Abstract
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder that was previously considered a lifelong disease and requires long-term reliance on hypoglycemic medications. However, recent studies have shown that certain patients may achieve remission through various intervention strategies, which may alter the ultimate therapeutic targets for clinicians. This review aims to summarize and update the latest clinical evidence and research advancements regarding remission strategies for T2DM, primarily including intensive lifestyle interventions, metabolic surgery, short-term intensive insulin therapy, and non-insulin hypoglycemic drugs (e.g., glucagon-like peptide-1 receptor agonists and dorzagliatin) treatments, to guide the selection of appropriate treatment modalities for diverse patient populations. Additionally, the durability of sustained diabetes remission is briefly discussed.
Introduction
Type 2 diabetes mellitus (T2DM), a metabolic disorder characterized by insulin resistance and progressive β-cell dysfunction, represents a major global public health challenge as evidenced by the latest International Diabetes Federation (IDF) data, which reported that the number of diabetic patients aged 20–79 years has reached 589 million worldwide in 2025, and is expected to rise to 853 million by 2050, accounting for approximately 13% of the global population. 1 T2DM has traditionally been regarded as an irreversible and progressive condition necessitating lifelong pharmacological management. In the early 1990s, this classical perception was challenged by the identification of diabetes remission among patients with T2DM after metabolic surgery. 2 In 2009, diabetes remission was first mentioned by a consensus group that was defined as “the achievement of glycemia below the diabetic range in the absence of active pharmacologic or surgical therapy.” 3 This seminal work introduced a stratified classification system distinguishing between partial (HbA1c <6.5% maintained ⩾1 year), complete (HbA1c <5.7% with normoglycemia ⩾1 year), and prolonged remission (sustained complete remission ⩾5 years) and emphasized “remission” over “cure” terminology to acknowledge the persistent risk of disease recrudescence. 3 Subsequently, the American Diabetes Association (ADA) in 2021 refined these criteria, emphasizing that HbA1c level <6.5%, measured at least 3 months after cessation of antidiabetic agents, should be used as the diagnostic criterion for remission of type 2 diabetes. 4
While current scholarly discourse on diabetes remission has extensively documented individual therapeutic approaches, existing reviews regarding diabetes remission mainly examined specific intervention modalities, such as intensive lifestyle intervention (ILI)5,6 or metabolic surgery intervention,7,8 lacking systematic comparative analyses. The current comprehensive review synthesizes evidence across ILI, metabolic surgery, and emerging pharmacotherapies (including glucagon-like peptide-1 (GLP-1) receptor agonists (GLP-1RAs), sodium-glucose cotransporter-2 inhibitors (SGLT2is), and dorzagliatin), makes a comparison of their therapeutic effectiveness, and delves into the underlying mechanisms. Moreover, by analyzing the unique characteristics of diverse patient populations, this review facilitates clinicians to make the personalized selection of optimal remission strategies.
Strategies for diabetes remission
Lifestyle intervention
Since the Diabetes Prevention Program (DPP) was designed and implemented in 1994, 9 multiple studies have been undertaken to evaluate the effectiveness and safety of ILI in the prevention of diabetes. The American College of Lifestyle Medicine clinical practice guideline has further standardized and recommended ILI developed by a multidisciplinary panel and drawing on robust data from 8 guidelines, 118 systematic reviews, and 112 randomized trials. 10 Rooted in the 6 pillars of lifestyle medicine (nutrition, physical activity, stress management, sleep, social connectedness, and avoidance of risky substances), this framework outlines 14 key actions, including assessing baseline habits, prescribing exercise, reducing sedentary time, addressing sleep disorders, formulating nutrition plans, promoting social support, and optimizing pharmacotherapy. 10
During the last 30 years, evidence has accumulated that demonstrates that ILI, especially dietary intervention and exercise intervention, may contribute to diabetes remission11–20 (Table 1). Look AHEAD (Action for Health in Diabetes), the first randomized controlled trial (RCT) of ILI for adults with T2DM, adopted the DPP protocol,21,22 which focused on calorie restriction (1200–1800 kcal/day) and 175 min of weekly exercise. 23 Gregg et al. analyzed the results from the Look AHEAD cohort and found that 9.2%, 6.4%, and 3.5% of patients with ILI had long-term remission for at least 2, 3, and 4 years, respectively, while the proportion in the control group was less than 2%. 12 Lean et al. conducted The Diabetes Remission Clinical Trial (DiRECT) study with the primary outcome of diabetes remission and 306 patients with type 2 diabetes and a body mass index (BMI) of 27–45 kg/m2 were included in the study, participants were randomly allocated to receive a weight management plan mediated by dietary and exercise interventions and general diabetes care, and the result demonstrated that 46% participants in the intervention group achieved diabetes remission, a significantly higher rate compared to 4% in the control group (odds ratio (OR) = 19.7; 95% confidence interval (CI) 7.8–49.8). 14 Similarly, the Diabetes Intervention Accentuating Diet and Enhancing Metabolism (DIADEM-I) study recruited 147 overweight diabetic patients with BMI ⩾27 kg/m² and disease duration <3 years (average 21.2 months). 13 After randomization to either an ILI group similar to DiRECT or standard care, 61% of the intervention group achieved remission and experienced an average weight loss of 11.98 kg at 12 months. 13 Further evidence from large-scale observational studies supports these findings. Kim et al. analyzed 138,211 patients with newly diagnosed T2DM from the Korean National Health Insurance Service. 24 This study found that 6.3% achieved remission during the follow-up period to 2017, which was positively associated with the initiation of regular exercise (OR = 1.54; 95% CI 1.46–1.63). 24 Iglesies-Grau et al. found that a 12-month structured multidomain lifestyle intervention, including expert-guided education, nutrition counseling, and personalized exercise, resulted in remission in 20% of prediabetic and 12% of diabetic participants. 18
Representative studies investigating ILI-induced remission of diabetes.
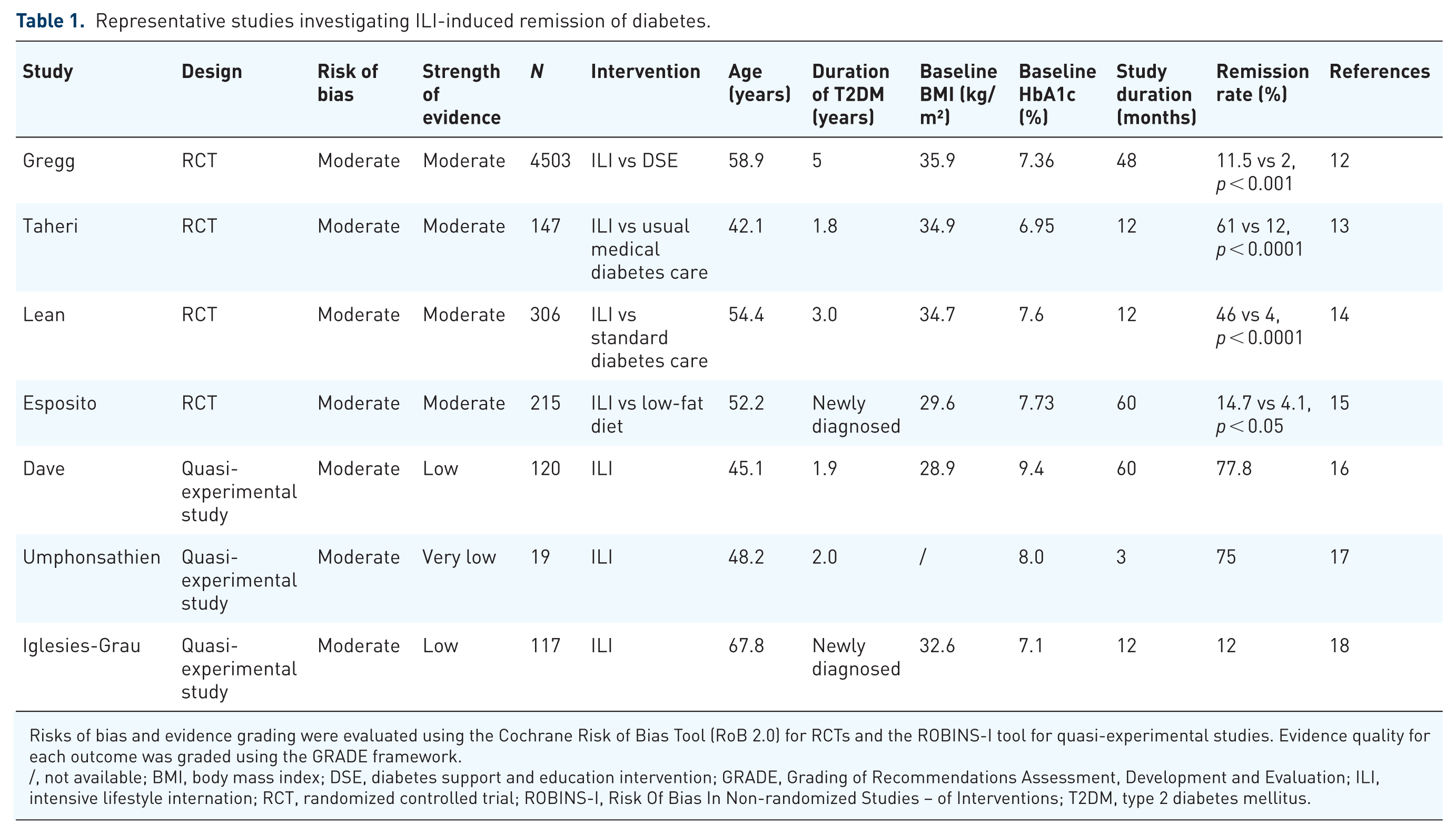
Risks of bias and evidence grading were evaluated using the Cochrane Risk of Bias Tool (RoB 2.0) for RCTs and the ROBINS-I tool for quasi-experimental studies. Evidence quality for each outcome was graded using the GRADE framework.
