Abstract
Background:
Hyperglycemic emergencies (HGEs) are the major deadliest acute complications of diabetes. HGEs have reached an alarming stage and increased year-to-year leading to increased morbidity, hospitalization, and mortality. Despite HGEs causing this increased healthcare, psychological, social, and economic burden, studies conducted to address this burden and its predictive factors remain limited. Thus, this study aimed to investigate the incidence and predictors of HGEs among adult diabetic patients.
Methods:
An institution-based retrospective follow-up study was employed on 538 systematically selected adult diabetic patients who had diabetic follow-up in Sidama region and Gedeo zone public hospitals from September 1, 2018, to September 1, 2022. The sample size was determined using STATA V-14. Data were collected using an extraction checklist, entered into EPI data version 4.4.2.2, and analyzed using STATA version 14. The Kaplan–Meier curve and log-rank test were used to determine the survival probabilities and to compare the survival status. The Cox proportional hazard regression model was used to determine the association and identify the predictor variables. A statistical significance was declared at a p-value of <0.05 in line with a 95% confidence interval (CI) and hazard ratios.
Results:
The study was conducted on 538 diabetic adult patients with a response rate of 100%. The mean age of study participants was 44.5 years, and more than 66.7% were males. The incidence rate of HGEs was found to be 29 (95% CI: 25.3–33.2) per 1000 person-months with a total of 7176.5 person-month observations. Being farmer (adjusted hazard ratio (AHR) = 6.47; 95% CI: 2.61–16.04), poor glycemic control (AHR = 6.84; 95% CI: 3.47–13.49), less frequent diabetic follow-up (AHR = 4.00; 95% CI: 1.02–15.57), and having hypertension (HTN) (AHR = 2.94; 95% CI: 1.62–5.34) were significantly associated with increased hazard of acquiring HGEs among adult diabetic patients. Conversely, the hazard of experiencing HGE was 63% lower among patients who had diabetic nephropathy relative to those without diabetic nephropathy (AHR = 0.35; 95% CI: 0.15–0.83). Hence, setting and strengthening specific diabetic management strategies focused on the identified predictors could be paramount to reducing HGEs and their unwanted effects. Moreover, it’s better to consider more frequent diabetic follow-up visits for all patients regardless of other complications.
Introduction
Diabetes mellitus (DM) is a top driver of mortality and morbidity worldwide, accounting for more than 6.7 million adult deaths globally. 1 Diabetes causes several acute complications, including hypoglycemia and hyperglycemic emergencies (HGEs).
Diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS) are the commonest HGEs. HGEs are among the deadliest short-term endocrinologic complications resulting mainly from poor glycemic control.2–4 HGEs have reached an alarming stage and increased year-to-year contributing to increased morbidity, hospitalization, and mortality.5–7 The mortality rate of diabetic patients who developed HGEs ranges from 5% to 20%. 8 This figure was raised to 34% in developing countries.9,10
Different risk factors are presented that strike HGEs unexpectedly. Poor glycemic control, newly diagnosed DM, interruption of therapy, patient nonadherence to therapy, infections, depression, alcohol intake, substance abuse, and comorbidities appear to play roles in the unexpected occurrence of HGEs. Generally, sociodemographic and behavioral, therapeutic, and clinical profiles were potential risk factors for HGEs.11–13 Prompt diagnosis and treatment of HGEs is paramount to decrease related morbidity and mortality. Insulin therapy, electrolyte, and fluid replacement are vital management strategies of HGEs.14–16
Delayed and improper management of HGEs is frequently associated with several complications including cerebral edema, pulmonary edema, hypoglycemia, thromboembolism, electrolyte disturbances, shock, and renal failure. HGEs can be prevented by improving healthcare access, identifying precipitating factors, and proper diabetic education.17,18 In addition to the unwanted health consequences, the socioeconomic burden of HGEs is indisputable. Because most HGE patients are managed in intensive care units (ICUs), the costs are markedly higher and even impossible to maintain in developing countries. 14 Nowadays, in Ethiopia, HGEs impose a huge challenge to cover medical costs and hospitalization, 19 which leads to increased mortality and lifestyle disturbances.
