Abstract
Background:
The etiology of type 2 diabetes mellitus (T2DM) is complex, with environmental factors playing a significant role in its pathophysiology. Nonsteroidal anti-inflammatory drugs usage and Helicobacter pylori infection are the two most frequent causes of peptic ulcer disease (PUD). The link between PUD and T2DM is unclear, and comprehensive analyses of anti-PUD medications’ impact on T2DM risk, especially in Asian populations, are lacking. This study aimed to determine the relationship between PUD, anti-PUD medications, and the likelihood of developing T2DM.
Objectives:
Using a population-based cohort study conducted in Taiwan, we investigated the impact of PUD and anti-PUD medications on the risk of T2DM.
Design:
This is a retrospective, population-based cohort study using the largest database used in Taiwan.
Methods:
An 18-year follow-up period study was conducted on a cohort of patients with PUD diagnosed between 2001 and 2018 using the Taiwan National Health Insurance Research Database. The risk of PUD as well as anti-PUD medications use were examined using Cox proportional regression model.
Results:
Based on multivariable Cox proportional hazards regression analysis, patients with PUD had a higher overall T2DM incidence (22.7 vs 21.3 per 1000 person-years) than patients without PUD. The adjusted hazard ratio was 1.12 (95% confidence interval = 1.10, 1.13). Patients with PUD have a higher risk of T2DM in both genders and age groups. Patients with anti-PUD medications, such as H2 receptor antagonists, proton-pump inhibitors, antibiotics, prostaglandin analogs, anticholinergics, and antacids usage, are associated with a lower risk of developing T2DM than those without. Patients with PUD who underwent surgery were found to have a higher risk of T2DM.
Conclusion:
Patients with PUD are more likely to develop T2DM. Nevertheless, patients receiving anti-PUD medications have a lower incidence of T2DM.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is a growing global health concern, currently affecting 537 million individuals worldwide, a number projected to rise to 643 million by 2030. 1 Characterized by peripheral insulin resistance, insufficient pancreatic β-cell mass, or inadequate insulin synthesis and secretion, the etiology of T2DM is complex, with environmental factors playing a significant role. 1 Identifying modifiable contributors to T2DM could enable effective prevention and management, reducing its medical and economic burden. Individuals with T2DM are at elevated risk for complications, particularly cardiovascular conditions, underscoring the importance of improving glucose regulation and reducing disease prevalence. 1 Lifestyle modifications, including medication, weight loss, exercise, and dietary interventions, remain the primary strategies for managing T2DM. 1
Peptic ulcer disease (PUD), encompassing gastric and duodenal ulcers, arises from an imbalance between protective and aggressive factors affecting mucosal integrity. 2 Causes include Helicobacter pylori infection, nonsteroidal anti-inflammatory drugs (NSAIDs), and gastric acid, alongside lifestyle factors like smoking, alcohol consumption, and stress. 2 Helicobacter pylori, a gram-negative bacterium infecting the gastric mucosa, affects 30%–40% of Americans and nearly half of the global population. 3 It contributes to 70% of gastric ulcers and 95% of duodenal ulcers and is associated with gastric adenocarcinoma and mucosa-associated lymphoid tissue lymphoma. PUD can also result from drug-induced ulcers, stress, or conditions like Zollinger-Ellison syndrome. 2 PUD treatments include H2 receptor antagonists, proton-pump inhibitors (PPIs), H. pylori eradication regimens, bismuth salts, prostaglandin analogs, anticholinergics, and antacids. 2
The lifetime prevalence of PUD is in the 5%–10% range, with annual incidences of 0%–3%. 4 Use of anti-PUD medications has transformed PUD management, with decreasing incidences since the 1950s and decreases in annual prevalence estimates (0.12%–1.5%) accompanied by reductions in complications and mortality. 2 In the United States, PUD-related hospitalizations decreased by 25.8% from 2005 to 2014. 2 PUD’s age-standardized prevalence rate fell from 143.4 per 100,000 people in 1990 to 99.4 per 100,000 people in 2019, declining in tandem with mortality and disability-adjusted life-years. 2 Long-term NSAID users develop gastric ulcers in 10%–30% of cases, with complications occurring in 2%–4% of cases. 2 PUD complications include bleeding, perforation, and gastric outlet obstruction. 4 Bleeding, often asymptomatic until presenting as melena or hematemesis, is a significant concern, with hospital admissions declining globally but mortality remaining at 5%–10% and it is more common among older individuals, high-dose NSAID users, and those taking concurrent corticosteroids. 4 Perforation, characterized by severe abdominal pain, has mortality rates of up to 20%, depending on age and comorbidities. 4 Anti-PUD medications significantly reduce hospitalization rates, a critical finding given the rising use of NSAIDs and aspirin, which independently increase PUD risk. With their widespread and growing use of anti-PUD medications, this analysis underscores the importance of thoroughly understanding their implications.
