Abstract
Background:
Radiofrequency ablation (RFA) has gained recognition as a highly effective treatment for benign thyroid nodules (BTNs). However, post-procedural thyroid function changes have been reported.
Objectives:
This study specifically focuses on the potential link between changes in thyroid function and the volume reduction ratio (VRR) of treated nodules. Additionally, it seeks to evaluate whether fluctuations in thyroid function at mid-term follow-up can serve as early indicators for the development of long-term hypothyroidism following RFA.
Design:
Retrospective cohort study.
Methods:
In this study, 50 euthyroid individuals (mean age = 47.1 years; 43 females, 7 males) with a total of 72 BTNs (median volume = 4.61 mL) undergoing RFA were evaluated. Comprehensive assessments, including clinical examinations, ultrasound imaging, and blood tests, were conducted at specific intervals (pre-RFA, and at 6 months, 12 months, and annually post-RFA).
Results:
The mean follow-up period was 22.3 months. Both medium-term and long-term follow-ups revealed significant reductions in triiodothyronine (T3) levels (p = 0.010, p = 0.036) and elevations in thyroid-stimulating hormone (TSH) levels (p < 0.001, p = 0.006) compared to baseline measurements. A negative correlation was found between medium-term T3 levels and long-term VRR (r = −0.475, p = 0.001). Furthermore, patients with lower T3 levels during medium-term follow-up demonstrated a significantly higher long-term VRR compared to those with higher T3 levels (0.89 vs 0.77, p = 0.030).
Conclusion:
Following RFA, notable alterations in thyroid function were observed, without meeting the criteria of hypothyroidism. Additionally, a lower mid-term T3 level may be indicative of a better VRR during long-term follow-up.
Plain language summary
This study looked at a treatment called radiofrequency ablation (RFA), which uses heat to shrink thyroid nodules. We wanted to see whether changes in thyroid hormone levels after treatment were linked to how much the nodules shrank over time. We followed 50 patients with normal thyroid function before treatment. They had regular check-ups, including ultrasound scans and blood tests, for more than a year after RFA. We found that, although some hormone levels changed slightly after the procedure, most patients’ thyroid function stayed within the normal range. Interestingly, patients whose T3 hormone levels were lower about six months after RFA had better long-term nodule shrinkage. This suggests that checking thyroid hormone levels a few months after RFA could help doctors predict the long-term success of the treatment.
Introduction
Ultrasound (US)-guided radiofrequency ablation (RFA) has emerged as an effective and minimally invasive treatment option for patients with benign thyroid nodules (BTNs). Studies have reported a significant post-ablation volume reduction rate, typically ranging from 50% to 80%, highlighting its efficacy in nodule size reduction and symptom relief.1,2 Compared to traditional surgical approaches, RFA provides the advantages of reduced invasiveness and fewer complications, making it a preferred choice for suitable candidates. 3 However, like any medical procedure, RFA carries risks, with primary complications including potential voice changes, rare occurrences of tumor rupture, and, in some cases, permanent hypothyroidism. 4
Hypothyroidism is a significant concern following thyroid surgery, particularly in patients undergoing hemithyroidectomy. Studies have shown that the incidence of post-hemithyroidectomy hypothyroidism varies widely, with reported rates ranging from 10.9% to 48.8%. 5 Research has also highlighted several predictive factors associated with an increased risk of developing hypothyroidism after surgery. These factors include higher preoperative thyroid-stimulating hormone (TSH) levels, a reduced volume of the remaining thyroid tissue, and the presence of Hashimoto’s thyroiditis, an autoimmune condition that affects thyroid function.6 –9
Although the incidence of hypothyroidism after RFA is lower than that following surgical intervention, instances of thyroid dysfunction have still been reported during post-RFA follow-up evaluations. 4 Identified risk factors for post-RFA hypothyroidism include higher baseline TSH levels and higher ablation volume ratios, both of which appear to influence the likelihood of thyroid dysfunction. 10 Despite these findings, there remains a lack of extensive large-cohort studies to thoroughly investigate these associations and establish a clear understanding of risk stratification in post-RFA hypothyroidism.