/, not available; BMI, body mass index; DSE, diabetes support and education intervention; GRADE, Grading of Recommendations Assessment, Development and Evaluation; ILI, intensive lifestyle internation; RCT, randomized controlled trial; ROBINS-I, Risk Of Bias In Non-randomized Studies – of Interventions; T2DM, type 2 diabetes mellitus.
Currently, the dietary intervention methods mainly include low-carbohydrate diet,25–28 ketogenic diet,29–32 Mediterranean diet,15,33,34 and intermittent fasting,35–37 which have been summarized in Table 2. Low-carbohydrate diet typically restricts a daily carbohydrate intake to less than 130 g or 26%–45% of the total energy, 26 effectively promoting rapid weight reduction and enhancing glycemic regulation, as reflected by reduced HbA1c levels. 27 However, the restrictive nature of low-carbohydrate diets may lead to micronutrient deficiencies. 28 The ketogenic diet, characterized by a macronutrient composition that is predominantly fat (70%), with moderate protein (20%) and low-carbohydrate (10%–15%) intake, 31 may confer greater short-term weight loss benefits compared to other dietary patterns. However, it can lead to early side effects, such as liver steatosis, headache, drowsiness, hair loss, and gastrointestinal reactions.31,38 In addition, long-term adherence is significantly lower than that of the carbohydrate diet pattern. 32 The Mediterranean diet, rich in plant-derived foods, including grains, vegetables, and olive oil, along with the replacement of red meat with poultry and fish, 15 has been related to more favorable outcomes in terms of reducing blood lipids and cardiovascular-related risk factors. 33 Of note, since the Mediterranean diet does not limit any particular food categories, it may not consistently lead to substantial weight reduction, and in certain instances, it may even lead to weight gain. 34 Intermittent fasting is defined as a dietary approach that alternates between phases of fasting and phases of unrestricted eating. Common intermittent fasting approaches include time-restricted eating, alternate-day fasting, and the 5:2 diet. 37 Patients generally exhibit good tolerance and compliance with intermittent fasting regimens. 35 Howbeit, the prolonged impacts of intermittent fasting on weight management are not well-established, and it may potentially increase the risk of developing eating disorders and associated complications. 36
Advantages and disadvantages of different dietary types.

Of note, ILI, particularly dietary interventions, are susceptible to sociocultural influences. Culturally tailored ILI can improve intervention adherence. 39 A qualitative study of the DiRECT intervention among Māori and Pacific populations in New Zealand showed that cultural norms around food, such as food-sharing, hospitality etiquette, and waste-aversion, posed challenges. 40 In addition, diabetes increases the risk of mental health issues such as depression and anxiety, often stemming from the burden of long-term dietary interventions and self-management. 41 A 2024 global survey by the IDF showed that 77% of people with diabetes had experienced anxiety, depression, or other mental health issues. 42 These mental health comorbidities reduce treatment adherence and thereby hinder diabetes remission. 43 Thus, the implementation of the ILI strategy for T2DM remission requires collaborative support from multidisciplinary professionals, including not only endocrinologists and dietitians but also psychological counselors.44,45 Furthermore, the multidisciplinary team, together with family support, ensures the sustainability of lifestyle modifications through regular follow-up, which is crucial for maintaining T2DM remission. 46
The American College of Sports Medicine (ACSM) and the ADA have recommended that exercise is a fundamental component of diabetes management.47,48 According to the guidelines of the ACSM and the ADA, the recommended types of exercise intervention for individuals with diabetes generally include aerobic exercise training, resistance exercise training, and joint training. 49 Numerous clinical trials and population studies have shown that physical exercise intervention could also contribute to the prevention and treatment of T2DM.50,51 A meta-analysis in 2006, including 18 RCTs and 1 randomized crossover trial, demonstrated that all exercise modalities effectively improved glycemic control parameters (e.g., HbA1c, insulin sensitivity, and fasting insulin), with potential additive benefits from combined exercise. 50 Another meta-analysis which included 47 RCTs with 8538 patients reported significant HbA1c reductions (mean difference −0.67%; 95% CI −0.84% to −0.49%) with aerobic exercise, resistance training or combined exercise training. 51 According to the ACSM and the ADA, patients with T2DM should receive joint training three times a week and at least 150 min of moderate-intensity or higher-intensity aerobic exercise (about 40%–60% of the maximum oxygen uptake) every week. 49 Physical exercise, when combined with dietary intervention, serves a crucial function in promoting the remission of T2DM as part of lifestyle intervention. However, there is a lack of clinical evidence to definitively assess the correlation between exercise intervention and diabetes remission.
Weight loss achieved through ILI has been demonstrated to be associated with diabetes remission. According to the results from DiRECT, 24% of the subjects undergoing ILI achieved a weight loss of 15 kg or more. 14 Furthermore, this trial proved that the duration and rate of diabetes remission were dose-dependently associated with the degree of weight loss. 14 The DiRECT trial found that the rate of complete remission for T2DM was 57.1% among individuals who attained a weight loss of 10–15 kg, while it increased to 86% for those who lost more than 15 kg. 14 These findings were corroborated by a large-scale population-based study involving 162,316 diabetic patients, which also reported the highest remission rates among those with ⩾15 kg weight loss in any age group. 20 A comment citing Kanbour et al.’s systematic review (22 RCTs, >12,000 participants) further supported this dose-dependent relationship, noting that complete remission rates at 1 year reached 0.7% with <10% weight loss, 49.6% with 20%–29% loss, and 79.1% with ⩾30% loss, aligning with DiRECT findings.52,53
Pathophysiological studies have demonstrated that the reduction of visceral fat resulting from weight loss may be the primary mechanism contributing to diabetes remission.54–56 Excessive fat deposition in organs and tissues, such as the liver and pancreas, can increase insulin resistance and hinder the β-cell redifferentiation in the body.57,58 The pathogenesis by which visceral adiposity induces insulin resistance is relatively multifactorial. A key underlying mechanism involves the release of adipocyte-derived mediators (e.g., ceramide, diacylglycerols, and acylcarnitines) leading to impaired transduction within the insulin signaling cascade. 59 Notably, chronic low-grade inflammation triggered by visceral adiposity also serves as a critical mechanism contributing to the development of insulin resistance. Lumeng et al. demonstrated that high-fat diet-induced obesity promoted a shift in adipose tissue macrophages from the anti-inflammatory M2 phenotype to the proinflammatory M1 state in obese mouse models. 60 Various proinflammatory mediators, such as tumor necrosis factor (TNF) and interleukin-1β (IL-1β), released by M1 macrophages inhibited the phosphorylation of insulin receptors and downstream signaling molecules, resulting in impaired intracellular insulin signal transduction. 61 According to the double circulation hypothesis put forward by Taylor et al., 57 negative energy balance induced by lifestyle interventions first leads to the reduction of fat accumulation in the liver, and further increases hepatocyte insulin sensitivity. In addition, decreased output of triglycerides to other body compartments, consequently reduces fat deposition in other organs, particularly islets and muscle tissues, and then alleviates systemic insulin resistance.
