Abstract
Objective:
To evaluate the residual effect of partial remission (PR) on immediate post-PR glycemic control according to its occurrence and duration in a cohort of children with type 1 diabetes mellitus (T1DM).
Patients and Methods:
Values of glycemic control parameters [i.e. HbA1C, insulin dose–adjusted hemoglobin A1C (IDAA1C), glycemic target–adjusted HbA1C (GTAA1C)] and data from glucose monitoring devices from 189 pediatric patients with new-onset type 1 diabetes were collected retrospectively from 24 months. Patients were characterized according to their remission status (PR+ and PR−). PR+ patients were subdivided into three subgroups regarding PR duration [i.e. short (⩾3–⩽6 months), intermediate (>6–⩽12 months), and long PR (>12–⩽14 months)]. We compared glycemic control data from each PR+ subgroup at +6 and +12 months post-PR with PR− patients at the same postdiagnosis time. Second, PR+ subgroups were compared with each other.
Results:
PR+ patients showed improved glycemic control (i.e. HbA1C, IDAA1C, and GTAA1C) at + 6 months post-PR when compared with nonremitters (PR−), independently of the PR duration subgroups (p < 0.05). Interestingly, patients in long PR+ subgroup exhibited higher positive residual effect than short PR+ subgroup with lower GTAA1C scores (p = 0.02), better time in range (TIR) (p = 0.003), less time in hypoglycemia (10.45
Conclusion:
This study supports the positive impact of PR occurrence and duration on short-term metabolic control (better HbA1C levels, IDAA1C and GTAA1C scores, TIR, and less glycemic variability) with the residual effect increasing according to PR duration.
Keywords
Introduction
Diabetes mellitus is a metabolic disorder characterized by chronic hyperglycemia resulting from dysfunction in insulin secretion, insulin action, or both. Type 1 diabetes mellitus (T1DM) results from irreversible immune-mediated destruction of pancreatic insulin-producing β cells.1,2 At the time of T1DM diagnosis (usually corresponding to the onset of insulinopenic symptoms), islet β-cell mass is reduced to 10–30%.1,3 Shortly after the initiation of insulin therapy, about 60% of patients with T1DM experience a ‘partial remission’ (PR) period also called ‘honeymoon period’.4,5
PR definition has evolved over the years and is still debated. 6 In 2009, based on the European Hvidøre Study Group, Mortensen and colleagues 7 suggested identifying PR using the insulin dose–adjusted hemoglobin A1C (IDAA1C) score, readily usable in clinics and integrating both HbA1C levels and daily insulin requirements. The IDAA1C score has now been validated in large pediatric patient cohorts8–11 and by the International Society for Pediatric and Adolescent Diabetes (ISPAD). 5 It is currently considered as the most recognized standard to define PR when its value is ⩽9. More recently, the glycemic target–adjusted HbA1C (GTAA1C) score has been suggested by our team as an alternative definition of PR that does not depend on insulin requirements and which relies on objective markers of glycemic homeostasis (HbA1c and percentage of normoglycemia). GTAA1C score predicts PR when its value is ⩽4.5 and is strongly correlated with IDAA1C score. 12
Mechanisms underlying PR remain controversial. A state of improved insulin sensitivity, decreased glucotoxicity, relative escape of β cells from the immune system (by decrease of HLA type I expression), and partial recovery of previously exhausted β-cell function are key metabolic aspects involved in the remission period.4,13–21 While a dichotomy prevails in the occurrence or absence of PR, this period is marked by heterogeneity in intensity and duration that may be short, intermediate, or long and which is influenced by an array of well-described clinical factors (e.g. age, gender, ketoacidosis at diagnosis).4,9,18,19,22–30
Subsidiary studies of the Diabetes Control and Complication Trial (DCCT) and the Epidemiology of Diabetes Interventions and Complications (EDIC) trial have highlighted the importance of early optimal glycemic control to prevent micro- and macro-vascular complications of T1DM.31,32 The PR period is key in the early management of T1DM but also as a target period for strategies aiming at preserving endogenous β-cell mass. However, apart from being potential leverage for therapeutic applications, little is known about whether the amplitude (i.e. intensity and duration) of PR influences short-term glycemic control in patients with T1D.