Helicobacter pylori infection can stimulate prolonged inflammatory responses, including elevated tumor necrosis factor-α (TNFα) and interleukin-6 (IL-6) levels, potentially altering systemic cytokine networks. 5 Chronic inflammation is a key factor in the pathogenesis of PUD and is similarly associated with the development of T2DM. This shared inflammatory basis has prompted researchers to investigate potential associations between PUD and T2DM, aiming to better understand the interplay between these conditions. Some studies suggest higher rates of H. pylori infection in T2DM patients,6,7 while others do not support an association between H. pylori infection and DM8,9 so the relationship remains inconclusive. NSAIDs and aspirin are major contributors to PUD and its complications. NSAIDs use quadruple PUD complication risks, while aspirin doubles the risk by prostaglandin inhibition, direct mucosal irritation, and increased acid exposure, compromising the stomach’s protective barriers. 2 PPIs reduce gastric acid secretion, increasing gastrin levels and potentially promoting β-cell production. 2 However, their role in T2DM development is debated. Whereas one cohort study linked PPI use to a higher diabetes risk, 10 other analyses have suggested protective effects in patients with upper gastrointestinal disorders,11,12 and a randomized trial found no significant association. 13 Surgical interventions for PUD, particularly gastric surgeries, may increase T2DM risk due to potential development of dumping syndrome, which causes rapid gastric emptying and spikes in blood glucose levels, impacting glucose metabolism. 14 Furthermore, alterations in gut hormones following surgery can exacerbate insulin resistance, heightening T2DM risk in these patients. 15
Considering such complexities, we hypothesize that PUD may influence T2DM risk. Despite existing studies, comprehensive evaluations of the impact of anti-PUD medications on T2DM risk are lacking, particularly in Asian populations. To address this gap, we conducted a retrospective study in a Taiwanese cohort to explore the relationships among PUD, its treatments, those who were not on these indicated anti-PUD medications, and T2DM risk, with the aim of generating hypotheses for further investigation.
Methods
Data source
The Taiwan National Health Insurance Research Database (NHIRD) was used for this study. Since 1996, more than 99% of Taiwanese residents’ health information has been collected and stored in the NHIRD. The database of Longitudinal Generation Tracking Database (LGTD2000) from NHIRD consists of data, including the demographic characteristics of the insured, records of admission and discharge, medicine, and treatment. The disease codes in this database were coded using the World Health Organization’s Ninth and Tenth Revision, International Classification of Diseases (ICD-9 and ICD-10, respectively). The Research Ethics Committee of China Medical University Hospital approved this study (CMUH111-REC2-109-CR-1).
Study population
In this study, the case cohort consisted of patients diagnosed with PUD with the disease codes ICD-9: 531-533 and ICD-10: K25-27, K31.82, and K56.6, between 2001 and 2018. Those who were not diagnosed with PUD were matched to the case cohort according to sex, age, comorbidity of hyperlipidemia, coronary heart disease (CHD), hypertension, obesity, sleep disorders, chronic obstructive pulmonary disease (COPD), stroke, and heart failure, and year of index date to identify the control cohort. The index date for control subjects was randomly appointed a month and day with the same index year of the matched PUD cases. Propensity score matching was performed at a ratio of 1:1. The first prescription date was the index date for patients with PUD, a random date between 2001 and 2018 was set as the index date for the controls, and the follow-up period was from the index date to until a new diagnosis of T2DM or until the subject was censored because of death, withdrawal from insurance, or the end of follow-up on December 31, 2019. Participants who were diagnosed with T2DM before the index date were excluded. In addition, the diagnostic criteria have been specified (e.g., ICD codes) and minimum occurrences within a defined time frame are indicated. For instance, we identified PUD, T2DM, and comorbidities with at least three claims for outpatient and/or hospitalization visits. To avoid subjects being misdiagnosed or coded incorrectly as PUD, T2DM, or comorbidities, we defined patients with at least three consensus diagnoses to ensure the validity of the diagnosis.
Main outcome and comorbidities
The study outcome of the interest was the occurrence of T2DM (ICD-9: 250.×0, 250.×2, and ICD-10: E11). In this study, we considered age, sex, T2DM-related comorbidities, and the use of anti-PUD medications as confounding factors. In addition to continuous values, age was also divided into four groups containing younger or equal to 34, 35–49, 50–64, and older or equal to 65 years old. The T2DM-related comorbidities were identified before endpoints and included hyperlipidemia (ICD-9: 272; ICD-10: E78), CHD (ICD-9: 411–414; ICD-10: I20, I24, I25), hypertension (ICD-9: 401–405; ICD-10: I10–I16), obesity (ICD-9: 278; ICD-10: E66), sleep disorders (ICD-9: 327.23, 780.51, 780.53, 780.57; ICD-10: G47.30, G47.33), COPD (ICD-9: 491, 492, 496; ICD-10: J41, J43, J44), stroke (ICD-9: 430–438; ICD-10: I60–I69), and heart failure (ICD-9: 428; ICD-10: I50) (the details have been documented as Supplemental Table 1). The anti-PUD medications that are available in Taiwan, contain H2 receptor antagonists (famotidine, cimetidine, nizatidine, ranitidine, and roxatidine), PPIs (esomeprazole, lansoprazole, pantoprazole, omeprazole, rabeprazole, and dexlansoprazole), antibiotics (clarithromycin, metronidazole, tetracycline, and amoxicillin), bismuth salts (bismuth subcitrate), prostaglandin analog (misoprostol), anticholinergics (dicyclomine and pirenzepine), antacids (aluminum hydroxide, magnesium hydroxide, calcium carbonate, and sodium bicarbonate), and other agent (carbenoxolone). The comparison between PUD patients with and without surgery was also conducted. The comorbidities, use of anti-PUD medications, and surgery of PUD were identified from the index date to endpoints.