Fat deposition in the islets also leads to the dedifferentiation of β cells. In 2012, Talchai et al. demonstrated for the first time that β-cell dedifferentiation served as a critical factor in the pathogenesis of diabetes. 62 Furthermore, it was observed that, in the diabetic mouse model, calorie restriction-induced redifferentiation of dedifferentiated β cells, thus restoring the expression of key transcription factors (e.g., Nk6 homeobox 1 (Nkx6.1), pancreatic and duodenal homeobox 1 (Pdx-1)) essential for β-cell maturation. 63
In addition, dietary intervention may also promote the remission of T2DM by modulating the composition of the intestinal microbiome.
64
Several clinical studies have demonstrated that dietary interventions can alter the diversity and abundance of oral and gut microbiota, which subsequently modulate the impacts on blood glucose concentrations and metabolic processes.65–67 Furthermore, a study involving 183 newly diagnosed individuals with T2DM found that the intestinal microbiome composition varied between the remission and non-remission groups following lifestyle intervention.
68
Specifically, the remission group had a higher abundance of the
Similarly, a clinical study included 39 overweight prediabetic men under a 12-week exercise intervention, which indicated that the relative abundance of intestinal microbial species changed significantly after the exercise intervention. 67 Notably, a significant decrease in Prevotella, a contributor to insulin resistance, and an increase in the Streptococcus group occurred only in responders. 67 However, the researchers did not establish whether the observed differences in the gut microbiome composition were directly attributable to the dietary intervention (Figure 1).

Mechanism of lifestyle intervention in alleviating T2DM. The mechanism mentioned in the figure does not mean to cover all relevant factors, but rather to describe the broad and complex mechanisms involved.
Metabolic surgery
Metabolic surgery was first introduced as a potential therapeutic approach for improving or alleviating T2DM in 1955,70,71 which was recommended by ADA in 2009 among individuals with T2DM and BMI of 35 kg/m² or higher.
72
Compared with nonsurgical strategies, metabolic surgery is recognized as more efficient treatment for facilitating weight reduction and diabetes remission (Table 3).73–84 A systematic review and meta-analysis encompassing 11 studies with 796 diabetic patients quantitatively synthesized the efficacy differences between metabolic surgery and nonsurgical treatments.
85
Compared with nonsurgical treatments, including ILI
40
and medical therapy,75,78–80 the metabolic surgery group achieved a significantly higher diabetes remission rate (risk ratio (RR) = 22.1; 95% CI 3.2–154.3,
Representative studies investigating bariatric surgery-induced remission of diabetes.

Risks of bias and evidence grading were evaluated using the Cochrane Risk of Bias Tool (RoB 2.0) for RCTs and the ROBINS-I tool for quasi-experimental studies. Evidence quality for each outcome was graded using the GRADE framework.
The study failed to provide detailed information.
/, Not available; AGB, adjustable gastric banding; BMI, body mass index; BPD, biliopancreatic diversion; GLP-1RAs, GLP-1 receptor agonists; GRADE, Grading of Recommendations Assessment, Development and Evaluation; ILI, intensive lifestyle intervention; RCT, randomized controlled trial; ROBINS-I, Risk Of Bias In Non-randomized Studies – of Interventions; RYGB, Roux-en-Y gastric bypass; SG, sleeve gastrectomy; T2DM, type 2 diabetes mellitus.
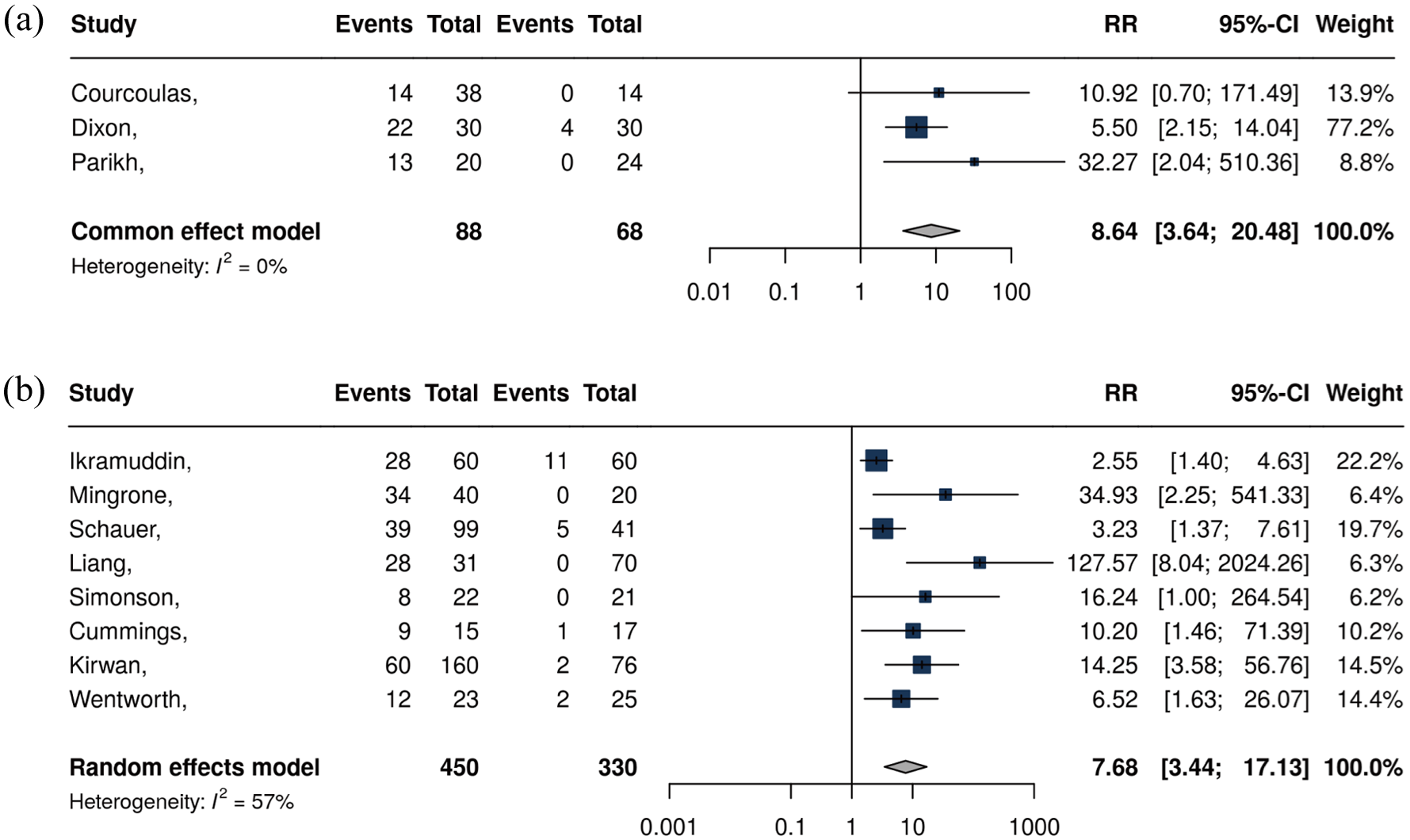
Diabetes remission rates with metabolic surgery versus nonsurgical controls. Comparisons are shown against structured ILI in (a) and medical therapy in (b).