Despite HGEs causing this increased healthcare, psychological, social, and economic burden at the individual, community, and country level, studies conducted to address this burden and its predictive factors remain limited. Available studies investigated the patient outcome, characteristics, and factors associated with mortality after HGEs.3,20,21 Moreover, earlier studies conducted in Ethiopia addressed only DKA without considering HHS, whereas some others investigated all acute complications of DM by merging both hypoglycemic emergency and HGEs simultaneously, which makes the interpretation of findings difficult. Therefore, this study aims to determine the incidence and identify predictive factors of HGEs among adult diabetic patients.
Materials and methods
Study area, design, and period
A retrospective follow-up study was employed among adult diabetic patients who had DM follow-up at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia, involving 4 years of data (from September 1, 2018 to September 1, 2022). Twelve hospitals in the Sidama region have separate chronic outpatient departments (OPD), and only three have separate diabetic clinics (Hawassa University Comprehensive Specialized Hospital, Adare Hospital, and Yirgalem General Hospital (YGH)). Overall, there were 1389 diagnosed diabetic patients in these hospitals. Gedeo zone has four hospitals. However, only Dilla University General Hospital has separate chronic OPD. Of the 330 registered DM patients, 303 had followed up at Dilla University General Hospital. 22 The study was conducted from September 18, 2023, to November 18, 2023.
Population of the study
All adults diagnosed with DM and having chronic diabetic follow-up at public hospitals in Sidama region and Gedeo zone public hospitals were the source population. All adult diabetic patients who had chronic DM follow-up at selected public hospitals in Sidama region and Gedeo zone public hospitals from September 1, 2018 to September 1, 2022. Patients who developed DKA at DM diagnosis were not considered during data extraction. Registries without follow-up data were excluded as well.
Sample size and sampling procedure
The sample size was determined using the Cox model by STATA version 14 by considering assumptions of the Cox proportional hazard (PH) model. Noncompliance to DM medications was used as a predictor variable with a probability of HGEs = 0.124 from a previous study conducted in Jimma, 3 which provides the largest possible sample size. The following parameters were considered: a 95% confidence level, 80% power, an estimated effect size = 2.0, and anticipated withdrawal = 0.01. Accordingly, the required sample size for the study was 538.
Of the 24 available hospitals, only 4 Hawassa Comprehensive Specialized Hospital (HUCSH = 546), YGH (YGH = 265), Adare General Hospital (AGH = 578), Dilla Referral Hospital (DUGH = 330) had organized chronic OPD with considerable patient flow. Hence, all the four hospitals were considered. The calculated sample size was proportionally allocated to each selected hospital based on patient flow. Medical records of adult DM patients were filtered from diabetic follow-up and discharge logbooks (medical ward, emergency room, and OPD). Then, individual patient recordings were selected using a systematic random sampling technique with replacement. Every three patient charts were selected after determining the sampling interval (K = 1719/538 ≈ 3) and listing in order of their medical record number. Missed patient charts and recordings with incomplete data were replaced with the next chart. Charts without records of definitive variables (date of DM diagnosis and date of the event or last observation) were considered incomplete (Figure 1).

Flowchart presenting the sampling and data extraction procedure to assess hyperglycemic emergency and its predictors among adult diabetic patients at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia, 2023/24 (n = 538).
Operational definitions
HGE (event) was declared when there was a diagnosis of DKA, HHS, or both confirmed by RBS >250 mg/dl and urine ketone body >+2 for DKA, and RBS >600 mg/dl with minimal or absent urine ketone body for HHS plus clinical diagnosis. 20
Data collection tool and procedure
The data extraction checklist was designed after reviewing related literature.3,11,24,25 The checklist comprised sociodemographic, behavioral, clinical profiles, and therapeutic variables. Patient charts were retrieved using the medical registration number. The extracted data was coded to avoid duplication. Six data collectors (nurses with bachelor’s degrees) and four supervisors (health professionals with master’s degrees) collected the data.