The intuitive clinical experience suggests that the level of glycemic control achieved during PR is difficult to maintain once remission ends – often abruptly, and the ensuing imbalance may become chronic. Furthermore, for some patients, PR appears to play a negative effect: the longer the PR, the greater the glycemic imbalance at the end of remission. Very few studies have evaluated the post-PR period and the underlying risks of diabetes-related complications. 33 Correlation between PR duration and postremission glycemic control was not extensively studied so far, especially in children. Our DIABHONEY study aims to assess the impact of the occurrence and duration of PR on metabolic control in pediatric patients with T1DM in the immediate post-PR period (i.e. 12 months post-PR).
Patients and methods
Study design and participants
We retrieved from our patient database a retrospective cohort of 398 children and adolescents diagnosed with type 1 diabetes between January 1997 and December 2018 and followed up in the pediatric diabetes clinic of our tertiary health care center (Cliniques universitaires Saint-Luc, Brussels). The study was approved by the local ethical committee (reference CHE:11/JUI/274) and conducted in accordance with the Declaration of Helsinki. Patients eligible were aged between 1 and 18 years and were diagnosed with new-onset T1DM. T1DM was established according to ISPAD guidelines, 3 based on symptoms of insulinopenia, elevated blood glucose (BG), positive anti-islet autoantibodies (i.e. GAD65, IA2, and insulin), and lack of family history of genetic diabetes. Exclusion criteria were diabetes onset before the age of 1 year, presence of severe chronic medical conditions before the diagnosis of T1DM (i.e. autoimmune diseases other than type 1 diabetes, active cancer, kidney, liver, or adrenal insufficiency) and use of medication that may affect insulin secretion and/or glucose homeostasis (i.e. corticosteroids, sulfonylurea, incretins, diazoxide, somatostatin, immunomodulatory drugs). Patients with a PR less than 3 months, above 14 months or ongoing at the time of study were also excluded. All patients performed carbohydrate counting and underwent similar dietary education at diagnosis.
Medical records of each patient were reviewed to collect demographic data at diagnosis [i.e. age, gender, date of diagnosis, height, weight, body mass index (BMI)] as well as quarterly follow-up data until 24 months. This included routine clinical and biological parameters [HbA1C levels (%), insulin doses in total daily dose in IU and IU/kg body weight, IDAA1C and GTAA1C scores, number of severe hypoglycemia] and data from glucose monitoring devices [using either continuous glucose monitoring (CGM) or self-monitoring of blood glucose (SMBG)]. The parameters retrieved from CGM or BG meter were average glucose (mg/dl), glucose variability [glycemic SD (mg/dl)], coefficient of variation of glucose (CV, %)], number of glucose measurements, time spent in hypoglycemia (below 70 mg/dl, % total time), number of severe hypoglycemia, time spent in hyperglycemia (above 180 mg/dl, % total time), and time spent in normoglycemia (70–180 mg/dl; % total time) also called time in range (TIR). Body mass index (BMI) was calculated using the formula = body weight (kg)/[height (m)].2
PR definition and groups
PR was defined by a combination of both IDAA1C and GTAA1C scores below their respective threshold (i.e. IDAA1C ⩽9 and GTAA1C ⩽4.5). IDAA1C was calculated according to Mortensen and colleagues, 7 as such: HbA1C (%) + [4 × insulin dose (U/kg/day)]. The GTAA1C 12 corresponds to HbA1C (%) − [3 × % of normoglycemic values (70–180 mg/dl)]. The end of PR period was defined as the first follow-up consultation where the patient exhibited both an IDAA1C >9 and a GTAA1C >4.5.
Patients were divided into two groups depending on the occurrence (i.e. positive cohort, PR+ group) or absence of PR (i.e. control or negative cohort, PR− group) at 3 months postdiagnosis. PR+ group was further divided into three subgroups according to their PR duration: short (PR duration ⩾3 and ⩽6 months), intermediate (PR duration >6 and ⩽12 months), or long PR (PR duration >12 and ⩽14 months). Patients with a PR period longer than 14 months were excluded as some might have been misdiagnosed with T1DM (e.g. presenting features of monogenic diabetes) (Figure 1). The control group (i.e. PR−) was determined as patients with IDAA1C (>9) and GTAA1C (>4.5) indexes.

Flowchart of patient groups in the DIABHONEY study.