Metabolic surgery primarily consists of four main procedures: Roux-en-Y gastric bypass (RYGB), adjustable gastric banding (AGB), sleeve gastrectomy (SG), and biliopancreatic diversion with duodenal switch, 87 among which RYGB and vertical sleeve gastrectomy (VSG) are the most widely conducted surgical techniques. 73 RYGB surgery involves creating a small gastric pouch from the upper stomach, which is then directly connected to a segment of the small intestine, bypassing the majority of the stomach and the duodenum. 88 This procedure exerts a dual effect, involving both restriction of food intake and malabsorption, via reconstructing the digestive tract and bypassing a significant section of the stomach and proximal small intestine. 88 VSG procedure involves the resection of a significant portion of the stomach, typically comprising approximately 75%–80% of its total volume, and the remaining stomach is reshaped into a tubular or sleeve-like structure, resembling a narrow and elongated pouch. 79
Several studies have demonstrated that RYGB is more effective than VSG in terms of weight loss and diabetes remission outcomes.89–92 A large observational cohort study conducted in Sweden found that people who received RYGB lost more weight on average (27% vs 18%) when compared with VSG. 89 Consistently, a systematic review that analyzed 15 RCTs with a total of 1191 patients, followed for a duration ranging from 6 months to 3 years, reported a significantly higher diabetes remission rate for RYGB (67.5%) than for VSG (51%). 90 Similar findings were reported from a meta-analysis that included 10 RCTs with a total of 705 patients (the rate of diabetes remission following 1-year of follow-up period: RYGB 57% vs VSG 47%). 91 However, no statistically significant difference in the long-term sustained remission rates between the two surgical interventions. 91
Despite its reliable and long-lasting effects on glucose control and diabetes remission, metabolic surgery is a complex procedure associated with a relatively high risk of perioperative complications, including postoperative bleeding, anastomotic leak, malnutrition, and even death. 93 In addition, bariatric surgery may cause early worsening of diabetic retinopathy, emphasizing the importance of preoperative retinal evaluation and close postoperative monitoring. 94
As a result, metabolic surgery was initially reserved for individuals with a BMI over 40 kg/m2 or those with a BMI over 35 kg/m2 and obesity-related complications. 95 However, this view has evolved as increasing evidence has verified its benefits of diabetes remission and the enhanced safety profile with the emergence of laparoscopic surgery. 96 A prospective multicenter observational study involving 4776 subjects undergoing metabolic surgery for the first time found that the mortality rate for laparoscopic minimally invasive surgery was 10 times lower than that for open surgery using the same procedure. 97 Aminian et al. analyzed data from T2DM patients who accepted laparoscopic RYGB or one of seven other surgical procedures as recorded in the American College of Surgeons database and found that metabolic surgery was relatively safe, with a perioperative mortality rate comparable to that of knee replacement surgery. 98
Another major issue with bariatric surgery is its high medical cost. 99 Compared with low-cost dietary and lifestyle modifications, bariatric surgery is regarded as a high-cost option, with socioeconomic factors being a major barrier to its promotion. 99 A systematic review and meta-analysis of 9 studies, encompassing over 64 million patients, revealed significant socioeconomic disparities (e.g., race, income, education level, and insurance type) in access to bariatric surgery. Among medically eligible patients, those who undergo bariatric surgery were predominantly white, of higher income, and privately insured. 100 Furthermore, a study conducted in Taiwan found that during the first year of follow-up, the bariatric surgery group incurred the highest medical expenditures at $3494 per person. This amount exceeded the costs observed in the metabolic syndrome group, the high BMI with comorbidities group, and the simple high BMI group. 101 However, the bariatric surgery group showed reduced comorbidities and treatment requirements, with its medical expenditures dropping to the lowest level among all groups during the 1-year follow-up after metabolic surgery. 101 Thus, while bariatric surgery entails substantial initial medical costs, it leads to dramatic health improvements and a significant decrease in future healthcare expenditures.
The mechanisms by which metabolic surgery induces diabetes remission are multifaceted. First, the loss of appetite and reduced gastric volume following metabolic surgery lead to caloric restriction, thereby inducing weight loss and diabetes remission. The loss of appetite is mainly due to the increase of anorexic hormones, such as oxyntomodulin (OXM) and peptide YY (PYY), and the decrease of food-stimulating hormone secreted by the gastrointestinal tract (e.g., Gastrin). 7
In addition to caloric restriction, this strategy can also achieve diabetes remission through other mechanisms.102,103 The foregut hypothesis, based on animal experiments conducted by Rubino et al., suggests that RYGB surgery bypasses the proximal part of the digestive tract, resulting in the elimination of the stimulation of food on the stomach and duodenum and the reduction of hormone secretion, including glucose-dependent insulinotropic polypeptide (GIP). 104 Ectopic fat accumulation is a key driver of insulin resistance. By promoting lipid storage in adipose tissue, GIP diverts lipids away from ectopic deposition and attenuates lipotoxicity in insulin-sensitive tissues such as the liver and skeletal muscle. 105 Corresponding to the foregut hypothesis, the hindgut hypothesis posits that food rapidly reaches the distal intestine after RYGB surgery, leading to increased secretion of hormones, including GLP-1. 106 Nannipieri et al found that the recovery of GLP-1 response after RYGB and laparoscopic sleeve gastrectomy (LSG) was a major determinant of diabetes remission. 107
There are also other gut hormones warranting discussion, including PYY and OXM. Similar to GLP-1, PYY is co-secreted by intestinal L cells.
108
Research by le Roux et al. demonstrated that elevated PYY levels following RYGB enhanced satiety by inhibiting the activity of neuropeptide Y (NPY)-expressing neurons and potentially influence gastrointestinal transit.
109
Laferrère et al. reported that OXM levels significantly increased by more than twofold after RYGB, with peak OXM positively correlated with peak GLP-1 (partial
The increase in bile acid levels in postoperative patients may also play an important role in diabetes remission. A clinical cohort study conducted by Gerhard et al. in 2013 has shown that patients who underwent RYGB experienced an increase in bile acid levels, and individuals who achieved remission of T2DM also exhibited elevated levels of bile acids than non-remission patients. 113 Bile acids can regulate metabolism through the Farnesoid X receptor (FXR) and the Takeda G-protein receptor 5 (TGR5). 114 Animal experiments have shown that mice lacking the FXR gene and TGR5 did not experience improvement in glucose metabolism after undergoing SG, while wild-type mice exhibited substantial enhancements in both weight and blood glucose levels.115,116 In addition, bile acids can also promote the secretion of GLP-1 from intestinal L cells, thereby promoting the alleviation of T2DM. 117
Furthermore, metabolic surgery may influence the composition and quantity of the intestinal microbiome. Arora et al. found that RYGB can change the diabetes-related microbiota in rats.
118
Consistently, several clinical trials have also confirmed significant changes in the gut microflora diversity after metabolic surgery.119–122 Graessler et al. sequenced the fecal samples of patients 3 months after RYGB surgery and found a notable increase in the number of
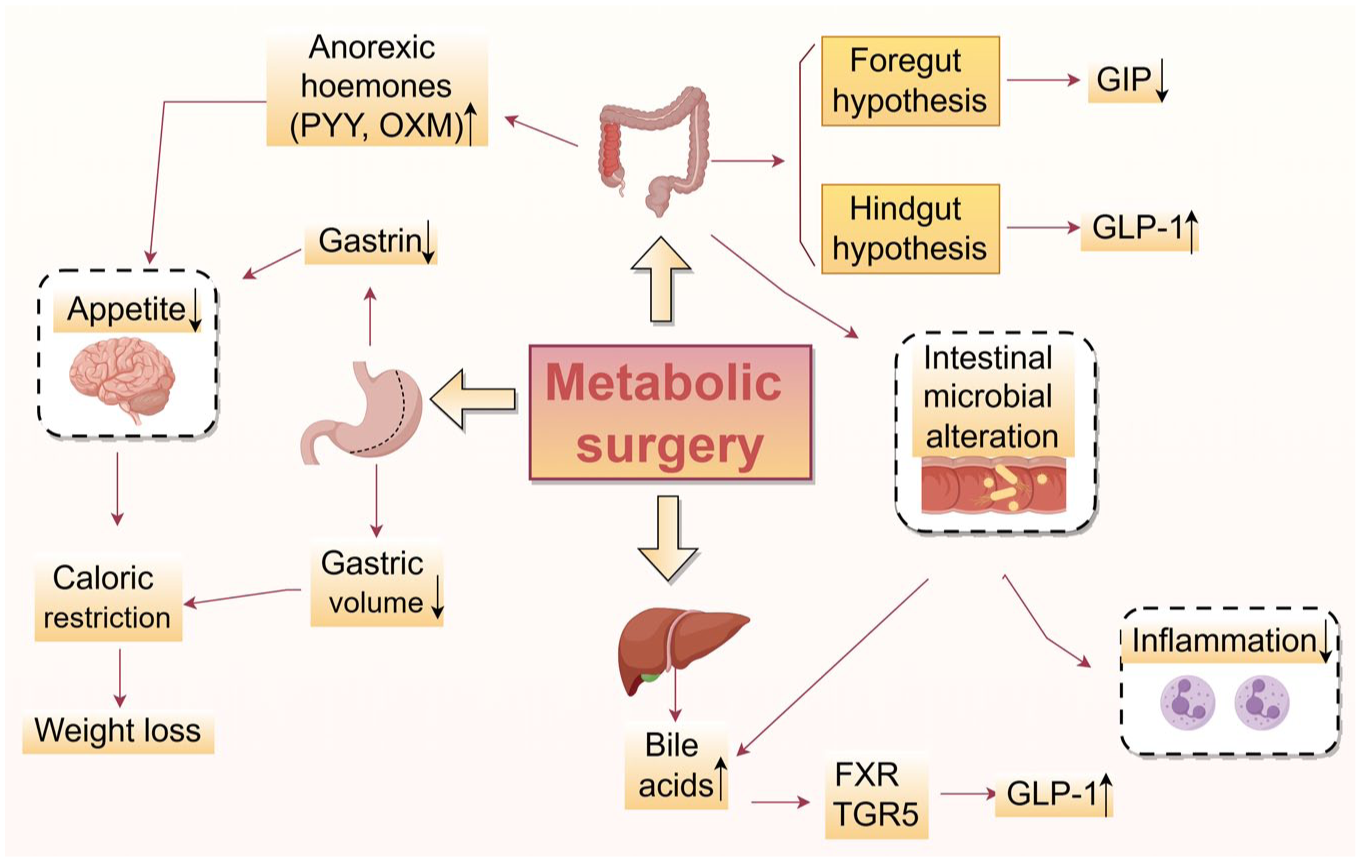
Mechanism of metabolic surgery in alleviating diabetes mellitus. The mechanism mentioned in the figure does not mean to cover all relevant factors, but rather to describe the broad and complex mechanisms involved.
