Abstract
Metabolic dysfunction-associated fatty liver disease (MAFLD), formerly known as nonalcoholic fatty liver disease, is becoming a significant contributor to chronic liver disease globally, surpassing other etiologies, such as viral hepatitis. Prevention and early treatment strategies to curb its growing prevalence are urgently required. Recent evidence suggests that targeting the gut microbiota may help treat and alleviate disease progression in patients with MAFLD. This review aims to explore the complex relationship between MAFLD and the gut microbiota in relation to disease pathogenesis. Additionally, it delves into the therapeutic strategies targeting the gut microbiota, such as diet, exercise, antibiotics, probiotics, synbiotics, glucagon-like peptide-1 receptor agonists, and fecal microbiota transplantation, and discusses novel biomarkers, such as microbiota-derived testing and liquid biopsy, for their diagnostic and staging potential. Overall, the review emphasizes the urgent need for preventive and therapeutic strategies to address the devastating consequences of MAFLD at both individual and societal levels and recognizes that further exploration of the gut microbiota may open avenues for managing MAFLD effectively in the future.
Keywords
Introduction
Metabolic dysfunction-associated fatty liver disease (MAFLD), formerly known as nonalcoholic fatty liver disease (NAFLD), represents a complex disease entity characterized by a broad range of manifestations from mild to severe hepatic steatosis, which can progress to fibrosis, cirrhosis, decompensated liver disease, hepatocellular carcinoma (HCC), and cardiometabolic-related events. 1 It is emerging as a significant factor in the global burden of chronic liver disease.2,3 It is poised to surpass other etiologies, including viral hepatitis, in becoming the leading cause for liver transplantation worldwide.4,5 The impact of MAFLD is exacerbated further by the growing prevalence of obesity, a leading etiology for metabolic dysfunction-associated steatohepatitis [MASH, formerly known as nonalcoholic steatohepatitis (NASH)].6,7
A multitude of factors contribute to the presentation and progression of MAFLD, such as age, gender, metabolic changes, activity, dietary habits, genetic predispositions, and the intricate balance of the gut microbiota.8–10 The latter plays a complex role in the gut–liver axis, where the gut microbiota, intestinal barrier, immune system, and liver interact intricately to influence the development and progression of MAFLD. 11
This review extensively explores the literature and new evidence to explain the factors influencing gut dysbiosis and its involvement in MAFLD pathogenesis. It summarizes techniques that aim to quantify gut microbiota parameters and use them as diagnostic tools for MAFLD. Lastly, the review discusses the emerging therapeutic interventions that target specific processes of gut dysbiosis to help alleviate symptom manifestations and disease progression of MAFLD. The gut microbiota may hold key mechanistic, diagnostic, and therapeutic opportunities for relieving the health and economic burden of MAFLD, and future studies and clinical trials should focus on these exciting possibilities.
Methods
A comprehensive literature search was conducted in May 2023 in PubMed and Embase databases using the keywords ‘metabolic dysfunction-associated fatty liver disease’, ‘nonalcoholic steatohepatitis’, ‘MAFLD’, ‘NASH’, ‘microbiota’, AND ‘microbiome’. The search encompassed articles published from January 2000 to May 2023. Preference was given to English language meta-analyses and randomized controlled trials (RCTs) in humans to ensure high-quality evidence. In cases where such studies were lacking, nonrandomized studies, controlled cohorts, or animal studies were also included, although with limitations. The search strategy aimed to identify relevant studies for an in-depth review, providing valuable insights into the relationship between MAFLD, the gut microbiota, and advancements in diagnosis and treatment approaches.
Results
From gut dysbiosis to MAFLD
The pathogenesis of MAFLD is complex and involves dynamic mechanisms. Accordingly, the proposed theory mandates a parallel and ‘multiple-hit’ dynamic interplay between multiple factors. One such factor that may strongly influence MAFLD symptoms and progression is gut dysbiosis, which includes dysregulation of gut permeability, diet, microbiome alteration, changes in gut receptors and biomarkers, endotoxemia, endogenous alcohol production, intestinal metabolites, short-chain fatty acids (SCFAs), and bile acids (BAs).12–14 The multilevel interactions and mechanisms of these factors that may collectively contribute to the development of MAFLD are illustrated in Figure 1. 15 The pathogenesis cascade can start with increased gut permeability, leading to enhanced bacterial translocation, and the release of toxic products, which in turn triggers inflammation mediated by multiple proinflammatory mechanisms, including Toll-like receptor (TLR) 4. The altered gut microbiota in patients with MAFLD includes an increased abundance of certain species (e.g., Proteus, Enterobacter, Escherichia, etc.) with a reduced population of others (e.g., Ruminococcus, Lactobacillus, etc.). In addition, disruption in the gut microbial balance is linked to changes in BA metabolism, modulation of farnesoid X receptor (FXR) stimulation, and alterations in fat and glucose homeostasis. The dysbiosis-induced release of metabolites, such as 2-butanone and 4-methyl-2-pentanone, along with ethanol production by gut bacteria, contributes to oxidative damage.16–24 Notably, variations in the gut microbiota composition and the Firmicutes/Bacteroidetes (F/B) ratio have been observed in different studies. In addition, they have not only been associated with MAFLD but also linked to its severity.21,25–29 Further comprehensive investigations are required to establish microbial signatures associated with MAFLD, enabling the development of targeted preventive and therapeutic strategies.

