Abstract
Transgender (trans) women (TW) were assigned male at birth but have a female gender identity or gender expression. The literature on management and health outcomes of TW has grown recently with more publication of research. This has coincided with increasing awareness of gender diversity as communities around the world identify and address health disparities among trans people. In this narrative review, we aim to comprehensively summarize health considerations for TW and identify TW-related research areas that will provide answers to remaining unknowns surrounding TW’s health. We cover up-to-date information on: (1) feminizing gender-affirming hormone therapy (GAHT); (2) benefits associated with GAHT, particularly quality of life, mental health, breast development and bone health; (3) potential risks associated with GAHT, including cardiovascular disease and infertility; and (4) other health considerations like HIV/AIDS, breast cancer, other tumours, voice therapy, dermatology, the brain and cognition, and aging. Although equally deserving of mention, feminizing gender-affirming surgery, paediatric and adolescent populations, and gender nonbinary individuals are beyond the scope of this review. While much of the data we discuss come from Europe, the creation of a United States transgender cohort has already contributed important retrospective data that are also summarized here. Much remains to be determined regarding health considerations for TW. Patients and providers will benefit from larger and longer prospective studies involving TW, particularly regarding the effects of aging, race and ethnicity, type of hormonal treatment (e.g. different oestrogens, anti-androgens) and routes of administration (e.g. oral, parenteral, transdermal) on all the topics we address.
Keywords
Introduction
In 2016, several groups published priorities for transgender (trans) health and research with a focus on collaboration and overcoming health disparities among this population.1,2 Despite the increasing presence of gender diversity in both mainstream media and medical literature, many barriers to optimal health among trans people still exist. For example, the Report of the 2015 United States (US) Transgender Survey 3 showed that healthcare coverage for trans people in the US remains precarious, with 25% of those who sought coverage for hormones in 2014 being denied and 23% of respondents not seeing a doctor out of fear of mistreatment as a trans person. Similarly, in Europe, a 2012 European Union Agency for Fundamental Rights survey found that 22% of trans respondents felt personally discriminated against by healthcare personnel because of their trans identity. 4 Many providers, including specialists, do not feel adequately trained to care for trans patients,5–7 which may result in patients seeking alternatives to the standard transition-related options that are described in international guidelines.
Trans people can experience social, hormonal or surgical transition depending on their level of self-acceptance and availability of support and resources. Gender-affirming hormone therapy (GAHT) is often an important aspect of transition-related care, improving quality of life (QOL) and mental health of trans people.8–17 GAHT should be accessible to, but not required of, all trans people. In this narrative review, we aim to provide readers with a comprehensive summary of health considerations related to TW, defined here as people assigned male at birth (AMAB) but who have female gender identity or expression. We also aim to identify areas to focus our research in order to answer the many questions that remain surrounding TW’s health.
We acknowledge that trans terminology is evolving. Though used with the best inclusive intention, terms and phrases to describe trans study populations may still be limited in definition. For example, some may use the phrase, ‘trans people AMAB’, to be inclusive of TW and gender nonbinary people, individuals who do not identify as strictly male or female. For the sake of consistency throughout this narrative review, we will use the abbreviation ‘TW’ to refer to a ‘trans woman’ or ‘trans women’. Table 1 contains additional terminology that will be used.
Terminology.
Part 1: feminizing GAHT
The recently updated Endocrine Society guidelines 18 provide recommendations for feminizing GAHT for TW (Table 2). The World Professional Association for Transgender Health (WPATH) Standards of Care (SOC), version 7, 19 The Center of Excellence for Transgender Health at the University of California, San Francisco 20 and additional reviews also provide summaries of these feminizing hormonal treatments.21–25 Typical feminizing GAHT includes oral, transdermal and parenteral oestradiol formulations in addition to anti-androgens [e.g. spironolactone, cyproterone acetate (CPA) and gonadotropin-releasing hormone agonists (GnRHas)]. Oestrogens and anti-androgens suppress serum testosterone levels and block androgen action, and it is expected that testosterone levels will decline on all forms of treatment.26,27 Transdermal oestradiol patches or gel (and depending on availability, parenteral oestradiol injections) may be safer with regard to triglycerides and clotting risk than oral oestradiol by bypassing first-pass hepatic metabolism. 28 Transdermal oestradiol use has been proposed and implemented particularly in older TW (e.g. age >40 years).29–31 We are unaware of any randomized controlled trials (RCTs) comparing oral, transdermal and parenteral oestradiol or the various anti-androgens in TW.
Feminizing gender-affirming hormone therapy for trans women.
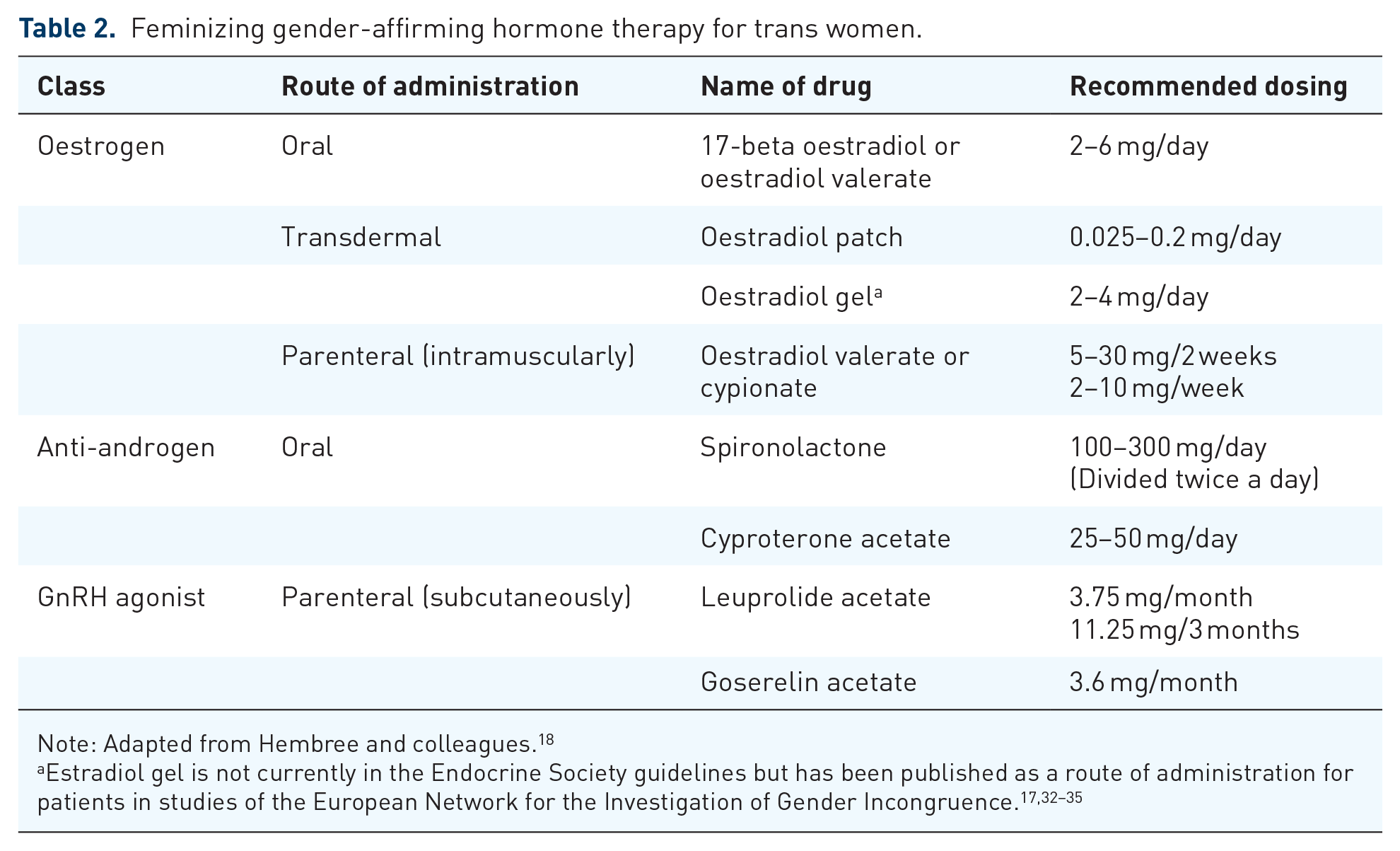
Note: Adapted from Hembree and colleagues. 18
Oestrogens not in the guidelines have caveats to their use. Conjugated equine oestrogens cannot be accurately measured in current serum oestradiol assays, making dose adjustments difficult based on serum oestradiol level. Addressed particularly in the cardiovascular disease (CVD) section, oral ethinyl oestradiol is no longer recommended due to the associated increased risks of blood clots and cardiovascular (CV) morbidity and mortality. We are unaware of specific studies looking at the safety and efficacy of sublingual and pellet formulations of oestradiol.
