Abstract
Objective
We aimed to analyze the clinical characteristics of patients with psoriasis and determine the predictive factors of psoriatic arthritis (PsA).
Methods
This retrospective cohort study was performed among patients with psoriasis. Demographic and clinical data were collected. Psoriasis treatment was categorized as topical agents, phototherapy, oral therapy, and biologics. Predictive factors of PsA development were determined using logistic regression analyses.
Results
We included 330 patients with psoriasis, and 83 (25%) patients developed PsA. Thirty-eight (45.8%) patients who developed PsA were Malay, 24 (28.9%) were Chinese, and 21 (25.3%) were Indian. The mean age of patients with PsA was 54.2 (±15.8) years, and the duration from diagnosis of psoriasis to diagnosis of PsA was 36 (3.5–114) months. Predictive factors for developing PsA were female sex (odds ratio [OR] = 3.33, 95% confidence interval [CI] 1.78–6.22), presence of nail involvement (OR = 5.36, 95% CI 2.50–11.51), severe psoriasis (OR = 27.41, 95% CI 7.58–99.11), and oral systemic therapy prior to PsA diagnosis (OR = 4.09, 95% CI 2.04–8.22).
Conclusion
Patients with psoriasis who are female, have nail involvement, severe skin psoriasis, and require oral systemic therapy for psoriasis may have an increased risk of developing PsA.
Keywords
Introduction
Psoriatic arthritis (PsA) is a type of chronic, inflammatory arthritis associated with psoriasis, and 10% to 30% of patients with psoriasis subsequently develop PsA. 1 This may result in substantial joint destruction with permanent damage if it remains untreated. The overall prevalence of PsA in patients with psoriasis is estimated to be approximately 19.7%, with a rate of approximately 14% in Asian patients. 2 Nearly one-third of patients develop PsA after being diagnosed with psoriasis for 30 years, and the risk appears to remain constant after the initial diagnosis. 3 This implies that in addition to psoriasis, the risk of developing PsA does not change with time and other factors may contribute to development of the condition.
In daily clinical practice, it remains a challenge for clinicians to identify patients with psoriasis who have a higher risk of developing PsA owing to its chronicity and insidious onset. Several studies have identified various risk factors associated with the development of PsA including the affected skin areas, severity of skin psoriasis, presence of nail involvement, sex, smoking, and body mass index (BMI).4–8 However, the current evidence regarding these factors is limited and inconclusive, particularly in the Asian ethnic population. PsA occurs in 6% to 42% of White patients with psoriasis. This figure is considerably lower at 1% to 9% in the Asian population, suggesting that differences in race and ethnicity may play a role in the development of PsA. 9 Even within the Asian population, differences in race and ethnicity exist, which may be important in predicting PsA among patients with psoriasis. However, there is a lack of data from the Asian region in this regard. To the best of our knowledge, only two multi-ethnic Asian studies from Singapore have reported that PsA is more prevalent among Indian patients than among Chinese and Malay ethnic groups.8,10
Recognizing predictive factors in the development of PsA may improve clinicians’ decision-making in the monitoring and management of patients as early treatment has been shown to improve long-term outcomes. 11 Thus, the objective of this study was to evaluate the clinical characteristics of patients with psoriasis who have developed PsA, and to identify factors that predict PsA in a relatively large and diverse group of patients with psoriasis.
Methods
Study population
We conducted a retrospective cohort study among adult (aged 18 years and above) patients with psoriasis who attended the dermatology clinic at University of Malaya Medical Centre (UMMC) in Kuala Lumpur. Patients with psoriasis were consecutively recruited if they visited the dermatology clinic at any time between 1 January 2010 to 31 December 2019. These patients were then followed up. All data collected from the electronic medical records were de-identified. Ethical approval was obtained from the Medical Research Ethics Committee at UMMC on 7 June 2020 (approval number: 202042-8431). As the study data were collected solely in a retrospective chart review of patient records and all patient data were de-identified, patient consent was not required by the ethics board. This study was performed in accordance with the Declaration of Helsinki. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 12
All patients in the study were diagnosed with psoriasis by a dermatologist based on clinical features or skin biopsy. Patients who subsequently developed PsA had a verified diagnosis from a rheumatologist and fulfilled the Classification Criteria for Psoriatic Arthritis criteria. 13 Patients with unconfirmed diagnoses of psoriasis or PsA or those with other overlapping conditions, including rheumatoid arthritis or other connective tissue diseases, were excluded from the study.