Interactions between gut microbiome, bile salts, lipid metabolism, inflammation markers, and hepatocytes in the pathophysiology of MAFLD 15
Gut permeability and endotoxemia
Multiple studies have established a connection between impaired gut barrier function and bacterial translocation.30–33 De Munck et al. 34 conducted a systematic review and meta-analysis of 14 studies addressing intestinal permeability and MAFLD. Increased permeability was noted on dual sugar tests and zonulin levels [0.79; 95% confidence interval (CI): 0.49–1.08 and 1.04 ng/mL; 95% CI: 0.40–1.68]. An association with hepatic steatosis was noted in four studies but not with hepatic inflammation or fibrosis. In individuals with MASH, compromised intestinal permeability is proposed as a potential mechanism, resulting in elevated serum endotoxin levels and subsequent liver injury. A total of 34 studies were included in the meta-analysis conducted by Soppert et al. 35 Serum endotoxin levels were notably elevated in simple steatosis versus healthy controls (0.86; 95% CI: 0.62–1.11) and in MASH versus MAFLD/MASH (0.81: 95% CI:0.27–1.35; p = 0.0078). Moreover, they observed an association between advanced disease histology and endotoxin levels, adding more evidence to the value of blood endotoxin levels as a potential future diagnostic and staging maker of MAFLD.
Diet and gut microbiota
Recent evidence has emphasized the significant impact of diet on the gut microbiota and its role in metabolic health. Diverse dietary regimens have the potential to induce varying alterations in the composition of the intestinal microbiome. Low-carbohydrate diets (LCD) and ketogenic diets (KD) have been shown to cause distinct alterations in the gut microbiota, including changes in Actinobacteria, Bacteroidetes, Firmicutes, and Bifidobacterium populations. For example, β-hydroxybutyrate synthesized during KD is linked to reduced Bifidobacterium abundance. Furthermore, the microbiota associated with KD has been found to reduce proinflammatory Th17 cells, suggesting potential anti-inflammatory effects. Another example is the ability of a high fructose diet to disrupt metabolism, increase energy intake, and induce microbiota dysbiosis through increasing intestinal permeability.36–40
The Mediterranean diet can positively alter gut microbiota composition, promoting intestinal barrier integrity and reducing inflammation and harmful bacteria like Escherichia/Shigella. Furthermore, high-fat diets (HFD) have been found to increase specific Lactobacillus species resistant to BAs, which may influence lipid metabolism and contribute to MAFLD development. 37 These findings highlight the dynamic interplay between diet, gut microbiota composition, and metabolic health, offering potential avenues for targeted interventions in patients with MAFLD.41–46
Toll-like receptors
The TLR signaling pathway plays a pivotal role in establishing a connection between gut dysbiosis and the initiation of MAFLD by detecting molecules from the gut microbiota.47–50 Activation of this pathway leads to the production of cytokines, and sustained elevation of these cytokines poses potential harm to the host. Recent literature highlights that TLR signaling contributes to the exacerbation of hepatic injury in various chronic liver diseases, encompassing conditions such as alcoholic liver disease (ALD), chronic viral hepatitis, and MAFLD/MASH.49,51–53 TLRs, including TLR2, TLR3, TLR4, TLR5, TLR7, TLR8, and TLR9, exhibit heightened sensitivity to microbial component alterations, such as peptidoglycans and lipopolysaccharides, distinguishing between physiological colonization and pathogenic presence and actively contributing to initiating inflammatory responses.49,54–56 Dysregulation of the lipopolysaccharide/TLR4 signaling pathway, affected by compromised gut barriers and dietary and microbial composition, emerges as a major mechanism in the pathogenesis and trajectory of MAFLD.49,57–59 Robust evidence supports the significant role of TLR4 in hepatic steatosis development, particularly through lipopolysaccharide-mediated activation, inducing NF-κB-dependent inflammatory cytokine production.49,60–62 Conversely, TLR9 signaling under chronic overnutrition stress serves as a driving force for hepatic steatosis progression, with decreased TLR9 expression mitigating steatohepatitis and liver fibrosis.49,63 However, TLR9 signaling can be associated with liver injury and may promote the progression of MAFLD.49,64–67 While the involvement of TLR2 in MAFLD remains controversial with variable outcomes, TLR3 signaling induces inflammatory processes, impacting cholesterol efflux genes and influencing proinflammatory cytokine expression.49,68–72
The progression from hepatic steatosis to liver fibrosis involves complex interactions between damaged hepatocytes, inflammatory signals, and hepatic stellate cells (HSCs).49,73 Contributing factors include genetic predisposition, advanced age, ethnicity, and comorbidities like obesity, dyslipidemia, and diabetes. 74 TLR4, activated by Lipopolysaccharides (LPS), plays a significant role in MAFLD-related fibrosis, regulating inflammatory responses.75–80 TLR2 and TLR3 exhibit conflicting roles in the literature, with studies demonstrating both profibrotic and antifibrotic effects in different models.69,81–84 TLR5’s role in liver fibrosis also conflicts, while TLR7 signaling demonstrates a protective role.49,85–87 TLR9, recognizing CpG-containing DNA, influences fibrogenic responses in HSCs, but its role is complex and conflicting, with both profibrotic and antifibrotic outcomes reported.49,51,88–90
