Abstract
Insulin allergy is a difficult challenge in treating patients with poor control of type 2 diabetes mellitus (T2DM), who are at risk for developing diabetic ketoacidosis (DKA) and hyperglycemic hyperosmolar syndrome (HHS). Currently, there are no established guidelines that provide standardized treatment protocols for this issue. We report the case of a 50-year-old man with T2DM and poorly controlled blood glucose who required insulin therapy but was allergic to insulin. This allergic reaction involved a progressive and severe rash and itch that occurred at the injection site and on the patient’s chest and back. After successful desensitization and 3 months of follow-up, we summarize the treatment protocol for insulin allergy in high-risk patients. We used a regimen of continuous subcutaneous insulin infusion (CSII) with Basal Insulin Desensitization, including CSII with rapid-acting insulin and the intradermal injection of long-acting insulin for simultaneous desensitization. After successful desensitization and 3 months of follow-up, the patient achieved stable glycemic control (HbA1c: 7.2%, fasting blood glucose: 6.5 mmol/L, postprandial glucose: 8.1 mmol/L) with no recurrence of allergic reactions. We summarize the treatment protocol for insulin allergy in high-risk patients.
Plain language summary
We report a case of successful treatment for insulin allergy in a patient who experienced a progressive and severe rash and itch at the injection site and on his chest and back after insulin injection. In this case report, we provide a flowchart summarizing the management of insulin allergy.
Keywords
Introduction
Poor glycemic control can cause acute metabolic complications such as diabetic ketoacidosis (DKA) and hyperglycemic hyperosmolar syndrome (HHS). 1 However, for insulin-allergic patients in a hyperglycemic crisis, glycemic control is difficult to achieve unless a nonallergenic insulin preparation is selected or if desensitization therapy with incremental dose enhancement for tolerance induction is successful. 2 Continuous Subcutaneous Insulin Infusion (CSII) stands out as the optimal desensitization strategy, 3 which permits progressively increasing, yet small, doses of insulin to maintain minimal yet gradually increasing blood concentrations until they reach therapeutic levels. 3 However, it is still unknown what management strategies should be employed for patients who have successfully undergone desensitization to rapid-acting insulin but later refuse long-term use of an insulin pump.4–6 In this report, we utilized CSII with Basal Insulin Desensitization (CSIIBID) to successfully treat diabetes mellitus (DM) complicated by insulin allergy. Current guidelines lack standardized protocols for insulin allergy in hyperglycemic crises. Our CSIIBID strategy uniquely combines continuous subcutaneous rapid-acting insulin infusion with intradermal long-acting insulin desensitization. This dual approach addresses both acute glycemic stabilization and long-term tolerance, particularly for patients reluctant to use insulin pumps post-discharge. To our knowledge, this is the first reported protocol integrating these modalities. Through a detailed history and focused testing, we have outlined a flowchart for insulin desensitization treatment, thereby providing a new therapeutic strategy.
Case presentation
Chief complaints
A 50-year-old man presented to the endocrinology department with signs and symptoms of poor glycemic control.
History of present illness
The patient had been diagnosed with T2DM 10 years prior (fasting blood glucose 25.7 mmol/L), with no polyuria, weight loss, polydipsia, or lethargy, and his body mass index was 22.99 kg/m2. He was prescribed insulin glargine and developed a pruritic rash at insulin glargine injection sites within 2 weeks of initiation. The rash and itch spread to the patient’s abdomen, chest, head, and face in 3 days. After insulin cessation, his allergic symptoms gradually improved in 1 week. Symptoms recurred with detemir (3 days) but resolved with insulin cessation and oral antidiabetic drugs therapy (metformin 2000 mg/day). Glycemic control deteriorated (HbA1c: 11.3%, fasting blood glucose: 10 mmol/L, and 2-h postprandial glucose: 15–20 mmol/L), prompting hospitalization.
History of past illness
The patients had no other specific medical history.
Personal and family history
The patient denied any family history of diabetes.
Physical examination
On physical examination, the patient’s vital signs were as follows: body temperature, 36.5°C; blood pressure, 122/61 mmHg; heart rate, 77 beats per min; respiratory rate, 18 breaths per min; height, 172 cm; and weight, 68 kg. He exhibited clear consciousness, moderate nutrition, normal skin elasticity, equal and round pupils, normal light sensitivity and light reflex, a supple neck, smooth breathing, strong heart sounds, a normal abdomen, a normal spine and limbs, normal muscle strength, and normal joint reflexes.
