Abstract
Background:
There is interest in repurposing bicalutamide for gender-affirming hormone therapy, but little data regarding efficacy and safety in the transgender population.
Objectives:
To determine the effect of bicalutamide on serum total testosterone concentrations and liver function. Given bicalutamide is a pure androgen receptor antagonist, we hypothesized that serum total testosterone concentrations would be higher than the cisgender female reference range and those recommended for transgender individuals in consensus guidelines.
Design:
Case series.
Methods:
We identified transgender people attending outpatient clinics who were using bicalutamide for gender affirmation. The primary outcome was serum total testosterone concentration. Secondary outcomes included serum alanine transferase (ALT) concentration.
Results:
Twenty-four transfeminine people prescribed bicalutamide were identified. The median (interquartile range) age was 29 (24–38) years, bicalutamide dose 25 (25–50) mg daily and bicalutamide duration 6 (4–10) months. The median serum total testosterone concentration was 7.7 (0.7–17.5) nmol/L, luteinizing hormone 2.9 (0.5–6.0) IU/L and ALT 20 (13–29) IU/L. One individual had asymptomatic transaminitis. Six individuals discontinued bicalutamide due to a lack of perceived benefit.
Conclusion:
Bicalutamide was associated with significant variation in serum total testosterone concentration, likely related to concomitant estradiol or previous anti-androgen therapy. Median serum ALT concentrations remained within the normal reference range. Guideline recommendations for serum total testosterone within the cisgender female reference range may be inappropriate for people using bicalutamide. Further research is needed to establish the longer-term efficacy and safety of bicalutamide in the transfeminine population.
Plain language summary
Bicalutamide is a drug that blocks the effect of testosterone. The transgender community are interested in its use for feminisation, but there is little research of its use in this population. In this study, we looked at blood test results of transgender people using bicalutamide for hormone therapy. We found that blood testosterone levels varied significantly between individuals and that overall markers of liver function remained normal. Guidelines for transgender hormone therapy recommending a target blood testosterone level in the female range may not be applicable to bicalutamide as it acts mainly by blocking the effect rather than production of testosterone. Reassuringly, bicalutamide appeared to be safe in our study. Further research is needed to see if it helps with feminisation compared to standard treatment.
Introduction
The use of steroidal anti-androgens cyproterone acetate and spironolactone for gender affirmation has been well described since the 1980s.1,2 However, while the doses of cyproterone acetate typically used today are now much lower than historically described (e.g. 10 mg daily vs 50 mg daily) some concerns remain about the risk of mood disturbance, dose-dependent hyperprolactinaemia 3 and an increased risk of meningioma with increasing cumulative dose exposure.4,5 Similarly, there are theoretical risks of hyperkalaemia, dehydration and renal impairment with the use of spironolactone.
Synthesis of testosterone by testicular Leydig cells is regulated by a negative feedback loop involving hypothalamic secretion of gonadotrophin-releasing hormone and pituitary secretion of luteinizing hormone. Testosterone, and its metabolite 5-dihydrotestosterone, bind to the androgen receptor to exert a variety of effects in different tissues. 6 Testosterone is also the major source of estradiol in men, derived from aromatization in tissues such as the testes, muscle and adipose. 7
Bicalutamide is a second-generation non-steroidal androgen receptor antagonist derived from flutamide which has been used for the treatment of prostate cancer since 1995. 8 While it still has a role in the treatment of castrate-sensitive metastatic prostate cancer, there has been recent interest in repurposing its use for gender-affirming hormone therapy.9 –11 Theoretically favourable properties of bicalutamide include potent androgen receptor antagonism and negligible off-target effects 12 compared to conventional anti-androgen therapy (e.g. spironolactone and cyproterone acetate interact strongly with the mineralocorticoid 13 and progesterone receptors 14 respectively).