First, the positive (PR+) and negative (PR−) cohorts were compared for age, sex, and BMI. Second, we compared clinical and glucose homeostasis data (listed above) from each of the three subgroups of PR+ patients (classified as having short, intermediate, or long PR) at +6 and +12 months after the end of their PR period, to PR− patients at the same time points after diagnosis (see Supplemental Figure S1). Finally, the follow-up data for children with short PR (>3 and ⩽6 months) were compared with children with intermediate (>6 and ⩽12 months) and long PR (>12 and ⩽14 months).
Statistical analysis
Data were analyzed using the JMP Pro 14.3.0 software. The values of categorical variables are expressed in absolute numbers (
Results
Characteristics of study participants
A total of 189 patients were included in the DIABHONEY study (Figure 1). PR occurred in 69.8% of the patients (132/189). Mean age at diagnosis was 9.0 ± 3.7 years. Patients in the PR+ cohort were statistically older than children in the PR− cohort (9.6 years
Characteristics of the patients at diagnosis.
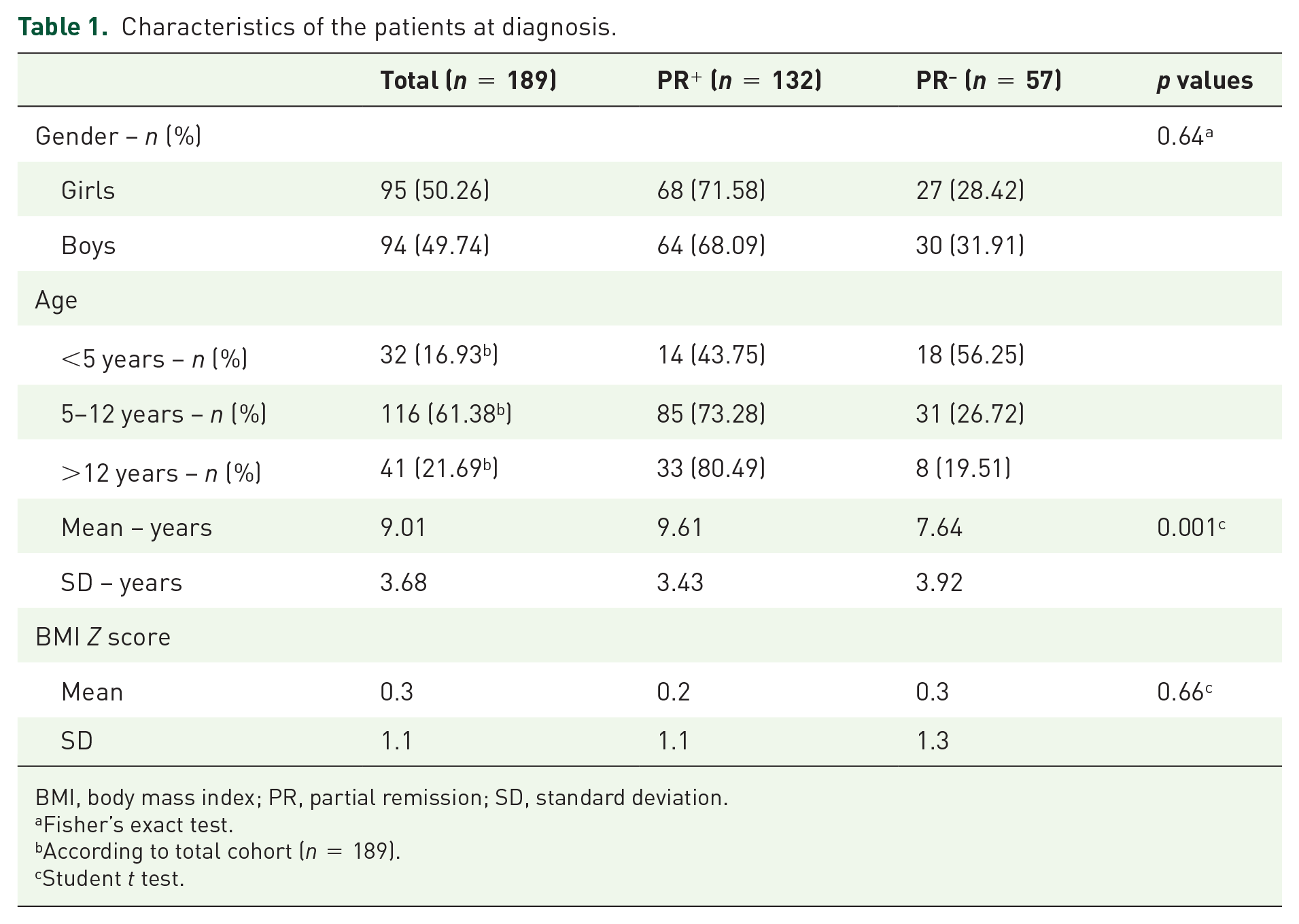
BMI, body mass index; PR, partial remission; SD, standard deviation.