Data quality assurance
Before collecting the data, the checklist was evaluated by experts and pre-tested on 5% of the calculated sample size. Afterward, the required amendments were made to the checklist. One-day training was given to data collectors and supervisors by the investigators before actual data collection regarding the purpose of the study, data collection tools, correct completion of the checklist, and ethical considerations to conduct the data collection. The collected data was checked daily for completeness.
Data management and analysis
The collected data was coded, cleaned, and checked for consistency and completeness. Then, it was entered into EPI data version 4.4.2.2, and exported into STATA version 14 for analysis. Descriptive statistics and summary measures were computed to determine the nature of data, frequency distributions, and summary statistics. The Kaplan–Meier (KM) curve was constructed to determine the time to HGE occurrence and cumulative survival probabilities at a point in time. The log-rank test was used to compare the survival status between different categories of covariates.
The Cox PH regression model was applied to conduct bivariable and multivariable analyses. Covariates with a p ⩽ 0.20 in the bivariable analysis were transferred to multivariable analysis for confounder adjustment. PH assumptions were checked using the Schoenfeld residuals test (global test = 0.0639), and the model fitness test was checked using Cox–Snell’s residual test (Figure 2). The findings showed that the PH assumptions were fulfilled, and the model was fitted well to the data. A multicollinearity test was also conducted using the variance inflation factor (VIF = 1.72), and no evidence of collinearity was found. Finally, statistical significance was declared at a p-value of <0.05 in line with a 95% confidence interval (CI). The hazard ratio was used to report the strength of associations.
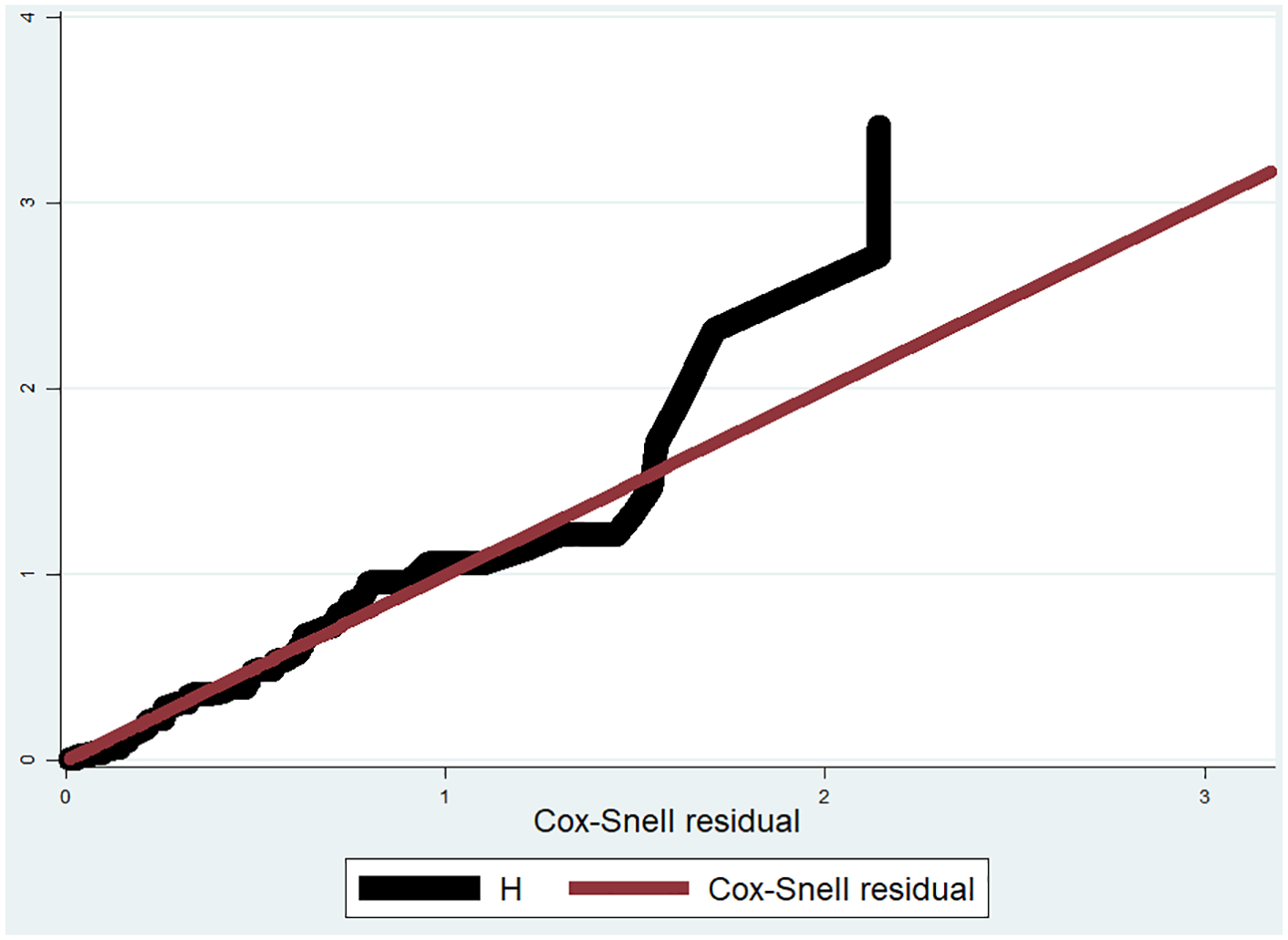
Cox–Snell residual cumulative hazard graph for hyperglycemic emergency and its predictors among adult diabetic patients at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia, 2023/24 (n = 538).
Ethics approval and consent to participate
An ethical approval letter was obtained from the Institutional Review Board (IRB) of Dilla University, College of Health and Medical Science (protocol unique number=
Results
Description of sociodemographic characteristics
