Abstract
Background:
Latent autoimmune diabetes in adults (LADA) is a form of diabetes that shares clinical features with type 2 diabetes mellitus (T2DM), often leading to misdiagnosis and delayed treatment. Early detection is critical to prevent the progression of the disease.
Objectives:
This study aims to analyze the risk factors of LADA and develop a predictive model to enhance early diagnosis.
Design:
A retrospective study was conducted on T2DM patients treated at our hospital between June 2019 and June 2024. The study focused on identifying risk factors for LADA and developing a predictive model.
Data sources and methods:
Clinical data of 728 patients (651 non-LADA, 77 LADA) were analyzed. LASSO regression was used for variable selection, followed by logistic regression to identify risk factors. The model’s performance was assessed using the receiver operating characteristic curve and the Hosmer–Lemeshow test.
Results:
Significant differences were found between the non-LADA and LADA groups in terms of thyroid disease history, diabetic ketoacidosis, fasting plasma glucose (FPG), 2-hour postprandial glucose (2hPG), and glycated hemoglobin (HbA1c) levels (
Conclusion:
The predictive model based on thyroid disease history, FPG, 2hPG, and HbA1c demonstrates excellent predictive ability in our cohort for early identification of LADA, suggesting its potential to aid in timely intervention and improved patient outcomes.
Plain language summary
Some adults who seem to have the common type 2 diabetes might actually have a different kind, called LADA. Telling them apart early is important because their treatments are not the same. This study tried to find simple clues from routine check-ups to help tell who might have LADA.
We reviewed the health records of 728 adults diagnosed with type 2 diabetes. We used a special analysis to pick out which patient features were most useful in identifying those who actually had LADA.
We found four key clues:
• A past history of thyroid disease.
• Higher sugar levels after fasting (not eating).
• Higher sugar levels after a meal.
•A higher long-term average blood sugar level (HbA1c).
We built a simple scoring tool using these four clues. This tool was very good at correctly pointing out patients with LADA in our study group.
Doctors can use our simple tool, based on four common tests, to quickly identify patients who need more specific checks for LADA. This can help people get the right treatment sooner.
Introduction
Latent autoimmune diabetes in adults (LADA) is a form of diabetes marked by a gradual autoimmune attack on the pancreatic β-cells, which do not necessitate insulin therapy during the initial clinical phase. 1 This condition poses various negative impacts on the health of individuals. Patients frequently experience inadequate control of blood glucose, accompanied by notable variations in glucose levels, which heighten the chances of both hypoglycemic and hyperglycemic events. Sustained irregularities in blood glucose can result in several complications, including retinopathy, diabetic nephropathy, and cardiovascular disorders, which significantly diminish the quality of life and lifespan of patients. 2 Because there is some overlap in clinical features between LADA and type 2 diabetes mellitus (T2DM), a significant number of patients are mistakenly identified as having T2DM in the early phases of the condition. This misdiagnosis contributes to unsuitable treatment strategies, hinders the protective measures for pancreatic β-cell performance, and hastens the deterioration of islet functionality.3,4 Thus, timely detection of LADA is vital. This article examines the risk factors associated with LADA and develops a predictive framework, intending to lay the groundwork for the early detection of this condition.
Materials and methods
This study was conducted and reported in accordance with the Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) statement. 5 The completed TRIPOD checklist is provided as Supplemental Material.
This retrospective observational study also adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.6,7 The completed STROBE checklist is provided as Supplemental Material.