Clinical characteristics and comorbidities
Demographic data for all recruited patients were collected from the electronic medical records and included patients’ sex, age, race and ethnicity, education level, marital status, smoking, and alcohol intake. BMI was categorized as normal, pre-obese, or obese according to the World Health Organization classification. 14 Comorbidity data collected in this study included hypertension, diabetes mellitus, ischemic heart disease, dyslipidemia, and osteoporosis, according to the International Classification of Diseases Tenth Revision 15 and were identified from patients’ electronic medical records.
Clinical characteristics of patients with psoriasis and/or PsA included age at diagnosis of psoriasis, duration of psoriasis, family history of psoriasis, severity of skin involvement, presence of nail involvement, and treatment for psoriasis prior to diagnosis of PsA. The extent of skin involvement was assessed using body surface area (BSA) and was categorized as mild (<10%), moderate (10% to 30%), and severe (>30%). 16
Patients diagnosed with psoriatic arthritis (PsA)
In patients who were diagnosed with PsA, the collected data included age at PsA diagnosis, duration between diagnosis of psoriasis and diagnosis of PsA, erythrocyte sedimentation rate (ESR) and/or C-reactive protein (CRP) upon diagnosis of PsA, and extent of joint involvement assessed according to the number of swollen and tender joints at the first presentation to the rheumatology clinic. To better distinguish between oligoarthritis and polyarthritis, we defined patients with active polyarthritis as having at least six tender and swollen joints, with an elevated inflammatory marker of either ESR or CRP.17,18
Treatment for psoriasis
The treatment used for psoriasis was categorized into four groups: topical agents (including those used currently or previously), phototherapy, oral systemic therapy (including conventional synthetic disease-modifying anti-rheumatic drugs [DMARDs] and acitretin), and biologic DMARDs. Topical agents refer to topical steroids, tar preparation, emollient, dithranol preparations, and calcipotriol. In patients diagnosed with PsA, we defined the use of oral systemic therapy and biologic DMARDs in this study as treatment initiated for psoriasis (not for PsA) before the diagnosis of PsA.
Statistical analysis
The collected data were analyzed using IBM SPSS version 26 (IBM Corp., Armonk, NY, USA). Descriptive data are expressed as mean ± standard deviation if normally distributed and median and interquartile range if non-normally distributed. Categorical data are described as frequency and percentage. For inferential analysis, categorical data are analyzed using the chi-square test. Independent variables were screened using simple logistic regression. Only variables with a p-value <0.25 or of clinical relevance were included in multiple logistic regression analysis to determine the predictive factors of PsA. A value of p < 0.05 was considered statistically significant.
Results
Patient demographics
A total of 330 patients with underlying psoriasis, with or without PsA, were included in the analysis. The mean age was 53 (±18.85) years and there were 180 (54.5%) male patients. As for race and ethnicity, 143 (43.3%) patients were Malay, 110 (33.3%) were Chinese, and 72 (21.8%) were Indian. Most patients (n = 245, 74.2%) had an education level of secondary school or above. The mean duration of follow-up for all patients in this cohort was 10.2 (±10.3) years (Table 1).
Sociodemographic characteristics of patients.

Chi square test was used, with the significance level set at 0.05.
Independent
*p-value <0.05.†Data available for N = 303 patients (Pso-only, n = 227; PsA, n = 76).‡Data available for N = 327 patients (Pso-only, n = 244; PsA, n = 83).§Data available for N = 321 patients (Pso-only, n = 239, PsA, n = 82).‖Data available for N = 324 patients (Pso-only, n = 241, PsA, n = 83).
PsA, psoriatic arthritis; SD, standard deviation.
Among 324 patients, nail involvement was observed in 191 (59%) patients; 220 (71.9%) out of 306 patients had mild skin involvement, 66 (21.6%) out of 306 had moderate, and 20 (6.5%) out of 306 had severe skin involvement in BSA assessment. Out of 317 patients, 122 (38.5%) patients had a normal BMI (BMI 22.54 kg/m2), 106 (33.4%) patients were in the pre-obesity category, and 79 (24.9%) patients were categorized as obese. The most prevalent comorbidity was hypertension (n = 148, 44.8%), followed by diabetes mellitus (n = 103, 31.2%), dyslipidemia (n = 124, 37.6%), ischemic heart disease (n = 32, 9.7%), and osteoporosis (n = 15, 4.5%). Nearly all patients (n = 325, 98.5%) were using topical therapy and 106 (32.1%) patients had received at least one oral systemic therapy (Table 2).