Given that changes in thyroid function may occur following RFA and are often related to the ablated volume, this study is designed to investigate the relationship between thyroid function alterations and the volume reduction ratio (VRR), a critical metric in assessing RFA efficacy. Focusing on euthyroid patients with BTNs, this research also aims to determine whether fluctuations in thyroid function observed during mid-term follow-up could serve as early indicators for the potential onset of long-term hypothyroidism post-RFA. By analyzing these trends, the study hopes to provide insights that may improve the safety and effectiveness of RFA in managing BTNs.
Materials and methods
Patients
This retrospective study was conducted at a single medical center in Taiwan. To minimize potential selection bias, we consecutively screened all adult patients with euthyroid status who were diagnosed with BTNs and underwent RFA between July 2017 and September 2021. The inclusion criteria were as follows: (1) age ⩾ 18 years; (2) confirmed euthyroidism through biochemical testing; and (3) documented diagnosis of BTNs via fine-needle aspiration or core-needle biopsy.
Subsequently, to ensure the internal validity of our efficacy analysis and reduce confounding bias, the following exclusion criteria were strictly applied: (1) patients who received multiple RFA sessions for the same lesion; and (2) patients with insufficient clinical or radiological data during the scheduled follow-up period. By excluding cases with missing data, we addressed potential information bias, ensuring a complete dataset for longitudinal comparison. After applying the exclusion criteria, a cohort of 50 euthyroid individuals, comprising 72 BTNs that underwent RFA, was included in the final analysis.
Pre- and post-ablation assessment
The benign nature of the thyroid nodules was established by achieving at least two benign cytology results through US-guided fine-needle aspiration cytology or core needle biopsy, which are standard methods for assessing thyroid lesions. In some cases, if only one benign cytological result was available, the nodule was deemed benign if it showed typical benign characteristics on US imaging, indicating a low risk of malignancy.
Prior to the RFA procedure, each patient underwent a comprehensive clinical evaluation, including physical examination, US imaging, and laboratory tests to assess thyroid function. Follow-up assessments were conducted at 6 months, 12 months, and then annually to monitor clinical outcomes, nodule volume reduction, and any potential complications.
The nodule-related symptom score was determined using a patient-completed questionnaire encompassing five clinical symptoms: compression, cough, dysphagia, voice alterations, and pain. Each positive symptom was assigned a single point, resulting in a potential symptom score range of 0–5. The cosmetic score was evaluated utilizing the following scale: 0 denoting the absence of a palpable mass, 1 indicating a palpable mass with no cosmetic concerns, 2 denoting a cosmetic issue during neck extension and/or swallowing, and 3 representing an easily detectable cosmetic problem.
Nodule volume was calculated employing the ATA thyroid nodule volume calculator (https://www.thyroid.org/professionals/calculators/thyroid-with-nodules/), incorporating measurements of length, width, and depth obtained via US scan. To evaluate the effectiveness and reduction rate of thyroid nodules post-RFA, the VRR was evaluated through US imaging and calculated using the formula: VRR (%) = )initial volume (mL) − final volume (mL)) × 100/initial volume. Additionally, blood tests were conducted to measure TSH, triiodothyronine (T3), and free thyroxine (FT4) levels, providing insights into thyroid function changes associated with RFA.
Ablation technique
Real-time US-guided RFA was performed as an outpatient procedure, ensuring precise nodule targeting and minimizing patient discomfort. Local anesthesia with 2% lidocaine hydrochloride was administered at both the puncture site and around the thyroid gland, providing effective pain control throughout the procedure.