Fisher’s exact test.
According to total cohort (
Student
Short PR improves glycemic control (IDAA1C, GTAA1C) 6 months after remission
To assess the influence of short PR (i.e. >3–⩽6 months) on T1DM control at 6 and 12 months postremission, patients experiencing short PR were compared with PR− controls at the same postdiagnosis time points (Figure 2, Supplemental Figure S1). Six months after the end of PR, HbA1C levels were significantly lower in PR+ children compared with PR− patients (
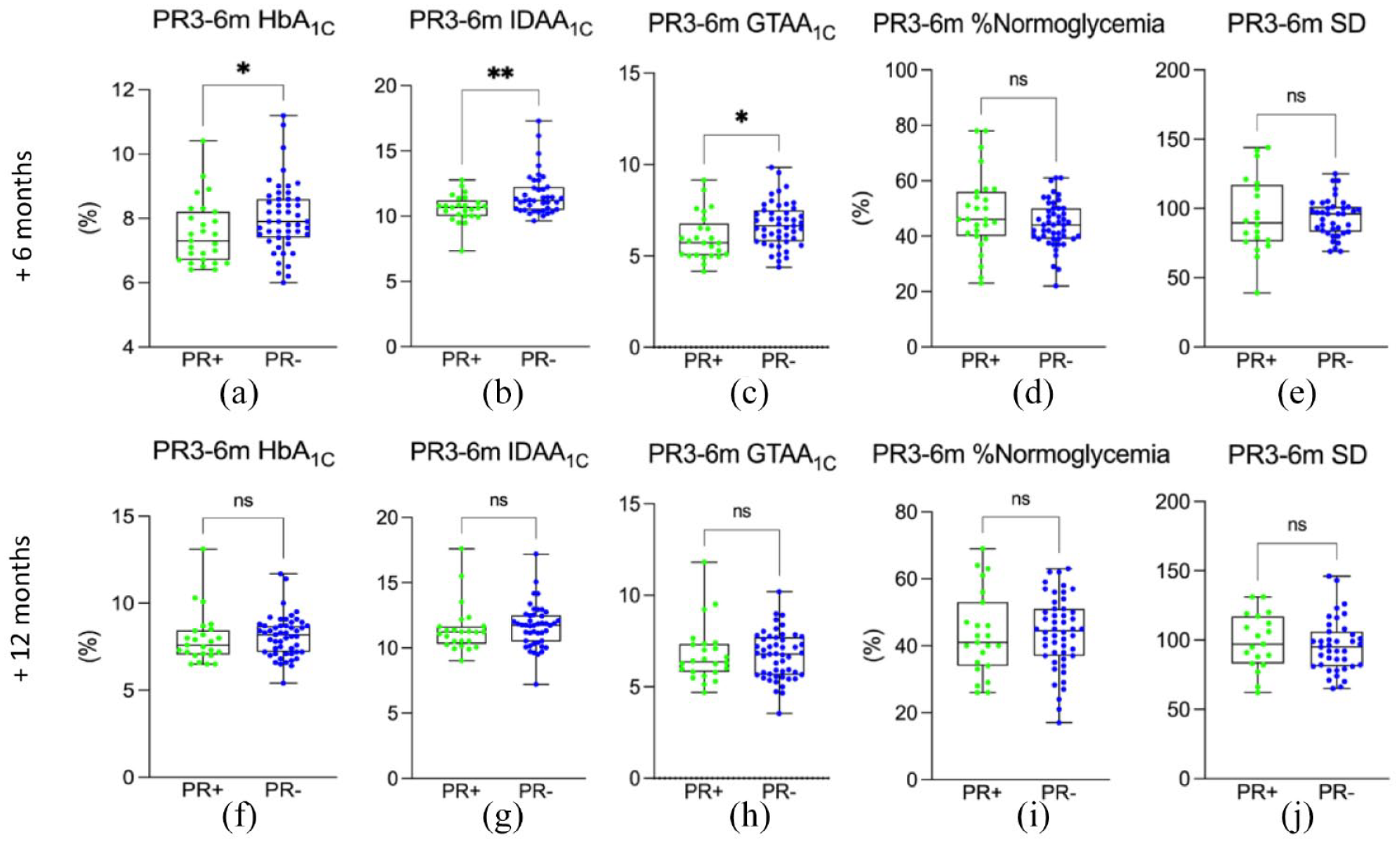
Assessment of the influence of short PR (>3–⩽6 months) at 6 and 12 months postremission. Comparison of the short PR+ cohort with the control PR− cohort, matched at the same postdiagnosis time for different parameters. Comparison of short PR+ cohort at 6 months post-PR