Baseline data
A single-center retrospective study was conducted at the First Affiliated Hospital of Ningbo University (Ningbo, China). The clinical data of patients with T2DM who received treatment between June 2019 and June 2024 were analyzed. The diagnosis of T2DM adhered to the “Chinese Guidelines for the Prevention and Treatment of Type 2 Diabetes (2020 Edition),” 8 while LADA diagnosis was based on the “Chinese Expert Consensus on the Diagnosis and Treatment of Latent Autoimmune Diabetes in Adults (2021 Edition)” 9 with the following criteria: (1) age of onset ⩾18 years; (2) positive islet autoantibodies (utilizing the antiglutamic acid decarboxylase antibody (GADA)) or positive islet autoimmune T cells; (3) no requirement for insulin therapy for at least 6 months after diabetes diagnosis. Additionally, in line with the consensus, the presence of residual β-cell function was evaluated, typically evidenced by fasting or postprandial C-peptide levels above the lower limit of the laboratory reference range at diagnosis. Inclusion criteria included: (1) fulfillment of the relevant diagnostic standards; (2) age of 18 years or older; (3) availability of complete clinical data. Initially, 812 patients were screened, of which 84 (10.3%) were excluded due to missing data in one or more key variables. The missingness pattern was assessed, and the rate of missing data for individual variables ranged from 2.1% to 8.7%, with apolipoprotein B (APOB) having the highest missing rate. The missing data were assumed to be missing at random (MAR), as the missingness was primarily related to logistical factors (e.g., specific tests not ordered based on clinical discretion rather than patient characteristics) rather than being systematically associated with LADA status or other outcome-related factors. The exclusion criteria encompassed: (1) individuals with other significant illnesses; (2) individuals with different autoimmune conditions; (3) pregnant or nursing individuals; (4) cases of secondary diabetes, among others. Ethical approval for this retrospective study was granted by the Ethics Committee of our hospital, along with a waiver of informed consent.
Sample size calculation
Generally, the final model incorporates 6–8 variables; referencing previous literature,10,11 the incidence of LADA in T2DM is approximately 5%—15%, and the sample size for modeling should be no less than 700 cases.
Indicator analysis
Gather the baseline information of patients, which include gender, age, body mass index (BMI), waist circumference, medical history of hypertension, thyroid disease, family history of diabetes, occurrences of diabetic ketoacidosis (DKA), smoking habits, alcohol intake history, symptoms categorized as “three more and one less” (i.e., polyuria, polydipsia, polyphagia, and weight loss), total cholesterol (TC), triglycerides (TG), levels of high-density lipoprotein cholesterol, low-density lipoprotein cholesterol (LDL-C), apolipoprotein A1, APOB, systolic blood pressure, diastolic blood pressure, fasting plasma glucose (FPG), glucose levels measured 2 h postprandial (2hPG), and glycated hemoglobin (HbA1c).
Statistical methods
Statistical analysis was performed using SPSS 26.0 and R 4.5.0 software. Measurement data conforming to a normal distribution were expressed as mean ± standard deviation (±SD) and compared using the
Results
Baseline patient characteristics
A total of 728 individuals diagnosed with T2DM participated in the study, including 651 non-LADA cases (89.42%) and 77 cases of LADA (10.58%). Significant differences (
Baseline characteristics of the patients.

2hPG, 2-h postprandial glucose; APOA1, apolipoprotein A1; APOB, apolipoprotein B; BMI, body mass index; DBP, diastolic blood pressure; DKA, diabetic ketoacidosis; FPG, fasting plasma glucose; HbA1c, glycated hemoglobin; HDL-C, high-density lipoprotein cholesterol; LADA, latent autoimmune diabetes in adults; LDL-C, low-density lipoprotein cholesterol; SBP, systolic blood pressure; TC, total cholesterol; TG, total triglycerides.
LASSO regression for variable selection
To address potential multicollinearity among certain variables, LASSO regression was employed for variable selection, alongside 10-fold cross-validation, with the minimum λ value identified as the optimal solution. The number of variables corresponding to this scenario was then counted. The results indicated that the λ value at the minimum error was 0.019, at which point six nonzero coefficient variables were selected: history of thyroid disease, DKA, “three more and one less” symptoms, FPG, 2hPG, and HbA1c (see Figure 1).