Drug intervention
Short-term intensive insulin therapy
Extensive clinical studies support the efficacy of short-term intensive insulin therapy (SIIT) in inducing remission of diabetes among patients in the early stage of T2DM126–131 (Table 4). A multicenter RCT involving 382 patients with newly diagnosed T2DM demonstrated that, following less than 3-week SIIT, the diabetes remission rate of patients after 1 year was 51.1% in continuous subcutaneous insulin infusion (CSII) group and 44.9% in the multiple daily insulin injections group.
126
Another study from Korea found that after 53.6 ± 38.9 days of CSII for intensive treatment, 34.4% of patients experienced diabetes remission, with a remission duration of 13.6 ± 8.9 months.
127
A meta-analysis involving 7 studies and 839 participants indicated that 2–3 weeks of intensive insulin treatment led to remission rates of 66.2% at 3 months, 46.3% at 12 months, and 42.1% at 24 months.
132
In addition, several studies verified the improvement of β-cell function and insulin sensitivity after SIIT.128,133,134 In a multicenter study including 124 individuals newly diagnosed with T2DM, insulin sensitivity was assessed using a clamp test before and after a 2-week SIIT. The researchers found that first-phase insulin secretion improved from −6.72 to 14.26 μU/min/mL, and insulin sensitivity increased by 2.43 mg/kg/min (
Representative studies investigating short-term intensive insulin therapy-induced remission of diabetes.

Risks of bias and evidence grading were evaluated using the Cochrane Risk of Bias Tool (RoB 2.0) for RCTs and the ROBINS-I tool for quasi-experimental studies. Evidence quality for each outcome was graded using the GRADE framework.
BMI, body mass index; CSII, continuous subcutaneous intensive insulin infusion; GRADE, Grading of Recommendations Assessment, Development and Evaluation; IIT, intensive insulin therapies; MDI, multiple daily insulin injections; OAD, oral antidiabetic drug; RCT, randomized controlled trial; ROBINS-I, Risk Of Bias In Non-randomized Studies – of Interventions; T2DM, type 2 diabetes mellitus.
The therapeutic effects of SIIT primarily stem from two key mechanisms. First, SIIT alleviates T2DM by eliminating glucotoxicity-related damage to β cells. 135 It is well known that glucotoxicity damages β cells through a multi-hit process involving oxidative stress, endoplasmic reticulum stress, formation of advanced glycation end-products (AGEs), and altered gene expression encoding insulin itself and other key transcription factors, such as Pdx-1 and V-maf musculoaponeurotic fibrosarcoma oncogene homolog A (MafA), leading to the progressive decline in insulin secretion and ultimately apoptosis.135,136 Thus, SIIT mitigates glucotoxicity, thereby attenuating the aforementioned detrimental processes in β cells and ultimately preserving their function.
Another major mechanism is the redifferentiation of dedifferentiated β cells, which lose the ability to secrete insulin. 62 After relieving the toxicity of high glucose, dedifferentiated islets β cells can differentiate again and improve secretion function.137–139 In a diabetic mouse model, Wang et al. found that the quantity of insulin-positive β cells in insulin-treated diabetic mice could be almost completely restored compared to untreated mice 139 (Figure 4). These findings highlight the critical role of SIIT in reversing the pathophysiological processes underlying T2DM, making it a valuable intervention for early disease management.
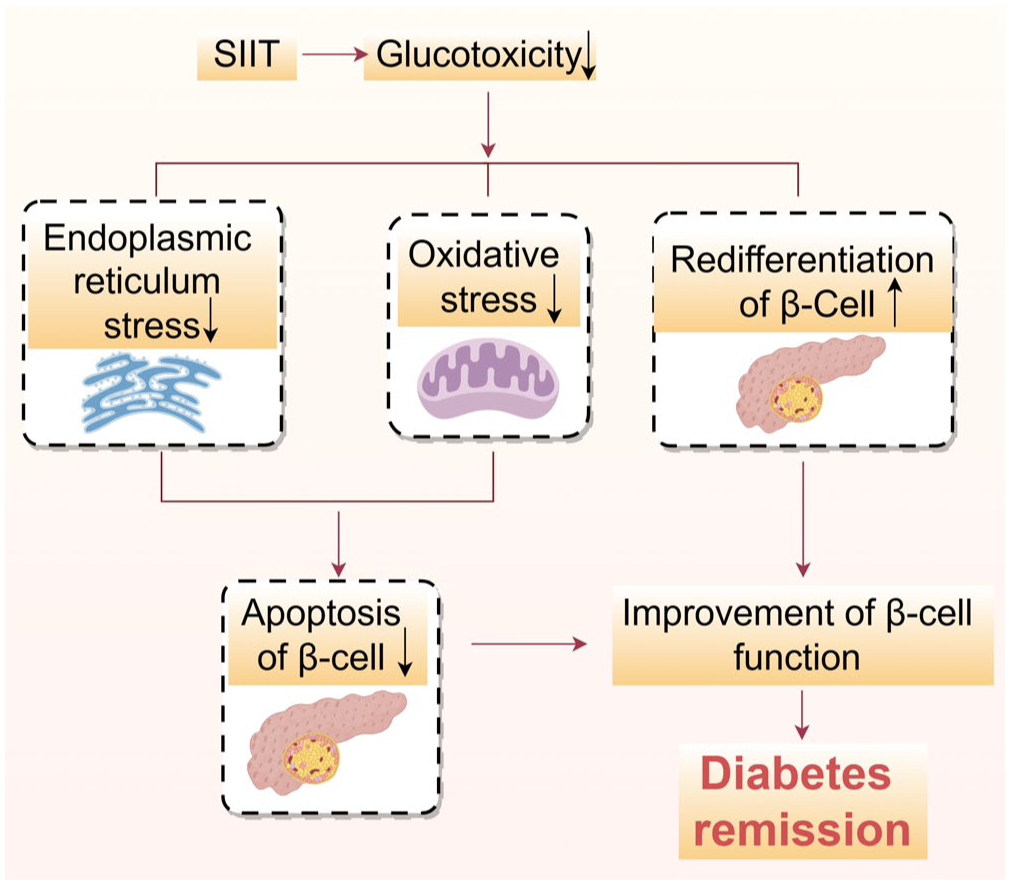
Mechanism of SIIT in alleviating diabetes mellitus. The mechanism mentioned in the figure does not mean to cover all relevant factors, but rather to describe the broad and complex mechanisms involved.
GLP-1 receptor agonists
GLP-1 is a peptide hormone secreted by the intestines that can inhibit gastric emptying, reduce appetite, and promote insulin secretion.
140
GLP-1RAs are a novel category of hypoglycemic medications that emulate the action of the hormone GLP-1, with a low risk of hypoglycemia and high efficacy in weight loss.
141
Currently, there are only limited clinical studies examining the effect of GLP-1RAs on diabetes remission.
142
A RCT included 103 T2DM patients aged 30–80 years with a disease course of less than 7 years, investigating the effect of glargine combined with exenatide on diabetes remission.
142
The results indicated the remission rate was 26.7%, which showed no statistical difference when compared with the intervention of glargine plus lispro (
The clinical implementation of GLP-1RAs is somewhat constrained by economic and practical barriers, especially in resource-limited settings. A cost-effectiveness analysis within the Chinese healthcare system concluded that GLP-1RAs for obesity management were not cost-effective compared to lifestyle intervention at current pricing. Only semaglutide demonstrated potential cost-effectiveness in patients with severe obesity and certain high-income subgroups. 146 Even in high-income countries, the budgetary impact remains substantial; projected coverage under US Medicare Part D (the prescription drug program for American seniors) would result in a net expenditure increase of $47.7 billion over 10 years. 147 The high cost of GLP-1RAs, along with significant out-of-pocket expenses, further restricts their use, particularly among socioeconomically disadvantaged populations. 148 Therefore, multifaceted strategies, including price reduction policies, improved patient support programs, and better prioritization of high-risk groups, are essential to enhance the affordability and accessibility of these therapies, especially in resource-limited settings.