Intermediate PR significantly improves glycemic control (HbA1C, IDAA1C, GTAA1C, TIR) 6 months after remission
To assess the influence of intermediate PR (i.e. >6–⩽12 months) on T1DM control at 6 and 12 months postremission, intermediate PR+ patients were compared with those in the matched PR− cohort at the same postdiagnosis time points (Figure 3, Supplemental Figure S1). As compared with results from short PR group, patients experiencing intermediate PR exhibited significative differences in their glycemic control 6 months after remission when compared with PR− group: HbA1C levels were significantly lower in PR+ group compared with the PR− control group (
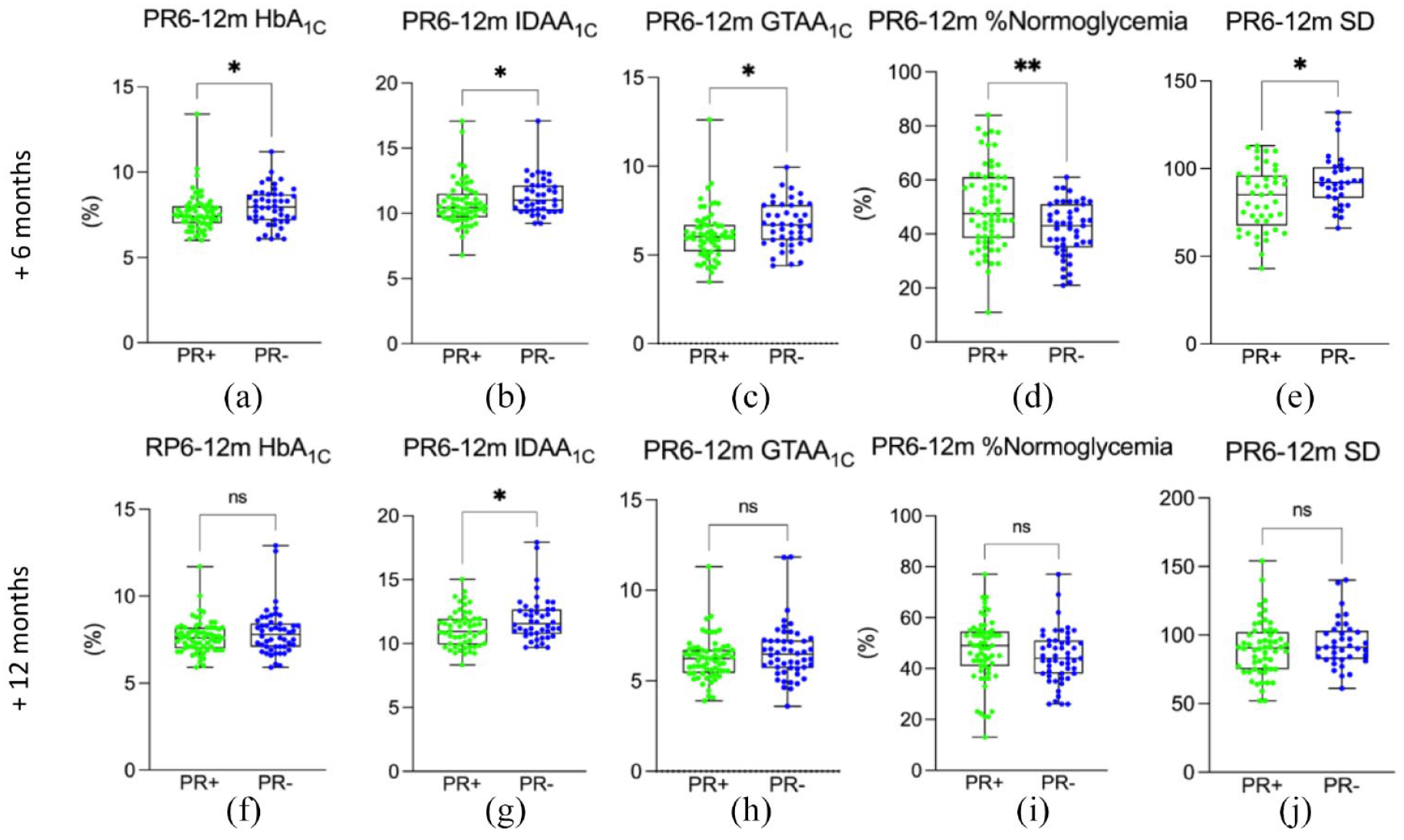
Assessment of the influence of intermediate PR (>6–⩽12 months) at 6 and 12 months postremission. Comparison of the intermediate