Hepatotoxicity is described as a rare complication of bicalutamide in the prostate cancer literature. Asymptomatic transaminitis has been reported in 6% of patients treated with bicalutamide and was typically self-limiting with continuation of treatment. 15 However, there are several case reports of severe liver injury (typically using doses of bicalutamide 50 mg daily or greater) 16 and recently the first case of suspected bicalutamide-associated hepatoxicity was reported in a 17-year-old transfeminine person. 17
Further research is needed to evaluate the efficacy and safety of bicalutamide as part of gender-affirming hormone therapy, particularly given treatment is typically intended to be long-term. In this retrospective case series, we aimed to explore the effect of bicalutamide on serum total testosterone concentrations and liver function in transgender people.
Methods
Using electronic medical records of the Austin Health Gender Clinic and private endocrinologists in Melbourne, Australia, transgender adults who used bicalutamide as an anti-androgen for gender affirmation between January 2020 and March 2023 (3 years, 2 months) were identified. Inclusion criteria included age ⩾18 years old and current or previous use of bicalutamide for gender affirmation. Exclusion criteria included no clinical and biochemical follow-up data following the commencement of bicalutamide. The retrospective audit was approved by the Austin Health Office for Research (Audit 21/32) and the need for informed consent was waived.
Deidentified data was retrieved including demographics, estradiol therapy, previous and current anti-androgen therapy and biochemistry (serum total testosterone, luteinizing hormone, estradiol and alanine transferase (ALT) concentrations). Treatment regimens and pathology results were recorded from the most recent clinical encounter where an individual was using bicalutamide. If present, a patient’s rationale for commencing bicalutamide was recorded and categorized by the authors as relating to ‘improved efficacy’ or ‘reduced side effects’ %). Serum total testosterone, estradiol and luteinizing hormone were measured by immunoassay and ALT by enzymatic assay in routine use for clinical care by National Association of Testing Authorities (NATA)-accredited laboratories. Changes in serum ALT were also categorized as per Common Terminology Criteria of Adverse Events (CTCAE), using the highest serum ALT observed during the data collection period, whereby grade 1 toxicity is defined as serum ALT >3.0 times the upper limit of normal (1.5–3.0 times baseline if baseline was abnormal). 18
Data was presented as a series and summarized with the median (interquartile range [IQR]) values for continuous variables and N (%) for non-continuous variables. No formal statistical comparison was made given the small sample size and significant heterogeneity in terms of estradiol, bicalutamide and other anti-androgen use.
Results
Twenty-four patients were identified from medical records with historical or current bicalutamide use (see Table 1 for summary statistics). The median age was 29 years (24–38), and with a predominant binary female gender (N = 21, 88%).
Summary statistics (overall).

Categorical variables are reported as N (%) and continuous variables median (interquartile range) unless otherwise stated.
There was significant heterogeneity in estradiol formulation and dosing, with a number of individuals using intramuscular estradiol (N = 3, 13%) or subdermal estradiol implants (N = 4, 17%). The majority (71%) of individuals had previously used other anti-androgens prior to commencing bicalutamide. The median bicalutamide dose and duration were 25 (25–50) mg daily and 6 (4–10) months respectively. Six (25%) individuals elected to cease bicalutamide due to a perceived lack of benefit at their most recent follow-up appointment.
The rationale for commencing bicalutamide was recorded in the majority of individuals (83%). Comments were categorized according to prevailing themes relating to a belief that bicalutamide was of greater efficacy (e.g. to improve breast growth, to decrease spontaneous erections; N = 12, 50%) or associated with reduced toxicity compared to other anti-androgens (e.g. effects on libido, erectile function, mood and co-existing conditions such as prolactinoma; N = 8, 33%).
Bicalutamide use was associated with highly variable serum total testosterone concentrations, with a median of 7.7 (0.7–17.5) nmol/L. However, in a subgroup of individuals with no previous anti-androgen use (Table 2), the median serum total testosterone concentration was 17.5 (0.7–34.2) nmol/L.
Subgroup analysis – anti-androgen naïve bicalutamide users.