The RFA procedures were conducted using an RF generator (VIVA, STARmed, and M2004; RF Medical, Seoul, South Korea) in combination with an 18-gauge internally cooled electrode, equipped with a 5 mm, 7 mm, or 1 cm active tip. The choice of tip size was tailored to the specific nodule size and the proximity of nearby critical structures, optimizing safety and efficacy. Utilizing a trans-isthmic approach along with a moving-shot technique, the electrode was carefully advanced to the deepest portion of the thyroid nodule. Ablation was concluded upon the transformation of all visually identifiable areas within the nodule into transient hyperechoic zones.
Statistical analysis
All statistical analyses were conducted using IBM SPSS Statistics 24 (IBM Corp., Armonk, NY, USA). The Shapiro–Wilk test was applied to evaluate data normality, ensuring a robust approach for handling different data distributions. For data that did not follow a normal distribution, results were expressed as medians with interquartile ranges, while normally distributed data were represented by means and standard deviations.
To assess changes in thyroid function over various time points compared to the baseline, the Wilcoxon signed-rank test was used. Additionally, a partial analysis was performed to explore the association between thyroid function and nodule volume. Patients were categorized into subgroups based on T3 and TSH levels during a 6-month follow-up, using the midpoint of the normal range as the cutoff. For continuous variable comparisons between groups, the Mann–Whitney U test was employed. Statistical significance was considered with a p-value of less than 0.05.
Results
Demographic characteristics and efficacy of RFA treatment
A total of 87 patients were initially assessed for eligibility. After excluding 37 patients who met the exclusion criteria (e.g., received multiple RFA sessions or had insufficient follow-up data), a final cohort of 50 patients with 72 BTNs was included in the analysis (Figure 1). Table 1 provides a summary of the patients’ demographic data and treatment efficacy. The average age of participants was 47.1 years, with a standard deviation of 10.66. The cohort included 43 women (86%) and 7 men (14%), and the average follow-up duration was 22.3 months, allowing for assessment of long-term RFA outcomes. Notably, all 50 patients (100%) completed the scheduled follow-up evaluations at 6, 12, and over 12 months, ensuring no loss to follow-up during