Other anti-androgens proposed by the Endocrine Society guidelines 18 are the GnRHas, which are well tolerated with satisfactory results. 36 Since some centres now use them as first-line therapy to suppress endogenous gonadotropins [follicle-stimulating hormone (FSH) and luteinizing hormone (LH)] and testosterone in TW, 36 it is increasingly important to know about a TW’s history of puberty suppression. For example, administration of GnRHas in early puberty can lead to underdevelopment of the penis, which can compromise future vaginoplasty. 37 If GnRHas are initiated as part of GAHT after secondary sexual characteristics are established, those pubertal changes do not regress. 38 Gava and colleagues 39 reported on CPA and leuprolide acetate being equally effective in the suppression of gonadotropins and testosterone after 12 months. We are unaware of any long-term outcome studies in TW who underwent pubertal suppression with GnRHas, stressing the importance of this as a research focus.
Alternative anti-androgen therapies exist that have not yet been substantiated for routine use by supporting research. Progesterone is discussed in the breast development section, but we do not recommend its routine use in TW until more rigorous studies demonstrate potential benefits outweigh any risks. 40 Some references mention finasteride and dutasteride, 5-alpha reductase inhibitors, as single agents for individuals seeking partial feminization of with persistent virilized features after complete androgen blockade or orchiectomy. 20 These medications block the conversion of testosterone to dihydrotestosterone but can actually raise serum testosterone levels, although it is unclear whether this has deleterious effects on feminization. 41 Data on bicalutamide, an androgen receptor antagonist used in the treatment of prostate cancer, in TW are nonexistent, but Neyman and colleagues 42 recently published on bicalutamide’s feminizing effects (particularly breast development) in AMAB trans adolescents. Another recent study in cisgender (cis) women, with polycystic ovary syndrome (PCOS) showed some improvement in hirsutism when bicalutamide was added to an oral contraceptive pill (OCP). 43 It should be noted, however, that bicalutamide has been associated with an increase in liver enzymes.
When barriers prevent trans people from obtaining GAHT from a licensed medical provider, they may search for other sources. Previous reports stated that 4.7–24% of TW have obtained hormones from friends,44–46 and 2–70% have purchased them online. 47 Anecdotally, some TW who utilize services at the Ghent University Hospital Transgender Infopunt have admitted to using OCPs obtained from their female partners. The use of GAHT obtained from nonmedical sources varies from 23% in a United Kingdom sample of trans people referred to a gender clinic, 47 to 26.8% in a community-based questionnaire in people who self-identified as trans in Canada, 45 to 48.0% in a Belgian sample of trans sex workers 44 and as high as 63% in urban groups of TW in New York City. 46 It is important to ask patients about nonprescribed medications in a nonjudgmental fashion with the overall goal of patient and provider shared decision-making regarding the risks and benefits of GAHT.
In summary, guideline-recommended feminizing GAHT incorporates bioidentical oestradiol, unlike ethinyl oestradiol, and anti-androgens to facilitate desired feminization in TW. While these oestradiols and anti-androgens are used off-label, studies have demonstrated their efficacy in suppressing endogenous testosterone levels and promoting feminization. Future research is needed in order for TW and their providers to better individualize GAHT: (1) RCTs to determine which route of oestradiol and type of anti-androgen are most effective; (2) predictors of feminization based on individual TW baseline characteristics; (3) long-term effects of pubertal suppression across the rest of a TW’s lifespan; and (4) reducing barriers to access to GAHT among TW. Additionally, there is a growing population of gender nonbinary patients who were AMAB but desire feminizing hormones even though they do not identify as TW. We need more studies in gender nonbinary patients to see how taking the feminizing GAHT discussed above affects their health outcomes, which doses are most appropriate for nonbinary goals and whether these outcomes are similar to those in TW.
Part 2: benefits associated with GAHT
QOL and mental health
Research on QOL in trans people has previously been published in reviews8,13 and a meta-analysis. 48 In studies with a cross-sectional design, it appeared that trans people receiving GAHT had higher overall as well as physical and mental QOL scores49–51 and better mental and general health compared with controls.12,52,53 Trans people not receiving GAHT experienced more limitations in work and other activities due to mental health problems (which reflected in lower emotional Short Form-36 scores) than controls.12,52 In addition, the initiation of GAHT results in an increase of overall QOL scores. 54 However, receiving GAHT is not the only factor influencing QOL in TW, as partnership status,16,51,55 social support from family, 50 employment status,50,56,57 high levels of hope 51 and undergoing gender-affirming surgery (GAS) 13 contribute to increased QOL. On the other hand, depression,12,58 anxiety58,59 poorer education, 55 unemployment,55,59 financial dependence,55,60 a history of negative healthcare experiences, 57 lack of practical or social support,49,57,60 desiring but not being able to affirm the legal gender, 57 insecurity about one’s own appearance, 59 sleeping problems 59 and illicit drug use 57 are associated with decreased QOL. Although gender-affirming care contributes to improved QOL in trans people, studies calculating a possible gain in quality-adjusted life years are limited, 61 and an incremental cost-effectiveness ratio has never been calculated for gender-affirming treatment. We stress the need for cost-effectiveness analyses in the field of trans health, as those types of studies can highlight interventions that are relatively inexpensive yet have the potential to reduce gender dysphoria burden substantially.