Statistical analysis
Between the PUD and non-PUD cohorts, we calculated the frequency and percentage for the categorical variables (such as sex, stratified age, comorbidities, and anti-PUD medications) and the mean and SD for the continuous variables. The distribution differences between the two cohorts were assessed using standardized mean differences (SMD). An SMD of ⩽0.1 indicates a negligible difference between the two cohorts. Unadjusted and multivariable-adjusted hazard ratios (aHR) with corresponding 95% confidence intervals (CIs) for T2DM risk between the two cohorts were analyzed using the Cox proportional hazard regression model. The aHR was obtained from a multivariate model including sex, age, and comorbidities. We measured the cumulative incidence curves between patients with PUD and comparison cohorts using the Kaplan–Meier method and tested the differences using the log-rank test. We test the proportional hazards assumption by creating a model to evaluate the T2DM risk throughout the overall follow-up period and conducted an analysis for death as a competing risk. All statistical analyses were performed using SAS, version 9.4 (SAS Institute Inc., Cary, NC), and plots were plotted using R software version 4.0 (R Foundation for Statistical Computing, Vienna, Austria). The statistical significance level was set at p < 0.05.
Results
A flowchart illustrating the cohort selection process was shown in Figure 1, to enhance transparency in the population selection, showing inclusion and exclusion criteria for both the PUD and control groups. Between 2001 and 2018, 629,920 participants were enrolled in this study, and both the case and control cohorts included 314,960 subjects. Table 1 shows the baseline characteristics and demographics of the participants including sex, age, comorbidities, and treatments. The proportion of female patients was lower than that of male patients. The mean age in patients with PUD and comparison group was 50.5 (±15.6) and 50.5 (±15.7), respectively. Compared with patients without PUD, the comorbidities between the two cohorts were similar with all of the SMD ⩽0.10. Regarding medication, there were more patients with PUD receiving treatment than were controls, except for H2 receptor antagonists (case: 13.6% vs control: 22.4%). In the model evaluating the T2DM risk throughout the overall follow-up period, test results revealed a significant relationship between Schoenfeld residuals for PUD and follow-up time. This suggests that the proportionality assumption was violated. In subsequent analyses, we stratified the follow-up duration to deal with the violation of the proportional hazard assumption (Supplemental Table 2). We conducted sensitivity analyses, excluding patients with follow-up periods <6 months and <1 year in the Supplemental Table 2. The results remained significant (Supplemental Table 2). After accounting for the competing risks of death, the Fine and Gray model (which extends the standard Cox proportional hazard regression model) was used to estimate the cumulative incidence of T2DM. After mutual adjustment for all relevant confounding factors in the competing risk regression model, the risk of T2DM increased significantly in the presence of PUD (adjusted sub-HR = 1.18, 95% CI = 1.17, 1.20) (Supplemental Table 3).

Flowchart illustrating the cohort selection process.
Demographic characteristics, comorbidity, and medication in patients with and without PUD.
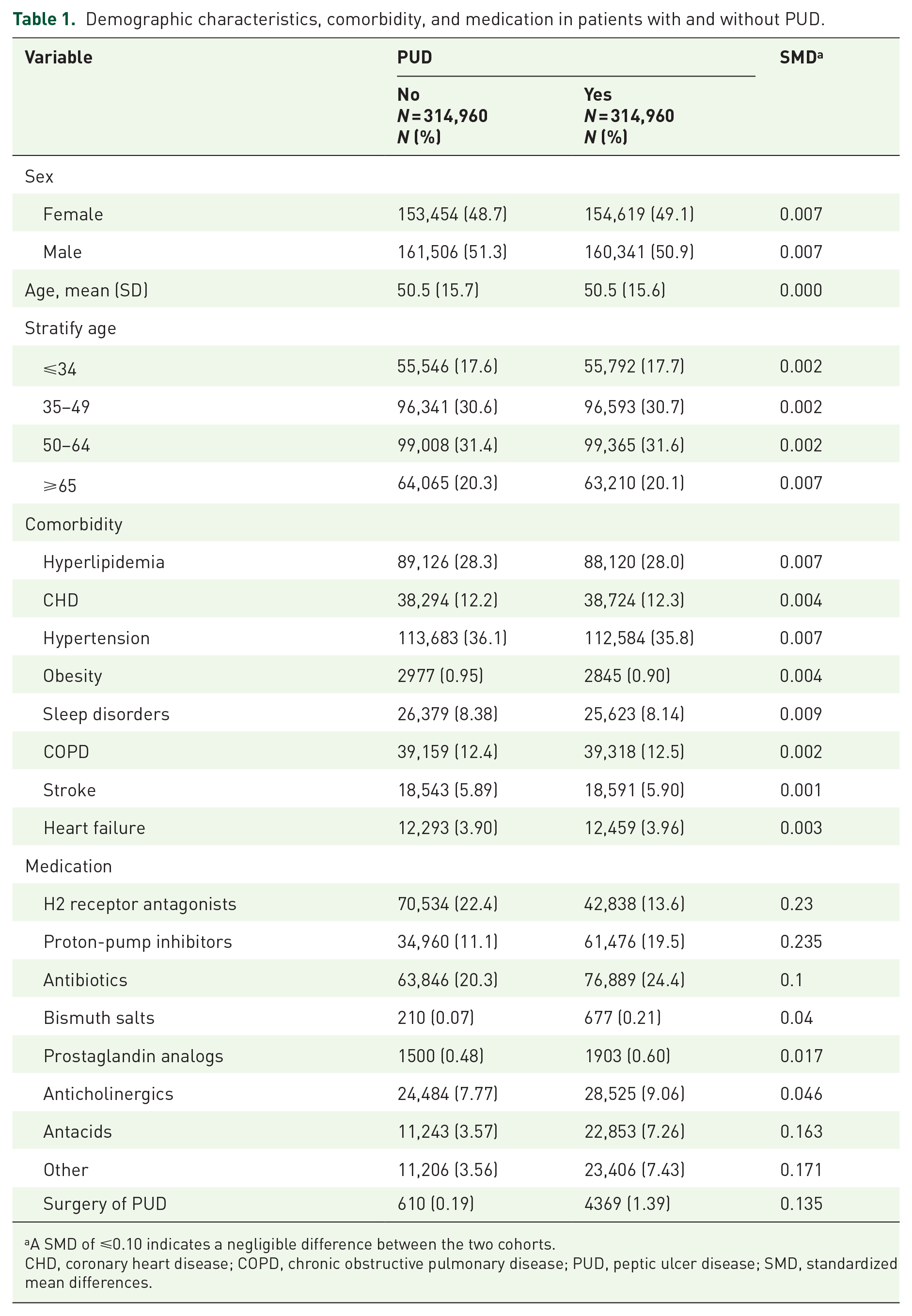
A SMD of ⩽0.10 indicates a negligible difference between the two cohorts.
