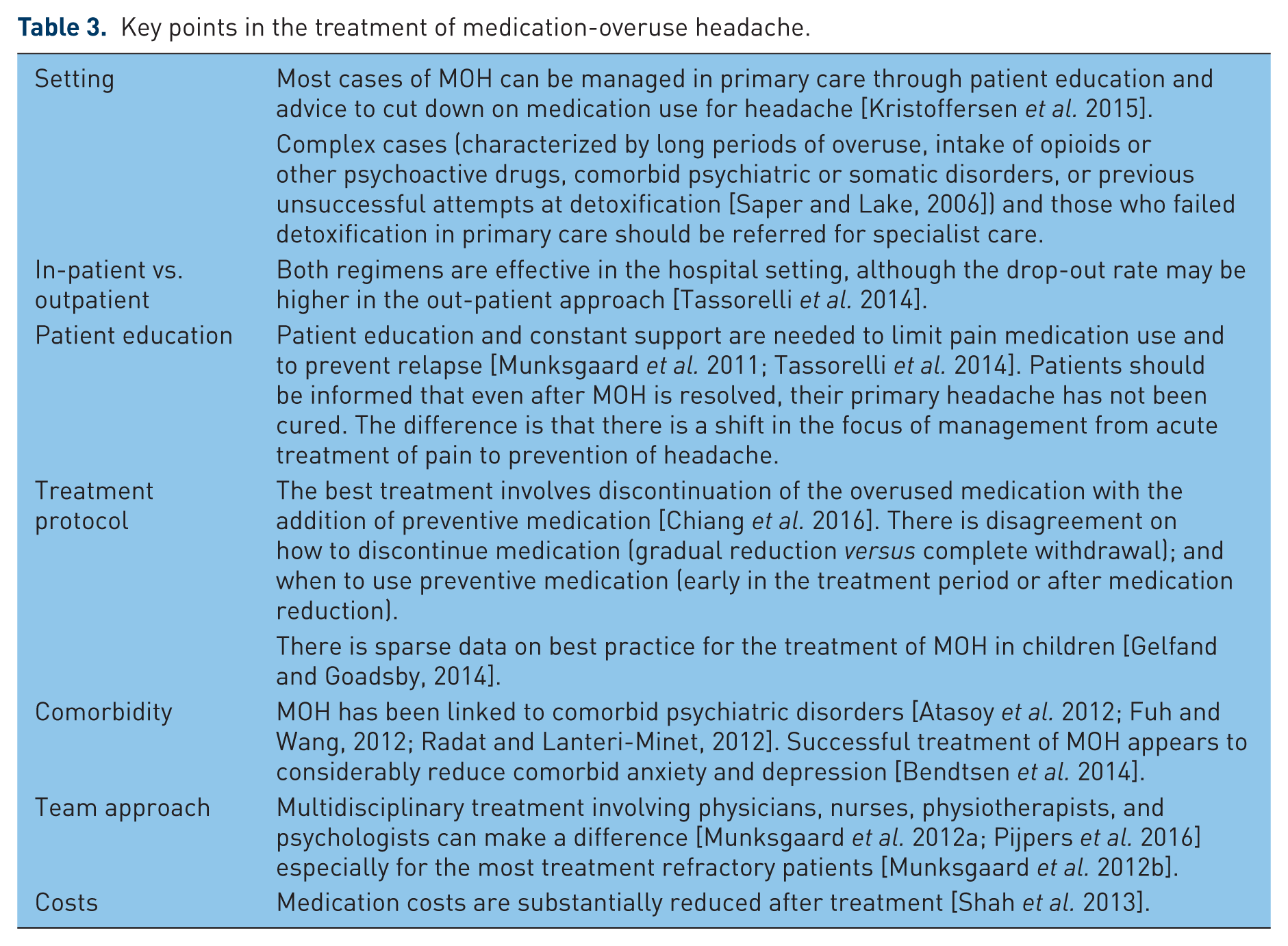
In specialist care, detoxification can be carried out through outpatient or inpatient programs. The results of a multicenter study conducted in Europe and South America, are promising: 62% were no longer overusers and 46% reverted back to an episodic pattern of headache after 6 months [Tassorelli et al. 2014].
MOH management protocols differ in two important ways: (1) the pace of detoxification (complete withdrawal
The Danish Headache Center currently advocates starting prophylactics after detoxification. Up to half of the patients may not even need migraine prophylactics if headache days are few [Munksgaard and Jensen, 2014].
TTH benefits from targeting the sources of stress and strengthening coping mechanisms, rather than reacting to the symptom of headache with an analgesic. For migraine, the focus shifts to optimizing acute treatment (for example, with triptans), and use of preventive medications: anti-epileptics (e.g. topiramate, valproic acid), tricyclic antidepressants (e.g. amitriptyline), and antihypertensive agents (e.g. beta-blockers).