Breast development
TW often consider breast development a key feature of feminization and it occurs predominantly during the first 6 months of GAHT, 62 with maximum effect expected by 2–3 years. 18 However, breast growth is often modest in TW.62–65 De Blok and colleagues 62 reported increasing breast-to-chest circumference difference and a bra cup size of less than an AAA cup in 48.8% of TW after 1 year of GAHT, with only 3.6% gaining a bra cup size larger than an A cup, independent of age, weight change, smoking, body mass index (BMI), serum oestradiol levels and oestrogen administration route. Assessing breast development only with Tanner staging and not measurements, Fisher and colleagues 10 showed breast development to Tanner 3 after 2 years of GAHT, with a shift from predominantly Tanner 2 after 6 months of GAHT to predominantly Tanner 3 after 12 months and almost all with Tanner ⩾3 after 24 months. Additionally, anatomical differences in the thoracic bone structure compared with cis women may result in breast size appearing smaller than the objectively measured volume. 66
The impact of various combinations of different oestrogens, anti-androgens and progestogens on breast development was previously reviewed by Wierckx and colleagues, 67 who concluded that current evidence did not support or refute any enhancement of breast development with progestogens. Pubertal data in people assigned female at birth (AFAB) (e.g. girls with Turner syndrome) argue for delaying progesterone as it causes ductal differentiation and may interfere with optimal breast development. 68 Due to unsatisfactory effects of GAHT alone on breast growth and development, 60–70% of all TW sought additional surgical breast augmentation. 67 Articles on breast development in TW report inconclusive results, possibly due to differences in methodology and the lack of prospective RCTs. Prior 69 recently published on the potential benefits of progesterone use on breast development (among other conclusions) in TW but the perspective was limited to clinical experience, assumptions taken from in vitro and animal data, and selected data from cis women. 40 It would be worthwhile for future studies to use volume measurements for examining hormonally induced breast development in TW over a longer follow-up period in a large cohort, which may result in the identification of predictive markers for breast development. 62 Additionally, rigorous prospective RCTs of oestrogen and oestrogen plus progesterone may provide more conclusive data to guide routine use of progesterone for breast development.
Bone health
Oestrogen serves as a key regulator of bone health, and oestrogen deficiency is thought to play an important role in both the rapid decline in bone mineral density (BMD) seen in postmenopausal women as well as the more gradual loss seen with aging in men.70,71 However, questions remain as to the overall prevalence of low BMD and fracture in TW and the timing and dosing of oestrogen to optimize bone outcomes.
Studies have shown a high prevalence of low bone mass in TW when compared with cis men both prior to and while on GAHT.33,72,73 The two studies that looked at baseline peripheral quantitative computed tomography (pQCT) found that TW had lower areal BMD (aBMD), smaller cortical bone area and a thinner radial cortex compared with cis men.74,75 Additionally, these studies showed lower grip strength and muscle mass.74,75 Genetics is thought to play a large role in attainment of peak bone mass, but dietary composition, exercise, puberty timing and lifestyle factors, such as smoking and alcohol use, are also influential. Some studies have shown low rates of physical activity and vitamin D in TW prior to GAHT. 75 There has also been a question as to the role of gonadotropins in bone health as FSH was slightly higher in TW at baseline in one study. 74 Some authors have argued that FSH may have effects on the bone, independently of oestradiol, although this is disputed.76,77 Early studies of TW treated with oestrogen also postulated a correlation between higher LH levels and lower BMD after GAS, 78 although more recent studies have not confirmed this.79,80
With the initiation of oestrogen, most studies report a positive change in BMD in TW81,82 and some have shown associations with oestradiol level attained. 80 A recent meta-analyisis 83 reviewed 13 studies with 392 TW and showed significant gains overall in lumbar spine (LS) BMD at 12 months [0.04 g/cm2; 95% confidence interval (CI) 0.03–0.060 g/cm2] and 24 months (0.06 g/cm2; 95% CI 0.04–0.08 g/cm2) which is the range often considered clinically significant. Changes at the hip were not significant in this group.
Body composition changes occur with GAHT and probably influence bone health. One review reported GAHT increases body weight 1–3 kg per year in TW in the setting of decreased lean mass and increased fat mass. 23 A study of 142 TW in Brazil, many of whom were already on GAHT (86%) or underwent GAS (33%), assessed appendicular lean mass, as an approximation of muscle mass, derived by dual-energy X-ray absorptiometry (DXA) measurements. Appendicular lean mass was similar in cis women to TW but lower than in cis men at the baseline measurement. There was a positive correlation between appendicular lean mass and total fat mass that correlated with L1–L4 BMD and explained 14.9% of BMD variation. 72 The question has been raised as to whether some of the improvement in DXA may be artefactual as it is affected by fat mass; however, the correlation between markers of bone turnover and BMD changes shown in some studies 74 would dispute this.
An area of concern for bone health is treatment with GnRHas for puberty blockade in trans adolescents. From their use in other medical conditions such as prostate cancer, their deleterious effects on the bone are well known, although these have the potential to be reversible if treatments are stopped or add-back therapies can be given.84,85 In a study of 15 young adult TW, similarly to older adults, baseline BMDs were lower than the population mean at baseline. 86 With GnRHas, BMD went down as expected; however, after initiation of GAHT, despite an increase in absolute aBMD, the Z-score at age 22 years was lower than at the start of treatment, with six people (40%) having an LS Z-score of <−2.0. 86 Similar findings were reported in a larger trial of 28 adolescents/young adults, with some overlapping study participants, where BMD Z-scores decreased during treatment with GnRHas then increased after initiation of GAHT, but remained below the population average and below baseline levels even after 24 months of GAHT. 87 Another recent study looked at the use of CPA in 21 late pubertal trans girls (Tanner 4 with mean bone age at study start of 17.1 ± 1.28 years) as a potential way to avoid the negative bone effects of GnRHas. 88 However, after 1 year of therapy there was a loss of lean mass and gain of fat mass as well as a decrease in Z-scores most profoundly at the LS, but also at the femoral neck and total hip. These findings all raise concerns, particularly since youth today are starting GnRHas earlier than in these studies (i.e. they may be taking a GnRHa for a longer duration), which may have more significant deleterious effects on skeletal development and will require further study.