An overall 538 diabetic patient records, which met the eligibility criteria were incorporated in the analysis. More than two-thirds of the samples were males whereas about 36% (193/538) reside in rural areas. Additionally, 66.2% were living with their spouse, and 15.8% were divorced or widowed. Furthermore, 43.5% of diabetic patients were not incorporated into the community health insurance program (Table 1). The average age of the study participants was reported to be 44.5 years with a standard deviation of (SD = 15.4). The mean age of patients who were diagnosed with HGEs was found to be 38 years (SD = 15.4), and this figure was escalated to 48.5 years (SD = 14) for those without HGE. Moreover, three-fourths of diabetic patients who developed HGE were under the age of 49 years. Contrarily, half of those who did not experience HGE were above 50 years of age (Table S1 in S1 File).
Sociodemographic characteristics among adult diabetic patients at public hospitals of Sidama Region and Gedeo zone, Southern Ethiopia, 2023/24 (n = 538).

Description of clinical characteristics
The majority (75%) of the patients were diagnosed with type 2 DM. Despite this, a high proportion (72.6%) of type 1 diabetic patients had developed HGE, while only 27.3% of type 2 diabetic patients had HGE. On the other hand, the majority (59.3%) of the patients used oral hypoglycemic agents (OHA; either metformin or Glibenclamide), whereas 8.9% of the patients used a combination of OHA and insulin. Besides, 42.4% of the patients had preexisting illness. HTN (39.4%) and ischemic heart disease (3.9%) were the most frequently identified comorbidities. Likewise, 12% of the patients had chronic diabetes complications (Table 2).
Clinical profiles of adult diabetic patients at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia, 2023/24 (n = 538).

BPH, benign prostate hyperplasia; CKD, chronic heart diseases; DM, diabetes mellitus; HHD, hypertensive heart diseases; HTN, hypertension; IHD, ischemic heart diseases; OHA, oral hyperglycemic agents.
HGE among study participants
Of the 538 patients, 208 (38.7%) developed either of the 2 HGEs. One hundred fifty-one (72.6%) patients who developed HGE had DKA and the rest 27.4% had a hyperosmolar HHS. Regarding the episode HGE, 18.3% of the patients experienced HGE at least 2 or more times. Again, 96.6% of patients diagnosed with HGE had 1 or more polysymptoms, whereas only 5.4% of diabetic patients without HGE had polysymptoms. Additionally, 41.8% of those who developed HGE experienced adverse health events/complications (Table S2 in File S1).