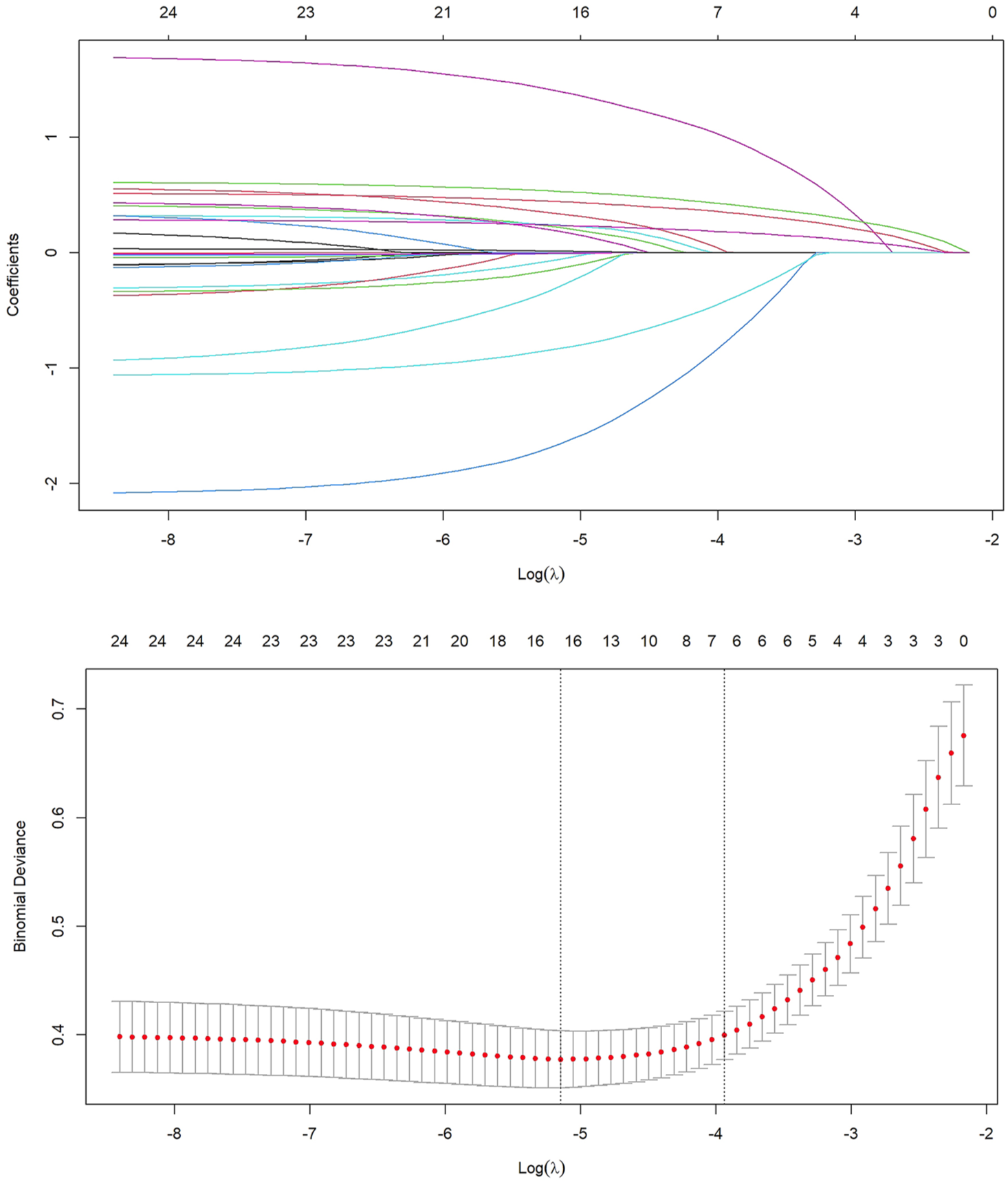
LASSO regression.
Logistic regression analysis
Thyroid disease history, DKA, “three more and one less” symptoms, FPG, 2hPG, and HbA1c were included as independent variables, with whether LADA was the dependent variable, into a multivariate binary logistic regression equation. The selection of the final four predictors (thyroid disease history, FPG, 2hPG, and HbA1c) was based on statistical significance in the multivariate analysis. DKA and “three more and one less” symptoms were excluded from the final model as they did not reach statistical significance (
Logistic regression analysis.

2hPG, 2-h postprandial glucose; CI, confidence interval; DKA, diabetic ketoacidosis; FPG, fasting plasma glucose; HbA1c, glycated hemoglobin; OR, odds ratio.