the study period. Regarding nodule composition, 63 nodules were classified as solid or predominantly solid, while 9 were predominantly cystic. Analysis showed that although the predominantly cystic group exhibited a higher long-term VRR compared to the solid/predominantly solid group (98.0% vs 85.0%), the difference was not statistically significant (p = 0.083), indicating that nodule composition did not significantly bias the primary outcomes in this cohort.

Flowchart of participant selection.
Baseline characteristics and treatment efficacy after RFA.
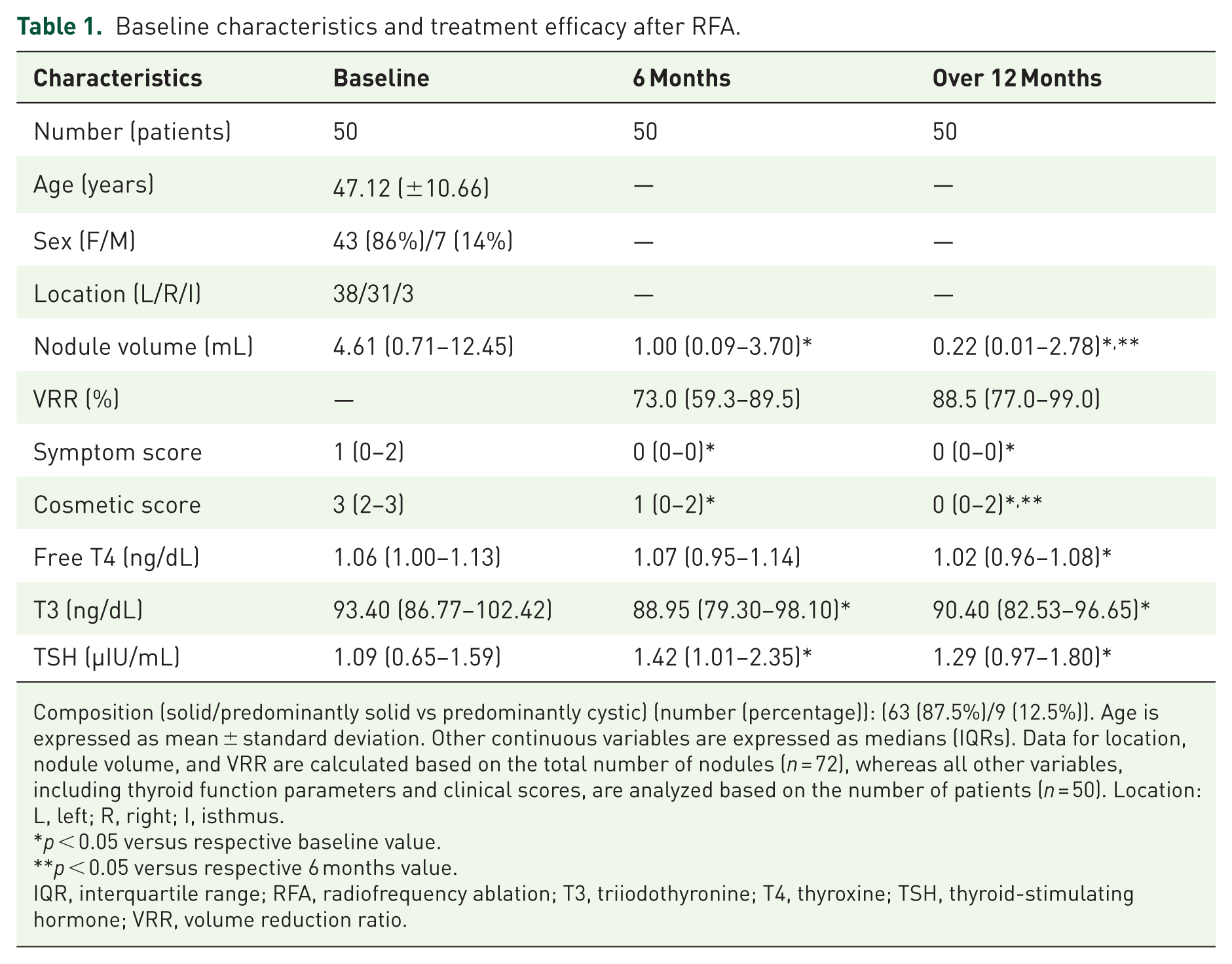
Composition (solid/predominantly solid vs predominantly cystic) (number (percentage)): (63 (87.5%)/9 (12.5%)). Age is expressed as mean ± standard deviation. Other continuous variables are expressed as medians (IQRs). Data for location, nodule volume, and VRR are calculated based on the total number of nodules (n = 72), whereas all other variables, including thyroid function parameters and clinical scores, are analyzed based on the number of patients (n = 50). Location: L, left; R, right; I, isthmus.
p < 0.05 versus respective baseline value.
p < 0.05 versus respective 6 months value.
IQR, interquartile range; RFA, radiofrequency ablation; T3, triiodothyronine; T4, thyroxine; TSH, thyroid-stimulating hormone; VRR, volume reduction ratio.
Prior to undergoing RFA, the median baseline volume of BTNs was measured at 4.61 mL. After RFA treatment, follow-up evaluations revealed substantial decreases in nodular volume, with the median volume reducing to 1.00 mL at 6 months and further shrinking to 0.22 mL over 12 months. This considerable reduction, significant at both medium-term and long-term follow-ups (p < 0.001), highlights the effectiveness of RFA in treating BTNs. Specifically, the median VRRs were 73.0% at 6 months and reached 88.5% beyond 12 months (Figure 2). Additionally, notable decreases were observed in both symptom and cosmetic scores post-RFA, with significant improvements sustained over time (p < 0.001) when compared to baseline values.

Serial ultrasound images demonstrate nodule volume reduction after RFA. Left image (Pre-RFA): Transverse ultrasound image showing a solid benign thyroid nodule with an initial volume of 5.01 mL. Middle image (Six-month follow-up): Significant volume reduction is observed (0.41 mL). Right image (Twelve-month follow-up): Continued shrinkage of the nodule is evident (0.01 mL), achieving a final VRR of 99.8%.