Although the body of clinical trials regarding the effects of GLP-1RAs on diabetes remission is not yet robust, their diverse biological actions indicate that GLP-1RAs hold significant potential for promoting remission. One of the key mechanisms is weight loss. 141 A meta-analysis including 76 RCTs with GLP-1RAs and 39,246 patients in 2024 summarized that GLP-1RAs can significantly reduce body weight. 149 Among them, CagriSema (semaglutide and cagrilintide) led to a mean weight reduction of 14.03 kg, followed by tirzepatide, inducing a mean weight reduction of 8.47 kg. According to the DiRECT study mentioned above, a weight loss of 5–10 kg can achieve a remission rate of 34%. 14 Therefore, it is reasonable to assume that the weight loss induced by GLP-1RAs may play a substantial role in facilitating diabetes remission.
Furthermore, substantial experimental studies150–159 and clinical research evidence160,161 have proved that GLP-1RA can enhance β-cell function by directly stimulating the proliferation of pancreatic β cells, attenuating β-cell apoptosis, and improving glucose-stimulated insulin secretion (GSIS). These actions are primarily attributed to the modulation of signaling pathways associated with apoptosis (e.g., p27, Bcl-2-associated X protein (BAX), Cidea, cyclin A2), key components governing GSIS such as transient receptor potential melastatin 5 (TRPM5) and receptor activity-modifying protein 3 (RAMP3), as well as transcription factors critical to β-cell function, including Pdx-1, MafA, and neurogenic differentiation factor (NeuroD).150–159 Similar findings were reported in some clinical trials. For example, an RCT randomly assigned 69 metformin-treated T2DM patients to receive either insulin glargine or exenatide.
160
After 52 weeks of treatment, exenatide treatment increased C-peptide secretion in the first and second stages by 1.5 and 2.8 times, respectively, compared with insulin therapy.
160
Another double-blind RCT of 51 patients with early T2DM showed that, following 4 weeks of SIIT to eliminate glucotoxicity, daily subcutaneous liraglutide sustained enhanced β-cell function (assessed by Insulin Secretion-Sensitivity Index-2) over 48 weeks (339.8 ± 27.8 vs 229.3 ± 28.4,
Interestingly, GLP-1RAs have been verified to have the potential to ameliorate inflammation in pancreatic islet cells. 162 In a nonobese diabetic mouse model, Hadjiyanni et al. found that after intervention with GLP-1RA exendin-4, the proportion of insulin-positive cells was significantly increased and the infiltration of pancreatic islet inflammatory cells was significantly reduced. 163 In addition, our previous research has shown that exenatide can ameliorate pancreatic islet β-cell dysfunction by restoring the balance of Th17/Treg cells. 164 Furthermore, an in vitro study identified that GLP-1RA exendin-4 can modulate immune responses, inhibiting the pro-apoptotic effect of IL-1β by suppressing the c-Jun NH2-terminal kinase (JNK) pathway 165 (Figure 5). To sum up, GLP-1RAs may exert beneficial effects on diabetes remission through multiple mechanisms at both cellular and molecular levels.
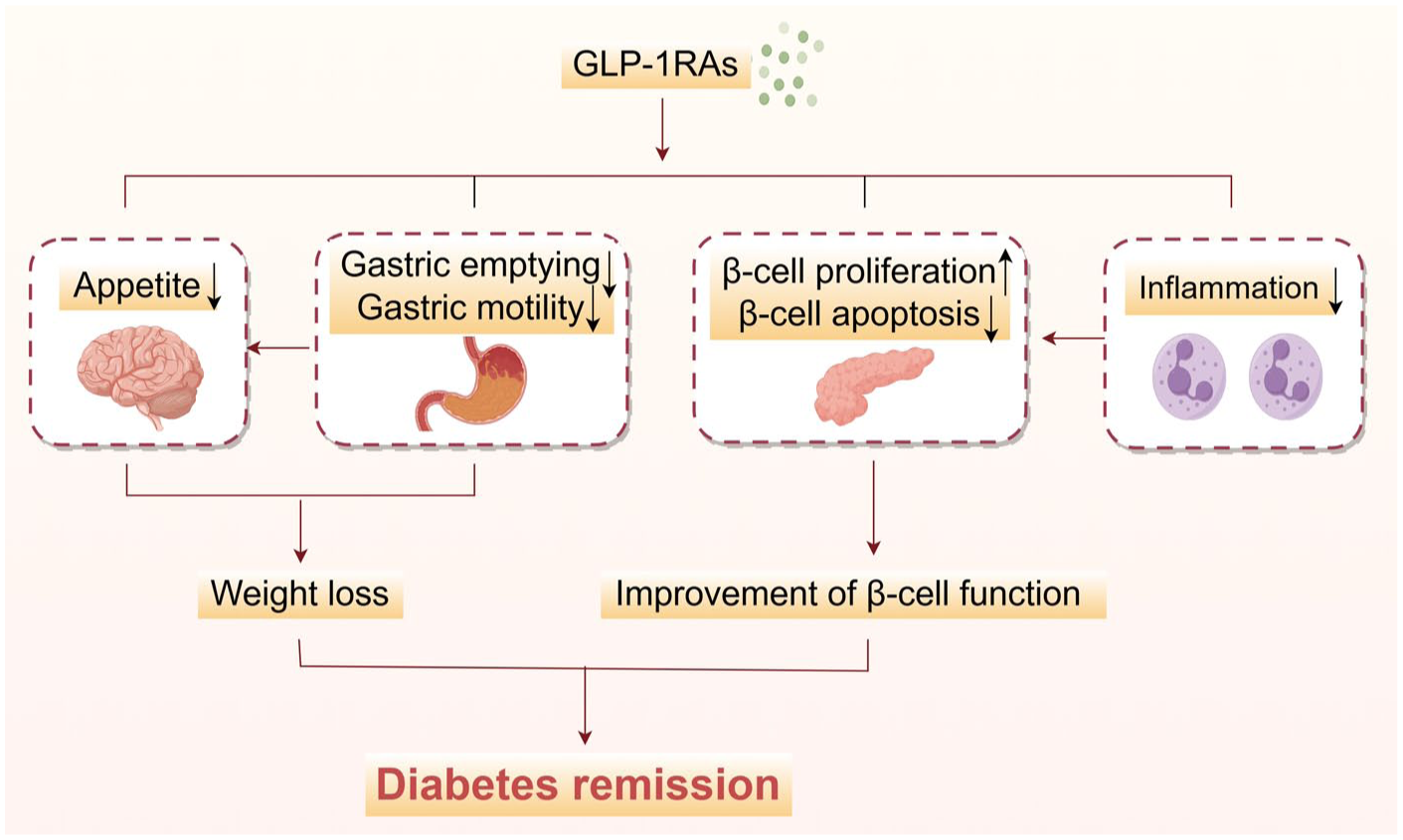
Mechanism of GLP-1RAs in alleviating diabetes mellitus. The mechanism mentioned in the figure does not mean to cover all relevant factors, but rather to describe the broad and complex mechanisms involved.
Others
SGLT2, a secondary active transporter located in the proximal renal tubules, mediates approximately 90% of renal glucose reabsorption.
166
SGLT2is exert hypoglycemic effects by reducing glucose reabsorption in the renal tubules and promoting urinary glucose excretion, thereby facilitating weight loss through additional caloric loss.
167
A recently conducted multicenter randomized placebo-controlled trial involving 328 participants with T2DM assessed the effects of SGLT2is on diabetes remission.
168
The study demonstrated that the combination of dapagliflozin and calorie restriction significantly enhanced the remission rates of T2DM in overweight and obese patients (44% vs 28% with calorie restriction alone,
In addition, significant advancements have resulted in the creation of new hypoglycemic drugs targeting various mechanisms, with glucokinase emerging as a significant target. As the initial enzyme in the glycogen synthesis pathway, glucokinase plays a pivotal role in regulating the secretion of GLP-1 and insulin, as well as glycogen synthesis and degradation in response to fluctuations in blood glucose concentrations.169,170 Clinical studies have demonstrated that dorzagliatin, a glucokinase activator (GKA), can significantly increase HOMA-β in patients with T2DM, indicating its ability to improve insulin secretion. 171 Furthermore, a prospective cohort study conducted in 2023 involved 69 patients who achieved glycemic stability (defined as HbA1c < 7.0% or 7.0% ⩽ HbA1c < 8.0%) under dorzagliatin intervention. 172 Notably, after discontinuation of dorzagliatin, the remission rate was 65.2% within a 52-week observation period, 172 indicating its potential efficacy in alleviating T2DM, although more clinical evidence is required.