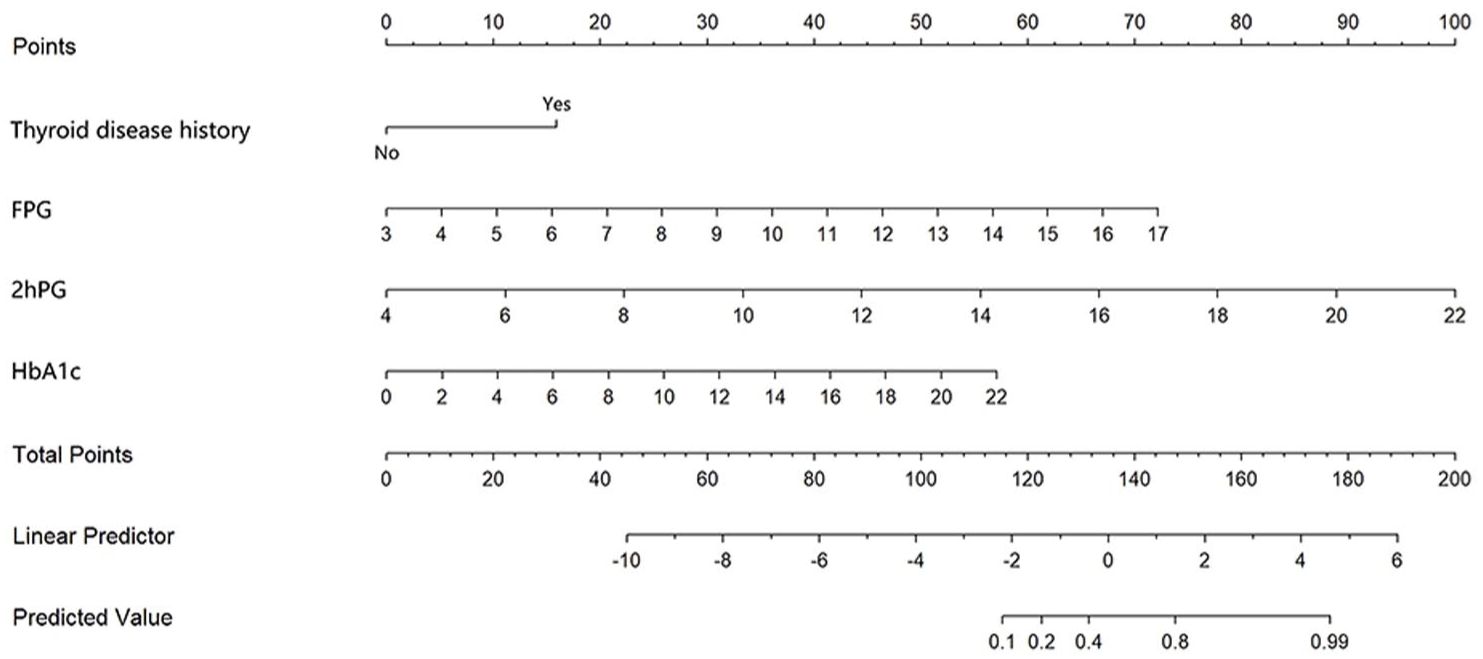
Nomogram of the prediction model.
Evaluation of prediction models
The area under the curve (AUC) of the predictive model is 0.907 (95% confidence interval (CI): 0.825–0.989). The optimal operating point was determined using Youden’s J statistic, which maximizes the sum of sensitivity and specificity. At the optimal cutoff probability of 0.18, the model achieved a sensitivity of 76.6% (95% CI: 67.8%—84.7%), a specificity of 91.9% (95% CI: 89.8%—93.9%), a positive predictive value (PPV) of 52.1% (95% CI: 43.8%—60.4%), and a negative predictive value (NPV) of 97.2% (95% CI: 95.9%—98.3%), indicating good discriminative ability (see Figure 3). Furthermore, internal validation using bootstrapping with 1000 resamples was performed to assess the model’s potential for overfitting and to evaluate its stability. The bootstrap-validated AUC was 0.892, indicating minimal optimism (0.015) and demonstrating that the model maintains good discriminative ability upon internal validation. The bootstrap validation also provided estimates of model calibration: the calibration slope was 0.94 (95% CI: 0.89–0.99) and the calibration intercept was −0.05 (95% CI: −0.11 to 0.01), indicating good agreement between predicted probabilities and observed outcomes. The Brier score was 0.072 (95% CI: 0.065–0.079), reflecting good overall model performance.