Alterations in thyroid function following RFA treatment
Figure 3 demonstrates the changes in thyroid function parameters post-RFA treatment observed in our study. Over long-term follow-up, we noted a significant decrease in T3 and FT4 levels (90.40 vs 93.40, p = 0.036 for T3; 1.02 vs 1.06, p = 0.002 for FT4) alongside an increase in TSH levels (1.29 vs 1.09, p = 0.006) compared to baseline, though remained within the normal physiological range. This indicates a measurable impact of RFA on thyroid hormone dynamics. Additionally, during mid-term follow-up, a significant decrease in T3 levels (88.95 vs 93.40, p = 0.010) and an increase in TSH levels (1.42 vs 1.09, p < 0.001) were observed, whereas FT4 levels remained relatively stable without notable reduction. Therefore, we concentrated our further study on mid-term T3 and TSH levels.

Alterations in thyroid function following RFA treatment.
Relationship between thyroid function and volume reduction
A partial analysis, adjusted for age and sex, demonstrated a negative correlation between medium-term T3 levels and the long-term VRR in thyroid nodules, with a correlation coefficient of r = −0.475 and statistical significance at p = 0.001 (Table 2 and Figure 4). Specifically, a more pronounced decrease in T3 levels during the mid-term follow-up period was associated with a greater reduction in nodule volume over the long term, suggesting a potential predictive role of T3 levels in treatment outcomes. This suggests that mid-term T3 levels serve as a robust predictive marker for treatment outcomes, even after accounting for individual demographic variations.
Partial analysis.

The values were denoted as “r value (p value).”
Bold values indicate statistical significance.