PR+ cohort with the control PR− cohort, matched at the same postdiagnosis time for different parameters. Comparison of intermediate PR+ cohort at 6 months post-PR
Long PR improves glycemic control (HbA1C, IDAA1C, GTAA1C, TIR) and glycemic variability 6 months after remission
To assess the influence of long remission (i.e. >12–⩽14 months) on T1DM control at 6 and 12 months postremission, long PR patients were compared with those in the PR− control group at the same postdiagnosis time points (Figure 4, Supplemental Figure S1). As observed previously for short and intermediate PR + patients, HbA1C, IDAA1C, and GTAA1C scores at +6 months were significantly lower (respectively,
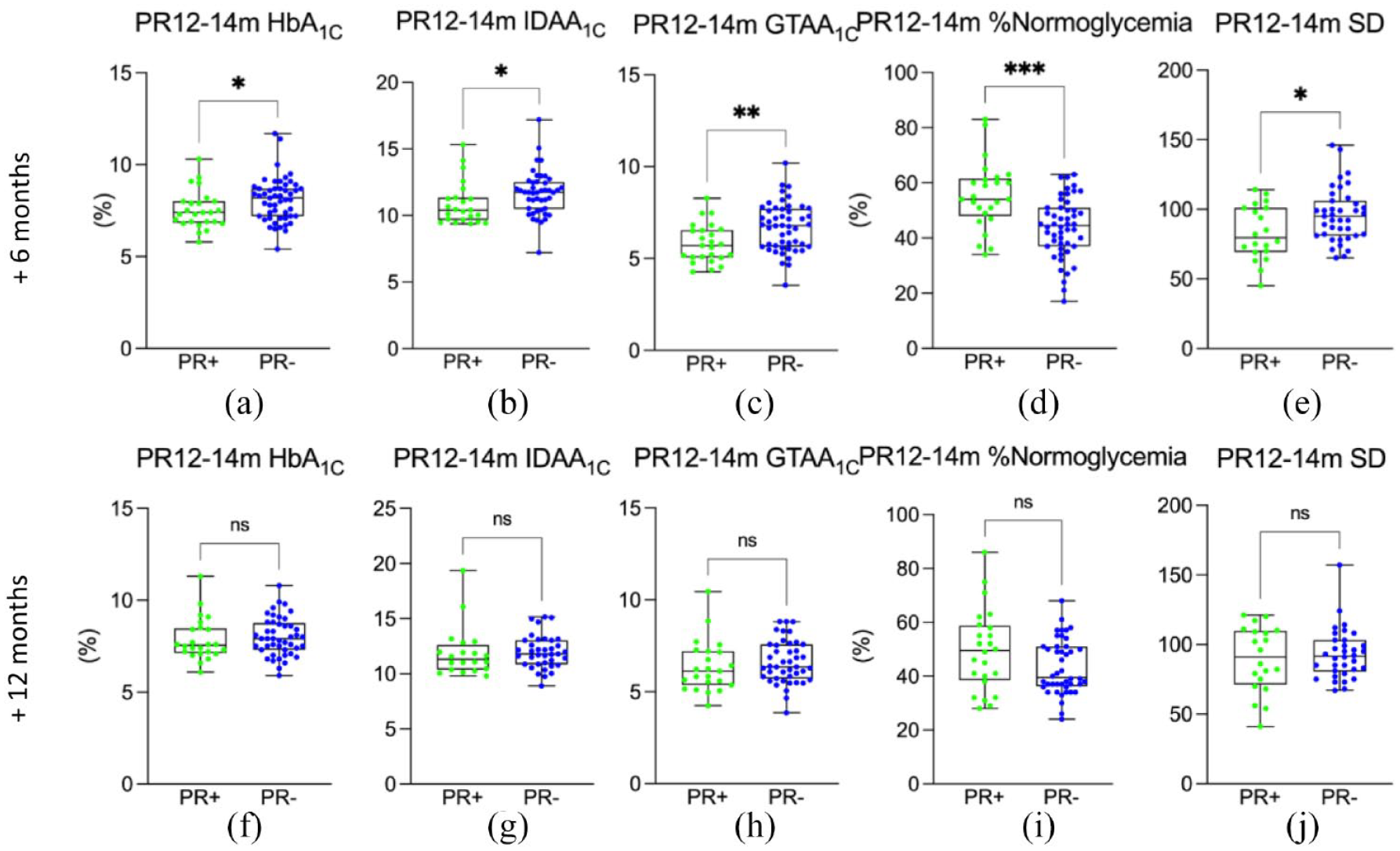
Assessment of the influence of long PR (>12–⩽14 months) at 6 and 12 months postremission.