In summary, bone density in TW may run low even prior to initiation of GAHT. Lifestyle factors probably contribute to this. When GAHT is initiated, there are positive changes in BMD as well as some measures of bone quality, however the effect on fracture rates is not fully known as studies have not been powered to examine this end point. Questions remain as to the effect of type and route of oestrogen utilized as well as the effect of GnRHas, gonadectomy and anti-androgens on bone health. The US population, which may have differences in ethnicity, average BMI, lifestyle habits, types of oestrogens and anti-androgens used, and rates of gonadectomy compared with European cohorts, has not been well studied and certain measures bear repeating. As far as clinical recommendations for patients, the Endocrine Society guidelines 18 suggest checking bone density in patients who have traditional risk factors for osteoporosis and specifically in those who stop hormones after gonadectomy. However, given the high prevalence of low BMD even prior to the initiation of GAHT or GAS in TW, it may be reasonable to assess a baseline DXA earlier.
Part 3: risks associated with GAHT
Cardiovascular morbidity and mortality
There have been systematic reviews,89–92 meta-analyses,89,93 and narrative reviews28,94 on CV morbidity and mortality in TW. Based on available evidence, no consistent increased risk for CV mortality and morbidity is observed when adhering to the current GAHT regimens, although studies assessing CV outcomes in TW are generally limited by the lack of RCTs, relatively short follow up, not controlling for traditional CVD risk factors and the reported use of ethinyl oestradiol instead of the currently preferred bioidentical oestradiols.31–33,91,95–103
Gooren and colleagues 104 published on the apparent reversal of the traditional sex differences in CVD patterns. In contrast with cis women having a lower incidence of heart disease compared with cis men (up to age 75 years), TW treated with oestrogens (plus anti-androgens) showed more CV pathology than trans men, individuals AFAB who have male gender identity, receiving testosterone. Aging and pre-existing CVD contributed to increased risk as well as ethinyl oestradiol (in doses 2–4 times of OCPs). 104 Elamin and colleagues 89 reported very few CV events [death, cerebrovascular accident (CVA), myocardial infarction (MI), venous thromboembolism (VTE)] in their 2010 review.
The longest follow up study (mean 19.4 ± 7.7 years) of GAHT in TW (mean age at hormone start 31.4 ± 11.4 years) was the Amsterdam cohort, reported by Asscheman and colleagues, 31 which revealed very few CV events (18 and 5 observed cases of mortality by ischemic heart disease and CVA, respectively, out of 1,331 TW). There was an increased risk of mortality by ischemic heart disease among TW compared with the general population [standardized mortality ratio (SMR) 1.64; 95% CI 1.43–1.87] but not an increased risk of CVA (SMR 1.2; 95% CI 0.93–1.64), and the ischemic heart disease risk was driven by continuous ethinyl oestradiol use. 31 In a study by Wierckx and colleagues, 32 TW experienced more MI than cis women but a similar proportion compared with cis men. Cerebrovascular disease was higher in TW than in cis men and there was more type 2 diabetes mellitus in TW than controls. 32
The data are more conclusive about the increased risk of VTE in TW. Wierckx and colleagues 32 found that 5% of TW had a VTE during GAHT (n = 5 in first year hormones, n = 3 during GAS). Getahun and colleagues 105 reported on acute CV events as part of the Study of Transition, Outcomes and Gender (STRONG) cohort, consisting of 6,456 Kaiser Permanente members [n = 2842 transfeminine (sic)], and found a higher incidence of VTE among transfeminine participants compared with reference men [adjusted hazard ratio (aHR) 1.9; 95% CI 1.4–2.7] and reference women (aHR 2.0; 95% CI 1.4–2.8), though the absolute rates were still very low and the increased risk was driven by those with longer (>2 years) follow up. Transfeminine participants had a higher incidence of VTE, with 2- and 8-year risk differences of 4.1 (95% CI, 1.6–6.7) and 16.7 (95% CI, 6.4–27.5) per 1,000 persons relative to reference men and 3.4 (95% CI, 1.1–5.6) and 13.7 (95% CI, 4.1–22.7) relative to reference women, respectively. Another caveat with the STRONG cohort data is that the authors did not adjust for duration, type (including ethinyl oestradiol) or route of administration of GAHT.
Cigarette smoking is an independent risk factor for CVD and stroke 106 as well as VTE.107–110 There are varying published rates of current smoking status among TW, including 15% in the STRONG study 101 and 38.6% in the Amsterdam cohort. 31 Wierckx and colleagues 32 reported that 7 of 11 cases of VTE, 3 of 3 cases of MI, and 4 of 5 cases of transient ischemic attack/stroke were current or former smokers. Although not considered an absolute requirement to start GAHT, smoking cessation should be encouraged at every visit for CVD and overall health benefits. Myers and Safer 111 reported a higher rate of smoking cessation among TW (64%) when they were strongly advised to quit before initiating GAHT compared with national smoking cessation rates (6.2%).
There has been a surge in reviews attempting to reassess CVD risk of GAHT, often reviewing the same mostly European studies on mortality and long-term safety in TW.28,93,94,104,112 More recently, data from the US have been published. Nokoff and colleagues 99 analyzed responses to the 2015 Behavioral Risk Factor Surveillance Survey (BRFSS) in the US and found that TW had higher odds of reporting MI than cis women (OR 2.9; 95% CI, 1.5–5.3) but not cis men. Getahun and colleagues 105 also reported the risk of ischemic stroke was higher among TW compared with cis women (aHR 1.9; 95% CI 1.3–2.6), again related to longer (>6 years) follow up, and the risk of MI was higher among TW overall compared with cis women but not higher among TW who initiated oestrogen during the enrolment period.