CHD, coronary heart disease; COPD, chronic obstructive pulmonary disease; PUD, peptic ulcer disease; SMD, standardized mean differences.
Figure 2 shows the cumulative incidence curves of T2DM in patients with and without PUD. The mean and standard difference in follow-up time for the case cohort and comparison cohort were 6.97 ± 5.04 and 7.44 ± 4.98, respectively. As Table 2 shows, patients with PUD had a higher risk of T2DM than did the controls (aHR = 1.12, 95% CI = 1.10, 1.13). This increased risk persisted across both male and female subgroups. Patients with PUD had higher aHRs for T2DM than did the control cohort in all stratified age groups. In both individuals with and individuals without comorbidities, the case group had relatively higher aHRs of T2DM.

Cumulative incidence of type 2 diabetes mellitus compared between the cohort with and without peptic ulcer disease using the Kaplan–Meier method.
Comparison of incidence and HR of type 2 diabetes mellitus stratified by sex, age, and comorbidity between patients with and without PUD.
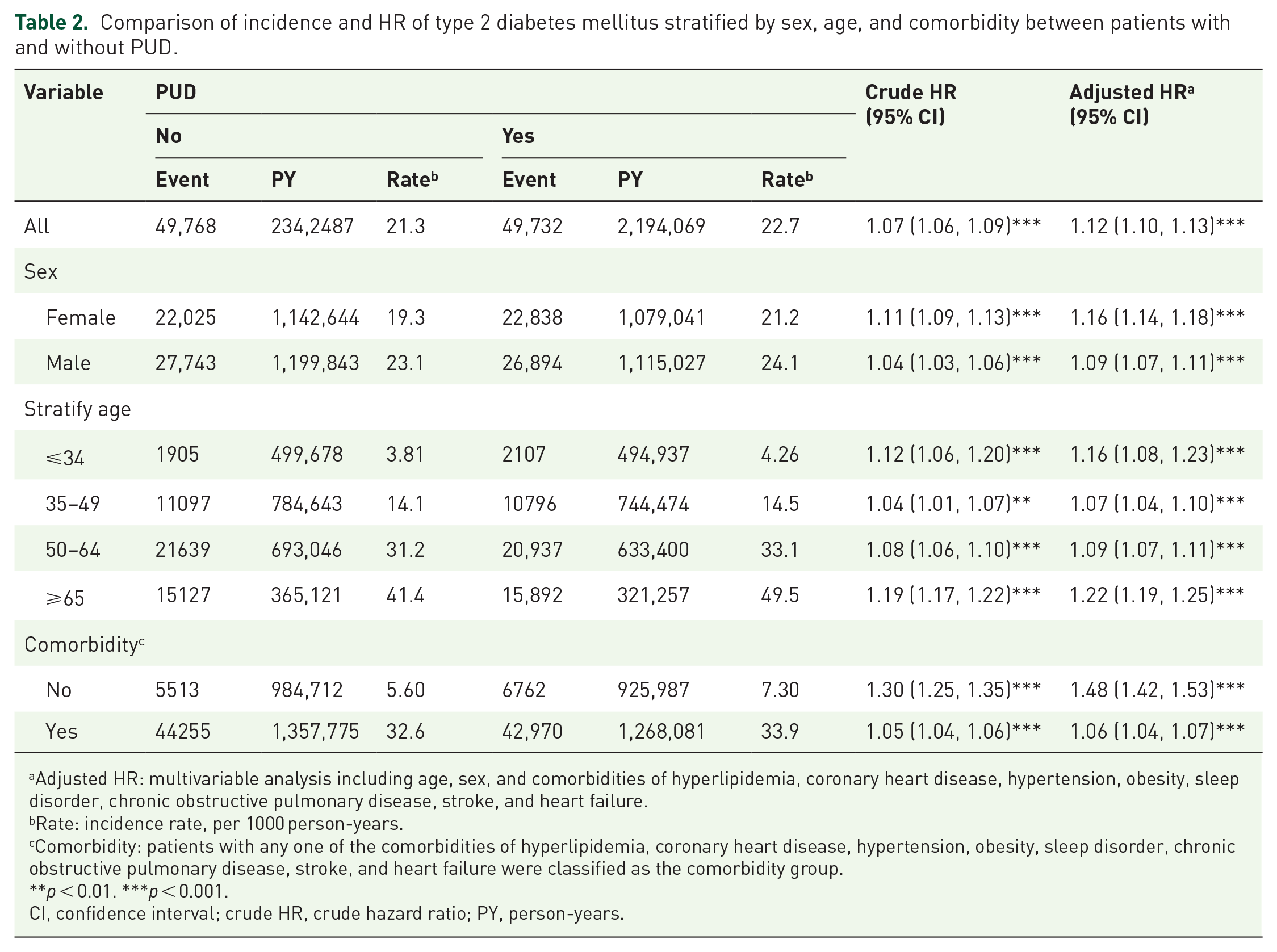
Adjusted HR: multivariable analysis including age, sex, and comorbidities of hyperlipidemia, coronary heart disease, hypertension, obesity, sleep disorder, chronic obstructive pulmonary disease, stroke, and heart failure.