Laboratory examinations
Laboratory investigations revealed a random blood glucose level of 26.8 mmol/L with undetectable blood ketones (<0.3 mmol/L), consistent with non-ketotic hyperglycemia. Persistently low C-peptide levels (0.37–0.7 ng/mL; normal range: 0.78–1.9 ng/mL) confirmed endogenous insulin deficiency, necessitating exogenous insulin administration despite allergy risks. Concurrently, an elevated HbA1c of 11.3% (100 mmol/mol) underscored prolonged poor glycemic control. Urinalysis demonstrated glucosuria (3+) without ketonuria, and autoimmune diabetes antibodies (including GAD65, IA-2, and ZnT8) were negative (Table 1).
Biochemical analysis of a patient with severe hyperglycemia and insulin deficiency.
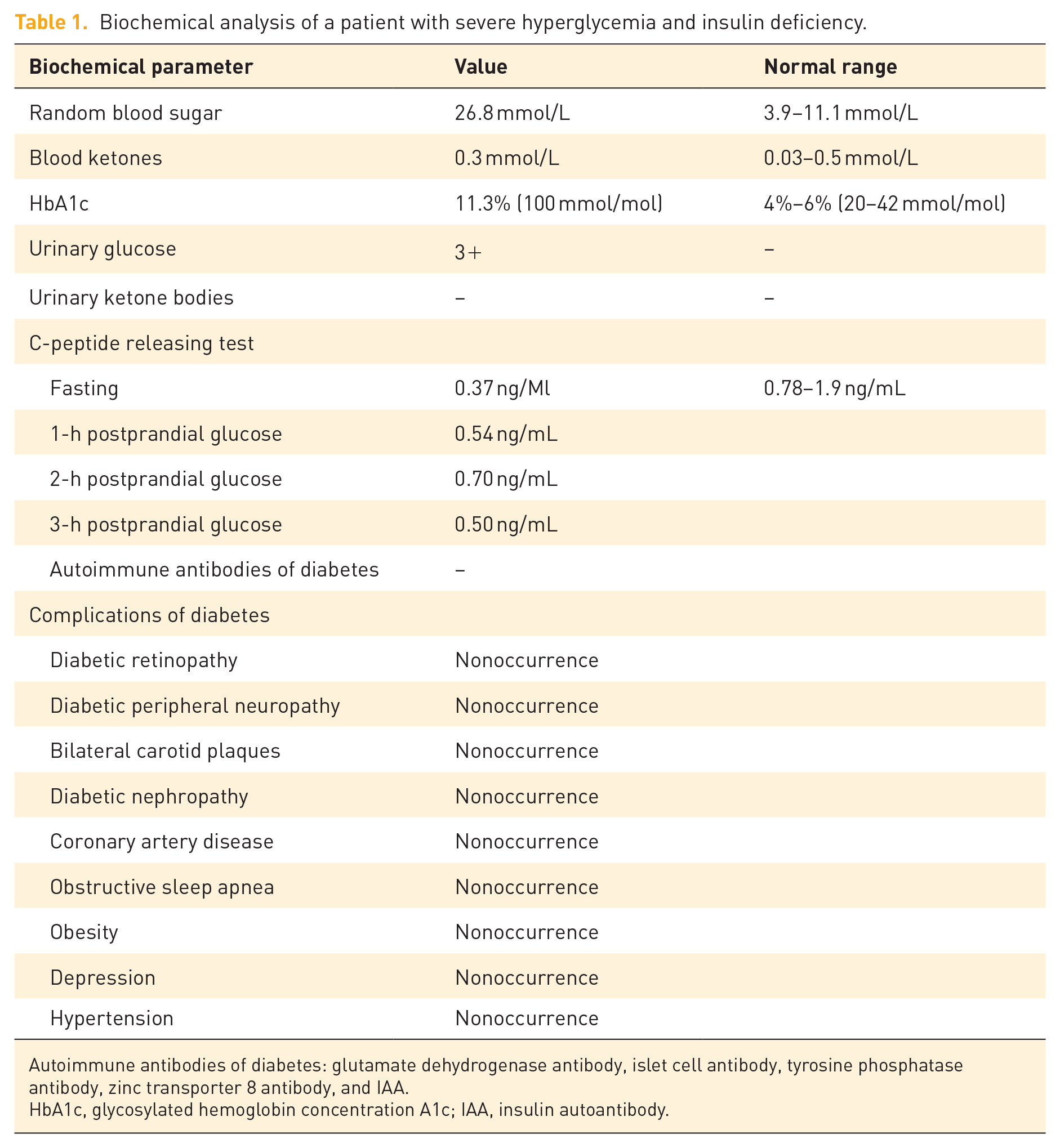
Autoimmune antibodies of diabetes: glutamate dehydrogenase antibody, islet cell antibody, tyrosine phosphatase antibody, zinc transporter 8 antibody, and IAA.