Corrective p < 0.00625.
T3, triiodothyronine; TSH, thyroid-stimulating hormone; VRR, volume reduction ratio.

Scatter plot.
Subgroup of low T3 level and high T3 level
During the medium-term follow-up, a comparison was conducted between groups with lower and higher T3 levels to evaluate initial nodule volume and the VRR over both medium- and long-term periods, as shown in Table 3. The initial volume did not differ significantly between the low T3 group (6.97 mL) and the high T3 group (10.72 mL), with a p-value of 0.439. However, the low T3 group demonstrated significantly higher VRRs at long-term (88.5% vs 76.5%, p = 0.030) follow-ups, indicating a more substantial volume decrease in nodules among lower T3 level patients.
Subgroup analysis of T3 level after RFA.
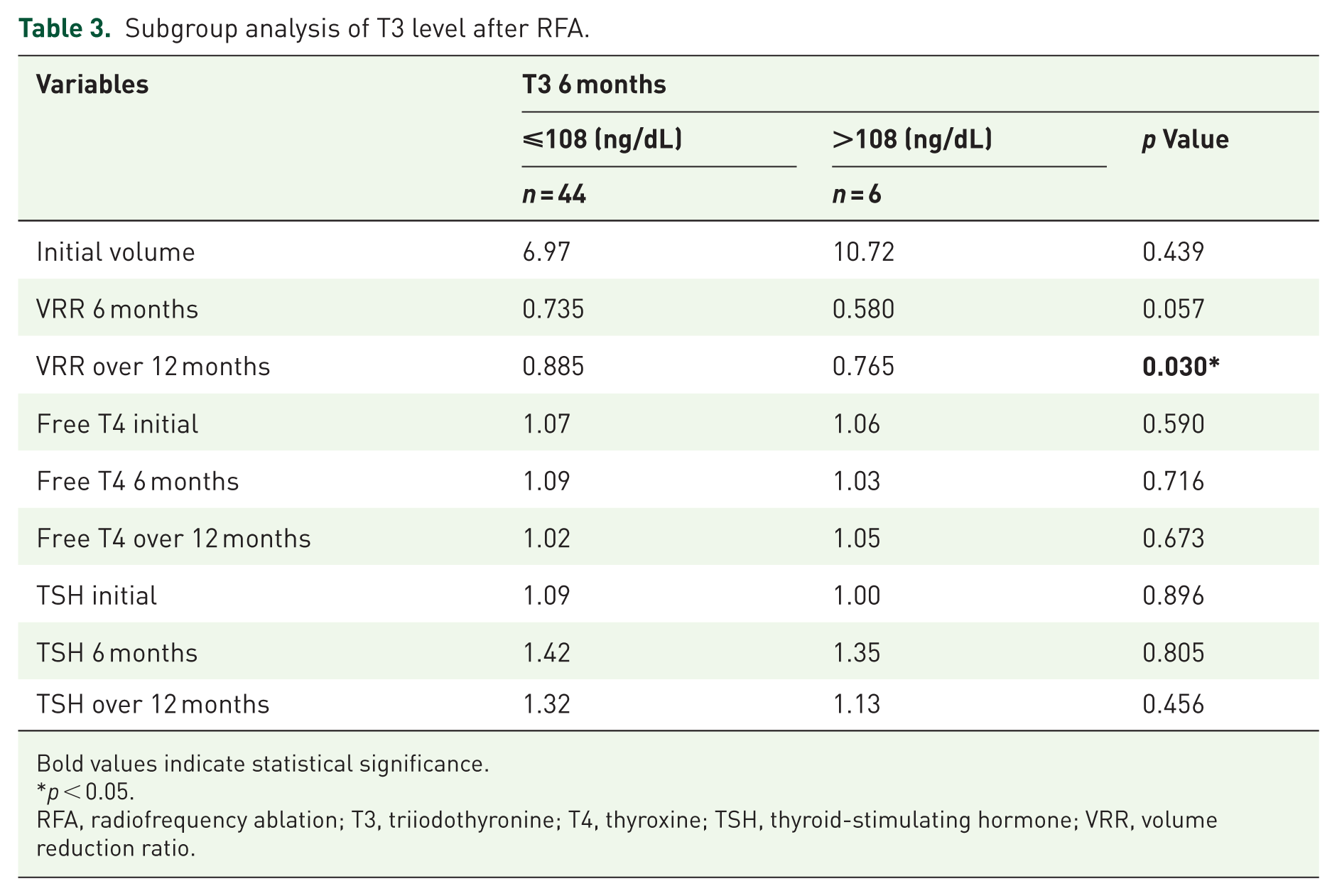
Bold values indicate statistical significance.
p < 0.05.
RFA, radiofrequency ablation; T3, triiodothyronine; T4, thyroxine; TSH, thyroid-stimulating hormone; VRR, volume reduction ratio.
Discussion
This retrospective study analyzed trends in thyroid function following RFA of BTNs in euthyroid patients treated at a single institution. The findings revealed a significant decrease in T3 levels, accompanied by an increase in TSH levels, in both medium-term and long-term follow-ups when compared to baseline levels. Notably, a more pronounced mid-term reduction in T3 was associated with a greater long-term VRR of the treated nodules. Moreover, when comparing patient groups with lower and higher T3 levels during the medium-term follow-up, those with lower T3 levels exhibited a significantly higher long-term VRR, suggesting a potential link between T3 dynamics and nodule shrinkage outcomes.
