Abstract
Objective
This study was performed to explore the effective management of bleeding associated with radiofrequency ablation (RFA) of benign thyroid nodules.
Methods
Thirty-five patients with benign thyroid nodules who were treated with ultrasound-guided RFA from July 2015 to December 2016 at the Third Affiliated Hospital of Sun Yat-sen University were retrospectively reviewed. The technique efficacy, bleeding, and other complications were assessed during the follow-up period.
Results
The mean technique efficacy was 55.6%±22.8% at 1 month and 24.1%±17.1% at 6 months after the procedure. One case of an intranodular haematoma and two cases of voice change (>1 month) were observed. All patients recovered with corresponding treatment.
Conclusion
Although the incidence of haemorrhage is low, serious haematomas are life-threatening. Therefore, having a comprehensive understanding of the potential complications, an accurate clinical strategy, and adequate technical skills may prevent or help to properly manage these complications.
Introduction
Thyroid nodules are extremely common, and the associated morbidity rate ranges from 19% to 68% according to high-resolution ultrasound (US) findings. 1 , 2 Most thyroid nodules are benign and require no intervention other than clinical follow-up. According to the 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer, thyroid-stimulating hormone suppression therapy for benign thyroid nodules (BTNs) is not recommended because the potential harm outweighs the benefit. 3 Radioiodine therapy was historically an effective treatment for thyroid hot nodules and a possible alternative to surgery. However, this technique has been proven to have uncertain efficacy and some adverse effects, such as hypothyroidism or recurrence.4–6 Surgery may be considered for growing BTNs with pressure-related symptoms, neck discomfort, cosmetic concerns, or decreased quality of life. 3 At present, partial/total thyroid surgery is considered the gold standard treatment. Surgery is associated with numerous complications, such as nerve injury, anaesthesia-related problems, long hospital stays, conspicuous scars, haemorrhage, and lesions of the parathyroid glands. 7 , 8 In addition, hypothyroidism is inevitable after total thyroidectomy and requires lifelong hormone supplementation. Hence, increasingly minimally invasive therapeutic strategies are currently used to treat BTNs. In most cases, several thermal ablation techniques such as laser ablation, microwave ablation, radiofrequency ablation (RFA), and high-intensity focused US have been shown to be effective in BTNs. Among these thermal ablation techniques, RFA is the most widely applied. 9 , 10
RFA of thyroid diseases, first reported in 2001,11–13 is considered efficacious and safe for treatment of BTNs. 14 , 15 To date, no life-threatening complications related to RFA have been reported. However, several cases of haemorrhage related to fine needle aspiration (FNA) or core needle biopsy (CNB) have been reported.16–18 Although a micro-invasive procedure such as FNA can result in massive uncontrolled bleeding resulting in upper airway respiratory obstruction, such uncontrolled bleeding is a rare but life-threatening complication of RFA. Thus, management of bleeding associated with RFA of BTNs is of vital importance. This study was performed to explore the effective management of bleeding associated with RFA of BTNs.
Materials and methods
This study was approved by the Ethics Committee of the Third Affiliated Hospital of Sun Yat-sen University, and written informed consent was obtained from all patients prior to the performance of US-guided FNA or CNB and RFA. The requirement to obtain informed consent for publication was waived because of the retrospective nature of the study.
Patients
All consecutive patients who underwent RFA of BTNs at our institution from July 2015 to December 2016 were analysed. The following inclusion criteria were applied: confirmation of benignancy (Bethesda Class II) by FNA cytology or CNB; complaints of pressure symptoms (compressive symptoms, neck discomfort, or foreign body sensation) or cosmetic problems; a >2-cm maximum diameter of the index nodule; anxiety about a malignancy, unsuitability for surgery, or unwillingness to undergo surgery; and a normal serum thyrotropin concentration, normal complete blood counts, and normal blood coagulation test results. The exclusion criteria were nodules showing malignant features (i.e., taller than wide, spiculated margin, marked hypoechoic appearance, or microcalcifications) on US imaging, 19 abnormal thyroid function, performance of other treatments for the thyroid nodules within 6 months before the procedure, pregnancy, and age of <18 years. For the present study, only patients with ≥6 months of follow-up after the procedure were included. Thirty-five patients met the inclusion criteria.
Pretreatment assessment
Before the procedure, conventional US findings, US-guided FNA findings, contrast-enhanced US (CEUS) findings, and laboratory and clinical results were evaluated. Two radiologists (T.W. and J.R. with 10 and 18 years of thyroid US experience, respectively) performed the US, US-guided FNA, and CEUS examinations using a Logiq E9 US device (GE Medical Systems, Milwaukee, WI, USA) equipped with an ML6-15 linear transducer with a centre frequency of 12 MHz (frequency range, 4–15 MHz). The US examination included characterisation of the location, shape, size, margins, solid/cystic proportions, echogenicity, calcification status, and internal vascularity of each nodule.