Incidence of HGE
The median survival time was 37.1 (95% CI: 35.6–39.4) with an overall 7176.5 person-month observations recorded. The incidence rate of HGE was found to be 29 (95% CI: 25–33) per 1000 person-month observations or 35.3 (95% CI: 31.0–40.0) per 100 person-year observations. Specifically, the incidence of HGE in the first week of follow-up was reported to be 109 (95% CI: 91–131) per 100 person-month observations. Yet again, the incidence rate of HGE in the first month was 31 per 100 person-month observations (95% CI: 26–37). The cumulative probability of developing HGE at the end of the first week, 6 months, 12 months, 24 months, 36 months, and 45 months was 0.22, 0.29, 0.34, 0.38, 0.46, and 0.60, respectively (Table S3 in File S1). When we see the occurrence rate for each type of HGE, the incidence of DKA and HHS was 28.6 (95% CI: 25–33) and 9.70 (95% CI: 7.0–12.0) per 100 person-year observations, respectively.
KM curve and log-rank tests
As we can note from the KM failure curve, the probability of developing HGE increased sharply in the first week of follow-up and then rose step-wise to 60%. This implies that as the follow-up time increases, the probability of developing HGEs increases as well. We can also note that the graph crosses the 50% survival probability at 37 months (Figure 3). As such, 50% of the participants will have the probability to develop HGE after being followed for 37 months. The survival difference between categories of each covariate was described using the log-rank test statistically and graphically (Figure S2).

Overall Kaplan–Meier failure estimate for HGE and its predictors among adult diabetic patients at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia, 2023/24 (n = 538). The Y-axis represents the probability of occurrence of HGEs whereas the X-axis indicates the follow-up time in months. The black vertical and horizontal lines were reference lines added to ease graph interpretation (median time to switch estimation).
PH assumption tests and model fitness
The Cox-PH assumption was assessed using the Schoenfeld residuals test. The output for all covariates was above the p-value of 0.05, and thus, none of the variables incorporated into the Cox model violated the PH assumptions. The overall global test was 0.0639, which implies that the assumptions were fulfilled and the model fitted to the data well. The Cox-PH model fitness was also checked graphically using Cox–Snell’s residual. The Cox–Snell graph runs tangentially to the hazard line (45-degree line) except for large values over time, which denotes the model fitted to the data successfully (Figure 2).
Predictors of HGE
The Cox PH regression model was used to identify the predictors of HGEs among diabetic patients. Variables that fulfilled the selection criteria based on the bivariable analysis output were fitted to the multivariable analysis. Consequently, being a farmer, having HTN, diabetic nephropathy, poor glycemic control, and longer diabetic visits were found to have a statistically significant association with the occurrence of HGE (Table 3).
Multivariable Cox proportional hazard regression analysis for the predictors of HGEs among adult diabetic patients in Sidama region and Gedeo Zone public hospitals, Southern Ethiopia, 2023/24 (n = 538).

Statistical significance at 95% CI.
CI, confidence interval; DM, diabetes mellitus; GO, governmental organization; HGE, hyperglycemic emergencies; NGO, non-organizational organization.
Diabetic patients who managed their lives by farming were 6.47 times at an increased hazard of developing HGE (adjusted hazard ratio (AHR) = 6.47; 95% CI: 2.61–16.04) compared to their counterparts. Likewise, having a co-existing HTN was linked to a tripled hazard of acquiring HGE (AHR = 2.94; 95% CI: 1.62–5.34) than those without HTN. Conversely, the hazard of experiencing HGE was 63% lower among patients who had diabetic nephropathy relative to those without diabetic nephropathy (AHR = 0.35; 95% CI: 0.15–0.83). Diabetic patients with poor glycemic control had 6.84 times escalated hazard of developing HGE (AHR = 6.84; 95% CI: 3.47–13.49) compared to their counterparts. The hazard of experiencing HGE among patients who attend diabetic follow-up every 4 months or longer was quadrupled compared to patients who had shorter diabetic visits (AHR = 4.00; 95% CI: 1.02–15.57).
Discussion
Determining the incidence of HGEs and identifying the predictive factors among diabetic patients is essential for the better prevention, diagnosis, management, and control of diabetes and its complications. The study findings will also aid clinicians and patients in recognizing the predictors and applying them in actual practical settings.