Receiver operating characteristic curve.
To evaluate the clinical utility of the model, decision curve analysis was performed across a range of clinically relevant probability thresholds (5%—50%). The model demonstrated positive net benefit compared to the “treat all” and “treat none” strategies across most thresholds, supporting its potential value in clinical decision-making for LADA screening.
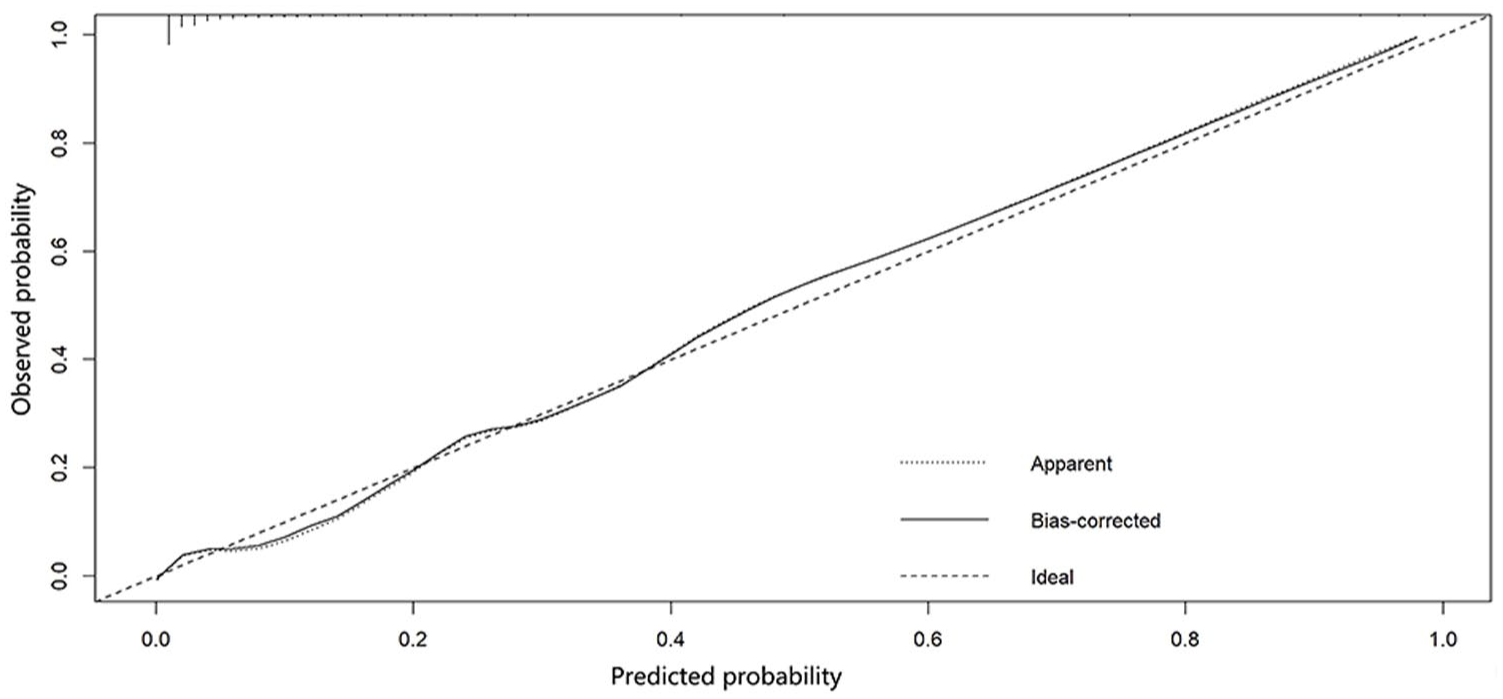
The Hosmer–Lemeshow test result (χ2 = 9.863,

Calibration curve.