Furthermore, TLRs are key players in the interplay between chronic hepatic inflammation and HCC pathogenesis. TLR4, implicated in HCC development, exhibits multifaceted roles by promoting proinflammatory and malignancy-related molecules (e.g., Treg cell counts) and contributing to HCC cell proliferation and resistance to apoptosis.91–93 TLR9 is associated with aggressiveness and poor prognosis in patients with HCC. 94 A similar link was noticed with TLR5.94,95 Literature on TLR3 signaling appears to show antitumor activity, suggesting therapeutic potential for HCC.49,93,96–98 Collectively, TLR4, TLR9, TLR5, and TLR3 emerge as potential targets for therapeutic interventions, highlighting the intricate involvement of TLRs in hepatic steatosis, progression to fibrosis, and HCC pathogenesis. 49
Macrophages
Macrophages emerge as pivotal contributors to the MAFLD and progression to steatohepatitis. Targeting their pathways emerges as a promising avenue for therapeutic strategies aimed at mitigating MAFLD progression. Clinical evidence highlights portal macrophage infiltration in the early stages of MAFLD, preceding overt inflammation and exhibiting association with hepatocyte damage and a positive correlation with disease severity.99–101 Depletion of macrophages through diverse methods showed potential protective benefits against steatosis development, highlighting the indispensable role of macrophages in the MAFLD dynamics.101,102 Additionally, proinflammatory macrophages contribute to hepatic insulin resistance, influencing the responsiveness of hepatocytes to insulin.101,103
In the broader context of chronic liver diseases, macrophages intricately interact with HSCs, establishing bidirectional signaling that profoundly influences inflammation and fibrosis.101,104 Notably, M2 macrophages, associated with hepatic injury in MAFLD, orchestrate a fibrotic response conducive to liver remodeling and tissue repair.101,105,106 The identification of ‘restorative’ hepatic macrophages in mice, Ly-6Clocells, introduces complexity with their human counterpart remaining to be clarified.101,107 Furthermore, macrophage autophagy has been implicated in attenuating liver fibrosis in mouse models.101,108 Overall, macrophages play important regulatory roles in inflammation, fibrosis, and fibrolysis at different stages of hepatic injury within the MAFLD spectrum.
Endogenous alcohol production
Several studies have proposed a connection between endogenous alcohol production and MAFLD. Studies have detected elevated blood alcohol levels in patients with MAFLD, indicating that gut bacteria, particularly Enterobacteriaceae, might contribute to endogenous alcohol production within the body.29,109,110 This internal alcohol production can lead to hepatotoxicity through both direct mechanisms and indirect pathways, including increased oxidative stress in the liver. Therefore, the alcohol produced by gut bacteria represents a potential factor in the development and progression of MAFLD and highlights the role of endogenous alcohol in the liver pathology associated with this metabolic disorder.29,111,112
BAs and SCFAs
The interplay between the gut microbiota, BAs, and their receptors plays a pivotal role in the development and progression of MAFLD. Patients with MAFLD show elevated levels of total fecal BAs, including cholic acid and chenodeoxycholic acid, along with an altered balance of primary and secondary BAs. Notably, BAs interact with FXR in the intestines, affecting BA absorption, transport, hepatic lipogenesis, lipid metabolism, and inflammation regulation.113,114 Manipulating the gut microbiota and antagonizing FXR in animal models have shown promising results in reducing hepatic lipogenesis and improving lipid metabolism. Moreover, stimulating the G-protein-coupled receptor 5 by gut-bacteria-derived secondary BAs influences glucose homeostasis through glucagon-like peptide-1 (GLP-1) action. Clinical trials using FXR agonists have demonstrated encouraging outcomes in ameliorating hepatic steatosis and reducing inflammation in patients with MAFLD. Collectively, these findings underscore the significant role of gut-microbiota-mediated alterations in BAs and their receptors in lipid metabolism, glucose homeostasis, and the pathogenesis of MAFLD.22,115–120
Microbiota-derived metabolites
Gut microbiota produces SCFAs like acetate, propionate, and butyrate through fermentation. SCFAs are important to maintain intestinal integrity and function.121,122 They serve as important precursors for gluconeogenesis and lipogenesis, providing energy in normal conditions. The activation of G-protein-coupled receptors by SCFAs triggers the release of peptide YY and GLP-1, contributing to feelings of satiety and reduced food intake.123–125 Moreover, SCFAs activate adenosine monophosphate-activated protein kinase, promoting hepatic autophagy and lipid oxidation. They also inhibit histone deacetylases, influence gene transcription, and exhibit potential anti-inflammatory properties.126,127 Studies have revealed variations in SCFA levels among individuals at different stages of MAFLD, and experiments involving SCFA supplementation in animal models have demonstrated beneficial effects on inflammation in the liver and adipose tissue.128–130
Genetics, microbiota, and MAFLD
Heritability of MAFLD, estimated to be around 20–70%, 131 is underlined by gene–environment interactions, supported by epidemiological, familial aggregation, and twin studies extending even to disease-related metabolic traits. Genes involved with steatosis pathogenesis may also be involved with fibrosis pathogenesis. Hepatic steatosis and fibrosis had a highly significant shared gene effect of 0.756 (95% CI: 0.716–1, p < 0.0001). 132 We are in an era of genome-wide association studies and gut microbiome signatures where many key genetic variants shaping MAFLD manifestations and severity have been identified, notably variants in PNPLA3, transmembrane 6 superfamily member 2 (TM6SF2), glucokinase regulator (GCKR), membrane-bound O-acyltransferase domain-containing 7 (MBOAT7), and hydroxysteroid 17β- dehydrogenase (HSD17B13). This is a major milestone in advancing the wheel of MAFLD treatment targets and personalized medicine approaches.