The study findings showed that the incidence rate of HGE was 35.3 (95% CI: 31.0–40.0) per 100 person-year observations. This finding was remarkably higher than the figure reported in previous studies conducted in Bahir Dar, Ethiopia, 26 Sao Paulo, Brazil, 13 and Thailand. 17 This discrepancy might be due to the difference in the age distribution of study participants. In our study, a significant proportion of the study participants were younger (mean age of 38 years among those who developed HGE) compared to previous studies (mean age ranges from 47 to 55 years in those studies). As per existing evidence, the incidence of HGE tends to be higher among the young population group. The other possible reason could be the difference in sample size where we have used a relatively larger sample size compared to the studies conducted abroad (these studies were conducted on just 52 and 83 samples).13,17 The advancement in diagnostic and therapeutic technologies and follow-up modalities might also enable better detection and prevention of HGEs in the developing world such as Thailand. Additionally, the study conducted in Brazil did not consider patients with a diagnosis of HHS, which might overlook the overall incidence of HGE.
The current study also identified pertinent predictive factors for HGE. Being a farmer is one of the predictive factors for HGE among diabetic patients. As per the researchers’ effort and knowledge, we could not access similar articles that directly evaluate and report the predictive effect of occupation on HGE. Nonetheless, some of these studies reported that the absence of a nearby health facility at a reasonable distance was associated with an increased risk of acquiring HGE. 27 This could be one of the justifications for our findings. Because, there is limited access to healthcare services (especially for services requiring specialty care such as chronic diabetics and cardiac follow-ups) in the rural parts of Ethiopia, where the majority of the farmers reside. 28 Consequently, farmers tend to have relatively infrequent follow-up visits or might miss their schedule (lost from DM follow-up), which will in turn lead to poor glycemic control and the rise of diabetic complications such as HGE. On the other hand, the most likely and convincing explanation could be the delay in the detection and diagnosis of DM among farmers due to the low utilization of healthcare services in the agrarian population. This was evidenced in the recent study conducted in the Sidama region. 29 Furthermore, farmers tend to have poor compliance with DM medications due to the lack of understanding compared to the urban population due to limited access to television, radio, or newspapers. Poor compliance with diabetic medications, in turn, could lead to poor glycemic control and hyperglycemic complications.3,26
Likewise, the study findings also showed that diabetic patients who had co-existing HTN were more likely to develop HGE compared to their counterparts. Findings of former studies conducted in Hawassa, 24 Bahir Dar, 26 and Jimma Medical Center 27 conformed with our study findings. The scientific explanation could be the increased insulin resistance secondary to increased peripheral arterial resistance in hypertensive patients. Mostly, diabetic patients experience vascular remodeling due to increased body fluid volume. This increased body fluid and vascular remodeling accounts for increased peripheral vascular resistance, which leads to elevated systemic blood pressure. Consequently, patients may experience insulin resistance and decreased insulin uptake due to this resistance, and thus, hyperglycemia may occur. 30 Similarly, evidence showed that hypertensive patients are more likely to experience elevated blood sugar levels due to its synergistic role in this mechanism. Both diabetes and HTN share similar precipitating factors such as inflammation, stress, obesity, blood vessel thickening, and immune function. 31 Furthermore, hyperglycemia could result from low insulin concentration and high counterregulatory hormones. 32 It is also linked with the increased release of inflammatory cytokines, lipid peroxidation, reactive protein, oxygen species, and cardiovascular pathology. 32
Patients who developed diabetic nephropathy during follow-up had reduced hazard of experiencing acute diabetic complications such as HGEs. The possible explanation could be that insulin clearance is reduced in patients with advanced kidney disease, which will, in turn, lower the incidence of diabetic emergencies in this special population. Even though diabetic nephropathy and other chronic kidney pathologies disrupt insulin uptake and are linked to unfavorable outcomes including hyperglycemia,33,34 the tendency to concurrently develop acute diabetic complications such as HGEs might be decreased owing to the active treatments provided, frequent and strict follow-up visits as well as cautious monitoring of the patient’s response to the treatments given. This magnifies the importance of close monitoring and frequent follow-up for all diabetic patients regardless of other complications and the severity of illness.