Clinical findings of patients with psoriasis only and psoriatic arthritis.
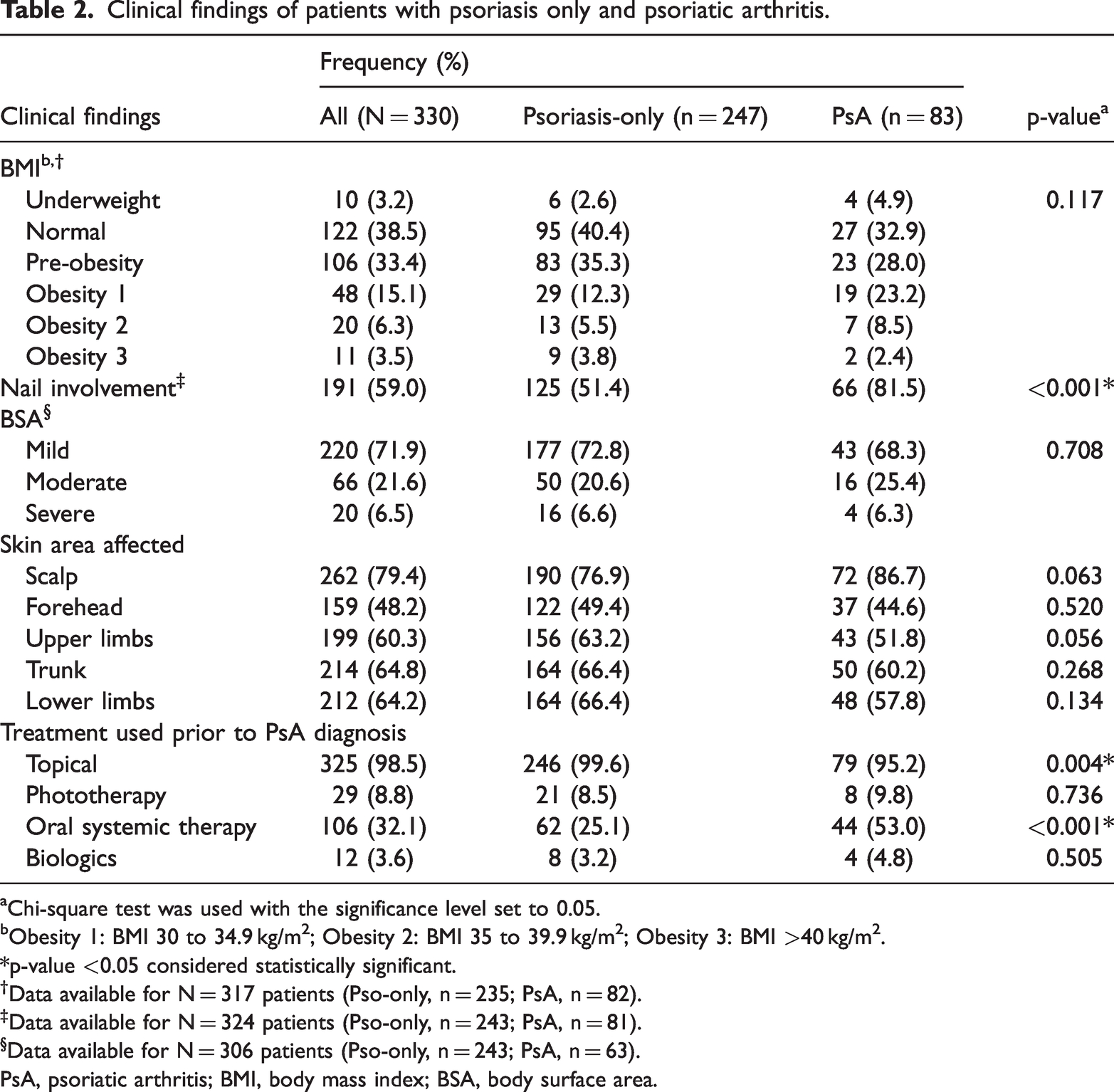
Chi-square test was used with the significance level set to 0.05.