There is an increasing role for psychological interventions for the management of chronic pain [Kaiser et al. 2015]. It is important to address psychiatric disorders (most commonly anxiety and depression) that are often comorbid with chronic headache and specifically MOH [Atasoy et al. 2012; Fuh and Wang, 2012; Radat and Lanteri-Minet, 2012]. Physiotherapy can reduce intensity, frequency and duration of migraine, TTH and cervicogenic headache [Luedtke et al. 2016].
There is no evidence on the effectiveness of occipital nerve stimulation and occipital nerve block for the treatment of MOH. Randomized control trials are needed to evaluate the usefulness of onabotulinumtoxinA alone or with early discontinuation [Chiang et al. 2016]. Neuromodulation is not recommended [Martelletti et al. 2013].
Current research
Current research on MOH ranges from basic science to clinic-based and population-based studies.
The Global Burden of Disease studies provide MOH prevalence estimates based on best available data, but representative studies are lacking in many areas of the world and there is hardly any research on children and adolescents. There are still many knowledge gaps in MOH epidemiology but an interesting and important avenue for prevention of chronic headache has been paved.
The pathophysiology of MOH is still unclear, with recent work pointing towards alterations in pain-related and reward-related systems: central serotonin (5-HT) or endocannabinoid-dependent modulating systems [Srikiatkhachorn et al. 2014, Kristoffersen and Lundqvist 2014]. Understanding the pathophysiology of opioid-induced hyperalgesia might also lead to an understanding of opioid-overuse headache [Johnson et al. 2013].
Different methods of measuring pain perception in patients with MOH all show alterations in pain perception between MOH patients and healthy controls. These findings indicate central sensitization [Ayzenberg et al. 2006; Coppola et al. 2010; Munksgaard et al. 2013; Perrotta et al. 2010, 2012; Zappaterra et al. 2011]. Patients with MOH appear to be more sensitive to pressure pain than patients with chronic TTH and chronic migraine [Zappaterra et al. 2011].
There are very few imaging studies on MOH. Studies have reported orbitofrontal hypometabolism [Fumal et al. 2006], reversible dysfunction in the ventromedial prefrontal cortex [Ferraro et al. 2012], changes in gray matter volumes in several brain areas including the midbrain and orbitofrontal cortex [Riederer et al. 2013; Chanraud et al. 2014], and changes in resting functional connectivity [Chanraud et al. 2014]. It is not known whether changes in the brain structure and function of people with MOH are the result or the cause of medication overuse.
In coherence with the changes seen in the fMRI scans of patients before and after detoxification [Grazzi et al. 2010], Perrotta and colleagues [Perrotta et al. 2010, 2012] found decreased thresholds for eliciting nociceptive withdrawal reflexes in MOH patients before detoxification compared with healthy volunteers. Furthermore, they found that the stimulation needed to elicit the reflexes increased after detoxification (after 10 days and after 2 months), indicating a decrease in sensitization after withdrawal. Munksgaard and colleagues [Munksgaard et al. 2013] showed that the pain intensity for pressure above the pain threshold is significantly higher in MOH patients before withdrawal, but continued to decrease during the first year after detoxification. After 12 months without overuse, these levels were similar to the pain intensity of healthy volunteers.
These findings in patients parallel data from animal studies which suggest that pre-exposure to acute medication decreases the electrical stimulation threshold to generate a cortical spreading depression, which increases activation of the trigeminal nucleus caudalis, thereby precipitating headache [Green et al. 2013].
The demonstration of central sensitization that decreases after withdrawal of overused medication shows how MOH is a condition that is treatable, and more importantly, preventable.
Randomized and follow-up studies on MOH treatment are needed. There are many unanswered questions: Is complete withdrawal of acute pain medication necessary, or is it enough to reduce medication days? Is abrupt cessation more effective than gradual reduction of an overused medication? Which types of patients benefit from prophylactics and when should these be introduced (before, during, or after detoxification)? Which types of patients are best managed by in-patient treatment? What are the best medical treatments for children? Can behavioral interventions help prevent headaches and reduce medication days? Do interventions such as physiotherapy and acupuncture improve outcomes? What are the protective factors in patients who do not relapse?
Conclusion
In summary, MOH is a debilitating condition of public health importance that has been included among the 20 most debilitating diseases in the GBD studies. It is the focus of increasing research activity. Prevalence is often reported as 1–2% but new country reports go as high as 7%. These data call for increased awareness of this unique chronic pain disorder that in principle is both preventable and treatable. Effective protocols for treatment have been developed but additional randomized and long-term follow-up studies are needed. A dedicated and international information campaign for prevention of MOH may reduce the burden of this costly and highly disabling type of headache. Further focused research could be of major importance for global health.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MLW received a research grant from Trygfonden to prepare the manuscript. This research received no specific grant from the commercial sector.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