HbA1c, glycosylated hemoglobin concentration A1c; IAA, insulin autoantibody.
Imaging examinations
The patient underwent no imaging examinations.
Final diagnosis
The final diagnosis was T2DM and insulin allergy.
Treatment
Rapid-acting insulin through the CSII desensitization method
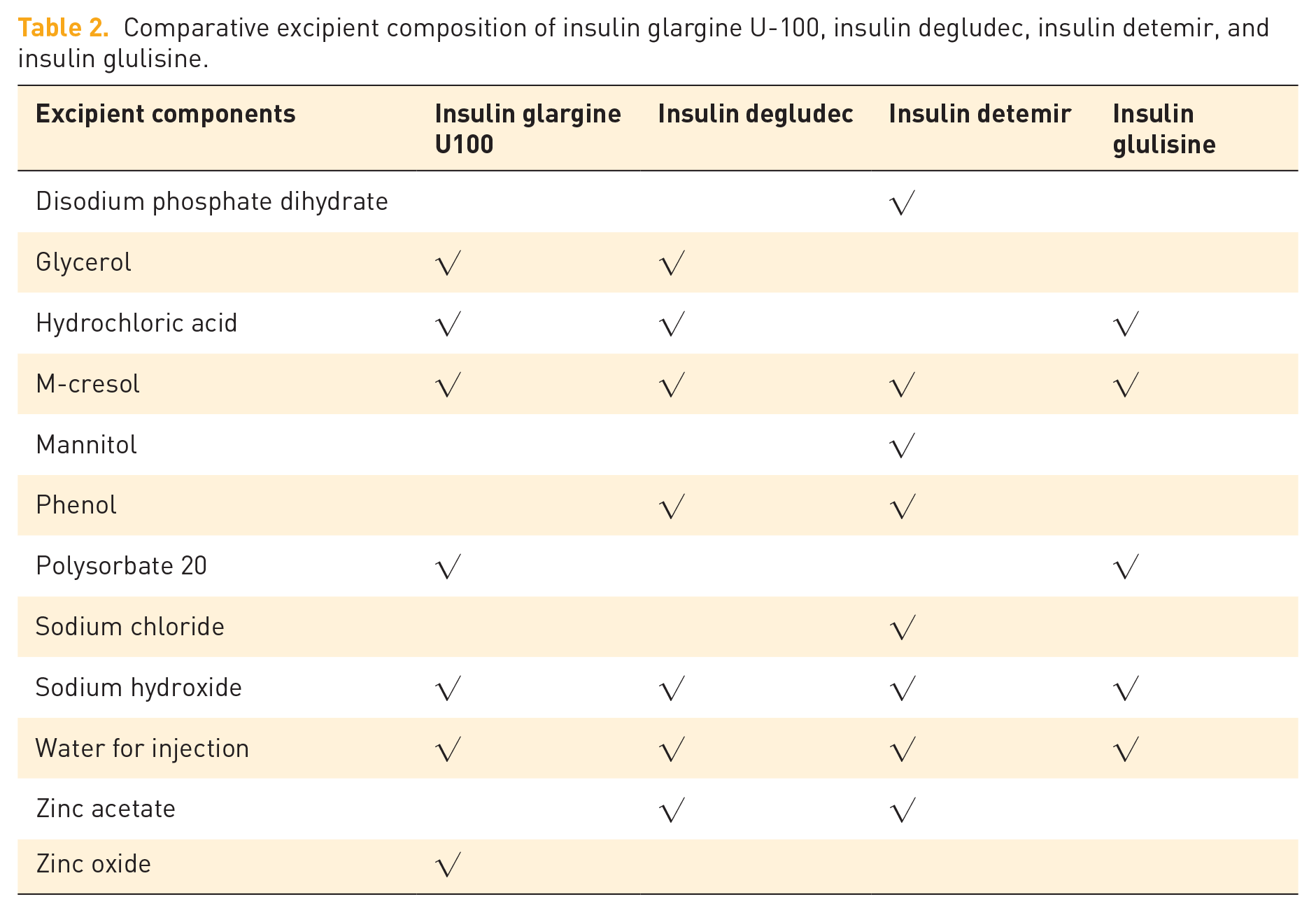
Considering the contribution of primary structure (amino acid replacement) and aggregates to immunogenicity,7,8 we selected insulin glulisine for desensitization, which showed minimal self-association and stability in monomeric and dimeric states in solution without zinc (Table 2).9,10 The initial hypoglycemic regimen was as follows: oral metformin tablets combined with insulin glulisine were injected subcutaneously through a pump at 0.1 IU/h continuously. The dose was gradually increased to 0.3 IU/h over 4 days to a maximum of 25 IU/day. The patient experienced mild rash and itching of the scalp with the injection, and no symptoms were detected the next day.