Furthermore, evidences regarding direct comparisons between ILI and pharmacologic interventions for diabetes remission remain limited, which may be attributed to substantial heterogeneity of the existing studies in patient baseline characteristics (e.g., disease duration, BMI, and comorbidities), intervention intensity (e.g., lifestyle intervention duration, pharmacologic dosage regimens), and outcome definitions (e.g., HbA1c thresholds for “remission”). Future research should prioritize well-designed, homogeneous RCTs with standardized baseline criteria and uniform outcome definitions to enable direct comparisons of the short-term efficacy, long-term durability, and cost-effectiveness of these strategies.
Patient characteristics of diabetes remission
Identifying the population characteristics associated with diabetes remission is crucial for predicting the probability of remission and guiding the selection of individualized therapeutic strategies in routine clinical practice. To date, there are a multitude of studies investigating the patient characteristics correlated with diabetes remission in both trial settings and normal care.12,13,20,128,173–180 According to a cross-sectional population-based study conducted in Scotland in 2019 including 162,316 people with a baseline HbA1c ⩾6.5%, population characteristics related to remission included age ⩾65 years, HbA1c <6.5% at diagnosis, weight loss since the diagnosis of diabetes, no history of glucose-lowering therapy, and a previous history of metabolic surgery. 20 Another cohort study by Karter et al. included 122,781 patients with T2DM who were not treated with metabolic surgery, and it indicated similar factors that were associated with diabetes remission, including lower baseline HbA1c levels, age >65 years, and no history of hypoglycemic drugs. 173 We also noticed the differences between the two research conclusions. The Scotland study additionally found a positive correlation between the possibility of diabetes remission and the extent of weight loss, 20 while findings from Karter and colleagues did not. Additionally, Karter et al. found that diabetes duration also exhibited a relationship with remission, and the duration <2 years was significantly correlated with a higher remission rate. 173 Two other population-based studies from Fujihara and Holman concluded similar results,174,175 which reported that a greater extent of weight loss was associated with a higher remission rate. The more than 10% reduction in BMI was correlated with a higher possibility of remission (OR = 3.57; 95% CI 3.42–3.72) when compared with a 5%–9.9% reduction in BMI (OR = 1.78; 95% CI 1.71–1.85). 175
Furthermore, the influence of sex on diabetes remission is also of considerable significance, yet neither the Scotland study 20 nor the research by Karter et al. 173 analyzed this potential association. Meanwhile, the more recent studies by Fujihara and Holman yielded inconsistent findings regarding the association between sex and diabetes remission. Specifically, Fujihara et al. reported a higher remission rate among male participants, 174 whereas Holman et al. observed the inverse trend, with females presenting a greater likelihood to achieve remission. 175 These discrepancies highlight the need for further investigation into the role of sex as a biological variable in diabetes outcomes. To date, the mechanisms through which sex influences remission remain poorly understood, underscoring the importance of sex-stratified analyses in future research to further optimize individualized treatment strategies.
Several RCTs have yielded findings regarding remission-related characteristics that are consistent with those observed in population-based studies.12,13,126,128,132,176–181 However, some paradoxical findings also emerge when comparing real-world evidence with controlled trials. One difference is in the age. RCTs generally found that younger age was associated with higher clinical remission rates, which is contrary to the conclusions of population-based studies. For example, Gregg et al. found that patients aged 44–56 exhibited a higher remission rate than those older than 56 (7.5% vs 6.5%). 12 It is speculated that the divergence between population-based studies and RCTs may be partially attributed to the study design and inclusion criteria. The age of patients included in RCTs typically ranged between 40 and 60 years. In contrast, population-based studies encompass broader demographics, representing a diverse age range.
In addition, different intervention strategies in RCTs exhibit distinct predictive factors for diabetes remission. In the study on ILI to alleviate T2DM, patients with shorter disease duration, lower baseline HbA1c levels, greater weight loss, no prior insulin therapy, and fewer hypoglycemic agents were more likely to achieve remission.12,13,180 The DiRECT trial has shown that weight loss is the strongest predictor of remission after ILI.
180
For each additional kilogram lost, there is a 24% increase in the 1-year remission rate (
Although these intervention strategies share certain commonalities in their remission predictors, their target populations and core advantages differ significantly. ILI serves as the fundamental strategy for T2DM remission, mainly through achieving sufficient weight loss in patients with obesity. 182 However, it has limited efficacy in those with long disease duration, severely impaired islet function, or complex comorbidities. 183 Metabolic surgery can rapidly achieve significant weight loss and improve complications, primarily indicated for patients with severe obesity (BMI >35 kg/m²) and/or complicated with severe metabolic syndrome. 184 A Meta-analysis of 11 RCTs showed that compared with nonsurgical treatments, patients undergoing metabolic surgery achieved an average weight loss of 26 kg at 2 years postoperatively (95% CI 21–31 kg). 85 However, in patients with BMI <27.5 kg/m², the risk of surgical complications increased, while the remission rate remained relatively low at approximately 35%, where risks may outweigh the benefits. 185 SIIT has been less extensively studied in weight-stratified analyses, as weight loss may not be a strong predictor of remission success. This strategy appears most appropriate for newly diagnosed patients. A multicenter RCT, Remission Studies Evaluating Type 2 Diabetes-Intermittent Insulin Therapy Main (RESET-IT Main) study, demonstrated that SIIT can only improve pancreatic β-cell function primarily in patients with a disease duration of less than 2.5 years, whereas showing limited efficacy in those with longer disease duration due to reduced reversible β-cell capacity. 186
According to the characteristics associated with diabetes remission, the selection of diabetes remission strategies could be guided in clinical practice (Figure 6). A fasting C-peptide level ⩾1.1 μg/L indicates preserved pancreatic β-cell function with potential for reversal. 14 Diabetes duration is also a critical prerequisite of remission. However, although the majority of studies consistently suggest that a shorter duration predicts higher remission likelihood, the specific cutoff values remain inconsistent. Nonetheless, a duration of under 2–5 years is widely accepted with greater remission potential.12,173–175,179,181,186 Once remission-induction therapy is initiated, ILI is recommended to be sustained throughout the entire treatment course. Patients with persistent severe obesity and/or complicated by refractory metabolic comorbidities may be considered for metabolic surgery. SIIT is indicated for patients with prominent hyperglycemia symptoms (HbA1c ⩾9.0% or fasting blood glucose ⩾11.1 mmol/L) to rapidly control glucotoxicity. 126 Otherwise, other pharmacological options, such as GLP-1RAs, SGLT2is, and GKA, may be considered. Following the achievement of diabetes remission, ongoing regular assessments of body weight, glycemic parameters, and β-cell function markers remain necessary. 4

Clinical pathway for the selection and management of diabetes remission strategies. This therapeutic pathway suggests preferred optional courses of action rather than a mandatory requirement.
Durability of sustained diabetes remission
Although diabetes remission can be sustained for an extended period, it does not equate to a permanent cure. Many clinical studies have observed the recurrence after achieving remission.12,128,178,187–190 The Look AHEAD trial reported that one-third of patients in the ILI group who had attained remission returned to T2DM status each year. 12 Another RCT evaluated the durability of T2DM remission following a structured weight management intervention. 187 The findings demonstrated that while 62% of subjects in the ILI group maintained remission at the end of the first year, this proportion declined significantly to 13% by the fifth year. 187 The Alliance of Randomized Trials of Medicine vs Metabolic Surgery in Type 2 Diabetes (ARMMS-T2D) study conducted a pooled analysis of four RCTs in the United States to evaluate the sustained durability of remission following metabolic surgery over a 12-year observation duration. It reported that the remission rates after metabolic surgery gradually declined from 50.8% in the first year to 12.7% at 12 years. 188 A more recent study conducted by Adams and the research team also identified a gradual decrease in the remission rate after metabolic surgery, albeit at a slower pace. 189 A study by Li et al. reported that following SIIT, the remission rate was 67% at 6 months, declining to 47.1% at 1 year, and stabilizing at 42.3% by the second year. 128 Although there is currently a lack of head-to-head RCTs comparing the durability of remission across different therapeutic interventions, metabolic surgery appears to offer a more durable remission compared to lifestyle modifications or pharmacological interventions, according to the available clinical data (Figure 7). Comparative researches are needed to further figure out the differences and effectiveness among different strategies, which will ultimately guide the development of more effective and sustainable management approaches for diabetes remission.