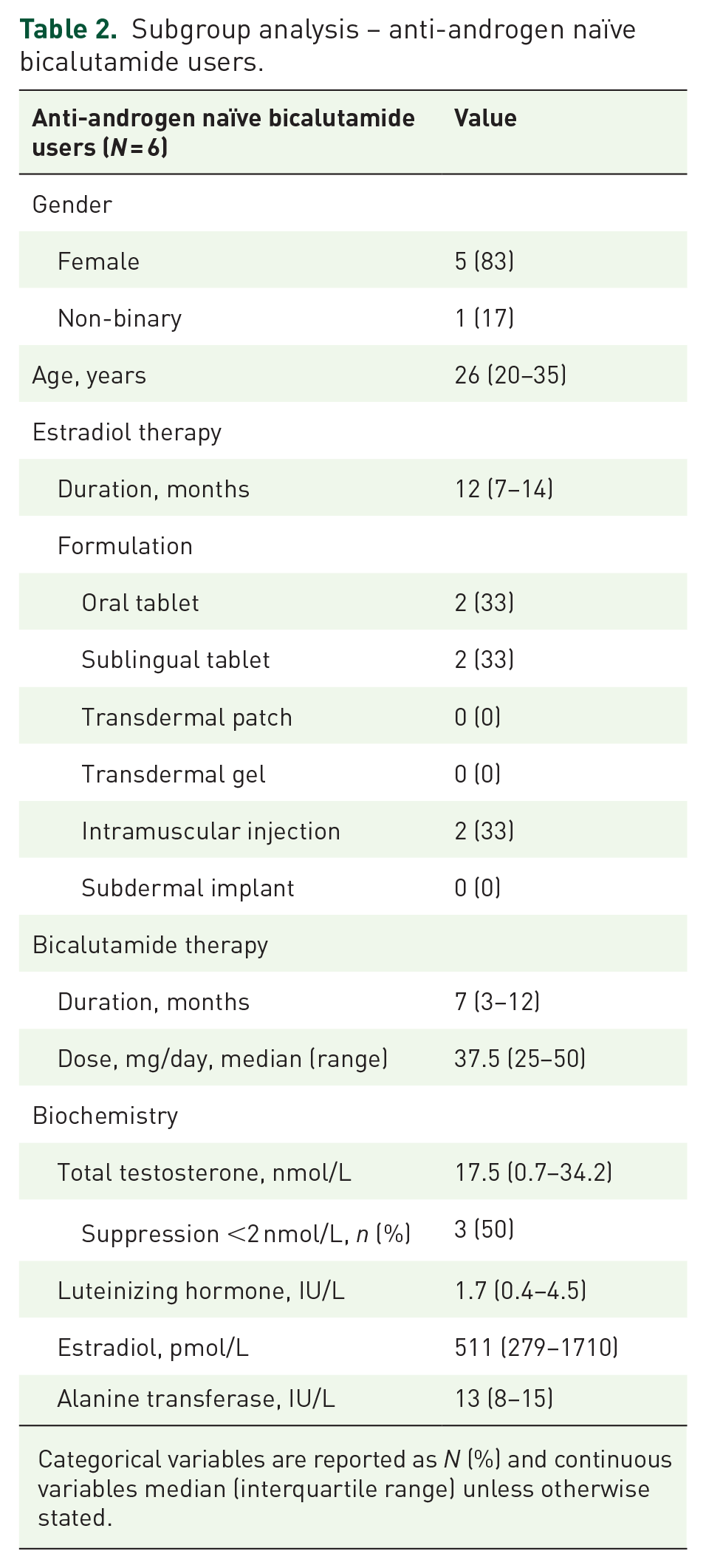
Categorical variables are reported as N (%) and continuous variables median (interquartile range) unless otherwise stated.
There was no signal of hepatotoxicity, with median serum ALT 20 (13–29) IU/L. Using CTCAE classification, one individual had a grade 1 toxicity (serum ALT >3.0 times baseline, or 1.5–3.0 times normal if the baseline was abnormal). This occurred in the setting of an intercurrent condition known to affect liver function and it was unclear whether bicalutamide was contributory.
Discussion
In this series of 24 transgender people taking bicalutamide, there was significant variation in serum total testosterone concentration, though this was higher in individuals who had not previously been treated with other anti-androgens. The median serum ALT was within the normal range, but one individual had asymptomatic transaminitis in the setting of polypharmacy overdose (grade 1 toxicity).