The PNPLA3p.I148M polymorphism is a cornerstone variant associated with the entire spectrum of MAFLD. It is also associated with an increased risk for disease progression and the occurrence of liver-related events and HCC throughout the literature.133,134 Elevated body mass index (BMI), the presence of the PNPLA3 risk variant, diminished relative abundances of Faecalibacterium sp. or Prevotella sp., augmented relative abundances of Gemmiger sp., and dietary patterns characterized by low fiber and specific vitamin content, coupled with enrichment in amino acids, uric acid, and purine, emerge as pivotal determinants influencing the severity of MAFLD. 135
The contribution of impaired TM6SF2, a major regulator of plasma lipids levels, function to MAFLD was described first in 2014.136,137 This has been further validated in the literature and a meta-analysis.138–142 Elevated levels of lipopolysaccharide-binding protein (LBP) were observed in individuals diagnosed with NASH. Intriguingly, individuals carrying the TM6SF2 rs58542926 T-allele, associated with susceptibility to NAFLD/NASH, exhibited higher LBP levels. TM6SF2 exhibits pronounced expression in the gastrointestinal tract, potentially surpassing its expression levels in the liver. This prompted the hypothesis that intestinal TM6SF2 plays an additional role in the progression of MAFLD by augmenting endotoxemia. 143 The link between TM6SF2 polymorphism and endotoxemia requires further investigations and studies.
Loss-of-function mutation (rs1260326) coding for the P446L protein in the GCKR gene variant, governing de novo lipogenesis, has been linked to MAFLD.144–146 MBOAT7 has a critical role in processes pivotal for systemic immune homeostasis, including for a broad range of TLR responses. These effects are further regulated by the genotype at MBOAT7 rs8736. Modulation of MBOAT7 may provide therapeutic benefits for suppressing inflammation in human diseases associated with dysregulation of the TLR signaling cascade, including in MAFLD. 147 HSD17B13 gene (rs72613567:TA) is strongly linked to decreased serum transaminase levels and a reduced risk of NASH through its lipid droplet-associated retinol dehydrogenase activity. Yet, further functional studies are required to clarify its role in MAFLD.148–150
The dynamic interplay between gut microbiome communities and their interactions with the host holds significant implications for the initiation and advancement of MAFLD, presenting a promising avenue for the identification of novel diagnostic and prognostic biomarkers. In the early stages of MAFLD, Desulfobacteraceae bacterium, and Mushu phage emerged as pivotal hub species. On the other hand, Fonticula alba, Faecalibacterium prausnitzii, and Mushu phage activity were identified as critical regulators influencing the progression toward steatohepatitis. 151
Lean MAFLD and microbiota
Lean MAFLD constitutes a globally acknowledged distinct pathophysiologic entity, with approximately 47–65% of cases exhibiting steatohepatitis. 152 While there is a recognized need for additional evidence to comprehensively characterize its spectrum, including disease pathogenesis, natural history, and prognosis, the prevailing definition centers on hepatic steatosis with a BMI below 25 kg/m² (or below 23 kg/m² in Asians).153–156 Chen et al. noted distinct disparities in gut microbiome constitution and bile acid profiles when comparing lean MAFLD to its nonlean counterpart. Notably, patients with lean NAFLD displayed elevated levels of total, primary, and secondary BAs compared to their nonlean counterparts, with statistical significance observed specifically for secondary BAs (p = 0.01). The composition of individual BAs also differed, as lean patients exhibited lower levels of deoxycholate, glycochenodeoxycholic acid, and chenodeoxycholic acid but higher levels of glycocholic acid compared to nonlean patients. Intriguingly, no significant difference between lean and nonlean patients was observed concerning more severe fibrosis. Patients with lean MAFLD exhibited enrichment in Erysipelotrichaceae UCG-003, Ruminococcus, Clostridium sensu stricto 1, Romboutsia, and Ruminococcaceae UCG-008. Conversely, Ruminiclostridium and Streptococcus were enriched in patients with obesity and MAFLD (Mann–Whitney test, p < 0.05). Notably, these changes remained significant for Ruminococcaceae UCG-008 even after correction for multiple comparisons (FDR p = 0.010). Consistent trends were observed in mice subjects with lean NAFLD, mirroring what was observed in humans, particularly the abundance of Ruminococcaceae bacterial family. These trends were further evident in several phylotypes within the Erysipelotrichaceae. 157 Duarte et al. observed that patients with NASH exhibited notable distinctions in the abundance of Faecalibacterium, Ruminococcus, Lactobacillus, and Bifidobacterium compared to the control group. Specifically, patients with lean NASH had a threefold reduction in the abundance of Faecalibacterium and Ruminococcus (p = 0.004). In contrast, patients with obesity and NASH demonstrated an abundance of Lactobacilli (p = 0.002), while patients with NASH who were overweight displayed a diminished abundance of Bifidobacterium (p = 0.018). Furthermore, patients with lean NASH displayed a deficiency in Lactobacillus in comparison to their overweight and obese NASH counterparts. Interestingly, this lean NASH subgroup exhibited a gut microbiome alpha diversity akin to that of the control group. Despite qualitative distinctions between lean NASH and overweight/obese NASH, these disparities did not achieve statistical significance (p = 0.618). 158
Macrophages derived from individuals with lean MAFLD exhibit heightened production of inflammatory cytokines, mirroring levels similar to their nonlean counterparts, suggesting a potential loss of metabolic adaptation over time. Comprehensive analyses of the transcriptome and chromatin landscape unveil that metabolic endotoxemia in lean MAFLD prompts a proinflammatory gene program and increases TLR4 production, consequently hindering BA signaling. 155 Long-term outcomes for patients with lean MAFLD prove comparable to or worse than those observed in nonlean MAFLD counterparts, signifying a gradual waning of early metabolic adaptation.156,157,159 The instigation of microbial dysbiosis induced by a Western-style diet contributes to endotoxemia, hepatic inflammation, and the infiltration of proinflammatory immune cells within the milieu of lean MAFLD.101,160
Both lean and nonlean patients manifest analogous macrophage responses, implying that macrophages may constitute early sites of adaptive mechanism exhaustion in the context of lean MAFLD. A metabolic–epigenetic axis governs macrophage inflammatory and metabolic responses, with lean MAFLD exhibiting modified chromatin accessibility and transcription factor networks.155,161 Overall, the complex interplay of microbial dysbiosis, compromised bile acid signaling, and altered epigenetic regulation contribute to the subtle progression of lean MAFLD, shedding light on potential avenues for therapeutic intervention.