PR duration also influences diabetes metabolic control
The data of the different PR+ subgroups were cross-sectionally compared with each other at the same time postdiagnosis of diabetes. Children in long PR+ subgroup showed better glycemic control when compared with the short PR+ subgroup at 18 months postdiagnosis (+12 months from the end of PR for short PR+ children and +6 months for those with long PR) (Figure 5). They exhibited lower GTAA1C scores (

Comparison of long PR cohort (>12–⩽14 months) at 6 months postremission to short PR cohort (>3–⩽6 months) at 12 months postremission.
Discussion
PR is a state of low glycemic variability, daily insulin needs, and HbA1C levels. Recent studies in young adults suggested that patients entering PR after diabetes onset were less at risk of vascular complications. 33 Currently, little is known about the influence of PR and its duration on short-term glucose homeostasis outcomes, especially in children.
We first characterized the cohort according to remission status (i.e. PR+ and PR−). Next, we subdivided PR+ groups regarding the duration of the PR (i.e. short, intermediate, and long) and evaluated whether the latter influenced glucose homeostasis at +6 and +12 months after the end of the PR. Our study shows that patients experiencing PR had improved glycemic control at +6 months when compared with nonremitters, independently of PR duration subgroups. Yet these results were not significant at +12 months. Finally, comparison between PR+ subgroups showed that experiencing a long PR allowed better glycemic control at +6 months compared with short PR.
Hallmarks of PR are a combination of low HbA1C, low insulin daily doses, and low glucose variability. In this context, both IDAA1C and GTAA1C scores highlight the positive influence of PR on diabetes control shortly after the end of PR (i.e. 6 months), each in a different way. An IDAA1C score ⩽9 strongly correlates with a stimulated C-peptide level ⩾300 pmol/l 7 reflecting residual β-cell secretion that characterizes PR. 13 This score depends on HbA1C and total daily insulin dose that reflects metabolic control in a broad sense. As previously suggested by our team, 12 the GTAA1C score allows an evaluation of PR independently of insulin requirements and based on an objective measure in addition to HbA1C: time in normoglycemia. GTAA1C provides a better reflection of glucose homeostasis and eventually a more clinically meaningful aspect of PR. Interestingly, IDAA1C and GTAA1C scores remained significantly lower in all three PR+ subgroups 6 months after the end of their PR period when compared with PR− patients.
Furthermore, the duration of PR influences the residual effect on glucose homeostasis: the longer the PR, the better the post-PR glycemic control at +6 months. Indeed, of all PR+ subgroups, long PR patients showed the best glucose homeostasis at 6 months after the end of the PR period with significantly lower GTAA1C score, better TIR, and less glycemic variability, than in the short PR+ subgroup. Our results additionally support the importance of prolonging PR period, as long PR improved glucose homeostasis and duration of the residual effects when compared with short PR.