Model specification and validation
The final logistic regression model equation is: logit (
The linearity assumption for continuous predictors (FPG, 2hPG, and HbA1c) was assessed using restricted cubic splines with three knots. The relationship between each continuous predictor and the log-odds of LADA was found to be approximately linear, as evidenced by nonsignificant deviation from linearity tests (
Multicollinearity among the predictors was assessed using variance inflation factors (VIFs). The VIF values were as follows: thyroid disease history = 1.08, FPG = 1.92, 2hPG = 2.15, and HbA1c = 1.84. All VIF values were below 5, indicating no concerning multicollinearity among the predictors in the final model.
Clinical implementation and risk stratification
The distribution of predicted probabilities (risk scores) from the model demonstrated clear separation between non-LADA and LADA patients, with LADA patients predominantly having higher risk scores.
For clinical implementation, we recommend a decision threshold of 0.15 for triage purposes. At this threshold, the model achieves a sensitivity of 83.1% and specificity of 87.4%, with a PPV of 45.3% and NPV of 97.8%. This threshold prioritizes sensitivity to capture the majority of true LADA cases while maintaining reasonable specificity. Patients with predicted probabilities above 0.15 should be considered for confirmatory islet autoantibody testing, especially in resource-constrained settings where universal antibody screening is not feasible.
Additionally, we propose a risk stratification system: low risk (<0.10), intermediate risk (0.10–0.30), and high risk (>0.30). In our cohort, 89.7% of LADA patients fell into the intermediate or high-risk categories, supporting the clinical utility of this stratification approach.
Missing data and sensitivity analysis
The multiple imputation analysis included all 812 initially screened patients. The results from the imputed datasets were consistent with the complete-case analysis. In the imputed data, the same four predictors (thyroid disease history, FPG, 2hPG, and HbA1c) remained statistically significant (
Discussion
The clinical manifestations of LADA partially overlap with those of T2DM, and many patients are frequently misdiagnosed as having T2DM in the early stages of the disease, leading to inappropriate treatment regimens and ultimately delaying LADA management. Therefore, early identification of LADA is of paramount importance.
Current clinical research has identified several risk factors for LADA. Wang et al. 12 demonstrated that age, ketosis, smoking history, 1-h postprandial glucose, and 2-h postprandial C-peptide concentration are risk factors for LADA development. Huang 13 reported that disease duration, hypertension, and LDL-C are risk factors for LADA with proteinuria. Li et al. 14 found that compared with T2DM patients, LADA patients exhibited lower BMI, C-peptide, and 2-h postprandial C-peptide levels, along with higher incidence rates of DKA and thyroid diseases.
This study demonstrates that thyroid disease history, FPG, 2hPG, and HbA1c constitute key risk factors for LADA. The significant association between thyroid disease history and LADA stems from their shared autoimmune pathological foundation. Both LADA and autoimmune thyroid diseases involve T lymphocyte-mediated immune dysregulation. 15 When patients have a history of thyroid disease, their immune system is already in an abnormally activated state, rendering them more susceptible to initiating autoimmune attacks against pancreatic β-cells, thereby increasing LADA risk. The emergence of glucose-related parameters (FPG, 2hPG, and HbA1c) as LADA risk factors is closely associated with the pathophysiological progression of the disease. In LADA patients, pancreatic β-cells undergo gradual autoimmune damage, resulting in progressive decline in insulin secretion. Initially, the body maintains glycemic stability through insulin resistance compensation; however, as β-cell function continues to deteriorate, this compensatory mechanism fails, leading to gradual elevation of fasting and postprandial glucose levels. HbA1c, serving as a comprehensive indicator of long-term glycemic control, reflects the sustained toxic effects of chronic glycemic fluctuations on β-cells in LADA patients. This hyperglycemic environment further exacerbates oxidative stress and inflammatory responses, establishing a vicious cycle of “hyperglycemia-islet damage” that accelerates LADA progression. From the perspective of immune-metabolic interactions, thyroid dysfunction can influence glucose metabolism through multiple pathways. Dysregulation of thyroid hormone levels alters insulin sensitivity: hypothyroidism increases hepatic glucose production and reduces peripheral tissue insulin responsiveness, while hyperthyroidism accelerates glycogenolysis and gluconeogenesis—both conditions can induce or aggravate hyperglycemia. This metabolic dysregulation interacts with autoimmune damage, potentially synergistically promoting LADA development and progression. Furthermore, hyperglycemia