Gut Microbiota and MAFLD diagnosis
Until now, no existing microbiota-derived testing has been validated for diagnosing or staging MAFLD. Currently, the most used diagnostic method is invasive (liver biopsy), and the available noninvasive biomarkers lack validity and an inability to evaluate a wide spectrum of MAFLD. Transient elastography offers the most reliable and consistent diagnosis. Therefore, there is a vast need for novel biomarkers that are accurate and reliable, and microbiota-associated biomarkers may hold some potential for diagnosing MAFLD.20,162 Table 1 lists the microbiota-associated biomarkers for MAFLD.
Microbiota-associated biomarkers for MAFLD.
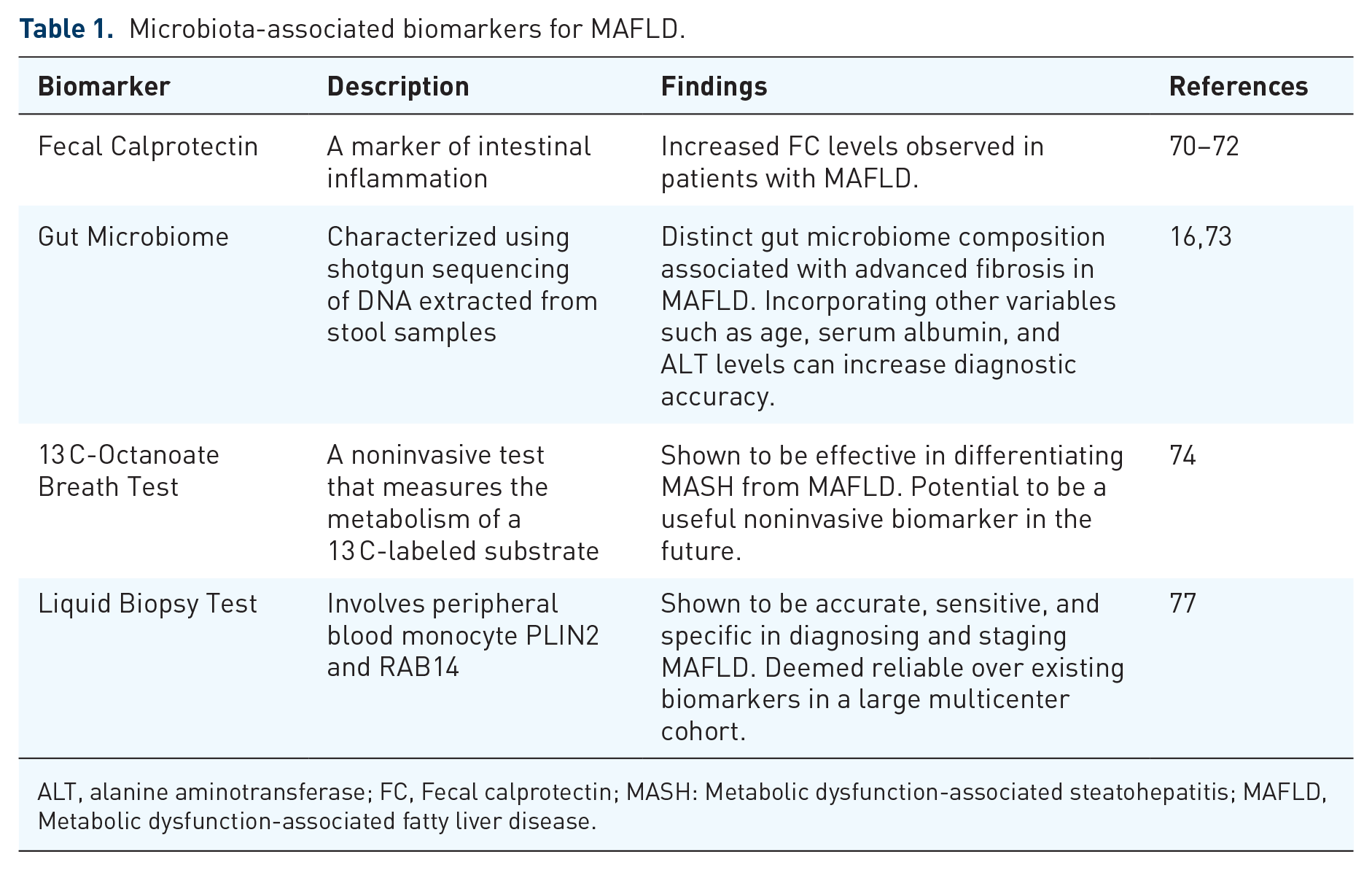
ALT, alanine aminotransferase; FC, Fecal calprotectin; MASH: Metabolic dysfunction-associated steatohepatitis; MAFLD, Metabolic dysfunction-associated fatty liver disease.