Laboratory tests included the levels of thyroid-stimulating hormone, free triiodothyronine, free thyroxin, and thyrotropin; a complete blood cell count; and a coagulation test (prothrombin time and activated partial thromboplastin time). The nodule volume was calculated using the following equation: volume = length × width × depth × 0.524. In addition, all patients underwent vocal cord function assessments by an experienced laryngologist before the ablation procedure. At enrolment, all patients were asked to rate their pressure symptoms on a 10-cm visual analogue scale (grade 0–10 cm), and the cosmetic grading score was assessed by the physician as described in the consensus statement. 20
Procedures and equipment
All RFA procedures were performed by one radiologist (J.R.) with 3 years of experience performing RFA in an outpatient clinic. We used an RF generator (VIVA RF System®; STARmed, Gyeonggi-si, South Korea) and an internally cooled 18-gauge, 70-mm-length, 7- or 10-mm, active-tip electrode (Star RF Electrode®; STARmed). Local anaesthesia with 2% lidocaine was applied to the puncture site. The hydrodissection technique was used under US guidance (5% glucose and norepinephrine were mixed and injected into the surrounding thyroid capsule), which provided a safe distance between the needle tip and adjacent critical structures. During the procedure, we paid special attention to the preservation of surrounding important structures to prevent significant complications. Therefore, two essential techniques were applied: the trans-isthmic approach and the moving-shot technique. 21 , 22 Ablation was suspended when the index nodule was covered by hyperechoic zones. The technique efficacy (TE) was then evaluated by CEUS at 5 to 10 minutes after RFA, until the hyperechoic zones disappeared. Technical success was defined as the change of an entire nodule to a no-enhancement zone on real-time CEUS. For nodules with an enhancement zone, an additional ablation was performed to destroy the nodule as much as possible. Complications were monitored immediately after the procedure and during the follow-up period. Major and minor complications and adverse effects were defined according to the criteria established by the Society of Interventional Radiology. 23 , 24
Follow-up evaluation
Any specific complaints or concerns were recorded for 1 month. Post-procedural follow-up was performed at 1 and 6 months after treatment. At each follow-up visit, a US examination, CEUS examination, and serum thyroid hormone measurements were performed; pressure symptoms and the cosmetic grading score were evaluated; and the volume of the treated nodule was calculated. The TE was calculated using the following equation: TE = (final nodule volume/initial nodule volume) × 100.
Statistical analysis
All statistical analyses were performed using SPSS software version 19.0 (IBM Corp., Armonk, NY, USA). Continuous variables are expressed as mean ± standard deviation. Quantitative data for volume and TE were analysed using a paired t-test. A P value of ≤0.05 was considered statistically significant.
Results
The patients’ characteristics are summarised in Table 1. Thirty-five patients underwent RFA, including 6 male and 29 female patients (mean age, 37.3 years). The mean largest BTN dimension was 36.5 ± 9.4 mm (range, 20–52 mm), and the mean BTN volume was 10.2 ± 7.0 mL. Twenty-two total complications (19 minor and 3 major complications) were observed among the treated patients. None of these complications was life-threatening, and all occurred without sequelae.
Patients’ baseline characteristics (n = 35)
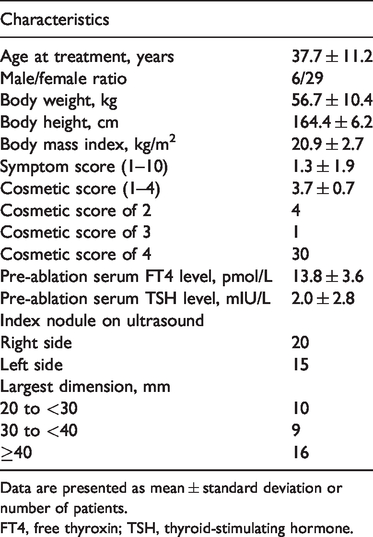
Data are presented as mean ± standard deviation or number of patients.
FT4, free thyroxin; TSH, thyroid-stimulating hormone.
Nodule volume
After treatment, the overall volume of the nodules significantly decreased (10.2 ± 7.0 mL at baseline, 5.4 ± 4.2 mL at 1 month, and 2.5 ± 2.3 mL at 6 months; P < 0.001), and the TE was 55.6% ± 22.8% at 1 month and 24.1% ± 17.1% at 6 months (P < 0.001) (Table 2). Figure 1 shows the shrinkage of the nodules at 1 and 6 months after the procedure compared with baseline; no hypoechoic blood supply was observed within the area of the nodules.