Rate: incidence rate, per 1000 person-years.
Comorbidity: patients with any one of the comorbidities of hyperlipidemia, coronary heart disease, hypertension, obesity, sleep disorder, chronic obstructive pulmonary disease, stroke, and heart failure were classified as the comorbidity group.
**p < 0.01. ***p < 0.001.
CI, confidence interval; crude HR, crude hazard ratio; PY, person-years.
Table 3 shows the risk of T2DM among the three groups of patients without PUD, those with PUD who did not undergo surgery, and those with PUD who underwent surgery. Compared with patients without PUD, patients with PUD with or without surgery had a higher risk of T2DM (with surgery, aHR = 1.41; 95% CI = 1.30, 1.52; without surgery, aHR = 1.12; 95% CI = 1.10, 1.13), and the risk of T2DM for patients with PUD who underwent surgery was 1.20 (95% CI = 1.11, 1.30) times that of the PUD patients without surgery.
Incidence, crude, and adjusted HR of type 2 diabetes mellitus compared among patients with PUD with and without surgery treatment compared to non-PUD controls.
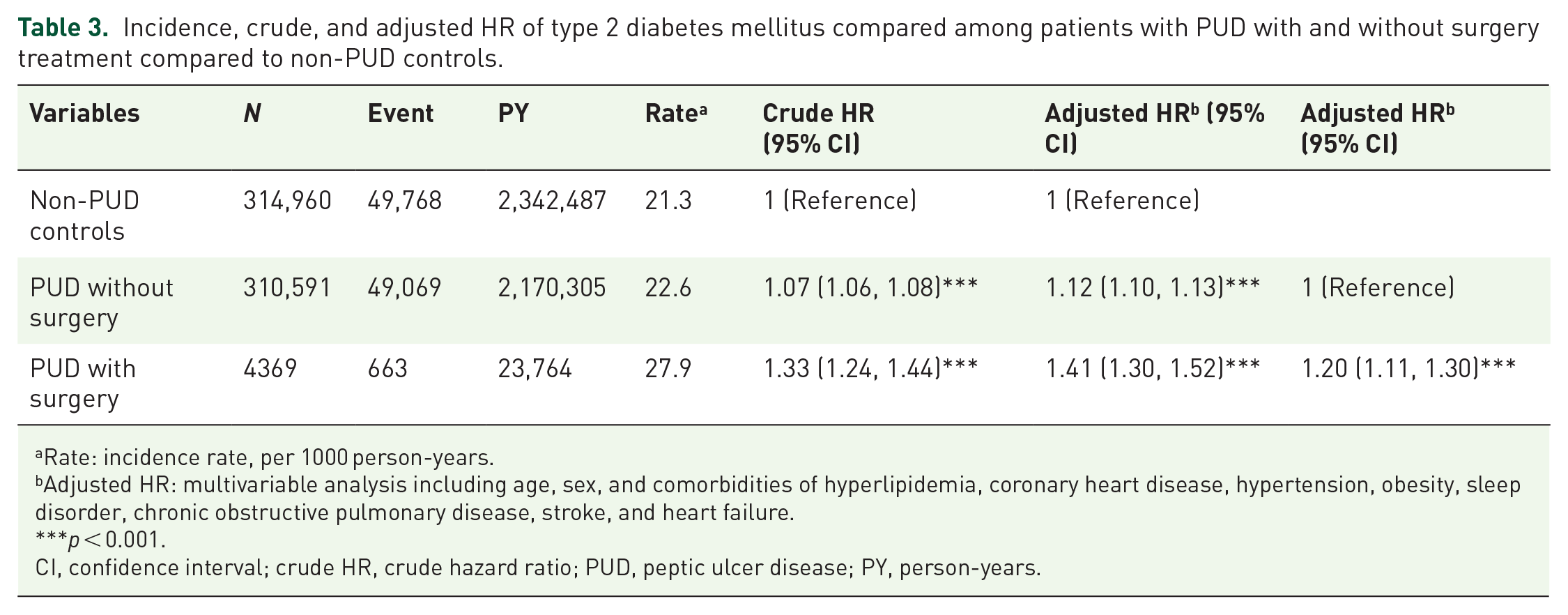
Rate: incidence rate, per 1000 person-years.
Adjusted HR: multivariable analysis including age, sex, and comorbidities of hyperlipidemia, coronary heart disease, hypertension, obesity, sleep disorder, chronic obstructive pulmonary disease, stroke, and heart failure.
p < 0.001.
CI, confidence interval; crude HR, crude hazard ratio; PUD, peptic ulcer disease; PY, person-years.