We hypothesize that the observed variability in serum total testosterone concentrations is related to the suppression of the hypothalamic-pituitary testicular axis mediated by supraphysiological serum estradiol concentrations, or previous anti-androgen exposure (e.g. cyproterone acetate). Of note, some documented estradiol preparations differed from Australian guidelines for transfeminine people, with several individuals taking multiple estradiol formulations concurrently and the number of individuals using intramuscular estradiol injections and subdermal estradiol implants which are available in Australia only as compounded medications. Several individuals also reported sublingual administration of estradiol tablets. This may suggest that patients requesting bicalutamide therapy are also more likely to seek alternative formulations of estradiol therapy.
Consensus guidelines typically recommend aiming for a serum total testosterone level within the reference range of the affirmed gender (i.e. serum total testosterone <2 nmol/L in transgender people desiring feminization).19,20 Previous cross-sectional analyses showed that the median serum total testosterone concentration was lower in those treated with estradiol in combination with cyproterone acetate than spironolactone or no anti-androgen. 21 However, a systematic review of anti-androgens identified no studies showing differences in clinically meaningful outcomes of feminization. 22 Given use of non-steroidal anti-androgens such as bicalutamide appears to result in feminization despite maintaining serum total testosterone concentrations in (or above) the typical cisgender male reference range, a more nuanced approach may be required in future guidelines considering the pharmacodynamics of different drug regimens.
As a potent androgen receptor antagonist with negligible off-target effects, the use of bicalutamide monotherapy is expected to increase serum total testosterone concentrations. 23 It is hypothesized that this more potent androgen receptor blockade and subsequent aromatization of testosterone to estradiol may assist with feminization and decrease estradiol requirements compared to conventional anti-androgens. This is supported by a case series of trans girls treated exclusively with bicalutamide 50 mg daily which described significant breast development in 11 of 13 individuals after 6 months of treatment, 9 and higher rates of gynaecomastia reported in men treated with bicalutamide compared to other forms of androgen deprivation therapy for prostate cancer. 24 Currently, we lack comparative studies assessing the effectiveness of bicalutamide on feminization compared to standard care.
Maintenance of higher serum total testosterone concentrations may negate some risks of conventional anti-androgen therapy such as bone loss and infertility. For example, in a 12-month open-label study of bicalutamide versus leuprolide in men with prostate cancer, use of bicalutamide was associated with an increase in bone density compared to a decrease in bone density observed in those treated with gonadotrophin-releasing hormone analogues. 25 Additionally, a case report described features of normal spermatogenesis in orchidectomy specimens of two men treated with bicalutamide for prostate cancer for greater than 4 years, presumably due to the maintenance of high intratesticular testosterone concentrations. 26 However, there is no data available on use of bicalutamide in combination with estradiol which may negate some of these theoretical benefits due to estradiol-mediated suppression of the hypothalamic-pituitary testicular axis.
There remains some apprehension in embracing bicalutamide for gender affirmation given documented cases of severe hepatotoxicity reported in the prostate cancer literature. However, toxicity is typically characterized by asymptomatic, mild and transient transaminitis, with onset within 2–3 months following commencement. 27 In our case series, despite the median duration of therapy of 6 months, there was only one episode of grade 1 toxicity in the setting intercurrent condition and it is unclear whether bicalutamide was contributory. While regular monitoring of liver function and counselling patients regarding potential symptoms of liver toxicity remains prudent, this data is reassuring and echoes the findings of other observational studies investigating bicalutamide in transgender youth.9,11
Strengths of this study include documentation of real-world bicalutamide use in adult transgender people and longitudinal data on the effects on serum sex steroids and liver function. Limitations include retrospective case note study design, variability in bicalutamide dosing and duration of therapy, small sample size, consistency of clinical documentation, a lack of clinically meaningful outcomes and potential confounding effects of previous anti-androgen treatment and concurrent estradiol therapy which may affect serum total testosterone concentrations.
Conclusion
In conclusion, we provide a series of transgender adults prescribed bicalutamide for gender affirmation and found significant variation in serum total testosterone concentrations, likely related to concomitant estradiol therapy and suppression of testicular function. The effect on surrogate markers of liver function was reassuring. Further research is needed to evaluate the efficacy and safety of bicalutamide in the transfeminine population in comparison to standard care.