Changes in volume before RFA and at each follow-up visit.

Data are presented as mean ± standard deviation (range).

(a, c, e) Ultrasound examination and (b, d, f) contrast-enhanced ultrasound examination of a 39-year-old woman treated with radiofrequency ablation. (a, b) Ultrasound and contrast-enhanced ultrasound revealed a cystic-solid nodule before ablation. (c, d) One month after ablation, ultrasound showed a hypoechoic nodule with a decreased volume. (d, e) Six months after ablation, the volume of the nodule had decreased further, and no blood supply was observed within the area of the nodule.
Bleeding complications
Twelve patients developed bleeding complications, including a perithyroidal haematoma (minor complication) in 11 patients and an intranodular haematoma (major complication) in 1 patient, as shown in Table 3. The haematomas were detected by US scans, which revealed gradual enlargement of a hyperechoic mass in or around the nodules (Figure 2). For the patient with intranodular haemorrhage, the haematoma was controlled through timely use of the ablation needle to coagulate the injured blood vessel and by injecting lyophilising thrombin powder into the haematoma (Figure 3). Most of the perithyroidal haematomas in this series required only observation with or without compression and disappeared within 1 to 2 weeks after the procedure. None of the 35 patients developed a subscapular haematoma.
Complications and adverse effects in 35 patients who underwent RFA of thyroid nodules.
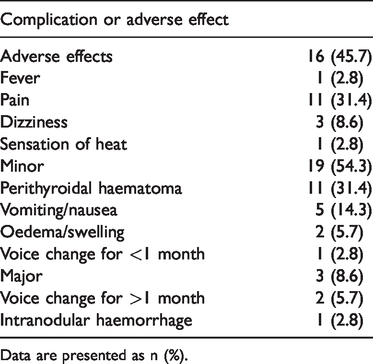
Data are presented as n (%).
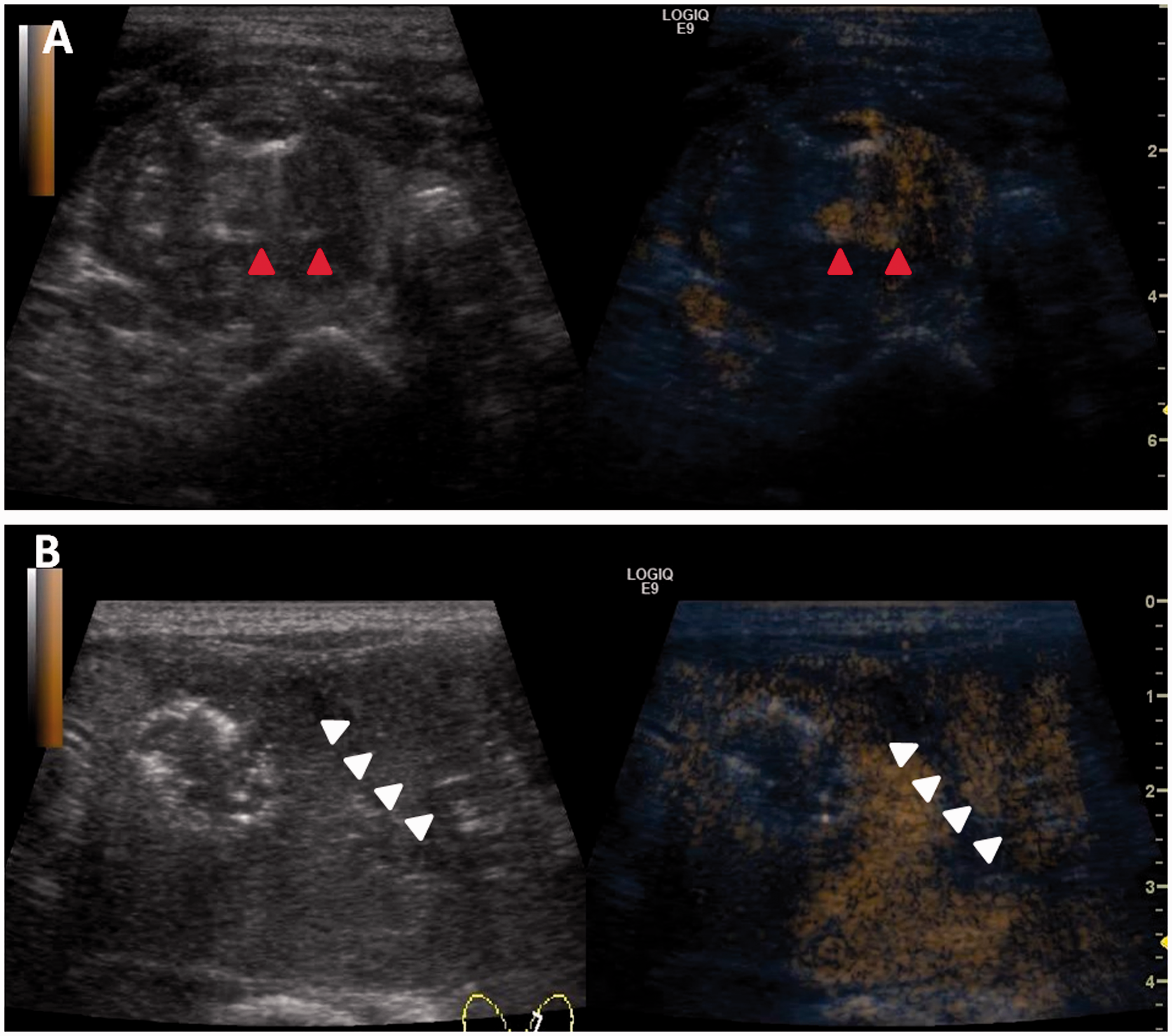
Ultrasound examination and contrast-enhanced ultrasound examination of patients with intranodular haemorrhage and perithyroidal haemorrhage. (a) Ultrasound and contrast-enhanced ultrasound revealed a hyperechoic mass lesion in the nodule. (b) Ultrasound and contrast-enhanced ultrasound revealed perithyroidal haemorrhage.

Ultrasound examination and contrast-enhanced ultrasound examination of patients with intranodular haemorrhage or perithyroidal haemorrhage during ablation. (a) Ultrasound revealed a hyperechoic mass lesion in the nodule. (b) Ultrasound showed an ablation needle inserted into the nodule to coagulate bleeding vessels. (c) After lyophilising thrombin powder was injected into the hematoma, ultrasound and contrast-enhanced ultrasound showed disappearance of the hyperechoic mass lesion and microbubble extravasation. (d) Ultrasound showed a hyperechoic mass lesion around the thyroid, and contrast-enhanced ultrasound showed no microbubble extravasation around the thyroid. (e) After lyophilising thrombin powder was injected into the haematoma, no microbubble extravasation was observed.