Obesity 1: BMI 30 to 34.9 kg/m2; Obesity 2: BMI 35 to 39.9 kg/m2; Obesity 3: BMI >40 kg/m2.
*p-value <0.05 considered statistically significant.†Data available for N = 317 patients (Pso-only, n = 235; PsA, n = 82).‡Data available for N = 324 patients (Pso-only, n = 243; PsA, n = 81).§Data available for N = 306 patients (Pso-only, n = 243; PsA, n = 63).
PsA, psoriatic arthritis; BMI, body mass index; BSA, body surface area.
Comparing patients with psoriasis and PsA
Among the 330 patients with psoriasis, 83 (25%) were subsequently diagnosed with PsA; the remaining 247 (75%) patients remained diagnosed with psoriasis only during the study period. The incidence of PsA was determined to be 2.52 per 100 patient-years (95% confidence interval [CI] 2.04–3.14). Among the 83 patients with PsA, the mean age was 54.2 (±15.8) years and there were 50 (60.2%) female patients. The duration between the diagnosis of psoriasis and PsA was 36 (3.5–114) months. According to race and ethnicity, 38 (45.8%) patients who developed PsA were Malay, 24 (28.9%) were Chinese, and 21 (25.3%) were Indian. There was no significant difference with respect to developing PsA among racial and ethnic groups (Table 1).
For patients with PsA, 43 (68.3%) had mild skin involvement based on BSA, 16 (25.4%) had moderate skin involvement, and four (6.3%) had severe skin involvement. The scalp (n = 72, 86.7%) was the most affected area for patients with PsA, followed by the trunk (n = 50, 60.2%), lower limbs (n = 48, 57.8%), and upper limbs (n = 43, 51.8%). Seven (12.3%) of 57 patients with available data presented with active polyarthritis at the initial diagnosis of PsA (Table 2).
The presence of nail involvement was higher in patients with PsA compared with those that had psoriasis only (p < 0.001). There was a higher rate of oral systemic therapy (prescribed for skin psoriasis) among patients who eventually developed PsA compared with those who only had skin psoriasis (p < 0.001), but no difference was found with respect to biologic therapy between these two groups (Table 2). There were no significant differences in comorbidities between patients with PsA and psoriasis only (Figure 1).

Comorbidities among patients with psoriasis only and those with psoriatic arthritis. PsA, psoriatic arthritis.
Predictive factors of PsA
In univariate analysis, female sex, the presence of nail involvement, use of topical treatment, use of oral systemic therapy prior to diagnosis of PsA, education level, marital status, presence of comorbidities, BMI, and area of psoriasis were identified as potential risk factors (Table 3). Further analysis in multivariate regression was adjusted for the following variables: sex, education, marital status, diabetes mellitus, nail involvement, affected body area, topical therapy, and oral systemic therapy use. The results showed that female sex (odds ratio [OR] 3.33, 95% CI 1.78–6.22), nail involvement (OR 5.36, 95% CI 2.50–11.51), severe skin psoriasis (OR 27.41, 95% CI 7.58–99.11), and prior oral systemic therapy use (OR 4.09, 95% CI 2.04–8.22) were significant predictive factors of PsA development (all p < 0.001) (Table 4).
Simple logistic regression analysis of factors associated with developing psoriatic arthritis.

Obesity 1: BMI 30 to 34.9 kg/m2; Obesity 2: BMI 35 to 39.9 kg/m2; Obesity 3: BMI >40 kg/m2.
PsA, psoriatic arthritis; BSA, body surface area; b, beta estimate; OR, odds ratio; CI, confidence interval; df, degrees of freedom.
Factors associated with developing psoriatic arthritis in multiple logistic regression analysis.