Fecal calprotectin
Recent studies show a correlation between MAFLD and inflammatory bowel disease, which supports the proposed theory of intestinal inflammation and permeability in the pathogenesis of MAFLD.163–165 Markers of systemic inflammation, like calprotectin, were studied in patients with MAFLD. Ponziani et al. 166 studied 41 patients and 20 healthy controls. They concluded that in patients with MAFLD cirrhosis, gut microbiota, and systemic inflammation were significantly correlated in the process of hepatocarcinogenesis. Moreover, Bourgonje et al. 167 found that higher plasma calprotectin levels are associated with suspected MAFLD and the risk of all-cause mortality. Demirbas et al. 168 also observed elevated fecal calprotectin (FC) levels in a pediatric cohort, including patients with obesity and MAFLD. Unfortunately, serum FC and myeloperoxidase levels did not show the same correlation as demonstrated by Bıçakçı et al. 169 Interestingly, Stehura et al. 170 added more complexity to the FC/MAFLD correlation question by investigating FC in 46 patients with MAFLD and coronavirus disease 2019 and observed higher FC levels. Further investigation is warranted to determine precise serum or fecal calprotectin levels before it can be used as a marker of MAFLD. However, it can still be used as a good indicator of gut inflammation.
Gut microbiome
Loomba et al. 22 developed a novel model that demonstrated robust diagnostic accuracy (area under the receiver operating characteristic curve: 0.94) by exploring a panel of gut microbiome-derived biomarkers to envision the presence of advanced fibrosis in 86 patients with biopsy-proven MAFLD. It was further validated in an independent cohort. Dong et al. 171 in 2020 recruited 50 patients with chronic liver diseases, including MAFLD, and 25 healthy controls. Microbiome composition was unique in patients with advanced fibrosis (p = 0.003), who had enriched levels of Prevotella copri, the most predictive microbiome in the classifier. Taking other biomarkers and variables, like age, serum albumin, and alanine aminotransferase levels, into account can increase the accuracy of detecting advanced disease stage and cirrhosis, as demonstrated by Oh et al. 21 All these findings stress the fact that a distinct microbiome signature is present and may play a diagnostic role in staging patients with MAFLD.
Other noninvasive tests
Several studies were conducted to identify novel surrogate biomarkers that could have diagnostic utility in MAFLD. Fierbinteanu-Braticevici et al. 172 demonstrated the efficacy of 13C-Octanoate breath test in differentiating MASH from MAFLD and its potential to become a useful noninvasive biomarker in the future. Octanoate is quickly absorbed in the small intestine and metabolized in the liver via beta-oxidation to acetyl-CoA and carbon dioxide (CO2). The exhaled CO2, collected at varied time points, allows for time-sensitive hepatic function evaluation. Other studies examined the potential effect of microbiota metabolites, such as amino acids, on steatosis in MAFLD.173,174 Women exhibiting steatosis demonstrated reduced microbial gene diversity alongside heightened endotoxin production, particularly from Proteobacteria. Additionally, these patients showed imbalances in the metabolism of aromatic and branched-chain amino acids. Another study showed the consistency of microbial metabolite, 3-(4-hydroxyphenyl) lactate and its significant association with MAFLD and liver fibrosis. Noteworthy, Angelini et al. 175 demonstrated in their large multicenter cohort that the novel PLIN2/RAB14- based liquid biopsy test was accurate, sensitive, reliable, and specific in diagnosing and staging MAFLD compared with existing biomarkers; however, the study was limited to Caucasians, and further investigations are required.
Gut dysbiosis and MAFLD: therapeutic interventions
There are several potential microbiota-related therapeutic targets for MAFLD. Some of those are listed in Table 2, while Figure 2 depicts the overview of current microbiota-related avenues for MAFLD regarding diagnosis, pathophysiology, and therapeutics.
Therapeutic MAFLD targets related to microbiota.

ALT, alanine transaminase; F/B, Firmicutes/Bacteroidetes; MASH, Metabolic dysfunction-associated steatohepatitis; NASH, nonalcoholic steatohepatitis.

Overview of the current microbiota-related avenues for MAFLD.