Other complications and adverse effects
The adverse effects of RFA included fever (n = 1, 2.8%), pain (n = 11, 31.4%), dizziness (n = 3, 8.6%), and a sensation of heat (n = 1, 2.8%). Minor complications included vomiting/nausea (n = 5, 14.3%), oedema/swelling (n = 2, 5.7%) and a voice change for <1 month (n = 1, 2.8%).
Discussion
Image-guided thermal ablation techniques, such as laser ablation, ethanol ablation, microwave ablation, high-intensity focused US, and particularly RFA, have recently become more widely used to treat thyroid nodules. Briefly, the basic mechanism of RFA involves the creation of thermal damage by friction and heat conduction, which is generated from an oscillating high-frequency alternating electric current produced by the RFA generator and then transferred through the electrode tip. The energy of RFA is powerful and accurate. 25 , 26 RFA is considered an effective and safe treatment for control of BTNs.
In most cases, the incidence of haemorrhage and other complications is low. 20 However, haemorrhage is sometimes life-threatening because serious haematomas may compress the upper airways. Many reports have described active bleeding during FNA of thyroid nodules and RFA of hepatocellular carcinomas,14–16,24–26 and some reports have described fatalities. 14 Thus, haemorrhage is an important complication.
Three types of haemorrhage may occur: perithyroidal, subcapsular, and intranodular.12,14,27–29 The mechanism of haemorrhage is thought to be related to the mechanical or thermal injuries induced by the RFA electrode tip. 30 , 31 Thyroid nodules reportedly have abundant capsular vessels that are usually anastomosed with vessels penetrating into the core. 32 These numerous vessels are abnormal, thin-walled, and susceptible to rupture. Large thyroid nodules are another cause of haemorrhage because multiple insertions are often required to treat such nodules. In addition, if the patient cannot coordinate with the physician during the RFA procedure, the perithyroidal or intrathyroidal vessels may easily be damaged by movement of the needle tip or production of heat energy.