Comparative excipient composition of insulin glargine U-100, insulin degludec, insulin detemir, and insulin glulisine.
Basal insulin desensitization
Following CSII desensitization, the patient’s blood glucose level was well controlled, and insulin glulisine was pumped. Reluctant to continue insulin pump therapy after discharge, the patient required one more basal insulin administration, involving intermediate or long-acting insulin. We desensitized the patient with basal insulin, including glargine, degludec, and detemir. Intradermal testing of these long-acting insulins was conducted along with CSII treatment, and the insulin that caused the mildest local reaction was selected for desensitization treatment (Table 3). First, a 1:1000 (0.1 IU/mL) dilution of insulin, with normal saline used as a control, was prepared. Each 0.1 mL volume of solution was injected within 5 min. The injection site was located 5 cm from the umbilicus, and the interval between the two adjacent injection points was more than 3 cm. The result was considered positive if the size of the wheal was greater than that of the negative control (3 mm) within 15 min. The wheal caused by insulin glargine disappeared after 19 min, that caused by insulin degludec disappeared after 30 min, and that caused by insulin detemir disappeared after 14 min. However, 24 h later, the injection sites of insulin glargine and insulin detemir, but not insulin degludec, presented new wheals (Table 3 and Figure 1).
Comparative allergic reaction characteristics (pruritus and rash size) following intradermal testing of insulin glargine U-100, insulin degludec, and insulin detemir over time.

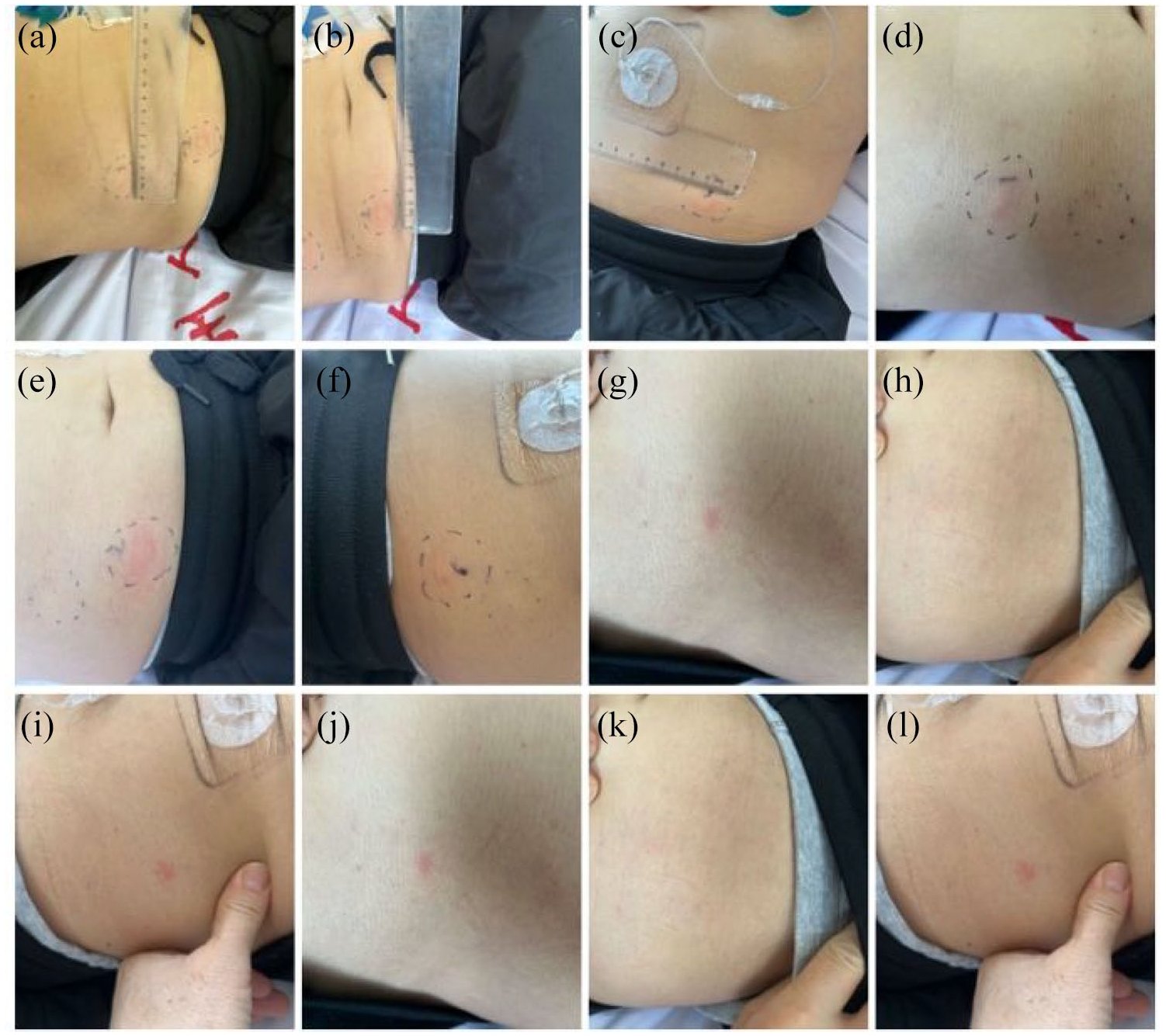
Time-course comparison of cutaneous hypersensitivity reactions to intradermal insulin glargine, degludec, and detemir (1:1000 dilution) at 10 min, 30 min, 2 h, and 24 h post-injection. Insulin preparations: (a) Glargine, (b) Insulin degludec, and (c) Insulin detemir in 10 min. (d) Insulin glargine, (e) Insulin degludec, and (f) Insulin detemir in 30 min. (g) Insulin glargine, (h) Insulin degludec, and (i) Insulin detemir at hour 2. (j) Insulin glargine, (k) Insulin degludec, and (l) Insulin detemir 24 h later.