Our study findings also indicated that less frequent DM follow-up visits (every 4 months or longer) showed a statistically significant association with the occurrence of HGE, which was supported by previous study findings.26,35 The possible justification might be frequent DM follow-up visits to enable timely detection and management of any fluctuation in the serum glucose level before complications occur. Besides, frequent visits are paramount to keep the patient attached to the care and, thus, enhance compliance with diabetic care recommendations and DM medications. Patients who had less frequent DM follow-up visits could miss such opportunities. This, in turn, could end up with a low treatment success rate and an increased chance of acquiring complications.
Additionally, patients with poorly controlled glycemic levels were at higher hazard of developing HGEs compared to their counterparts. This association was consistent with reports of earlier studies conducted at Jimma Medical Center 27 and Bahir Dar. 26 Glycemic control is the mainstay of diabetes management. Poorly controlled serum sugar levels can lead to chronic and acute diabetic complications. Poor glycemic control could result in hyperglycemia, one of the mechanisms that underlie the pathophysiology of DKA and HHS. Especially, in patients who had infrequent visits and were noncompliant with diabetic care instructions, there is no chance to manage the glycemic fluctuation, and hence, end up with hyperglycemic crisis.
The study finding generates pertinent points that could be used as an asset to the current science to improve the effectiveness of DM management. Considering censored data and time variables in the analysis and applying a relatively large sample size could increase the validity of our findings. Nevertheless, the study was not free of limitations, and some points need to be considered while interpreting and replicating the findings. First, since the study was conducted on secondary data via patient chart review, the effect of some possible predictor variables was not assessed. For instance, patients’ compliance with diabetic care packages and substance use were missed since they were not documented in the patient’s charts. Moreover, some patients might remain undiagnosed while having the disease for several months. Those patients might develop HGEs soon after the diagnosis of DM, which might inflate the incidence of HGE in our study. Unlike prospective studies, we had no suitable control/filtration mechanism for such cases during data extraction.
Conclusion
The incidence of HGEs among diabetic patients was found to be remarkably high compared to previous study findings and national as well as global reports. Being a farmer, having co-existing HTN, diabetic nephropathy, poorly controlled blood sugar, and less frequent DM follow-up were identified as independent predictors of HGEs. Hence, setting and strengthening specific diabetic management strategies focused on the identified predictors could be paramount to the existing science. Moreover, it’s better to consider more frequent diabetic follow-up visits for all patients regardless of other complications.
Supplemental Material
sj-docx-1-tae-10.1177_20420188241303416 – Supplemental material for Incidence and predictors of hyperglycemic emergencies among adult diabetic patients at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia: a survival analysis
Supplemental material, sj-docx-1-tae-10.1177_20420188241303416 for Incidence and predictors of hyperglycemic emergencies among adult diabetic patients at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia: a survival analysis by Kasie Gebeyehu Tiruneh, Migbar Sibhat Mekonnen, Addisu Getnet Zemeskel, Mequanint Ayehu Akele, Abinet Meno Abose and Ashenafi Zewde Wendimu in Therapeutic Advances in Endocrinology and Metabolism
Supplemental Material
sj-docx-2-tae-10.1177_20420188241303416 – Supplemental material for Incidence and predictors of hyperglycemic emergencies among adult diabetic patients at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia: a survival analysis
Supplemental material, sj-docx-2-tae-10.1177_20420188241303416 for Incidence and predictors of hyperglycemic emergencies among adult diabetic patients at public hospitals of Sidama Region and Gedeo Zone, Southern Ethiopia: a survival analysis by Kasie Gebeyehu Tiruneh, Migbar Sibhat Mekonnen, Addisu Getnet Zemeskel, Mequanint Ayehu Akele, Abinet Meno Abose and Ashenafi Zewde Wendimu in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
Appendix
Acknowledgements
The authors acknowledged data collectors, supervisors, hospital staff, and administrators for their unwavering commitment and cooperation. On top of this, the authors appreciate Dilla University for covering the data collection cost.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