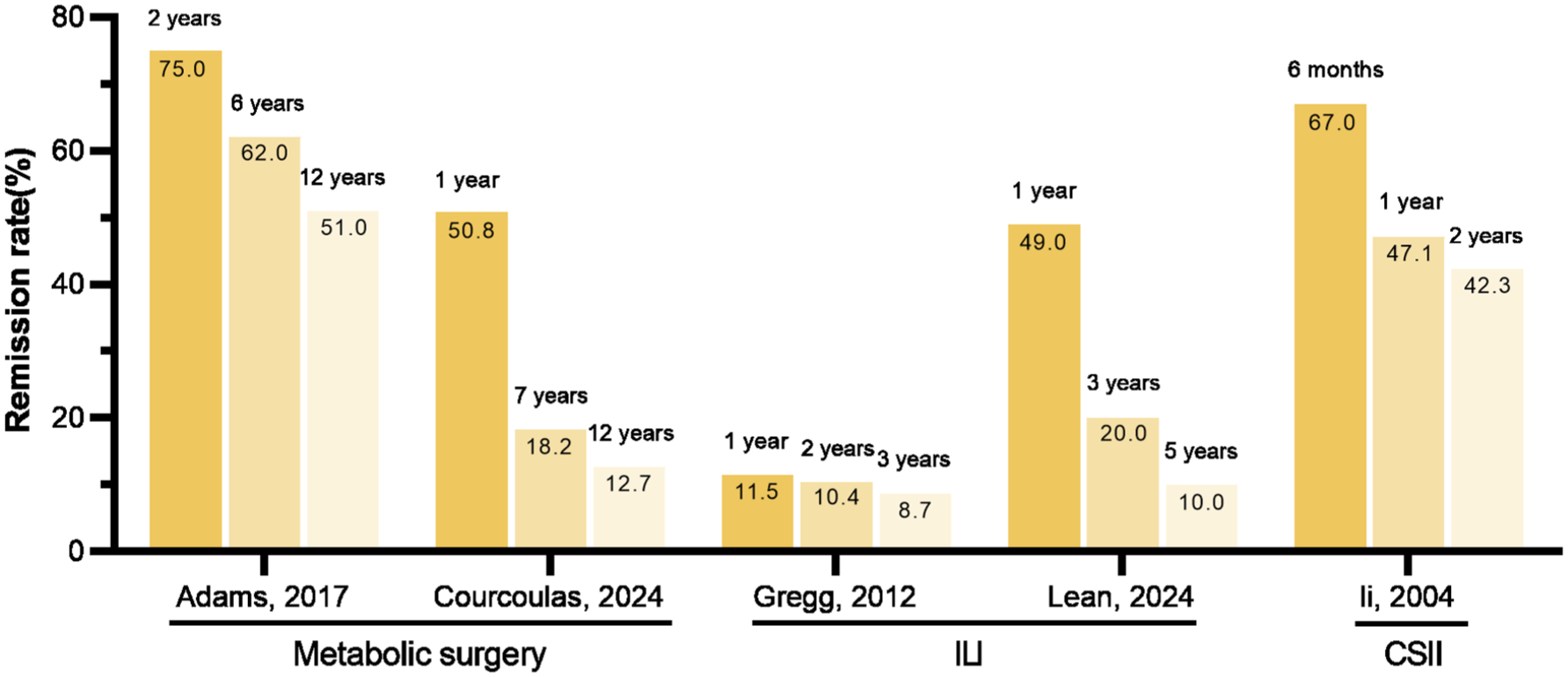
Changes in diabetes remission rate with time under different intervention measures.
Understanding the potential predictors of relapse under different interventions is essential for maintaining long-term remission. The Look AHEAD study identified that long-term remission following ILI may be associated with diabetes duration of less than 2 years, baseline HbA1c levels <6.7%, and weight loss >6.5% in the first year. 12 A multivariate analysis according to a follow-up study of 4434 T2DM patients who achieved complete remission after bariatric surgery identified potential predictors of recurrence including older age at surgery (HR for 5-year increase in age: 1.09; 95% CI 1.04–1.15), poor preoperative glycemic control (HbA1c ⩾6.5%), preoperative insulin use, and longer diabetes duration (HR for each additional year of diabetes: 1.13; 95% CI 1.09–1.17). 191 Therefore, identifying these intervention-specific predictors is crucial for stratifying relapse risk and personalizing long-term management strategies for patients who achieve initial remission.
Furthermore, although long-term clinical effects on macrovascular and microvascular complications have not been extensively studied in clinical trials with remission induction as the primary endpoint, we can still observe significant clinical benefits of related comorbidities following these remission-inducing interventions. ILI has demonstrated long-term benefits in reducing cardiovascular risks.192,193 The Da Qing Study, with 6-year ILI and 30-year follow-up in participants aged 25–74 years, showed that it reduced cardiovascular event risk by 26%, microvascular complication incidence by 35%, and all-cause mortality by 26%. 193 Similarly, studies have shown that bariatric surgery, compared with nonsurgical interventions, more effectively reduced the incidence of both outcomes.194,195 In a retrospective matched cohort study of patients with severe obesity and T2DM, bariatric surgery was associated with a lower incidence of 5-year composite macrovascular events and coronary artery disease compared with usual care. 194 In addition, a real-world study enrolled 5424 patients with newly diagnosed T2DM, and long-term follow-up (median 6.2 years) showed that early SIIT reduced the risk of stroke by 31% and the risk of hospitalization for heart failure by 28%. 196
Taken together, the durability of diabetes remission is concerning, highlighting the necessity of continuous monitoring of glucose parameters in the population that has achieved diabetes remission. In addition, investigation on the association between diabetes remission and macrovascular/microvascular complications is highly warranted.
Conclusion and outlook
The achievement of diabetes remission has long been a shared objective between healthcare professionals and individuals undergoing clinical treatment. This review comprehensively summarized the definition of diabetes remission, population characteristics, and potential interventions to achieve remission. Currently, research on strategies for diabetes remission predominantly centers around weight loss, with its efficacy well-established. Previous studies have demonstrated that metabolic surgery is the most efficient approach to induce diabetes remission, characterized by a high remission rate and relatively long-lasting effects. However, metabolic surgery is an invasive treatment associated with a certain risk of complications and low compliance, making it suitable only for a select group of patients. Although the emergence of laparoscopy has reduced the risk of surgery significantly, the risk of side effects after surgery still exists. In addition to surgical interventions, dietary intervention stands out as the most precise approach for achieving T2DM remission, while evidence supporting the reversal of T2DM through exercise intervention alone remains relatively limited. The research on drug intervention measures mainly focuses on SIIT, the effect of which on alleviating T2DM is relatively clear. However, research on other hypoglycemic drugs to reverse T2DM remains limited. Therefore, the development of novel hypoglycemic agents, especially those with weight loss effects such as GLP-1RAs, holds great promise. Howbeit, there is a notable absence of relevant clinical studies regarding these agents with diabetes remission as the primary endpoint.
Additionally, the predictive factors for alleviating diabetes vary among different population characteristics and intervention strategies. Tailoring interventions to the specific characteristics of diverse patient populations is essential for achieving diabetes remission. Notably, the association between sex and diabetes remission remains understudied, making it a critical research priority. Future work should include large-scale cohorts to systematically assess sex-specific differences in remission rates, alongside mechanistic studies investigating potential biological or behavioral mediators such as sex-based variations in insulin sensitivity, fat distribution, and response to weight loss interventions.
Although a complete cure for diabetes is currently unavailable, optimizing remission strategies for well-defined patient cohorts can significantly enhance both the rate and durability of remission, leading to a better quality of life for individuals with T2DM. Sustained weight loss is widely regarded as the cornerstone of achieving and maintaining diabetes remission. Thus, individuals with T2DM should adopt a sustainable long-term management regimen for weight control. Nevertheless, weight management is not the sole determinant for remission maintenance. Preserving pancreatic β-cell function and attenuating systemic/local inflammatory status are also critical to preventing disease recurrence.
Future studies are needed to elucidate the underlying mechanisms linking gut microbiota to remission, develop targeted microbial modulation strategies (e.g., probiotics, prebiotics, or fecal microbiota transplantation), and evaluate their synergistic effects with existing interventions, such as dietary or pharmacological approaches, to promote and sustain remission. Moreover, well-designed clinical trials to assess the long-term efficacy and safety of emerging interventions will be critical to inform more targeted therapeutic approaches.