To confirm this result, we repeated the experiment (Figure 2). Interestingly, the wheals caused by insulin glargine and insulin detemir disappeared within 2 h but reappeared after 24 h, similar to the initial results (Figure 3). The wheal caused by insulin degludec disappeared within 2 h, and no wheals reappeared after 24 h. Therefore, insulin degludec was selected for further desensitization.

Delayed wheal reactions to intradermal basal insulins: comparative 24- and 48-h hypersensitivity patterns for insulin glargine, degludec, and detemir with safety confirmation. Delayed wheal reactions (rectangle = 24 h reaction, circle = 48 h reaction) after intradermal injection of basal insulins: (a) glargine, (b) degludec, (c) detemir. Degludec showed no recurrence, confirming safety.

Integrated management protocol for insulin allergy complicated by hyperglycemia with risk of DKA and HHS.
Insulin degludec desensitization therapy
After written informed consent was obtained, insulin degludec desensitization was performed (Table 4). From Day 1 to Day 4, incremental insulin doses were given subcutaneously every 4 h from 0.001 IU to a total dose of 8 IU by the end of Day 4, with no allergic reactions, including itch or skin rashes. Finally, the hypoglycemic regimen was formulated as follows: metformin 500 mg orally, triple daily; three boluses of insulin glulisine injected before three meals: 5, 4, and 3 IU; and insulin degludec injected 13 IU before bedtime.
Daily desensitization protocol for insulin degludec: dosage escalation, cumulative dose, and local reaction monitoring (units: IU/mm).

Interval between injections: 4 h.
Outcome and follow-up
During follow-up over the next 3 months, no allergic reactions were detected, and good glycemic control was achieved.
Discussion
This case demonstrates the successful application of the CSIIBID protocol in managing a high-risk patient with insulin allergy and impending hyperglycemic crisis. By combining continuous subcutaneous rapid-acting insulin infusion with intradermal long-acting insulin desensitization, we achieved sustained glycemic control (HbA1c reduction from 11.3% to 7.2%) and allergy resolution over 3 months. This approach addresses a critical gap in current guidelines, which lack standardized strategies for insulin-allergic patients at risk of DKA/HHS.