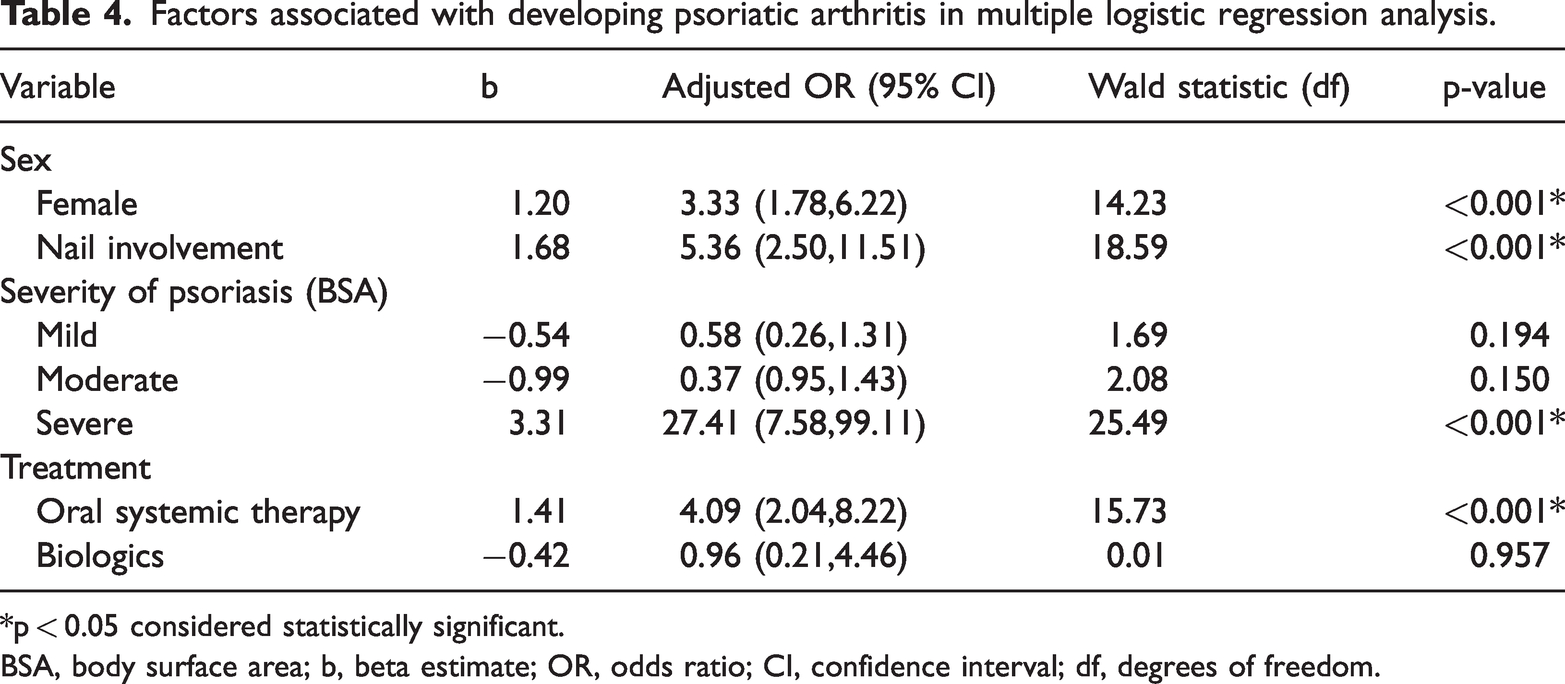
*p < 0.05 considered statistically significant.
BSA, body surface area; b, beta estimate; OR, odds ratio; CI, confidence interval; df, degrees of freedom.
Discussion
In this study, conducted among a varied group of Asian patients with psoriatic disease and including data over a span of nearly 10 years, we found that female sex, the presence of nail involvement, severe skin psoriasis, and the use of oral systemic therapy prior to diagnosis of PsA were significant predictive factors for developing PsA.
Our study findings suggest that female sex is a predictor of PsA development in patients with psoriasis. However, the available evidence on how sex affects the risk for PsA and the reasons behind this is scarce. In one study conducted in Scandinavia, there was a higher prevalence of PsA among female individuals, 19 whereas other large population-based studies found that PsA was nearly equally distributed between women and men.20,21 Although the reason for female sex being a risk factor remains speculative, it is possible that hormonal differences between the sexes as well as variations in the distribution of genes and polymorphisms within the major histocompatibility complex could play a role in the development and detection of this condition.22–24
Our study showed that patients with psoriatic nail involvement have a significantly higher risk of developing PsA. The results in our study population are in line with those of previous studies reporting similar findings.6,25,26 McGonagle and colleagues put forth a hypothesis suggesting an intimate anatomical connection between the nails and the entheses connecting to the extensor tendons and collateral ligaments. 27 This hypothesis and the inflammatory changes that occur could potentially explain the increased risk of developing PsA in patients with psoriatic nail involvement. Nevertheless, more comprehensive assessment in psoriatic patients with nail involvement is strongly advised in routine clinical practice as this may aid in detecting PsA at an earlier stage.