Thyroid function alterations following RFA treatment
Thyroid RFA is theorized to potentially affect thyroid function due to the induced damage to targeted parts of the gland. This damage could, in theory, lead to thyrotoxicosis, resulting from the sudden release of thyroid hormones in the ablated tissue, or to hypothyroidism if extensive areas of the thyroid are destroyed.11,12 Studies have examined these potential changes, with some evidence of transient alterations in thyroid function immediately post-RFA. For instance, a prospective study conducted on 22 BTNs reported significant changes in thyroid hormone levels shortly after RFA, with a decrease in TSH and increases in free T3 (FT3) and FT4 observed 1 week post-procedure. These values, however, returned to baseline at follow-ups conducted at 1, 6, and 12 /months. 10 Similarly, a retrospective analysis observed a case of transient hypothyroidism in one out of 875 patients following RFA of BTNs, which resolved within a year without long-term consequences.12,13 Nevertheless, evidence from multiple studies indicates that RFA of BTNs does not significantly impact long-term thyroid function, with TSH, FT4, and FT3 levels remaining within normal ranges over time.11,14,15
In contrast, our study observed a long-term trend characterized by significant reductions in FT4 and T3 levels accompanied by a mild elevation in TSH relative to baseline. While these changes remained within the euthyroid range, this trend differs from the transient thyrotoxicosis reported in early-phase studies. 10 This divergence may be attributed to the “volume-reduction effect” on the functional thyroid mass. By effectively neutralizing hyper-metabolic nodular tissue, RFA reduces the total hormone-secreting capacity of the gland, particularly when the treated nodules contributed significantly to the baseline hormone pool. The mild TSH elevation likely reflects a compensatory physiological feedback mechanism of the pituitary-thyroid axis to maintain homeostasis following the reduction in circulating FT4 and T3. These findings align with larger retrospective analyses12 –15 which suggest that while thyroid function remains stable and safe, a subtle recalibration of the thyroid axis is common as the ablated nodular tissue is resorbed and replaced by inactive fibrosis.
Mechanism linking mid-term thyroid function to long-term volume resorption
In our study, we observed a significant negative correlation between mid-term T3 levels and the long-term VRR. Specifically, patients whose mid-term T3 levels fell below the 108 mg/dL cutoff exhibited superior long-term VRR. While previous research has primarily focused on baseline TSH or ablation volume as risk factors for post-procedural thyrotoxicosis,10,16 our findings fill a critical clinical gap: in euthyroid patients, these hormonal fluctuations are not merely transient side effects but serve as a valuable prognostic indicator for therapeutic success.
This correlation can be explained through the pathophysiological mechanisms of thermal destruction and subsequent tissue remodeling. A mid-term T3 decline likely acts as a systemic surrogate marker for the “net ablation effect,” representing the completeness of follicular destruction within the targeted nodule. In contrast, technical parameters such as total energy delivery showed no significant correlation with VRR, likely due to the heat-sink effect, where blood flow in adjacent vessels dissipates thermal energy and limits the consistency of tissue necrosis. By monitoring T3 levels, we can more accurately account for these individual biological variations in response to thermal insult.
Furthermore, we analyzed the impact of nodule composition, as cystic nodules are traditionally expected to yield higher VRR. However, our analysis showed no significant difference in VRR between solid and predominantly cystic groups. This underscores that the thoroughness of functional tissue destruction—represented by the mid-term T3 decline—is a more potent driver of long-term volume reduction in our cohort than the initial cystic component. A more pronounced decline in T3 reflects a higher efficiency of the initial ablation in neutralizing the nodule’s cellular matrix, which in turn triggers a more robust localized inflammatory response. This biological process recruits macrophages for the effective phagocytosis of necrotic debris, followed by substantial fibrotic contraction—the essential pathway for achieving a high long-term VRR.
By shifting the clinical focus from simply monitoring post-procedural safety to utilizing biochemical markers like T3 as early indicators of long-term success, this study provides a novel framework for post-RFA counseling and evaluation. Despite these temporary fluctuations, the absence of permanent hypothyroidism in our cohort is reassuring for both clinicians and patients, suggesting that a single RFA procedure effectively balances therapeutic efficacy with functional preservation.