Diet
Multiple scientific studies have emphasized the importance of weight loss, aiming for a 7−10% reduction in body weight through a hypocaloric diet (aiming for an energy deficit of 500–1000 kcal/day) in addition to exercise to create a caloric deficit.78,79 However, there are divergent opinions on specific dietary recommendations (e.g., processed food and alcohol consumption). Limited evidence exists for some diets, like LCD or low-fat diets. Certain associations specifically highlight the potential benefits of the Mediterranean diet for patients with MAFLD. It is worth mentioning that similar diet recommendations apply to patients with MAFLD and type-2 diabetes, emphasizing that an individualized approach that focuses on calorie restriction and adherence to the Mediterranean diet could be hugely beneficial.162,176–180
Dietary interventions can target gut dysbiosis and restore gut homeostasis. In rodents, chronic administration of an HFD was associated with an increased abundance of Firmicutes and a decreased presence of Bacteroidetes species, resulting in a higher F/B ratio. 181 On the other hand, a high-fiber diet has shown protective effects against hepatic inflammation and has been linked to an increased presence of Akkermansia muciniphila. 182
Caffeine consumption is suggested to protect against MASH and its progression. Various mechanisms have been proposed, including glutathione production, scavenging reactive oxygen species, and gut microbiota modulation, driven by the active alkaloids and phenolic compounds in coffee and its ability to restore the F/B ratio and promote the growth of Bifidobacterium species.183–187
Exercise
Exercise training has been found to induce changes in the gut microbiota composition. Clarke et al. 188 showed evidence that athletes and healthy persons with a low BMI have different microbiota compositions with higher proportions of Akkermansia than healthy controls with a high BMI. Animal models have demonstrated that exercise is associated with a decreased abundance of certain species (e.g., Lactobacillaceae, Proteobacteria, Bacteroidetes, Flavobacterium, Alkaliphilus, F/B ratio, etc.) and increased numbers of others (e.g., Verrucomicrobia, Turicibacteraceae, etc.). The effects of exercise can go beyond those of diet alone, as shown when comparing exercise to calorie restriction in high-fat diet-fed animals. Exercise not only improved insulin sensitivity but also resulted in a greater reduction of low-density lipoprotein (LDL) cholesterol, primarily due to exercise-induced changes in the gut microbiome. These microbiota modifications have been associated with improved serum LDL cholesterol levels, liver fat mass, and triglycerides.189–191 In humans, we already know that exercise is associated with reduced rates of MAFLD, 9 but future studies are needed to confirm the link between exercise and the effects on the microbiome in patients with MAFLD.
Antibiotics
Antibiotics and their effect on gut microbiota have been explored as therapeutic targets in MAFLD. Norfloxacin and neomycin improved liver function by altering microbiota and causing bacterial translocation. On the contrary, another study showed no hepatic benefit for norfloxacin in patients with MAFLD. 192 Metronidazole has shown a positive effect on alanine transaminase (ALT) levels in combination with inulin supplementation compared to placebo (mean ALT change −19.6 versus −0.2 U/L, respectively; p = 0.026). 193
Several studies examined the use of rifaximin, a nonabsorbable antibiotic, in MAFLD. Kakiyama et al. 194 documented the effect of rifaximin in patients with cirrhosis, including the reduction in the ratio of secondary BAs to primary BAs. However, no substantial alteration was seen in the gut microbiota’s bacterial composition, apart from the decrease in Veillonellaceae. It is pertinent to highlight that most of the study participants were patients with hepatitis C and ALD. Gangarapu et al. 195 showed the beneficial effect of 4 weeks of rifaximin (1200 mg/day) on liver functions, such as ALT. Interestingly, Cobbold et al.’s 196 study showed no alteration in ALT following 6 weeks of rifaximin (800 mg/day). Notably, Abdel-Razik et al. 197 showed that rifaximin (1100 mg/day) significantly decreased serum ALT (from 64.6 ± 34.2 to 38.2 ± 29.2; p = 0.01) and serum aspartate transaminase (AST) (from 66.5 ± 42.5 to 41.8 ± 30.4; p = 0.02) after 6 months in patients with MAFLD. In addition, rifaximin reduced serum endotoxin and improved insulin resistance, proinflammatory cytokines, CK-18, and NAFLD liver fat score. Evidence suggests the benefits of rifaximin for MAFLD, but more studies are needed to establish the length and dose of administration.