It is important to manage bleeding associated with RFA of BTNs. Based on our experience, we suggest several steps to prevent such bleeding. Physicians should obtain a thorough medical history of each patient before the procedure. All risk factors for bleeding, including drugs (antiplatelet drugs, nonsteroidal anti-inflammatory drugs, and anticoagulants) and diseases affecting coagulation, should be recorded. 33 In addition, the patient’s coagulation function should be thoroughly evaluated. All patients with clinical coagulation disorders should be excluded. Even when coagulation indices are normal in patients with high risk factors for bleeding, such as liver cirrhosis, end-stage renal disease, anticoagulant use, or hypertension, 34 sufficient preoperative preparation should be emphasised. A patient with active bleeding in the present study met these conditions. Although his coagulation indices were normal, he had a subclinical coagulation disorder due to end-stage liver disease. Fresh frozen plasma or human prothrombin complex should be used in patients with liver cirrhosis, and anticoagulants should be withdrawn in these patients, which will help to improve coagulation function before the procedure. If a possibility of bleeding exists, Reptilase (haemocoagulase atrox for injection; Pentapharm, Basel, Switzerland) can be used preoperatively.
During the RFA procedure, an effective clinical strategy and adequate technical skills are both essential. Patient cooperation is the first requirement. When the needle tip is in the patient’s body, any uncooperative motion of the patient may lead to injury of vessels or other structures. Most patients can endure the procedure under local anaesthesia; however, anxious patients may require general anaesthesia to achieve cooperation. If possible, small-bore electrodes should be chosen to decrease the risk of bleeding. 35 It is necessary to cauterise the supplying vasculature of nodules to avoid recurrence and residue. However, the puncture route should be carefully designed to avoid pericapsular vessels, and the electrode tip should be closely monitored. Active bleeding during needle puncture is visible as a rapidly expanding hypoechoic or anechoic signal. Locating the haemorrhagic focus is not difficult with CEUS guidance. The bleeding point can be blocked by RF electrode tip insertion and direct ablation. When the bleeding is too rapid to control with the RF electrode tip by increasing the power, drug injection is a suitable alternative. Lyophilised thrombin powder can be dissolved in normal saline and then injected at the bleeding point through a syringe with US guidance. One report also described haemorrhage treated by local injection of hypertonic saline and epinephrine solution in a patient with hepatocarcinoma. 36 Mild bleeding, which appears as a hypoechoic layer, can mostly be controlled using ice and compression of the neck for several minutes after the procedure. 30 All bleeding can be controlled by conservative methods; thus, no surgical intervention is needed. Ecchymosis can be found after the procedure and usually disappears in approximately 2 to 3 weeks.
Post-procedure CEUS is indispensable for all patients, regardless of whether bleeding occurs. CEUS is an objective evaluation tool for active bleeding. 37 Close clinical observation for 4 hours postoperatively is recommended in our department because most bleeding occurs during the first 4 hours after thyroid lobectomy. 38 Observation of the neck can help to detect a haematoma early and may aid in preventing serious adverse effects.
Conclusion
Acute thyroid bleeding is one possible complication of RFA; although rare, it is potentially life-threatening. Proper selection of patients and sufficient preparation are essential. During the RFA procedure, both an effective clinical strategy and adequate technical skills are indispensable. The physician should trace the electrode tip using real-time US and sufficiently manage bleeding. Mild bleeding has limited morbidity and can be easily controlled by compression. Active bleeding tends to be rare; however, it may be disastrous if the operator is unaware or careless. Direct ablation with the RF electrode tip and drug injection into the bleeding focus are effective modalities for active bleeding. CEUS and close observation are also recommended after the procedure to detect abnormalities early. RFA is an effective and relatively safe alternative for selected patients with BTNs if performed by skilled physicians.
Footnotes
Author contributions
(I) Conception and design: Jie Ren and Bo Liu. (II) Administrative support: Jie Ren. (III) Provision of study materials or patients: Kunpeng Hu and Yufan Lian. (IV) Collection and assembly of data: Jinfen Wang and Wenchao Li. (V) Data analysis and interpretation: Wenchao Li and Zhicheng Yao. (VI) Manuscript writing: All authors. (VII) Final approval of manuscript: All authors.
Data availability
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National Natural Science Foundation of China (CN) (No. 81702375), the Natural Science Foundation of Guangdong Province (No. 2016A030313200), the Science and Technology Project of Guangzhou City (No. 20160701002), the Hengrui Foundation of Hepatobiliary and Pancreatic Cancer Research (No. CXPJJH11800001-20183331), the Natural Science Foundation of Guangdong Province (No. 2017A030313580), the Fundamental Research Funds for the Central Universities (Sun Yat-sen University) (No. 17ykpy67), and the 5010 Clinical Research Project of Sun Yat-sen University (No. 2016016).