In addition, there is currently no evidence of increased risk for arterial hypertension and dyslipidaemia in TW,32,39,91,98,99,113–120 despite indicators of a possible increased cardiometabolic risk (e.g. increasing BMI,115,121 percent visceral fat121,122 and insulin resistance 115 ). In contrast, other markers suggest a decreased risk compared with reference men and women as well as trans men [e.g. less insulin resistance,34,39,113,115,118 decreasing/stable blood pressure,116,118,119 decreasing LDL34,115,117,118 and smaller waist-to-hip ratio (WHR) 122 ]. Some conflicting results can be explained by the older mean age of TW compared with trans men, geographical location, GAHT regimen, minority stress (e.g. feelings of discrimination)123–126 and lifestyle factors.127–129
Studies on the effect of GAHT on weight among TW have revealed no change72,130 or increase in weight; 131 however, there has been consistency in body composition findings of decreased lean mass and increased fat mass.72,130–133 Klaver and colleagues 132 reported that body composition changes were similar between oral and transdermal oestradiol after adjustment for baseline BMI and age. There were higher odds of obesity among all trans US Veterans compared with ‘nontransgender’ (sic) US Veterans (aOR 1.58; 95% CI 1.48–1.70), 134 lower rates of obesity among TW (23%) compared with cis women (27%) and cis men (28%) in the STRONG cohort, 105 and higher odds of self-identified obesity among TW compared with cis women (OR 1.77; 95% CI 1.11–2.82) but not cis men (OR 0.93; 95% CI 0.58–1.48) in the 2015 BRFSS. 99
We are unaware of any weight loss or obesity intervention studies among TW in the literature but there are a few insights into the behavioural modification topics of diet and physical activity. The Colorado Transgender Health Survey reported 76% of trans adults said they had ‘exercised in the past month’ compared with 82% of all Coloradans. 135 The BRFSS 2015 analyses also found similar rates of physical activity between US TW, cis women and cis men (defined by the variable, ‘Does not meet physical activity aerobic recommendations’): TW had an OR of 1.05 (95% CI 0.73–1.51; p = 0.78) and 1.13 (95% CI 0.79–1.62; p = 0.52) compared with cis women and cis men, respectively. 99 Regarding dietary considerations for trans individuals, the literature includes specific attention to eating disorders, 129 weight gain and chronic diseases. 136 The Health Risk Questionnaire above revealed that a lower percentage of TW reported eating five or more servings of vegetables or eating three or more servings of fruits per day, and a higher percentage of TW reported drinking caloric beverages, all compared with cis women and cis men. 137
Given the lack of RCTs explaining the conflicting CV morbidity results among the available literature and the scarcity of data on older trans people,91,93,104 the importance of ongoing large-scale prospective studies, such as STRONG, 101 the European Network for the Investigation of Gender Incongruence (ENIGI) 138 and GEnder Dysphoria Treatment in Sweden (GETS), 139 and optimal management of conventional risk factors (e.g. weight, LDL cholesterol, blood pressure, smoking) cannot be overemphasized. The Endocrine Society guidelines 18 suggest monitoring weight, blood pressure and lipids at regular intervals. The absolute CV risk calculation should be performed according to local guidelines for the general population, prior to the initiation of GAHT as well as during follow up. As reference ranges for trans people are not yet defined, CV risk calculation using ‘male’ provides a sensitive estimate for TW but may lack specificity. 92 If CV risk factors are present, they should be managed according to the established population-based guidelines. In addition, as psychological stress can be as detrimental to CV health as physical stress, 140 we recommend addressing psychological wellbeing. Table 3 summarizes CVD risk factors in TW.
Cardiovascular disease risk factors in trans women: effects of feminizing GAHT.

Infertility
Due to the potential risk of infertility in TW taking oestradiol, it is imperative to discuss fertility preservation prior to the initiation of GAHT and throughout the transition process. Previous studies assessing testis biopsies in TW who underwent orchiectomy report conflicting results on the presence of spermatogenesis, mainly due to diverse methodologies. Leavy and colleagues, 141 reported severely impaired spermatogenesis in TW treated with oestrogens. Jindarak and colleagues 142 described maturation arrest in 36.4%, hypospermatogenesis in 26%, Sertoli cell-only syndrome in 20.2%, normal spermatogenesis in 11%, and seminiferous tubule hyalinization in 6.4% of the specimens, although, only 10.4% had been using anti-androgens in addition to oestrogens and data about GAHT was missing in 39.0%. Schneider and colleagues 143 reported normal testicular function in 48% of TW undergoing gonadectomy after stopping GAHT 2 or 6 weeks before gonadectomy or not stopping at all. However, only 71% were taking anti-androgens and no information was given about the duration or effectiveness of anti-androgen therapy before gonadectomy. As studies assessing the effects of GAHT on fertility in TW are currently inconclusive, the WPATH SOC, version 7, 19 encourages healthcare professionals to discuss reproductive options before initiating treatment that may reduce fertility.
Orchiectomy leads to irreversible loss of natural reproductive capacity. Trans people generally access reproductive medicine for two reasons: for fertility preservation prior to/during the transition process and for fulfilling the parental desire. Even after orchiectomy, it remains possible to have genetically related children if gametes have been preserved beforehand. Fertility options in trans people are discussed in full detail by De Roo and colleagues. 144
Fertility preservation utilization rates are considered low in trans people (9.6–15% in adults, 2.8% in adolescents),145–148 although reported parental desire rates in TW range from 28.8% to 58%,145,149,150 which is often higher before transition. 150 Although there are many options for fertility preservation in trans people, 151 many trans people still consider fertility the price to pay for transition 152 and cost remains an incredible barrier to undergoing fertility preservation. 153 For TW who underwent pubertal suppression, fertility preservation was often not possible at pubertal developmental Tanner 2 stage. 147 Adolescents are offered the choice of postponing pubertal suppression in order to proceed with fertility preservation or to initiate pubertal suppression at Tanner 2 and to interrupt the therapy for fertility preservation prior to initiation of GAHT. Therefore, it is important for healthcare providers to enquire about the fertility needs of trans people and to discuss reproductive options in each stage of the gender-affirming process. 154 Future research should further evaluate the effects of oestradiol in the presence and absence of different types of anti-androgens on fertility in TW and better characterize the cost-effectiveness and live birth rates of different fertility preservation options for TW.
Part 4: other health considerations for TW
HIV/AIDS
HIV prevalence rates for TW have varied widely across studies likely due to limited sampling with a focus on TW who engage in sex work and heterogeneous data collection methods.155–160 For example, Becasen and colleagues 158 recently published a meta-analysis of 88 US-based trans studies revealing that, among TW, laboratory-confirmed HIV prevalence was 14.1% (95% CI 8.7–22.2) and self-reported HIV infection was 21.0% (95% CI 15.9–27.2). A systematic review by Herbst and colleagues, 159 published 10 years earlier, reported higher prevalence of TW testing positive for HIV infection (27.7%; 95% CI 24.8–30.6) and lower self-reported HIV (11.8%; 95% CI 10.5–13.2). These estimates, regardless of publication year, are much higher than the estimated HIV prevalence in the general US population (<0.5%).161–164 Baral and colleagues 165 published an earlier systematic review and meta-analysis of studies from the US, Asia-Pacific, Latin America and Europe, finding a pooled HIV prevalence of 19.1% (95% CI 17.4–20.7), with higher prevalence associated with higher-income countries.