Comparison of various treatments for BTNs
RFA has emerged as a minimally invasive alternative to traditional treatments for thyroid nodules, offering distinct advantages in efficacy, risk profile, and thyroid function preservation.1,17 Compared to surgery, which remains the standard treatment for large or symptomatic nodules, RFA effectively reduces nodule size with a lower risk of complications.18,19 Surgery carries risks of nerve damage, bleeding, and hypothyroidism, whereas RFA is less likely to impair thyroid function due to its localized tissue ablation, preserving surrounding healthy tissue. A study involving 50 patients revealed a complication rate of 8% for RFA compared to 37.1% for surgery, with no cases of hypothyroidism reported in the RFA group. 20 In comparison with microwave ablation (MWA), RFA provides comparable outcomes in terms of nodule volume reduction and symptom relief.21,22 A prospective multicenter study involving 1252 patients demonstrated that the RFA group achieved a superior VRR compared to the MWA group at the 6 and 12 months (84.1% vs 78.4%, 89.6% vs 82.5%), as well as at the last follow-up. 23 Additionally, previous studies have indicated no significant differences in complication rates between RFA and MWA.23,24 Transarterial embolization (TAE) is a promising alternative for managing large BTNs, including those with substernal extension. By occluding the thyroid’s blood supply, TAE significantly reduces nodule volume.25,26 Although complication rates, such as post-embolization thyroiditis and transient thyroid dysfunction, range from 5% to –10%, stroke remains a major concern.27,28 Overall, RFA stands out as a safer, less invasive option that maintains thyroid function, making it a preferable choice for patients with benign nodules, especially when preserving thyroid function is a priority.
Study limitations and future directions
Our study presents several limitations that should be considered when interpreting the findings. Firstly, the retrospective design may not fully capture the actual treatment outcomes. Patients who experience marked improvement in cosmetic or symptomatic issues may choose not to continue with follow-up appointments, potentially leading to an underestimation of the treatment’s overall effectiveness. Secondly, our study lacks short-term data, specifically within the initial 6-month period, during which additional changes in thyroid function could emerge and offer further insights. Furthermore, while our overall cohort was sufficient for primary analysis, the sample size of specific subgroups was small. Notably, the high T3 subgroup included only six patients, which may result in a lack of statistical power for this specific category. Therefore, results derived from this subgroup should be interpreted with caution. Moreover, while we included all eligible consecutive patients during the study period, a formal pre-study power analysis was not performed, and our findings should be viewed as exploratory in nature. To overcome these limitations, future research should focus on prospective studies with larger patient populations and a particular emphasis on gathering short-term follow-up data to assess treatment outcomes and potential thyroid function alterations more accurately.
Conclusion
In conclusion, RFA is an effective treatment for BTNs, achieving significant long-term nodule volume reduction without causing permanent hypothyroidism. Notable alterations in thyroid function were observed, including a reduction in T3 levels and an increase in TSH levels at both medium- and long-term follow-ups. Importantly, lower mid-term T3 levels were associated with a higher VRR, suggesting that mid-term thyroid function monitoring may serve as a useful indicator for predicting long-term outcomes. Despite these thyroid function changes, all patients maintained normal thyroid function throughout the study period. 29
Supplemental Material
sj-docx-1-tae-10.1177_20420188261432727 – Supplemental material for Thyroid function alterations after radiofrequency ablation in benign thyroid nodule management
Supplemental material, sj-docx-1-tae-10.1177_20420188261432727 for Thyroid function alterations after radiofrequency ablation in benign thyroid nodule management by Yi-Han Chen, Pi-Ling Chiang, Cheng-Kang Wang, An-Ni Lin, Yen-Hsiang Chang, Chen-Kai Chou and Wei-Che Lin in Therapeutic Advances in Endocrinology and Metabolism
Supplemental Material
sj-xlsx-2-tae-10.1177_20420188261432727 – Supplemental material for Thyroid function alterations after radiofrequency ablation in benign thyroid nodule management
Supplemental material, sj-xlsx-2-tae-10.1177_20420188261432727 for Thyroid function alterations after radiofrequency ablation in benign thyroid nodule management by Yi-Han Chen, Pi-Ling Chiang, Cheng-Kang Wang, An-Ni Lin, Yen-Hsiang Chang, Chen-Kai Chou and Wei-Che Lin in Therapeutic Advances in Endocrinology and Metabolism