Mechanisms of desensitization
The CSIIBID protocol integrates CSII with intradermal desensitization of long-acting insulin, leveraging synergistic mechanisms to address both acute glycemic stabilization and long-term tolerance. Continuous infusion minimizes antigen exposure by maintaining low, incremental insulin doses, thereby depleting local hypersensitivity mediators (e.g., histamine) and avoiding IgE-mediated degranulation.4,11 Recent studies highlight that sustained insulin delivery through CSII reduces systemic inflammation by modulating cytokine profiles, such as downregulating CXCL10 expression, which is linked to β-cell apoptosis.12,13 In addition, intradermal desensitization induces gradual immune tolerance through dendritic cell modulation, a process enhanced by the structural stability of insulin analogs like glulisine.2,14 Behavioral interventions, such as exercise, further amplify this effect by upregulating protective factors such as Metrnl, which reduces cytokine-induced β-cell apoptosis by 46%.15,16
Comparison of insulin preparations
Structural and additive differences among insulin analogs critically influence their immunogenicity. Glulisine (rapid-acting) lacks zinc and incorporates methionine substitutions (B28/B29), reducing aggregate formation and epitope exposure. 17 Degludec (long-acting) features a hexamer-stabilizing deletion (B30), which lowers immunogenicity compared to glargine and detemir. The latter’s fatty acid side chains and acidic formulations (pH 4.0 in glargine) may promote epitope recognition.18–20 Additives also play a role: m-cresol in glargine/detemir is a known allergen, whereas degludec uses phenol-glycerol stabilizers with lower sensitization potential. Clinical data from a cohort of 162 patients with early onset diabetes revealed that T2DM patients exhibited higher fasting C-peptide levels (median 1.48 ng/mL) but lower insulin sensitivity indices, emphasizing the need for tailored insulin selection. 21
Immunological considerations
While IgE-mediated type I hypersensitivity was presumed in this case (acute onset <2 h), the delayed recurrence of wheals with glargine/detemir at 24 h suggests mixed type I/IV mechanisms. 22 The absence of IgE testing—a limitation—underscores the need for accessible immunophenotyping in resource-limited settings. 23 Future protocols should integrate skin prick tests, basophil activation assays, and IgG/IgE profiling to stratify patients by hypersensitivity type. Notably, the successful desensitization with degludec implies that T-cell tolerance (type IV) may dominate in prolonged protocols, offering a therapeutic target for refractory cases. 24
This patient’s allergic reaction was characterized by a rash and itch that occurred within 2 h at the insulin injection site, so we inferred that this patient had a type I hypersensitivity reaction. As the immunoassay was not available in our hospital, the IgE of the patient was not assessed. While IgE testing was unavailable, the temporal onset of symptoms strongly supports a type I hypersensitivity reaction. Future studies should validate this with immunologic assays.
Therapeutic algorithm
If a patient experiences hyperglycemia accompanied by DKA or HHS, the treatment goals should generally include the correction of dehydration, hyperglycemia, hyperosmolality, electrolyte imbalance, and increased ketonemia, as well as the identification and treatment of precipitating event(s). 1 Moreover, a rapid-acting insulin should be promptly selected for desensitization treatment via CSII. The choice of insulin should prioritize the preparation with the lowest immunogenicity. 25 After successful desensitization, the blood glucose levels should be maintained within a safe range. This article outlines the therapeutic algorithm for managing patients with insulin allergy who are at risk of DKA and HHS.
Conclusion
As anaphylaxis, DKA and HHS are all life-threatening conditions, it is important to expand the literature on successful insulin desensitization protocols for patients with DKA and HHS. Therefore, we propose a desensitization model of CSIIBID for patients who have extremely high blood glucose levels but who are allergic to insulin. Patients with insulin allergy at risk of acute DKA and HHS should be managed. CSIIBID may provide a practical and scalable approach to managing insulin allergy in hyperglycemic emergencies
Supplemental Material
sj-pdf-1-tae-10.1177_20420188251362093 – Supplemental material for Management strategies for patients with insulin allergy at risk of acute complications: experience from a case report
Supplemental material, sj-pdf-1-tae-10.1177_20420188251362093 for Management strategies for patients with insulin allergy at risk of acute complications: experience from a case report by Min Wen, Chun-Yu Song, Rui Wang, Bo-Rong Qiang and Guo-Li Du in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
Acknowledgements
We gratefully acknowledge all the physicians and nurses in our department who participated in the clinical and biochemical data collection, diagnosis, and therapy of these insulin-allergic patients.
Declarations
Supplemental material
Supplemental material for this article is available online.