In our study, we found a significant association between the severity of skin psoriasis and the development of PsA. It is widely known that more severe skin involvement is associated with PsA, which has also been evaluated in a recent meta-analysis of 29 studies. 28 However, it is unclear whether the severity of skin psoriasis can predict which patients with psoriasis will transition to PsA. The same meta-analysis also examined this issue; based on three prospective studies that reported inconsistent results, the authors could not conclude whether the severity of skin psoriasis predicts the future development of PsA.6,29,30 Despite the limitations of our retrospective study, we demonstrated that patients with severe skin psoriasis have a greater risk of developing PsA. This adds to the body of evidence that severe skin psoriasis may be a strong predictor of PsA development.
Our study showed that patients with psoriasis who received oral systemic therapy for skin psoriasis had a significantly greater likelihood of developing PsA whereas prior use of biologics was not a predictive factor. Oral systemic therapy is used for psoriasis when topical therapy and/or phototherapy do not result in or maintain skin clearance. 31 It is therefore logical to assume that patients who were prescribed oral systemic therapy for psoriasis in our cohort were those with more severe skin involvement. Because we have shown that more severe skin psoriasis is a predictor of PsA development, it is expected that oral systemic therapy is also a predictive factor for developing PsA. This finding can also be explained by a protopathic bias owing to reverse causality because skin psoriasis is an early manifestation of PsA.32,33 In our study, we analyzed the oral systemic therapy group separately from the biologic treatment group to eliminate any potential protective effects of biologic therapy. However, skin assessments were only conducted at the time of diagnosis and there was no available information on continuous assessments. With regard to biologic therapy, the number of patients on biologic therapy was small in both the psoriasis-only and PsA groups because access to funding for biologic therapy is limited locally; this could have potentially affected the results. Recent studies have reported conflicting results regarding the protective effect of biologic therapies against PsA development.34–37 Although the link between the two remains unclear, further investigation is needed regarding the paradoxical effect of advanced systemic therapies on the onset of PsA.29,38
The prevalence of PsA has been previously reported to differ among different racial and ethnic groups; however, treatment tends to be similar across all races and ethnicities.39,40 Our study did not find any association between race and ethnicity and the risk of developing PsA, nor did we observe any differences in race and ethnicity between the psoriasis-only and PsA groups. This contrasts with a previous study in Singapore reporting that Indian patients were more prone to developing PsA. 8 Nonetheless, environmental factors such as smoking and alcohol intake as well as genetic differences may play a role in PsA development. These factors should be taken into account as previous studies have linked smoking and alcohol consumption to psoriasis and treatment outcomes, which may explain the divergent findings. 41
Our study has several limitations. This was a single-center, retrospective study, which relied on a review of patients’ electronic medical records. A longer follow-up period could improve the accuracy of the data as patients may develop PsA beyond the study duration. Additionally, we could not account for other confounding factors, such as medication adherence or compliance. Protopathic bias could also affect the results on oral systemic therapy use in this study. We have taken steps to reduce the effects of confounding with use of a logistic regression model to estimate the propensity score; however, because this was an observational study, the true propensity score is unknown. Other assessment measures, such as the Psoriasis Area and Severity Index 42 or Disease Activity in Psoriatic Arthritis scores 43 were not available for use in this study.
Conclusion
The findings of this study suggested that patients with psoriasis who are female, have nail involvement, severe skin psoriasis, and use of systemic therapies for psoriasis are at greater risk of developing PsA. Patients may benefit from increased screening, which can lead to earlier diagnosis and treatment. More extensive studies are needed to better understand the predictive factors of PsA.
Footnotes
Acknowledgments
We thank Dr Ruhana Che Yusoff, biostatistician at the Department of Medicine, Faculty of Medicine, University of Malaya for her assistance in performing the statistical analyses for this study.
Authors’ contributions
Conceptualization: WYL, YCT, FY
Data curation: WYL, YCT, LSLP, WHH, NAAF, SSY, ZK
Formal analysis: WYL, YCT
Investigation: WYL, YCT, FY
Methodology: WYL, YCT, ZK, FY
Project administration: WYL, YCT
Resources: YCT, LSLP, FY
Supervision: YCT, FY
Writing-original draft: WYL, YCT, LSLP
Writing-review & editing: LSLP, FY
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