Current mechanisms underlying the residual effect of PR on short-term glucose homeostasis remain poorly understood. PR is characterized by decreased glucotoxicity (i.e. decreased glucose variability and increased euglycemia), 36 decreased lipotoxicity [e.g. decreased low-density lipoprotein (LDL) levels], 37 and increased immunotolerance (e.g. increased FoxP3 cells, decreased HLA-I expression on β cells) 21 jointly concurring to a reduction of β-cell destruction. These phenomena might together lead to improved residual secretion and increased insulin sensitivity that are known to have a long-term beneficial impact on micro- and macro-vascular diabetes complications 33 and are suspected to play a role in metabolic memory.38,39 We may assume that the combination of these mechanisms might also influence short-term glycemic control and influence the heterogeneity of PR duration. Taken together, these might partially explain the positive residual effect of PR on short-term metabolic balance that demonstrated to be proportional to its duration.
Another hypothesis behind this remanent phenomenon might be that it could be influenced by behavioral components (e.g. healthy diet and/or regular physical activity fostering residual insulin secretion). Yet this aspect of remanent stability of glucose homeostasis is difficult to demonstrate in our cohort, given the monocentric retrospective design of our study (all of our patients count carbohydrates and were given the same dietary education). Even though our study did not evaluate these aspects, it was previously shown that patients with T1DM who engage in regular physical activity have a higher incidence of PR (i.e. 44
Our initial hypothesis based on a clinical intuition was that a longer PR might be associated with a higher risk of glycemic imbalance shortly after PR ends as remitters would less be keen to strictly monitor their diabetes (and could adopt unhealthy habits during PR). Conversely, our results demonstrated that PR was significantly associated with improved glycemic control at 6 months. In addition, no differences were observed in the number of glycemic tests between remitters and nonremitters patients at 6 and 12 months post-PR (independently of the PR+ duration subgroup). This observation could support that the management of T1DM during the remission period (i.e. stability of glycemic control) did not modify the habits of glucose self-monitoring after PR ended.
Finally, it is also important to emphasize that there was no difference among PR+ subgroups regarding the occurrence of severe hypoglycemia when compared with children without remission (and no difference when comparing the three PR+ subgroups with each other). This supports recent results from our group which found no significant differences in the daily rate of grade II hypoglycemia (i.e. <54 mg/dl) between PR+ and PR− patients (Polle
Our study demonstrates several strengths. To the best of our knowledge, our cohort is the largest pediatric population to study for the first time the influence of PR on short-term glycemic control. Implementing a long-term evaluation of the effects of PR duration in longitudinal cohorts of patients with T1DM would be required to confirm our findings. This could be the subject of a study in the context of a national register of children with diabetes in Belgian centers.
Our study also exhibits several limitations, the main one being the single-center retrospective design of our data collection. Although we focused on objective biological parameters (such as HbA1C and the resulting IDAA1C score), glycemic follow-up data (even those collected from CGM methods) still partly depended on the regularity of individual daily monitoring. Moreover, as most patients were diagnosed with type 1 diabetes between 1997 and 2014 (implementation of CGM in Belgium), BG data were not collected in a standardized way in all patients because we included patients with both glucometers and CGM. For this reason, we separately analyzed data collected from CGM and SMBG and obtained similar results in glucose homeostasis parameters (all
In conclusion, our study confirms the previously observed frequency of PR occurrence in European pediatric patients with type 1 diabetes (>60%) and the lowest incidence of PR in young-onset children (<5 years). Our results also emphasize the positive impact of this PR period on short-term metabolic control (better HbA1C levels, IDAA1C and GTAA1C scores, TIR, and less glycemic variability) without increasing the number of hypoglycemia. This favorable effect seems to last at least 6 months after remission, but a significant influence at 12 months post-PR was not observed. The duration of PR is nevertheless associated with a more pronounced residual effect: the longer the PR, the better the post-PR glycemic control (at 6 months). This supports that prevention protocols that aim at prolonging PR may also improve short-term metabolic control, even after PR ends.
Supplemental Material
sj-pptx-1-tae-10.1177_20420188221145550 – Supplemental material for Influence of the occurrence and duration of partial remission on short-term metabolic control in type 1 diabetes: the DIABHONEY pediatric study
Supplemental material, sj-pptx-1-tae-10.1177_20420188221145550 for Influence of the occurrence and duration of partial remission on short-term metabolic control in type 1 diabetes: the DIABHONEY pediatric study by Laure Boutsen, Elise Costenoble, Olivier Pollé, Kezban Erdem, Céline Bugli and Philippe A. Lysy in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