Probiotics, prebiotics, and synbiotics
Researchers have increasingly investigated the therapeutic potential of microbiota modulators, such as probiotics, prebiotics, and synbiotics, to shape gut microbiota for hepatic health. Probiotics, beneficial microorganisms, can potentially improve hepatic inflammation, oxidative stress, and steatosis. Prebiotics serve as nourishing substrates for beneficial gut bacteria, while synbiotics, which combine probiotics and prebiotics, provide an innovative strategy to restore gut dysbiosis and bolster the survival and function of beneficial gut microbes. Keeping in mind that microbiota modulators do not possess curative effects, they hold potential as adjunct therapies to reach therapeutic goals in MAFLD. Escouto et al. 198 observed a significant improvement in AST to platelet ratio index after 6 months of probiotics supplementation. Manzhalii et al. 199 conducted an open-label trial using a probiotic cocktail over 12 weeks, which showed improved liver inflammation. Unfortunately, only a few research works have explored the role of probiotics on histologic markers of MAFLD and MASH. Focusing more on cardiovascular risk factors, Barcelos et al. showed that 24 weeks of supplementation with probiotics was not superior to placebo in reducing cardiovascular risk markers in MASH. 200
Preclinical studies of prebiotics have demonstrated their potential to improve biochemical and histologic markers of MAFLD. 201 A randomized trial with a placebo crossover design involving patients with biopsy-proven MASH (n = 7) received prebiotic administration, specifically oligofructose (16 g/day). The results showed a significant reduction in hepatic levels of AST compared to placebo (p < 0.05) and a nonsignificant decrease in triglyceride concentrations after 8 weeks of treatment. 202 However, a systematic review encompassing four clinical studies involving patients with obesity-related MAFLD did not provide substantial support for using prebiotics, primarily due to limitations in study quality. 203 A recent systematic review that included 13 trials showed that probiotics could reduce BMI, total fat percentage, total cholesterol, triglycerides, fasting insulin, lipopolysaccharide, homeostatic model assessment-insulin resistance, AST, ALT, gamma-glutamyl transferase (GGT), tumor necrosis factor-alpha (TNF-α), interleukin-6, liver stiffness, fat fraction, fat liver index, vaspin, and Clostridia and Erysipelotrichia classes. Prebiotics can reduce intra-hepatocellular lipids, NASH score, Roseburia, and Dialister and increase Bifidobacterium levels. Synbiotics can reduce BMI, AST, ALT, GGT, TNF-α, NAFLD fibrosis score, and liver stiffness, as well as improve Bifidobacterium levels. 204 A larger systematic review of 26 RCTs showed that synbiotics and probiotics are potentially the most effective therapies that can reduce AST and ALT in adult patients with MAFLD, respectively. 205
In a study by Malaguarnera et al., 206 66 patients with histologically diagnosed MASH were randomly divided into two groups. One group received a synbiotic treatment consisting of Bifidobacterium longum and a prebiotic (fructooligosaccharides), while the other group received placebo. Both groups went through lifestyle modifications and received a vitamin B regimen. The results showed that the active treatment group had significantly lower levels of TNF-α and C-reactive protein (CRP), as well as histologic improvement with decreased hepatocellular injury, inflammation, and steatosis after 24 weeks of treatment. In the largest double-blind, placebo-controlled trial, patients with ultrasound-diagnosed MAFLD (n = 80) were randomized to obtain an 8-week synbiotic treatment (probiotics including Lactobacillus casei and others) or placebo. At the end of the intervention period, the synbiotic group showed a significant reduction in steatosis as determined using an ultrasound scan, while those who received placebo showed no significant improvement. 207 Although there were no significant differences in CRP, ALT, or AST levels between the synbiotic and placebo groups (adjusted for energy intake), another study of 28-week supplementation in 50 lean patients with MAFLD showed a significant decrease in fibrosis, hepatic steatosis, fasting blood sugar, triglyceride levels, and inflammation markers. 208
GLP-1 receptor agonists
Numerous studies have highlighted the impact of the gut microbiota on glucose regulation and satiety.209–212 GLP-1 agonists have demonstrated their effectiveness in curbing calorie intake, promoting weight loss, improving glucose tolerance, and reducing cholesterol levels and cellular apoptosis in both MASH and obesity.213,214 Hupa-Breier et al. 215 examined the effects of GLP-1 in nondiabetic mice with MASH, finding that dulaglutide, alone and in combination with empagliflozin, led to significant weight loss, improved glucose regulation, and reduced anti-inflammatory and antifibrotic responses.
In addition, liraglutide has shown promising results in enhancing glucose and lipid metabolism in obese rat groups, regardless of their hyperglycemia status. Notably, liraglutide induced significant alterations in the gut microbiota composition, decreasing its diversity and abundance while promoting lean-related microbial characteristics and reducing obesity-related phenotypes. 216 These findings suggest that GLP-1 agonists may play a role in preventing weight gain by modulating the gut microbiota, but further in-depth investigations are required to fully comprehend the underlying mechanisms responsible for these weight-controlling effects.
Fecal microbiota transplantation
Fecal microbiota transplantation (FMT) has been investigated as an intervention for MAFLD, with multiple trials registered. However, the results have been contradictory. In a study by Craven et al., a single FMT infusion did not lead to a reduction in liver steatosis. 217 In contrast, another study involving a 3-day FMT infusion demonstrated a modest yet significant decrease in the severity of steatosis. 218 In a >12-month follow-up study by Bajaj et al., patients in the FMT arm showed sustained improvement in clinical and cognitive function parameters, with no recurrent hepatic encephalopathy occurrence and hospitalizations due to liver complications. 219 These findings highlight the variability in outcomes observed with FMT as a therapeutic approach for MAFLD up to this point.
Conclusion and future perspectives
In the field of MAFLD, there remains a significant need for improved diagnostic and therapeutic approaches. Currently, available methods are limited, emphasizing the importance of early detection for effective interventions. Well-designed, randomized studies exploring microbiota, microbiome signatures, and metabolites are essential to uncover underlying disease mechanisms and identify individuals at risk of MAFLD at an early stage. While the gut–liver axis has shown promise in managing MAFLD through interventions like rifaximin, prebiotics, probiotics, GLP-1 agonists, and fecal transplantation, the effects are often indirect, and individual responses can vary. More comprehensive studies are needed to precisely characterize microbial changes at different disease stages, including bacteria, viruses, and fungi, and to assess the benefits, dosages, duration of supplementation, long-term effects, and safety of probiotics, prebiotics, and synbiotics for preventing and treating MAFLD. Large-scale intervention studies with well-defined patient groups and reproducible endpoints are necessary to assess the effectiveness of microbiota-based interventions in managing MAFLD and its potential as a therapeutic avenue. Despite challenges, innovative diagnostic and therapeutic approaches focusing on the gut microbiota hold the potential to tackle the complex and challenging spectrum of MAFLD, emphasizing the need for continuous research and exploration in this field.