Table 4 shows the association of T2DM and anti-PUD medications in patients with PUD. Among those with taking H2 receptor antagonists (aHR = 0.66; 95% CI = 0.64, 0.67), PPIs (aHR = 0.86; 95% CI = 0.84, 0.88), antibiotics (aHR = 0.79; 95% CI = 0.77, 0.81), prostaglandin analogs (aHR = 0.65; 95% CI = 0.52, 0.80), anticholinergics (aHR = 0.67; 95% CI = 0.65, 0.70), antacids (aHR = 0.76; 95% CI = 0.73, 0.78), and other (aHR = 0.81; 95% CI = 0.79, 0.84) had significantly lower risk of T2DM than those who did not undergo these treatment. “No” subgroup in each category includes patients who did not take the specific type of anti-PUD medication in question. In contrast, the risk in patients who received bismuth salts was 0.94 (95% CI = 0.80, 1.10) times that of those who did not although it is not significant.
Cox model with HRs and 95% confidence intervals of T2DM associated with anti-PUD medications among PUD patients.
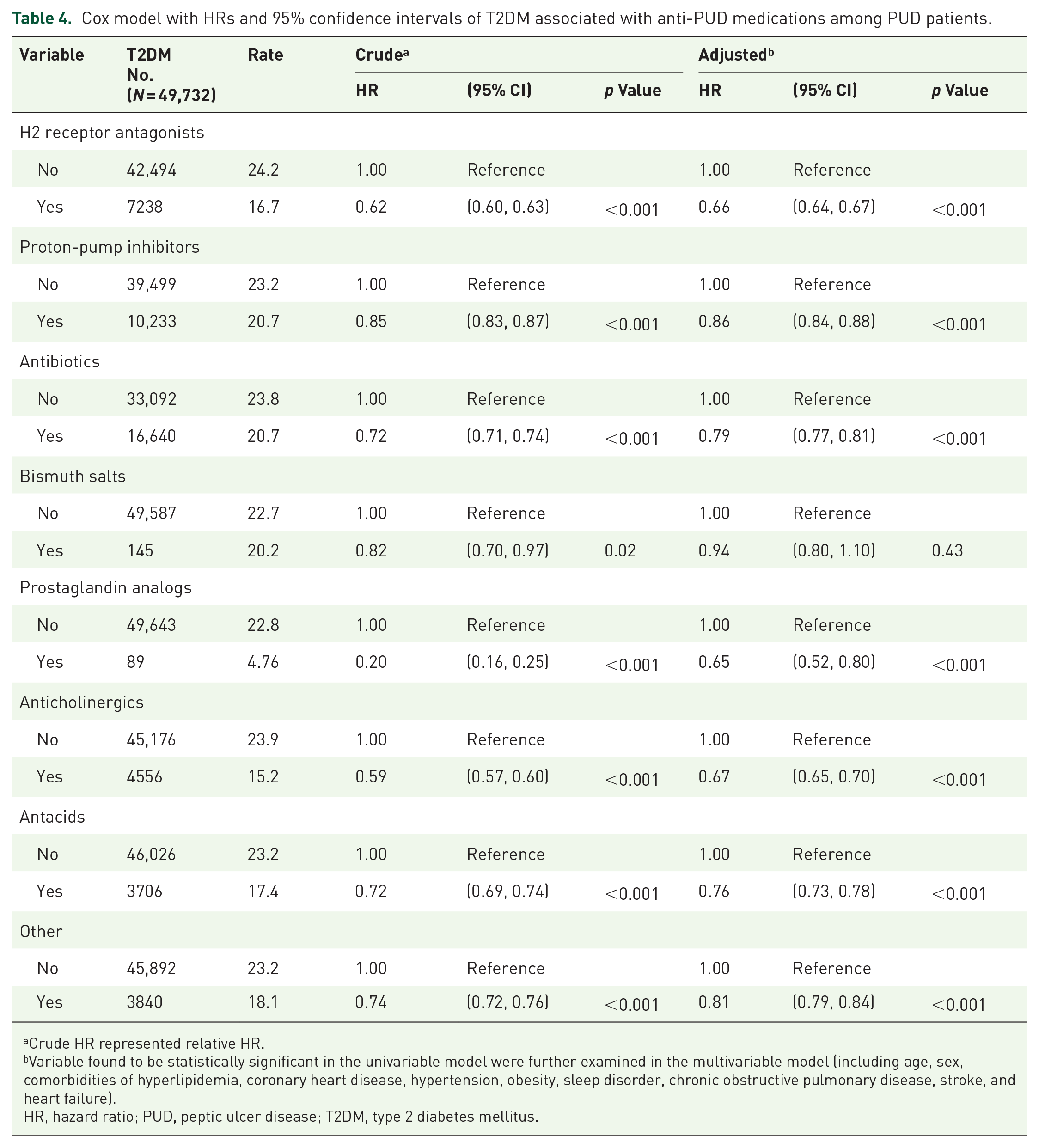
Crude HR represented relative HR.
Variable found to be statistically significant in the univariable model were further examined in the multivariable model (including age, sex, comorbidities of hyperlipidemia, coronary heart disease, hypertension, obesity, sleep disorder, chronic obstructive pulmonary disease, stroke, and heart failure).
HR, hazard ratio; PUD, peptic ulcer disease; T2DM, type 2 diabetes mellitus.
Discussion
This retrospective cohort study is the first to comprehensively assess the impact of anti-PUD medications on the risk of T2DM development in patients with PUD. The study found that PUD patients have an elevated T2DM risk compared to those without PUD. However, patients receiving most anti-PUD medications showed significantly reduced T2DM risk.