High-risk sexual behaviour, a risk factor for HIV infection, appears to be common among TW. Inconsistent condom use with primary as well as nonprimary and commercial partners, unprotected sex in sero-discordant relationships (i.e. one partner is HIV-positive) and sex while using substances of abuse are frequently reported.159,166–168 Engagement in sex work, another independent increased risk factor for HIV, is estimated to be high among TW. The disparities in housing, employment and access to healthcare and social services that many TW face may lead to economic marginalization and the feeling of needing to engage in sex work. 169 HIV prevalence is also estimated to be higher for TW of colour,158,159,170,171 which may be partially explained by more TW of colour being involved in sex work 170 and facing double stigmatization for their race/ethnicity and gender identity.157,172
The higher rates of HIV in TW are also related to increased morbidity and mortality. Asscheman and colleagues 31 reported that the higher risk of mortality among TW compared with the general population was mainly due to AIDS (SMR 30.20; 95% CI 26.0–34.7). HIV prevention, treatment and care utilization should be priorities around the world, but TW experience many barriers to treatment even after receiving an HIV diagnosis. There have been concerns about hormone–drug interactions between GAHT and medications used to prevent and treat HIV. Radix and colleagues 173 reviewed the literature on antiretroviral therapy (ART) use among TW and found that most studies have addressed interactions between ethinyl oestradiol (no longer recommended for GAHT as described above) and non-nucleoside reverse transcriptase inhibitors and protease inhibitors. Preexposure prophylaxis (PrEP), consisting of a tenofovir disoproxil fumarate/emtricitabine combination pill, is effective in preventing HIV infection though uptake in use has been limited. 174 There are calls for increased PrEP utilization in TW when clinically indicated. Research efforts in this area should continue to focus on TW’s unique barriers to optimal HIV screening, prevention and treatment as well as potential interactions between guideline-recommended oestradiol formulations/routes of administration and ART.173,175,176
Breast cancer
The long-term use of oestrogen in TW enables them to develop and maintain breasts but also raises concern about the development of cancer in the breast and other oestrogen-sensitive tissues. Retrospective cohort studies have demonstrated no cases31,91 or few cases177–180 of breast cancer but similar incidences to the general population,179–181 depending on study period, follow up and whether rates were compared with cis women or cis men. Rates from a large Dutch TW cohort between 1975 and 2011 (n = 2,307 and 52,370 person-years of GAHT exposure) found 4.1 cases per 100,000 person-years (95% CI 0.8–13.0) which was lower than expected if the one confirmed and second suspected cases were regarded as female breast cancer (170 per 100,000 person-years), but similar to that expected if regarded as male breast cancer (1.2 per 100,000 person-years). 179 A more recent retrospective analysis of the Dutch TW cohort between 1972 and 2016, but with fewer TW included, showed an increased risk of breast cancer in TW compared with Dutch cis men [standardized incidence ratio (SIR) 46.7, 95% CI 27.2–75.4] but a decreased risk compared with Dutch cis women (SIR 0.3, 95% CI 0.2–0.4). 180 The cases occurred at a younger age and after short exposure to oestrogen ± progestins. In the US, Brown and Jones 181 reported a higher incidence of breast cancer among TW and trans men veterans compared with published European cohorts, but a similar rate to an age-standardized US national sample.
As there is currently no evidence that breast cancer risk among TW is higher compared with cis women, we recommend following local population guidelines for breast cancer screening in women. Other recommendations for breast cancer screening in TW on continuous GAHT have included annual mammogram in those aged >50 years with additional risk factors such as having used oestrogen ± progestin for >5 years, a BMI >35 kg/m2 or a family history of breast cancer.177,182 A recent review by Sonnenblick and colleagues 183 provided radiologists with recommendations for culturally sensitive care for TW (and trans men) with regard to screening and diagnostic breast imaging as well as noted histologic and imaging changes due to GAHT. Noted caveats to breast cancer screening in TW include lack of evidence for these recommendations and how to manage TW with breast implants. 184 Mammography becomes less sensitive in the presence of implants, 185 therefore it is imperative to remind TW with implants to provide complete implant histories to the ordering providers and imaging technologists. Although mammography with additional views is still suggested for routine breast cancer screening in patients with breast implants regardless of gender identity, ultrasound is recommended as first-line imaging to investigate implant complications in those with pain, lumps or asymmetries. 186 Magnetic resonance imaging (MRI) can be performed after ultrasound especially if there is concern about intracapsular rupture not visible by ultrasound. 186 Others recommend comprehensive and individually tailored screening programs for trans people (e.g. possible gradual reduction of exogenous oestrogen doses with age and stimulating menopause in TW). 177 Additional prospective research is needed to better characterize predictors of breast cancer in TW as well as if there is different cost-effectiveness among screening modalities based on gender identity.
Other tumours
The long-term follow-up study of mortality that included 966 TW in the Netherlands who started GAHT prior to 1 July 1997, and with at least 1 year of follow up, did not find an increase in overall mortality from malignant neoplasm in TW (SMR 0.9; 95% CI 0.88–1.08). 31 However, among the reported deaths from a malignant neoplasm, there was a higher SMR for lung (1.35; 95% CI 1.14–1.58) and haematological (2.58; 95% CI 1.97–3.30) malignancies but lower SMR for digestive tract (0.42; 95% CI 0.28–0.60) malignancies compared with the general population. 31
GAS for TW generally does not include prostatectomy and questions arise about prostate cancer screening in TW. Prostate ultrasonography and biopsy demonstrated atrophy in nine TW aged 51–71 years with a mean of 15.8 years after oestrogen initiation and 13.4 years after orchiectomy, with few androgen receptors but ample oestrogen and progesterone receptors in stroma and epithelium. 187 Gooren and Morgentaler 188 reported one case of prostate cancer in a 63 year-old TW [among 2,307 TW (incidence rate 0.04%) who had a mean follow up of 21.4 ± 8.7 years] who had initiated GAHT at age 53 years and underwent orchiectomy at age 55 years. Additional cases of prostate cancer have been reported more recently.189–192 A total of 11 case reports have been published in TW with age at diagnosis between 54 and 78 years, the majority being metastatic on presentation (none of the 3 cases published after 2016), and years on GAHT between 6 and 41 years. 191 There is no consensus on routine prostate cancer screening among TW, with a caveat that prostate-specific antigen (PSA) cut-offs in TW are unknown. PSA should be low or undetectable in the setting of suppressed serum testosterone due to feminizing GAHT or orchiectomy, but we also do not have data to determine PSA utility in TW who are unable to achieve suppressed serum testosterone levels before orchiectomy. Following individualized population guidelines for screening in cis men may be appropriate for TW with prostate tissue present as suggested in the Endocrine Society guidelines. 18
There have also been some studies on brain tumours in TW. Meningiomas, benign tumours that are twice as likely in cis women compared with cis men and thought to be hormonally sensitive, were reported in 3 of 2,810 TW in the Amsterdam cohort treated between 1975 and 2011. 193 In reviews of the literature, CPA dosage was high, mostly 100 mg/day (but as low as 10 mg/day) and time from CPA start to meningioma diagnosis ranged from 1 to 25 years.193,194 Prolactinoma in TW has also been reviewed and associated with high-dose CPA. 189 While the recent Endocrine Society guidelines 18 suggest periodic monitoring of serum prolactin, a downgraded recommendation from the previous rendition, future studies will help us determine whether routine prolactin screening is needed particularly in TW who do not take CPA. Defreyne and colleagues 195 revealed that CPA increased prolactin levels in TW but the levels decreased when TW discontinued CPA. Fung and colleagues 117 demonstrated higher prolactin levels in TW receiving CPA than those taking spironolactone. Given these newer data, we recommend checking prolactin only in those patients with signs or symptoms of hyperprolactinaemia before or after starting feminizing GAHT.