itself serves as an inflammatory signal that activates the immune system and intensifies immune attacks on pancreatic β-cells, further elucidating the intrinsic link between glycemic parameters and LADA risk. Additionally, the role of C-peptide as a marker of endogenous insulin secretion has been highlighted in previous studies on LADA. For instance, Wang et al. 12 and Li et al. 14 identified postprandial C-peptide levels as significant predictors, with lower C-peptide indicating reduced β-cell function in LADA patients. In our study, C-peptide data were not routinely collected in the retrospective dataset, which limited its inclusion in the current model. Incorporating C-peptide measurements in future prospective studies could potentially enhance the robustness and discriminative power of the prediction model by providing direct insight into residual β-cell function. Furthermore, recent advances in diabetes risk prediction have utilized continuous glucose monitoring (CGM) and machine learning to identify early glycemic dysregulation and immunological risk. For example, Montaser et al. 16 used a 1-week CGM home test combined with machine learning to classify autoantibody status in at-risk individuals, achieving high predictive accuracy. Another study by Montaser et al. 17 further enhanced immunological risk prediction by integrating CGM-derived glycemic features with a type 1 diabetes genetic risk score, demonstrating the potential of multimodal data integration for early risk stratification. These approaches highlight promising avenues for future LADA prediction models, potentially incorporating dynamic glycemic monitoring and genetic profiling to improve early detection and intervention.
This study also developed a predictive model based on thyroid disease history, FPG, 2hPG, and HbA1c using a multifactor binary Logistic regression model. The model demonstrates good discrimination and strong predictive capability. Notably, the performance of a predictive model must be carefully interpreted in the context of class distribution. Our cohort included a minority of LADA cases (10.58%), which is the clinically relevant group for early intervention. In such imbalanced settings, we paid close attention to the model’s sensitivity (recall, 76.6%) and specificity (91.9%). The substantial sensitivity ensures that a high proportion of true LADA patients are correctly identified, which is paramount for a screening tool aimed at preventing misdiagnosis. The concurrently high specificity minimizes false alarms, reducing unnecessary psychological burden and clinical work-up for non-LADA individuals. The strong performance on both fronts, as reflected in the high AUC (0.907), indicates that the model is effective and reliable despite the imbalanced data. The model aids physicians in more intuitively assessing the risk of LADA, thereby facilitating its early identification.
Limitations
This study has certain limitations that should be considered. First, the inclusion criterion requiring “availability of complete clinical data” may introduce selection bias, as patients with missing data might systematically differ from those with complete records. Although this approach ensured data integrity for model development, it potentially limits the representativeness of our cohort. However, our sensitivity analysis using multiple imputation demonstrated that the results were consistent between the complete-case and imputed analyses, with nearly identical effect estimates and model performance. This suggests that the impact of missing data on our primary conclusions is minimal. We assumed the data were MAR, which appears reasonable given the similar results between analytical approaches. We acknowledge that patients with incomplete data, who were excluded, might have different clinical characteristics that could affect the model’s generalizability. The model was developed and validated internally on a dataset from a single center. We acknowledge the concern regarding potential overfitting given the limited LADA cases. However, our use of LASSO regression with 10-fold cross-validation for variable selection specifically mitigates this risk. The final parsimonious 4-variable model with good calibration (
Conclusion
In summary, the LADA prediction model established based on thyroid disease history, FPG, 2hPG, and HbA1c demonstrates good predictive capability. Its robust sensitivity and specificity balance the need to capture the critical minority class (LADA) while maintaining overall diagnostic accuracy, supporting its potential for clinical application.
Supplemental Material
sj-docx-1-tae-10.1177_20420188261423784 – Supplemental material for Analysis of risk factors and establishment of a prediction model for latent autoimmune diabetes in adults
Supplemental material, sj-docx-1-tae-10.1177_20420188261423784 for Analysis of risk factors and establishment of a prediction model for latent autoimmune diabetes in adults by Haiyan Yan, Jiarong Lv, Lingling Miao and Lei Shi in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