PUD symptoms are nonspecific and vary, with duodenal ulcers causing night-time discomfort and hunger, whereas gastric ulcers lead to weight loss, nausea, and postprandial pain. Untreated PUD often recurs due to persistent underlying causes such as H. pylori infection or NSAID use, particularly in asymptomatic elderly individuals. 2 More than 95% of duodenal ulcer patients and 70% of gastric ulcer patients exhibit antral gastritis caused by H. pylori infection. Helicobacter pylori contributes to the pathophysiology of PUD by establishing a microenvironment conducive to its survival, which inadvertently causes mucosal damage. 2 This damage triggers the release of pro-inflammatory cytokines, a process associated with the development of insulin resistance and T2DM.2,16,17 Similarly, NSAIDs, aspirin, smoking, stress, and alcohol compromise mucosal integrity by disrupting protective mucus glycoproteins and prostaglandin synthesis, while also promoting free radical formation and cellular autolysis. 4
Among PUD patients, we observed that, regardless of gender, age, or comorbidities, there is an increased risk of developing T2DM. Several long-term studies have linked H. pylori infection to an increased risk of T2DM, supported by findings of higher HbA1c levels in older individuals.6,17,18 Such findings are consistent with those of earlier research by Gasbarrini et al. 19 and So et al., 20 who discovered a positive correlation between H. pylori infection prevalence and diabetes in cross-sectional studies and a meta-analysis reported a significant association (odds ratio (OR) = 2.00, 95% CI = 1.82, 2.20). 21 However, research on the East Asian CagA strain of H. pylori found no link to diabetes, suggesting its inflammation may not exacerbate insulin resistance despite its role in gastric cancer. 9 The findings highlight the complexity of the H. pylori-T2DM relationship and its variability across strains and populations.
Anti-PUD medications focus on reducing gastric acid and enhancing mucosal defense. H2 receptor antagonists, such as famotidine, cimetidine, nizatidine, ranitidine, and roxatidine, inhibit the H2 receptor, reducing 90% of basal, food-stimulated, and nocturnal acid production. 2 While effective, long-term use often leads to ulcer recurrence. 2 H2 receptor antagonists have additional benefits, such as reducing hunger and postprandial blood glucose levels. Previous studies have reported an inverse relationship between H2 receptor antagonist use and insulin resistance (OR = 0.22, 95% CI = 0.05, 0.95). 22 Cimetidine amplifies insulin secretion and, along with ranitidine, reduces postprandial glucose levels significantly.23 –25 In the present study, H2 receptor antagonist treatment was associated with a 34% lower risk of developing T2DM, further highlighting their potential therapeutic benefits.
PPIs, including esomeprazole, lansoprazole, pantoprazole, omeprazole, rabeprazole, and dexlansoprazole directly inhibit the stomach’s proton-pump, blocking the final stage of acid secretion and controlling both basal and nocturnal acid release. 2 PPIs also inhibit H. pylori growth and are currently first-line PUD therapy, surpassing H2 receptor antagonists. Studies have explored the relationship between PPIs and glycemic control, with mixed results.11,26 For example, Singh et al. 27 conducted a randomized, placebo-controlled study assessing pantoprazole’s effects on glucose-insulin homeostasis in T2DM patients, finding reduced HbA1c levels and increased gastrin levels. It suggests that PPIs may enhance β-cell activity, stimulate insulin production, and slow gastric emptying, reducing postprandial hyperglycemia.2,28 This mechanism resembles incretin-based therapies, as gastrin, such as incretin hormones, promotes insulin secretion following meals. 29 However, a meta-analysis of one randomized trial and seven cohort studies found no significant association between PPI use and T2DM risk. 30 Conversely, a large Italian population-based study linked prolonged PPI use to a higher risk of T2DM, with increased risks of 19%, 43%, and 56% for use durations of 8 weeks to 6 months, 6 months to 2 years, and over 2 years, respectively. 31 These conflicting results, driven by variations in ethnicity, sex, diagnostic criteria, follow-up duration, and PPI assessment methods, highlight the critical need for standardized long-term studies to clarify PPIs’ role in T2DM management or risk.
Hypoglycemic episodes have been linked to several medications; the use of antibiotics in addition to insulin or sulfonylureas may increase the hypoglycemia likelihood. Triple-treatment prescriptions consisting of amoxicillin, clarithromycin, and a PPI increased the probability of hypoglycemic episodes significantly in individuals with diabetes and peptic ulcers. 32 Clarithromycin inhibits the cytochrome P450 3A4 (CYP3A4) enzyme. 33 This can potentiate the effects of sulfonylureas, meglitinides, or insulin in patients concurrently using the medications, increasing hypoglycemia risk. Amoxicillin is not directly linked to hypoglycemia but may contribute to gut microbiota changes or enhance the effects of other components of the triple therapy. 34 Thus, hypoglycemia during triple therapy is linked primarily to clarithromycin’s effect on insulin secretion and CYP3A4 inhibition, compounded by PPI-induced hypergastrinemia and potential gut hormone modulation. However, a two-treatment protocol with omeprazole, metronidazole, amoxicillin, and bismuth subcitrate and omeprazole, clarithromycin, and amoxicillin showed no significant differences in mean fasting plasma glucose and HbA1c levels. 35 Another study administered a 2-week treatment with metronidazole, amoxicillin, and bismuth subcitrate, and found no significant alteration in baseline insulin levels following triple medication during the follow-up year. 36 In our study, we observed a significantly decreased risk of T2DM in those who take PPIs and antibiotics medications, with aHRs of 0.86 (p < 0.001) and 0.79 (p < 0.001), respectively.