Other reproductive cancers in TW have been reviewed and include neovaginal and testicular. 192 There was also a case report of an oestrogen receptor beta-positive mature testicular teratoma in a TW taking oestradiol and progesterone. 196 There is still no consensus on the screening of these type of tumours, probably given their rare occurrences, and ongoing long-term studies will shed more light on whether or not routine screening for these tumours is needed in TW.
Voice therapy
In TW who went through puberty according to their sex assigned at birth, exposure to androgens has resulted in acoustic changes, which become noticeable from a testosterone concentration of 150 ng/dl and higher. Repeated exposure to concentrations higher than 200 ng/dl results in irreversible changes. 197 Certain characteristics of voice and speaking are more frequently perceived as male or female, such as frequency ranges [male (<145 Hz), overlap (145–165 Hz) and female (>165 Hz) range],198,199 resonance (which is typically higher in voices perceived as female), inflections (more frequent and upward gliding perceived as a feminine characteristic of voice), articulation (more articulation perceived as a feminine characteristic of voice), speaking intensity (speaking more quietly is perceived as a feminine characteristic of voice) and breathiness (perceived as a feminine characteristic of voice). 200
Patient-reported outcomes have shown an improvement in TW’s voice-related gender identity perception, voice-related anxiety, avoidance of situations due to their voice and voice quality during the first year of GAHT, even in TW who did not undergo voice therapy or voice surgery. 197 However, these improvements were unrelated to prospective changes in serum testosterone levels and influences other than the GAHT (e.g. more gender-congruent feelings because of breast development) may play a role in the improving self-perception of voice occurring in TW.
Still, many TW reported voice-related problems in everyday life, especially when communicating over the telephone, when they sense irritation and hesitation in their conversation partners or when addressed as male.201–203 Aiming for a more feminine voice, TW sometimes perform vocal compensations, such as elevation of the larynx in speech, which may result in vocal hyperfunction and a higher occurrence of vocal complaints. 204 Being perceived as male is negatively correlated to personal satisfaction of the voice, even if the fundamental frequency is within the female range. 205 However, Hancock and colleagues, 206 reported that QOL ratings in TW are strongly correlated with their self-rated perception of voice and only moderately related to how others perceive their voice.
The goal of speech-language therapists and pathologists is to help TW develop voice and communication that reflects their gender identity without causing laryngeal injury, a goal that is beyond achieving fundamental frequency within the female range, 200 which is often the goal of phonosurgery. 207 Ultimately, the patient’s goals and perceptions are most important 207 and an individualized approach is preferred. Additional research can help us identify predictors of attaining the desired voice pitch among TW and which vocal therapy techniques achieve the best patient-reported outcomes.
Dermatologic considerations
There have been several reviews of dermatologic and hair disorders in trans populations.208–211 Among TW taking GAHT, oestrogens slow down male pattern hair growth, decrease sebum production and possibly improve acne 212 but can also increase xerosis and brittle nails.211,213 The onset of skin softening and decreased oiliness starts in 3–6 months but the time to maximum effects is unknown. 18 Meanwhile, decreased terminal hair growth begins at 6–12 months with maximum effect at >3 years. 18 GAS may require preoperative hair removal to facilitate better postoperative outcomes and reduce the risk of scarring or developing hair in undesired areas. 209 TW who underwent pubertal suppression with GnRHas would not have had development of androgen-dependent hair nor need laser/electrolysis treatments for hair removal. 37 In those already with or at high risk of contracting HIV or other sexually transmitted infections, skin manifestations related to those infections may arise.209,210 There was also a report of squamous cell carcinoma arising from the penile skin in a neovagina 20 years after a trans woman underwent GAS. 214
Another dermatologic consideration is the use of silicone. In a Belgian cohort of sex workers, 65% reported having silicone injections in one or more parts of the body (buttocks, legs, face, hips, breasts), frequently injected by a friend without medical training (53%). 44 The use of silicone injections among TW has led to reports of Mycobacterium abscessus cellulitis and abscesses of the breasts, 215 granuloma formation and hypercalcaemia 216 and lymphoedema from migrating silicone. 217 Injecting silicones can have major complications that can arise any time after the injections took place,218,219 and as such should be discouraged according to healthcare specialists. 220
Facial hair growth may persist despite the use of oestrogens and anti-androgens; therefore, TW may resort to various cosmetic procedures including shaving, waxing, plucking, electrolysis, and laser or intense pulsed light hair removal,210,211,221 though no studies have been done comparing the efficacy of different methods nor the timing of the procedures with hormone initiation. Eflornithine cream has been used for facial hair removal in cis women but not studied in TW, though effects are not permanent and may be a less desirable option. 210 More studies are needed to determine the most effective cosmetic procedures to facilitate unwanted hair removal, to identify predictors of achieving desired feminizing skin features, and how to better provide TW with access to these cosmetic procedures.
The brain and cognition
Recent studies have investigated baseline differences between the brains of trans versus cis individuals as well as the effect of GAHT on the brain and cognition. The white matter microstructure of TW prior to initiation of GAHT has been characterized by diffusion tensor imaging to be between control females and control males.222–225 Hahn and colleagues 226 also described different structural connectivity networks in 21 TW prior to initiating GAHT compared with control females and males. Seiger and colleagues 227 analyzed brain MRIs of 14 TW (mean age 26.9 ± 6.1 years) at baseline and after at least 4 months (169 days ± SD 38 days) of continuous oral or transdermal oestradiol and anti-androgens (cyproterone acetate ± GnRHa ± finasteride) and found decreases in the hippocampal region, increases in the ventricles and a correlation between progesterone levels and changes in grey matter structure. Mueller and colleagues 228 found neuroanatomical volume differences in the amygdala, putamen and corpus callosum in TW compared with cis women but not cis men, suggesting the possibility of localized influence of sex hormones on neuroanatomy. These studies suggest there are hormonal influences on cortical and subcortical structures related to cognition, memory and emotional processing. Reviews stress that additional short- and long-term studies are needed to better understand the role of sex hormones on the adult human brain structure and function and how they relate to psychological differences between females and males.229,230
Brain-derived neurotrophic factor (BDNF) is involved in neurogenesis, neuronal maturation and synaptogenesis, influencing brain plasticity. A study of 10 TW from Belgium showed a significant decrease in BDNF levels after 12 months of GAHT (p = 0.014) independent of age, weight, BMI, total fat mass, total lean mass, LH, FSH, oestradiol, testosterone, cortisol, physical activity or smoking. 231 Therefore, it was concluded that the decreased BDNF in TW after GAHT resulted from the GAHT rather than as a consequence of or risk factor for gender identity. 231
Nguyen and colleagues 232 published a recent review of cross-sectional and longitudinal studies from the last 5 years, nearly all from European cohorts, and summarized fewer depressive symptoms, anxiety, problems with socialization and interpersonal functioning, global functional impairment, interpersonal sensitivity and hostility in TW on GAHT compared with baseline. They also summarized the general findings above that GAHT is correlated with changes in global and regional brain volumes, white matter microstructure and in cognitive performance on sex-biased tasks requiring verbal and visual memory in the direction of gender identity rather than assigned sex. 232 However, no studies have examined how GAHT influences executive function and cognitive domains used for daily living, and additional research will provide valuable data on these and other brain processes.