Prostaglandin analogs such as misoprostol reduce acid production and protect against NSAID-induced ulcers by enhancing mucus and bicarbonate secretion, although they are effective only for gastric ulcers. 2 A study found that acute misoprostol treatment does not enhance insulin-mediated glucose elimination in diabetic or nondiabetic groups, 37 though prostaglandins of the E series improve muscle glucose absorption in vivo and in vitro. 38 Pirenzepine, a muscarinic (M1) receptor inhibitor and an anticholinergic, lowers acid secretion and protects against mucosal lesions caused by irritants including alcohol and sodium hydroxide. 2 It effectively lowered blood glucose in T1DM patients, 38 while carbenoxolone improved glucose processing by increasing GLUT4 expression in skeletal muscle. 39 Our results showed that PUD treatment with prostaglandin analogs, anticholinergics, antacids, and carbenoxolone reduced T2DM risk by 35%, 33%, 24%, and 19%, respectively. We also observed that the reduced risk of T2DM in the non-PUD control cohort (Supplemental Table 4), while they were using the anti-PUD medications may reflect the similar underlying conditions and mechanisms among controls that prompted the use of these medications.
The correlation between PUD surgery and T2DM is multifactorial, involving changes in gastric physiology, hormonal regulation, and nutritional status. While some aspects of surgery may reduce T2DM risk (e.g., weight loss), others, such as altered incretin response or dumping syndrome, can disrupt glucose metabolism and potentially increase the risk.40,41 We found a significant difference in patients with PUD surgery in the increased risk of T2DM with aHR of 1.20 (p < 0.001).
The present study had several limitations. The primary drawback of this retrospective chart audit is its inability to establish a causal relationship between reduced T2DM risk and anti-PUD medications, as it only identifies statistical correlations. The analysis is based on documented intentions rather than controlled experimental data. First, the validity and reliability may have been compromised by the database utilized, which misclassified PUD and T2DM. However, we identified PUD, T2DM, and comorbidities with at least three claims for outpatient and/or hospitalization visits. To avoid subjects being misdiagnosed or coded incorrectly as PUD, T2DM, or comorbidities, we defined patients with at least three consensus diagnoses to ensure the validity of the diagnosis. In addition, Taiwan’s universal health insurance system incorporates peer review methods to minimize false positives. 42 Second, key T2DM risk factors, such as dietary habits, lifestyle choices (e.g., alcohol and tobacco use), and body mass index, were not accounted for. Lifestyle factors associated with PUD can increase T2DM risk. Smoking and excessive alcohol consumption impair glucose metabolism and increase insulin resistance, increasing the chances of contracting T2DM.43,44 Unhealthy diets high in refined carbs and fats worsen both PUD and T2DM by promoting obesity and insulin resistance.45,46 In addition, chronic stress and poor sleep, common in PUD patients, contribute to insulin dysregulation and glucose intolerance, further elevating T2DM risk.47,48 Genetic predispositions can increase T2DM risk in PUD patients through shared pathways involving inflammation and metabolic regulation. Genes such as IL-1, TNF-α, and IL-6 contribute to chronic inflammation, impacting insulin resistance in both conditions. 49 Variants in PPARγ and TCF7L2, associated with glucose metabolism, also affect insulin sensitivity, increasing T2DM susceptibility. 50 NOD1 and NOD2 polymorphisms linked to H. pylori infection in PUD have been shown to elevate T2DM risk by altering immune responses. 51 Some patients may have used over-the-counter PPIs, commonly taken for gastroesophageal reflux, which may not have been captured in the database. In addition, PUD severity could not be evaluated due to a lack of data on disease severity. Third, the database was originally designed for billing purposes, limiting access to personal information and anonymizing data, which prevented direct patient contact. Furthermore, clinical characteristics, such as blood glucose, HbA1c levels, imaging results, glycemic control measures, and serum ghrelin/GLP-1 data were unavailable, restricting the ability to assess T2DM severity in PUD patients. 52 Also, cases of T2DM were inferred from physician-documented medical records rather than comprehensive clinical data. Nevertheless, the diagnoses of T2DM and PUD were based on conventional symptomatic criteria, typical side effects, and imaging results, reviewed by experts to ensure reliability. 52 By controlling for comorbidities, the study minimized the confounding effects of drugs. Despite such efforts, retrospective studies inherently carry biases, including categorization bias, which can compromise statistical quality. However, Taiwan’s NHI program has high coverage and thorough claim reviews by reimbursement professionals and peer reviewers, ensuring the accuracy of diagnoses and disease codes. 52 To strengthen the findings, prospective studies or randomized controlled trials using additional data sources are required to better evaluate the relationships among PUD, anti-PUD medications, and T2DM. While retrospective analyses have intrinsic limitations, the results remain valid due to measures taken to mitigate non-differential categorization biases.
Conclusion
In conclusion, in the Taiwanese population, PUD was significantly associated with a higher risk of T2DM development. Individuals taking most anti-PUD medications showed a greater decrease in the incidence of T2DM risk compared to those not taking anti-PUD medications. Further research is necessary to determine whether certain anti-PUD medications could be clinically employed as novel antidiabetic medications and T2DM preventive agents.
Supplemental Material
sj-doc-1-tae-10.1177_20420188251323945 – Supplemental material for Impact of anti-peptic ulcer disease medications on type 2 diabetes mellitus risk in patients with PUD: a population-based retrospective cohort study
Supplemental material, sj-doc-1-tae-10.1177_20420188251323945 for Impact of anti-peptic ulcer disease medications on type 2 diabetes mellitus risk in patients with PUD: a population-based retrospective cohort study by Yi-Jen Fang, Hui-Hsia Hsieh, Cheng-Li Lin, Wan-Yi Lee, Chi-Hua Chen, Fuu-Jen Tsai, Bang-Jau You, Ni Tien and Yun-Ping Lim in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