Aging
Regarding older TW (arbitrarily defined as aged >50 years), very little data exist on the short- and long-term effects and safety of GAHT.1,2,23,30,233 Aging-related questions yet to be answered include: (1) Do older TW on chronic oestrogen therapy (± anti-androgens) have similar biological–endocrinological status as age-matched cis women?; (2) Which doses and modes of administration are the safest in older age?; (3) Is there an age at which GAHT initiation is not safe to initiate?; and (4) For how long should hormones be continued in the elderly? Answers to these questions will help us address both chronic effects as well as older age-specific effects from GAHT.
Despite the valuable data on the risks of VTE, CV events and breast cancer with conjugated equine oestrogens ± medroxyprogesterone acetate in postmenopausal women aged 50–79 years during the Women’s Health Initiative (WHI) in the US,234–237 extrapolating these data from WHI to TW cannot be done completely due to sex chromosomes, anatomic and physiological differences, and the administration of oestrogens ± progestins not routinely used in GAHT for TW. More research is needed in this area of short- and long-term safety of oestradiol formulations in older TW, particularly with dosing, goal hormone levels, routes of administration and duration of treatment.
The US Veterans Health Administration (VHA) has been and will be a significant source of data among older trans people;238–240 however, published retrospective studies on mortality and other medical and mental health disparities among VHA veterans have mostly looked at the general trans veteran population (rather than TW specifically) and did not take into account presence or type of GAHT nor gonadectomy status. Nonetheless, important data include: (1) similar top 10 causes of death among trans VHA veterans (mean age of death 64.2 years; range 25.0–91.0) and the US population at the time (with diseases of the circulatory system and neoplasm being the top 2); 241 (2) higher crude suicide rates among the trans cohort compared with the general VHA and US populations and occurred at a younger mean age (49.4 years; range 25.0–73.0) than the general VHA population; 241 and (3) higher odds of 10 mental health diagnoses and 14 of 17 medical diagnoses, including HIV and multiple CV risk factors, among the trans veterans compared with the cis veterans. 134
Other trans cohorts with promising proportions of older TW to allow for additional assessments of age-related comorbidities and associations with GAHT include the Amsterdam Gender Clinic, which reported 700 of 4,432 (15.8%) TW reached the age of 60 years or above in 2015, 30 and the STRONG study that will follow a transfeminine cohort of 3,475, of whom 16% were aged 46–55 years and 14% were aged >55 years at study enrolment. 101 In contrast, the European Network for the investigation of Gender Incongruence (ENIGI) has often published results on a variety of topics discussed above in mostly younger cohorts, though the ENIGI Endocrine protocol, which began in 2010, had a wide range of ages in the TW cohort (median 30 years; range 16–65) that was described in 2015. 138
The TW population continues to age and there are increasing numbers of older TW in many of the cohorts described above. Older TW deserve as much thoughtful and sensitive care as younger TW and nontrans older adults. The intersection of aging and transgender health is a novel area of research that can focus on the impact of aging on TW health outcomes, individual goals and desires associated with aging, and end-of-life and palliative care issues.
Conclusion
Trans health is an evolving field that includes physiological, psychological and societal considerations for the optimization of health. In addition to the increasing presence of gender diversity in the mainstream media, the medical literature has benefited from more publications on the risks and benefits of GAHT on the lives of trans people. In this narrative review, we aimed to comprehensively summarize important health considerations specific to TW that are important to them and their providers. We included the consistent evidence that GAHT improves QOL and mental health, has modest effects on breast development and positively impacts bone health in TW. Careful interpretation of TW’s baseline characteristics and type of GAHT regimen is needed when evaluating studies showing increased relative (albeit still low absolute) risks of VTE and stroke in TW compared with cis women and men. Nonetheless, CVD risk factors (e.g. obesity, smoking, diabetes, hypertension, hyperlipidaemia) should be optimized if present. Infertility risk and fertility desire should ideally be addressed prior to the start of GAHT. TW appear to have higher risk of breast cancer compared with cis men but not cis women, so we recommend following local population guidelines for breast cancer screening in women. Although we could not address all aspects related to TW’s health, we included topics not often addressed or elaborated on in other reviews such as HIV/AIDS, other tumours, voice therapy, dermatologic considerations, the brain and cognition and aging.
Additionally, we identified areas of future research (Table 4) that will hopefully provide answers to persistent questions surrounding TW’s health. We strongly advocate for more RCTs and large prospective cohort studies in order to further build confidence that the benefits of feminizing GAHT outweigh the risks for TW. We anticipate rigorous data to continue to be shared from ENIGI, STRONG, GETS and any other cohorts that may be getting underway soon. We also eagerly await the new WPATH SOC, version 8, to provide its up-to-date global perspective of evidence-based recommendations for trans health, adding to a growing literature that is helping healthcare providers take care of their trans patients.
Suggested areas of research still needed to address health considerations for TW.

AMAB, assigned male at birth; BMD, bone mineral density; CVD, cardiovascular disease;
DXA, dual-energy X-ray absorptiometry; GAHT, gender-affirming hormone therapy; GAS, gender-affirming surgery; GnRHa, gonadotropin-releasing hormone agonist; PSA, prostate-specific antigen; RCT, randomized controlled trial; TW, transgender women.
Footnotes
Acknowledgements
This collaboration was supported in part by an American Association of Clinical Endocrinologists and American College of Endocrinology (AACE/ACE) Lewis E. Braverman, MD, MACE, Education Fund (SJI; co-mentors: GT and MSR).
Author’s Note
SJI conceived of the review, participated in the design and coordination and drafted the manuscript. JD conceived of the review, participated in the design and drafted the manuscript. MSR participated in the design and drafted the manuscript. JVS drafted the manuscript. LVB drafted the manuscript. JM participated in the design and drafted the manuscript. GT participated in the design and drafted the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This perspective review received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. SJI is supported by an Advanced Fellowship in Geriatrics from the Geriatric Research, Education, and Clinical Center, Rocky Mountain Regional VA Medical Center, VA Eastern Colorado Health Care System, Office of Academic Affiliations, US Department of Veterans Affairs.
Conflict of interest statement
GT has scientific grants as Principal Investigator from Ipsen, Bayer Shering and Sandoz. GT receives a consulting fee as an advisory board member for Ipsen and Novartis. GT receives a lecturer fee as a speaker for Ferring and Novartis. The remaining authors declare that they have no conflict of interest.
